Abstract
Synaptic plasticity is widely believed to be the cellular basis of learning and memory. It is influenced by various factors including development, sensory experiences, and brain disorders. Long-term synaptic plasticity is accompanied by protein synthesis and trafficking, leading to structural changes of the synapse. In this review, we focus on the synaptic structural plasticity, which has mainly been studied with in vivo two-photon laser scanning microscopy. We also discuss how a special type of synapses, the multi-contact synapses (including those formed by multi-synaptic boutons and multi-synaptic spines), are associated with experience and learning.
Keywords
Introduction: A Brief History of the Synapse and Synaptic Plasticity
The term “synapse” was first introduced to describe the junction between nerve cells by the eminent British neurophysiologist Sir Charles Sherrington in A Textbook of Physiology [1]. There are two major types of synapses: chemical and electrical [2]. Electrical synapses are found in both neurons and glia [3, 4]. Cells connected through electrical synapses are directly linked by an intercellular specialization called gap junction. Gap junctions are intercellular channels composed of six protein units that allow electrical current to pass through, thus electrically coupling the connected cells and allowing bi-directional signal flow. In contrast, chemical synapses are composed of distinct pre- and post-synaptic elements, with a synaptic cleft in between. Depolarization of the presynaptic membrane induces the release of chemicals (neurotransmitters) from the pre-synaptic terminal, causing depolarization, in the case of an excitatory synapse, or hyperpolarization, in the case of an inhibitory synapse, of the postsynaptic membrane. The signal flow is thus unidirectional. In the central nervous system of vertebrate animals most synapses are chemical, and we shall focus on them in this review.
Chemical synapses are composed of asymmetrical pre- and postsynaptic structures. The postsynaptic components of most excitatory synapses in the mammalian brain reside on the spiny protrusions from dendrites, which were first observed by the great Spanish neuroanatomist Santiago Ramón y Cajal with Golgi's silver staining method [5]. He concluded that these protrusions were not artifacts but real structures of the neurons, and named them espinas (“thorns”), hence the English term “spines”. Having acknowledged that spines mature much later than axons and collaterals, Cajal considered them the indicator of cortical neuron maturation [6]. The association between spine structure and function was first proposed by the Chinese neuroscientist Hsiang-Tung Chang, who, calling spines “gemmules”, described them as “minute lateral projections on the surface of the dendrite which have a thickened terminal bulb connected with the dendritic process by a very slender stem having a diameter less than half a micron” [7]. Moreover, Chang correctly suggested that “if the end bulbs of the gemmules are the receptive apparatus for presynaptic impulses, the process of postsynaptic excitation initiated there must be greatly attenuated during its passage through the stems of the gemmules which probably offer considerable ohmic resistance because of their extreme slenderness.” This prescient statement was recently confirmed directly using spine voltage imaging [8]. In 1971, Rall and Rinzel suggested that the morphology of dendritic spines may relate to memory storage [9]. In 1982, Sir Francis Crick raised the “spine twitching hypothesis”, suggesting that the change in spine shape underlies the change in synaptic weight [10]. Indeed, accumulating experimental evidence suggests that spine morphology is indicative of synaptic function. Spine enlargement is associated with an increase in α-amino-3-hydroxy-5-methyl-4-isoxazolepropionic receptor (AMPAR)-mediated currents [11], and the spine volume is strongly correlated with the number of functional AMPARs, but not N-methyl-D-aspartate receptors (NMDARs): AMPARs are abundant in mushroom spines but sparsely distributed in thin spines and filopodia [12–14]. In contrast, most inhibitory interneurons do not harbor spines, and inhibