Abstract
A ventriculoperitoneal shunt (VPS) is a medical device used to deflect cerebrospinal fluid from cerebral ventricles to the peritoneal cavity. Since 1908, ventriculoperitoneal shunts have been considered the best option in hydrocephalus management. Manifestation related to VPS may arise anywhere whilst tunneling the VPS from the ventricle to the peritoneal cavity, but complications associated with the distal catheter are especially common and a leading cause of revision surgeries. Among the many complications associated with peritoneal shunts, migration of the catheter into other parts of the body is still common. Scrotal migration mainly takes place in children aged 4 days to 5 years. Patent processus vaginalis, formation of an inguinal hernia, and increased intraabdominal pressure are leading risk factors associated with migration of the peritoneal catheter into the scrotum. Thoracic migration of peritoneal catheters predominantly occurs in adult patients. Important mechanisms in thoracic migration include (1) iatrogenic perforation of the external or internal jugular veins by the surgeon during tunneling and (2) gradual destruction of the venous structures by the catheter due to routine extension and flexion of the neck. After entering the vessel, negative intrathoracic pressure combined with positive intraabdominal pressure aids migration into the thoracic cavity. Sharp tips or shunt types contribute to intraabdominal migration and perforation.
Introduction
A ventriculoperitoneal shunt (VPS) is a medical device that deflects cerebrospinal fluid (CSF) from cerebral ventricles to the peritoneal cavity (Fig. 1). Since 1908, VPSs have been considered the best option in hydrocephalus management [1]. Manifestation related to may arise anywhere whilst tunneling the VPS from the ventricle to the peritoneal cavity, but complications associated with the distal catheter are common, and a leading cause of revision surgeries [2–4]. Migration of the peritoneal catheter into different sites, such as the scrotum [5], pelvis [6], thoracic cavity [7], and abdomen [8], has been discussed extensively within the literature.
Among the many complications associated with peritoneal shunts, migration of the catheter into other parts of the body is reported in approximately 8.6% of cases [9]. This manuscript aimed to explore the possible mechanisms and important locations of distal catheter migration (Table 1).
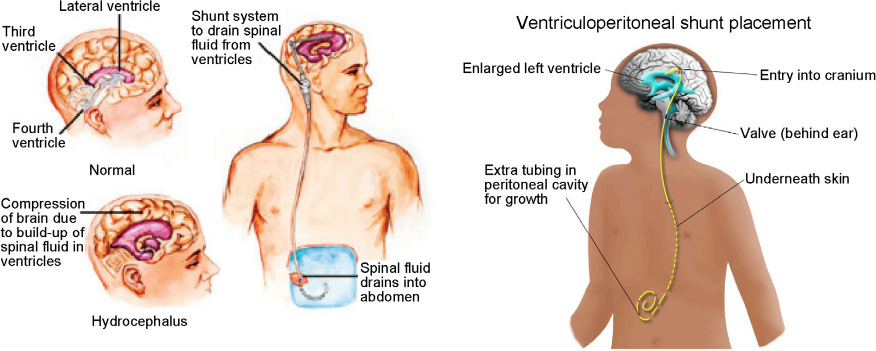
A ventriculoperitoneal shunt is a medical device that deflects cerebrospinal fluid from cerebral ventricles into the peritoneal cavity.
Review of the migration of distal ventriculoperitoneal shunt catheters into different sites.

Scrotal and vaginal migration
In their case report, Oktem et al. discussed the migration of abdominal catheters into the scrotum of four male children, aged 9 days to 10 months. Among the four patients, three had Pudenz catheters and one had a Radionics distal catheter. Abdominal radiography revealed that the Pudenz shunts were in the right scrotum of all three patients, whilst the Radionics shunt was in the left scrotum. The patency of the processus vaginalis is approximately 60%–70% in the first three months of an infant's life, and 50%–60% at 1 year of age; thus, Oktem et al. suggested that migration into the scrotum was due to a patent processus vaginalis, and that development of postoperative scrotal hydrocele, or swelling in infants with a VPS, should be taken as an indication of possible distal shunt manifestation [5, 10].
In a case report by Crofford and Balsam [10], four male patients, aged 2 weeks, 1 month, 3 months, and 4 years, were diagnosed with hydrocephalus and managed by VPS surgery. Upon physical examination, scrotal swelling and hydrocele were found, and abdominal radiography revealed that the tip of the catheter was in the right scrotum in all cases except the latter (4-year-old), in which it was present in the left scrotum. Three patients required repositioning of the VPS and herniorrhaphy, whilst one patient, in whom the catheter returned spontaneously to the abdomen, required herniorrhaphy only. Crofford and Balsam considered the processus vaginalis and increased intraabdominal pressure among the likely causes of scrotal migration in these cases [10, 11], hypothesizing that surgical repair is needed once a hydrocele, with or without a hernia, is diagnosed, and that shunt revision itself is seldom necessary, though repositioning is vital [12].
A 4-week-old boy was admitted for signs of hydrocephalus, which was later confirmed by computed tomography (CT). VPS surgery was performed using Pudenz ventricular and Raimondi peritoneal catheters. Five months after the operation, the Raimondi catheter had dislodged from the valve and was found via abdominal radiography, on the left side of the peritoneal cavity. The shunt was replaced with a radio-opaque Pudenz catheter, and the Raimondi shunt was left
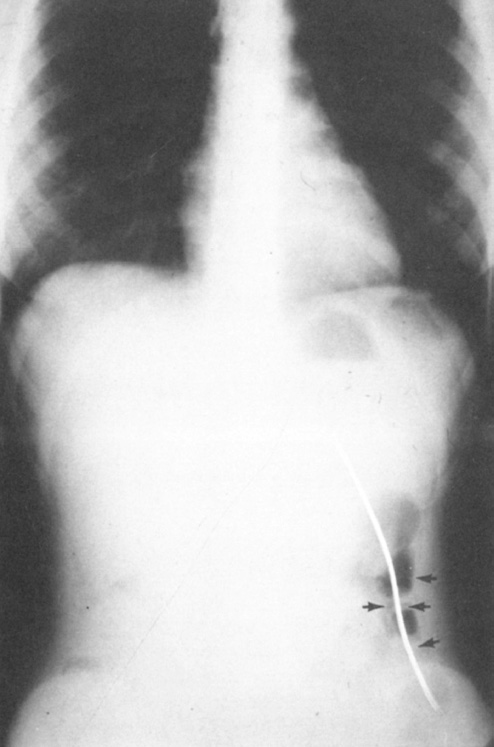
Peritoneal shunt migration was reported in a 5-year-old boy with scrotal swelling and hydrocele 4 months after VPS implantation. Abdominal radiography provided evidence of coiled peritoneal catheter migration into the left scrotum (Fig. 2). The catheter migration was resolved by manual compression of the swollen scrotum, and repair of the patent processus vaginalis. In this series, the authors described entrance of the catheter into the scrotum via the patent processus vaginalis, which became the scrotal tunica vaginalis; thus, surgical obliteration of the processus vaginalis and catheter repositioning are strongly suggested to remove the risk of catheter migration and malfunction [14].
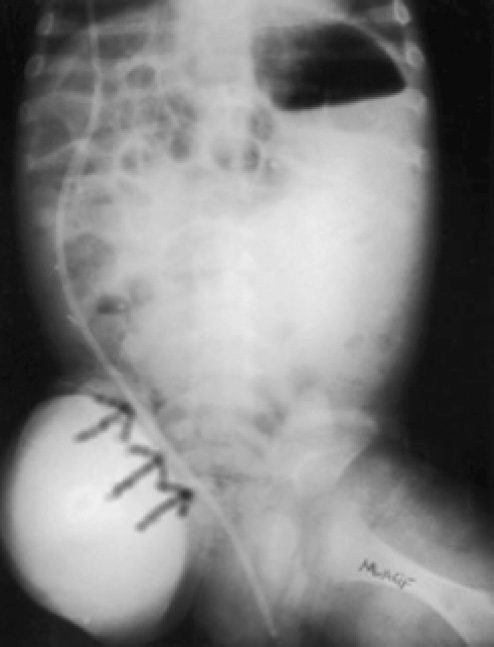
Plain abdominal radiograph shows the catheter migrated into the right scrotum.
Two cases with histories of VPS surgery presented with complaints of right inguinoscrotal swelling. In both, a tube-like object was palpated within the swelling, suspected, and confirmed via abdominal radiography, to be the previously inserted shunts. Right inguinal herniotomies were carried out, along with repositioning of the shunts into the abdominal cavities. A fascinating observation, in the radiographs of both patients, was that the abdominal end of each shunt coursed through the right paracolic gutter to enter the patent processus vaginalis on the right side. In this manuscript, the writers illustrated the mechanism of catheter migration, from the starting point to the final position, in three steps: (1) displacement of the catheter from the initial position; (2) migration of the catheter; (3) arrival at the patent processus vaginalis. Various factors, including the up and down movement of the liver during respiration and forceful peristalsis of the pylorus and duodenum, are involved in displacement. Vigorous peristaltic movement of the small bowel aids migration [15]. A neonatal girl baby endured darning of a huge lumbar myelomeningocele, followed a few weeks later by ventriculoatrial shunt for hydrocephalus. After period of 7th months, as a result of blockade at the atrial end, the shunt was converted into a ventriculoperitoneal shunt with a Pudenz valve and a Raimondi catheter; the patient was readmitted to the hospital because of vomiting, stiff neck, and fever, after 4 months of this surgery. A cerebrospinal fluid gained for culture profile from the valve reservoir grew enterococci. The patient was carried on high doses of antibiotics. During third day of admission, the responsible nurse noted that the child's bed nappies were frequently wet; assessment of that day discovered the tip of the Raimondi catheter lying in the vagina. Under general anesthesia investigation ended it clear that had of catheter perforated post-vaginal wall and the end had been extruded throughout vagina (Fig. 3). After x-ray (Fig. 4) the previous VPS system was removed. The patient was discharged in satisfactory condition. A new VPS system was operated at a later period after the infection had been controlled.
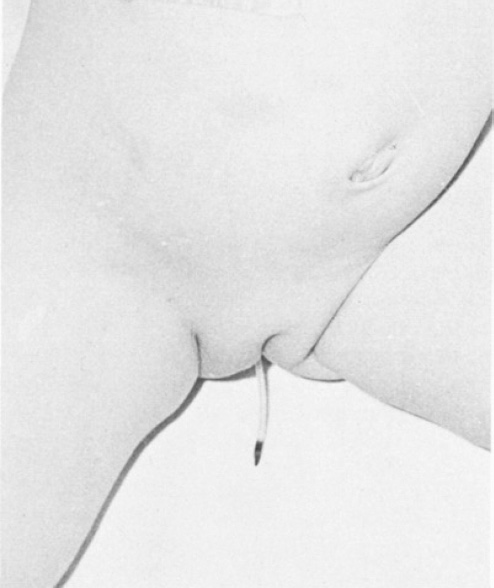
The end of a Raimondi ventriculoperitoneal shunt catheter projecting from the vagina after a displacement period of 4-months (self-photograph).
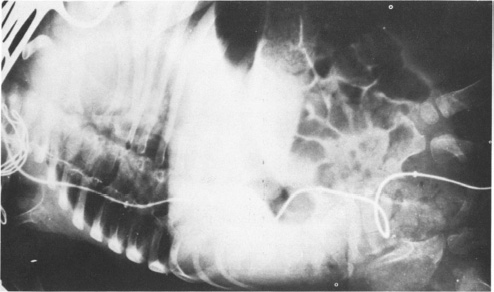
Vaginal protrusion of the catheter was discovered immediately after radiography clearly showed the course of the shunt system, from the neck to the vaginal outlet.
A 71-year-old man underwent VPS surgery after being diagnosed with hydrocephalus; however, 3 weeks after shunt placement, the patient was readmitted due to recurring gait difficulty, abdominal pain, sleep apnea, and thoracic fullness. A thoracic computed tomography (CT) scan revealed distal catheter migration through a right internal jugular vein into the heart, causing a blockage in the pulmonary artery (Fig. 5). The authors doubted that this intravascular catheter migration was due to the two mechanisms previously described by Morel Robert et al. [16]; rather, they postulated the following two mechanisms: (1) direct iatrogenic perforation of the external or internal jugular veins by the surgeon during tunneling; or (2) destruction of venous structures by the catheter over time, as a consequence of routine extension and flexion of the neck. After entering the vessel, intrathoracic negative pressure combined with positive intraabdominal pressure may have aided this catheter migration into the heart [17, 18].
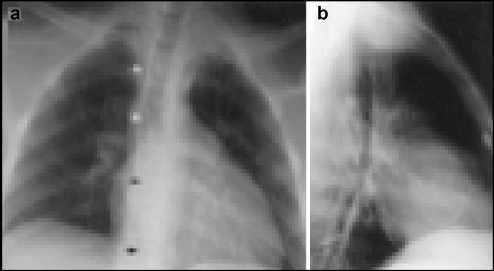
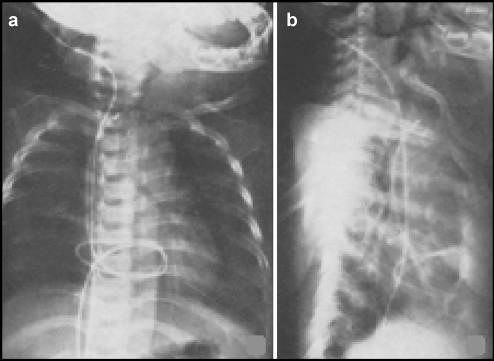
Plain chest radiographs. (a) Posterioanterior view showing the distal catheter stalking down toward the peritoneum (line of arrows). (b) Lateral view showing the tubing coils within the pericardial shadow.
A 51-year-old woman was diagnosed with normal pressure hydrocephalus, which was managed by VPS surgery; however, 5 months later, the patient has re-hospitalized with symptoms of fever and headache. Routine chest radiographs revealed migration of the catheter into the thoracic cavity, whilst Duplex echography revealed that the shunt had migrated into the right external jugular vein. CT scans showed the tip of the shunt coiled inside the pulmonary artery. The surgeons replaced the catheter to prevent further complications [19]. The mechanism behind this migration was thought to be the transvenous positioning of the catheter, whilst negative inspiratory pressures were implicated in the migration of the distal-end into the heart [19–21].
Congenital hydrocephalus of a 1-month-old baby boy was treated by VPS surgery with a medium-pressure Raimondi peritoneal catheter. At 12 years of age, the boy was readmitted for complaints of headache and vomiting. Thoracic radiographs were obtained, and the distal catheter was identified in the right lower phrenic region of the chest cavity. Shunt revision confirmed the peritoneal catheter lying in the dilated subcutaneous thoracic tract. Two months after the revision surgery, the boy was readmitted with vomiting, headaches, chest distress, and fever. This time, upon abdominal radiographic examination, the catheter tip was found to be piercing the diaphragm, penetrating the heart through the vena cava. The abdominal shunt was replaced for the third time, with a 20-cm-long shunt in the peritoneum, fixed within the subcutaneous tissue. The authors proposed two possible mechanisms and leading elements: (1) kinking of the catheter in the supraclavicular fossa during the tunneling process, with negative inspiratory pressure drawing it into the jugular vein; and (2) iatrogenic perforation of the jugular vein by the catheter guide [22].
A 12-year-old boy, who had undergone VPS surgery at 10 days old, was admitted to hospital due to intermittent frontal headaches and vomiting. CT examination revealed moderate ventricular enlargement, and shunt series showed the shunt entering the vena cava, right atrium and ventricle, and pulmonary artery. These findings were confirmed by echocardiography, and mild tricuspid regurgitation was found due to the catheter. Dislodgement of the coiled catheter from the pulmonary artery was attempted repeatedly (with Cook deflector wire), but the shunt was adherent to the pulmonary artery wall and only one-third of the shunt was retrieved. Gradual erosion into the vein or transvenous placement of the shunt, secondary to subcostal tunneling, were the proposed mechanisms for triggering migration [16].
A 14-year-old girl was hospitalized for complaints of papilledema, headaches, and vomiting. Magnetic resonance imaging revealed tetraventricular hydrocephalus. VPS implantation was performed, and no complications were noted during the operation. One month after surgery, the girl had abdominal pain and retro-auricular swelling. Abdominal radiography revealed that the distal part of the catheter was As revealed by neurological examination, the condition of the patient had significantly improved before her arrival (she moved all four extremities with strength). Patient was noted to have persistent hypertension, and the electrocardiogram demonstrated normal sinus rhythm and prominent left ventricular forces. Transthoracic echocardiography shown normal heart and valve function, but a linear density was noted in the patient's right ventricle, which was suggestive of a retained clot from an old central line. Plain x-rays of the chest revealed VP shunt tubing traveling down the chest; and coils of tubing were within the patient's superior vena cava and pericardium (Fig. 5). The x-rays images were initially read as a normal distal catheter with a second piece of tubing originating from either a retained catheter or central line coiled within the pericardium. A subsequent nuclear medicine patency study of the VP shunt demonstrated that all of the visualized tubing was connected to the distal catheter, and the shunt was observed to be functioning well with good flow of radiotracer into the peritoneum. CT examination of the chest demonstrated the exact landscape of the distal catheter's path (Fig. 6). The catheter moved down the neck deep to the patient's sternocleidomastoid muscle. There it stabbed the right internal jugular vein, inserted the superior vena cava, and looped within the patient's right atria and ventricle. The catheter then traveled back upward the superior vena cava and internal jugular vein, where it exited above the clavicle, and it then traveled back downward the patient through the subcutaneous tissue along the patient's chest before entering the peritoneal space. Several days before conclusive treatment, the distal catheter tip was observed to have migrated from the peritoneum to the subcutaneous space of the chest (Fig. 7 and Fig. 8). A collaborative procedure with the pediatric cardiothoracic surgery team was deliberate because of the probability that the distal catheter had tangled around the tricuspid or pulmonic valves, knotted from the multiple coils within the ventricle wall.
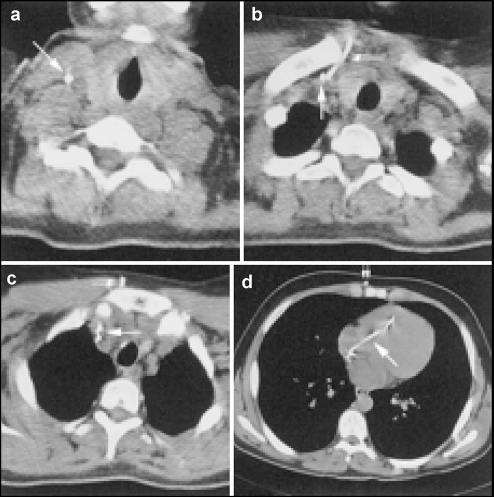
Computed tomography scans of the chest. (a) Distal catheter running deep into the sternocleidomastoid muscle (arrow). (b) Downward distal catheter within the internal jugular vein (single arrow) and ascending distal catheter surrounding the internal jugular vein, above the clavicle (double arrows). (c) Descending and ascending distal catheters within the superior vena cava (single arrow) and descending distal catheter tracking within the subcutaneous tissue toward the peritoneum (double arrows). (d) Loops of distal catheter within the right atrium and ventricle (single arrow) and descending distal catheter within the subcutaneous tissue (double arrows).
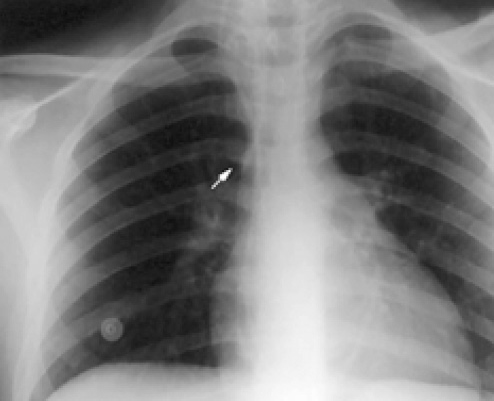
One-week later, plain chest radiographs revealed the distal tip of the catheter (arrow) had migrated from the peritoneum to the subcutaneous tissue of the chest.
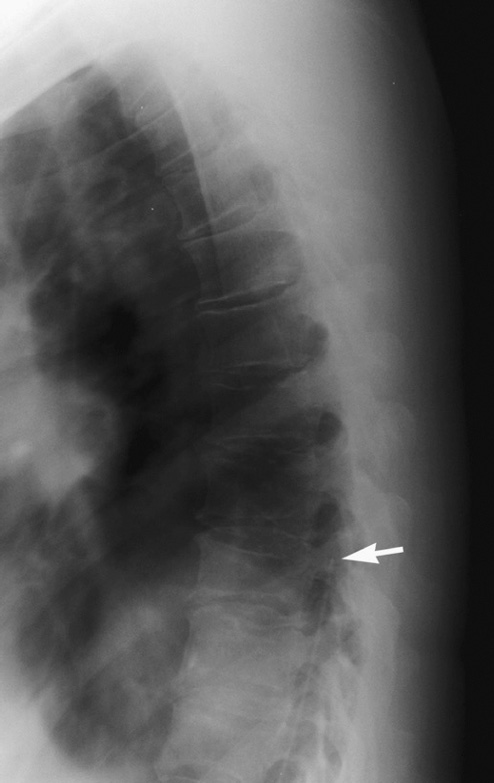
Thoracic lateral plain film spine radiograph showing the tip of the baclofen pump catheter at T9 (arrow), 1 month prior to removal.
The patient was placed in the supine position, and the previous right occipital scalp incision was reopened to define the shunt valve and distal catheter. Simultaneously, the cardiac surgeons made a subxyphoid incision and shaped a pericardial window through the diaphragm to visualize the pericardium while the distal catheter was slowly retrieved through the occipital incision.
Our Cardiothoracic team decided that a thoracotomy was unnecessary, because the pericardium was filled with blood when the distal catheter was drawn out. A new distal catheter was located into the peritoneal cavity. The patient's postoperative course was uneventful, and a postoperative plain x-ray of the chest demonstrated proper placement of the distal catheter in the peritoneum.
Consistent with Morell et al. [16], the authors concluded that the catheter passer could have pierced the internal jugular vein during the tunneling process, with retrograde blood flow and negative intrathoracic pressure pulling the catheter into the heart [23].
When another shunt malfunction was suspected, we performed a shuntgraphy using Conray 60. There was no occlusion on the ventricular side. It revealed, however, that the abdominal catheter was within the mediastinum (Fig. 9 and Fig. 10). The catheter returned at the diaphragm, and the tip remained at the neck. Palpation of the skin of the chest did not reveal any tube subcutaneously.
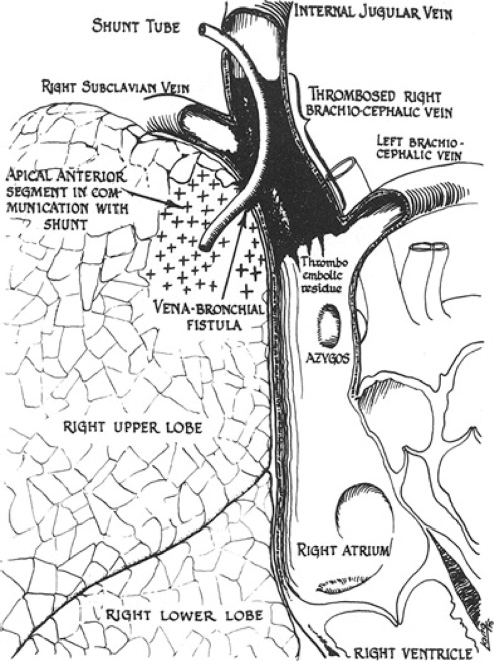
Anatomy of the cerebrobronchial fistula, thrombosis of the right internal jugular vein, and pneumonia of the right apical anterior lung segment.
(a) An anteroposterior view of the shunt gram of the abdominal catheter looped down along the right border of the sternum and back upwards at the diaphragm; the tip remained at the neck. (b) Lateral view of the shunt gram; the abdominal catheter is situated between the sternum and the thoracic vertebrae.
The second shuntography after two days showed complete obstruction of the abdominal catheter, and replacement of the abdominal catheter and flushing device was performed. The catheter was removed without any difficulty. The new abdominal catheter was fixed to the peritoneum and abdominal fascia. After the operation, vomiting ceased, and the baby was discharged.
Hydrocephalus in a 47-year-old man was treated by insertion of a VPS using a pressure-adjusted valve (Codman HAKIM Programmable Valve; Medos S.A., Le Locle, Switzerland). The location of the distal catheter was checked via abdominal radiography, and no complications occurred postoperatively. After 4 years, the man complained of abdominal pain for 1 month. CT and gastroscopic findings were normal, and intravenous infusion and antibiotics resolved the problem. However, 2 months after the abdominal discomfort, he was readmitted with symptoms of headache and fever. Abdominal (Fig. 11) revealed that the shunt had migrated inside the gastric lumen, and gastroscopy found that the anterior stomach wall had been perforated by the catheter. In the operating theatre, the shunt was extracted through the scalp insertion site. It was concluded that either the sharp tip of the catheter [24] or coiled spring-shape and rigid tip of the shunt [25, 26] had caused gastric perforation.
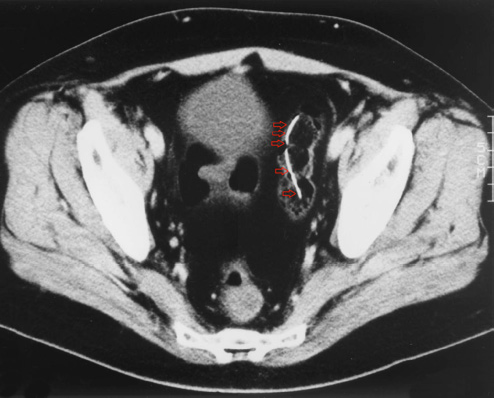
Abdominal computed tomography scan showing a tube in the lumen of the bowel (arrows).
A VPS implanted into a 3-year-old boy failed 2 years after surgery. He was hospitalized for abdominal pain, fever, irritability, and vomiting. The shunt tract was inflamed, and there was left upper quadrant abdominal tenderness. Upper gastrointestinal barium fluoroscopy confirmed that the tip of the distal catheter had moved to the lesser curvature of the stomach (Fig. 12 and Fig. 13). The shunt was withdrawn and 0.6 mL of fibrin glue was injected along the track. Meningeal signs and severe abdominal pain, when present together, are signs of shunt failure in patients treated previously with VPS implantation for hydrocephalus management. Less flexible shunts, such as silastic or spring-loaded catheters, are risk factors for migration [8, 27]. Dementia, gait disturbance, and urinary incontinence in a 69-year-old patient disappeared after VPS surgery with a silastic (Dow Corning Corp., Midland Michigan) catheter. Two months later, she complained of abdominal pain and fever without meningeal signs for one week. Seven months after the operation, she presented with abdominal pain, urinary incontinence, and meningeal signs. Her symptoms dissolved after antibiotic therapy, but her state of consciousness gradually decreased to a level of akinetic mutism. After her death due to pneumonia, autopsy examination revealed perforation of the peritoneal catheter into the stomach, and the sharp catheter tip was deemed to have caused the gastric perforation. The authors commented that viscus perforation in shunting systems is rare, but nevertheless should be considered and abdominal CT scans performed in cases of abdominal pain with meningeal signs [24].
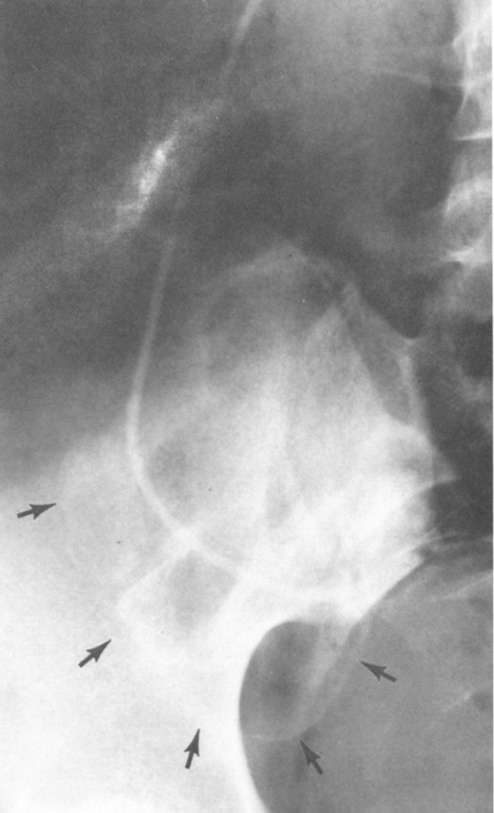
Roentgenogram of the abdomen showing metrizamide outlining the haustral markings of the ascending colon (arrows).
Abdominal roentgenogram showing the shunt catheter within the lumen of the descending colon (arrows).
VPS surgery using a low-pressure Pudenz shunt was performed (in the Neurosurgical Department of Osaka Neurological Institute) for the management of dilated ventricles in a 66-year-old man with signs of dysarthria, meningeal irritation, and drowsiness. The peritoneal end was inserted into the abdomen through a small right upper quadrant abdominal incision. In a few postoperative days, ventricular dilation decreased and the patient's consciousness level increased. Drowsiness was observed again 26 days after surgery, and ventricular dilation was reveled via abdominal CT examination; the catheter tip was observed in the liver parenchyma. The following day, shunt revision surgery was performed and the previous shunt was withdrawn from the liver parenchyma without any difficulty. Various potential mechanisms were involved in this migration, including: (1) the presence of a foreign body in the abdominal cavity (which is the most commonly observed mechanism) [28]; (2) an anchoring effect caused by fibrous encasement of the abdominal catheter, pressurizing the organ parenchyma at specific point, causing perforation [29]; and (3) the development of necrosis [30].
A teenage-boy had a VPS operation (in New York hospital) for hydrocephalus. He suffered from right lower quadrant abdominal pain, emesis, and headaches two years after the surgery. The skin covering the shunt tubing was erythematous; thus, vancomycin was administered intravenously at another hospital. The boy was taken to New York hospital again, but he was asymptomatic at that time. Physical examination was normal, but abdominal CT scans revealed a right lower quadrant peritoneal pseudocyst, with the shunt located inside the cyst. CSF and pseudocyst fluid cultures were sterile; therefore, he was discharged after vancomycin infusion. He experienced headaches with no other symptoms for four subsequent months.
Migration of the tip of distal ventriculoperitoneal shunt through a well-healed abdominal incision was previously reported in an adult patient [33] (Figs. 14 and 15). It perforated the skin, leading to an infection of the still draining shunt through the peritoneal catheter plus ventriculostomy drainage. It is probably desirable if the catheter has been exposed for any length of time. Abdominal complications have been re-viewed by Davidson [34]. In an adult, alternative sites for placement of the distal end of the catheter should be considered.
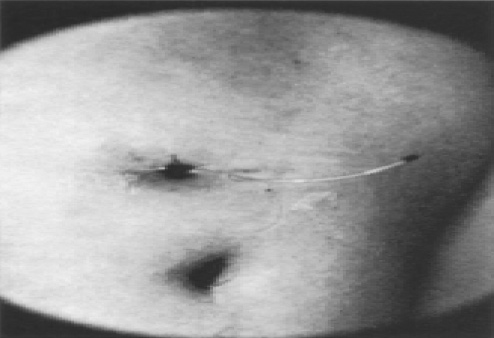
10 cm peritoneal catheter extruded from the center of the abdominal skin incision.
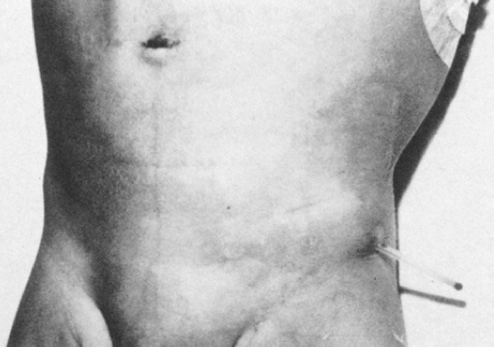
Extra-abdominal migration. The end of the Raimondi catheter is shown protruding from the abdominal wall. The surgical scar is visible at the right upper abdominal wall, near the umbilicus.
The ventricles markedly enlarged after overly vigorous drainage of CSF. Subdural hematomas or hygromas may occur (Fig. 16). If they are sufficiently large or are rapid in onset, it may cause increased intracranial pressure that may subsequently result in death. In the majority of cases, these fluid collections are small and asymptomatic and may resolve on their own [35–37].
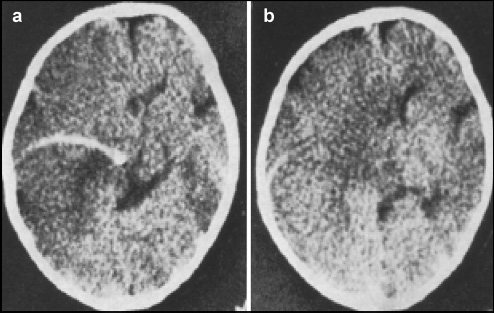
Plain computed tomographic scan revealing a marked midline shift to the right, and large low-density area in the left frontal lobe. The “subdural” catheter was positioned within the brain substance; however, complete migration of the ventriculoperitoneal shunt tubing occurred, with reservoir into the lateral ventricle.
A 6-month-old boy born by cesarean section at 34 weeks gestation on base of ultrasound evidence of progressive hydrocephalus. A ventriculoperitoneal shunt was placed without difficulty and the intraoperative CSF was sterile. At 1- and 4-month follow-ups, they found a functional shunt and a normally growing child. The patient developed seizure 5 months after delivery. with CT scans revealing an enlarged ventricular system consistent with the clinical diagnosis of shunt obstruction. In addition, the white matter seemed dystrophic. The seizures were controlled medically, and a gadolinium-enhanced magnetic resonance imaging (MRI) scan was obtained, (Fig. 17), which shown a multilobed ring enhancing lesion consistent with a brain abscess surrounding the proximal shunt catheter.
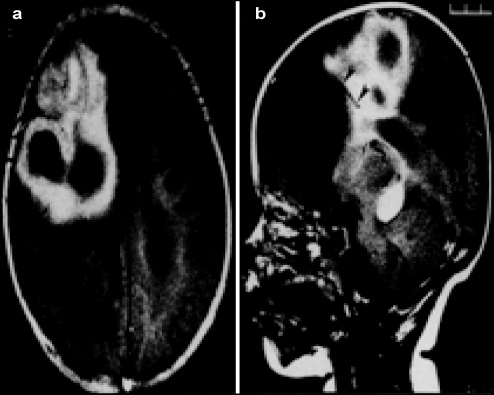
Gadolinium-enhanced axial magnetic resonance imaging scan. (a) A multiloculated abscess is visible in the right frontal region of the brain, surrounded by white matter edema. (b) Sagittal image showing the position of the tip of the ventriculoperitoneal shunt catheter in relation to the abscess cavity (arrowheads).
At the time of surgery, the shunt system was removed with evacuation of the abscess and portion of the capsule. The bile salt-stained distal tubing was pulled from the abdomen through a superficial incision. Cultures from the abscess grew Escherichia coli and Ellterobacter cloacae.
Antibiotics was given 21 days of appropriate, and external drainage of CSF was used to control the hydrocephalus.
The child didn't develop peritonitis.
A new shunt system was implanted after the repeat CSF cultures remained negative, and the child has done well.
A baclofen pump was implanted in a 44-year-old man patient with multiple sclerosis to treat his intractable spasticity, which had previously been managed by his neurologist with oral baclofen.
The patient was placed in the right lateral decubitus position. An InDura 8711 catheter (Medtronic/Sofamor Danek, Memphis, TN) was fixed at the L4–L5 level and attached to a SynchroMed pump (Medtronic), implanted in the left lower quadrant. The procedure were without complications.
Three years later, a plain x-ray of the system was obtained when he noticed that the pump was twisting in its subcutaneous pocket, as a plain x-ray showed a break in the catheter at L4–L5. The most cephalad extent of the catheter free fragment was at T9 (Fig. 18). The patient was given the option of having his pump explored and the catheter replaced. He decided to have the entire device removed because of the high cost of maintaining the pump. Pump was removed from his abdomen, and the lower portion of the spinal catheter was removed.
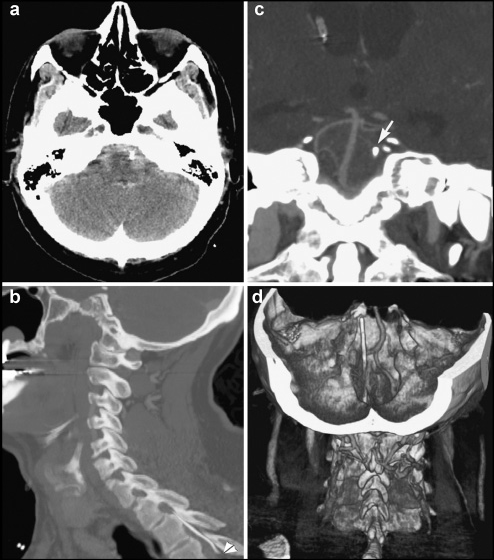
(a) Computed tomography scan of the posterior fossa showing an aneurysmal pattern of subarachnoid hemorrhage. (b) Sagittal modernization demonstrating the top of the catheter of a ventriculoperitoneal shunt. (c) Computed tomography scan (coronal view) showing the catheter rising next to the left of the basilar artery (arrow). (d) Computed tomography image showing a reconstructed three-dimensional view of the posterior fossa, and an angiogram demonstrating the nearby catheter, lateral to the basilar artery and close to the level of the superior cerebellar artery (the ventriculoperitoneal shunt catheter lies posterior to the vertebral artery).
No symptoms of baclofen withdrawal after his pump was removed. After ten days removal of pump and catheter, he sought treatment at the emergency department, reporting the sudden onset of a severe headache. A CT scan showed SAH (Fig. 18a) in the suprasellar, prepontine, and perimesencephalic cisterns as well as in the ventricles, with associated hydrocephalus. The tip of the baclofen pump catheter was just left of the basilar artery at the level of the superior cerebellar artery (Fig. 18, b–d).
Scrotal migration mainly takes place in children, aged 4 days to 5 years. Patent processus vaginalis, the formation of an inguinal hernia, and increased intraabdominal pressure are the leading risk factors for migration of a peritoneal catheter into the scrotum. Thoracic migration of a peritoneal catheter predominantly occurs in adults. Important mechanisms in thoracic migration include (1) iatrogenic perforation of the external or internal jugular veins by the surgeon during tunneling and (2) gradual destruction of venous structures by the catheter over time, due to routine extension and flexion of the neck. After entering the vessel, intrathoracic negative pressure combined with positive intraabdominal pressure aids migration into the thoracic cavity. Sharp tips or types of shunt contribute to intraabdominal migration or perforation.
Footnotes
The authors have no conflicts of interest to declare.
This study was supported by the Jiangsu Science and Technology Project-Clinical Medicine Special Grant (BL2014076).
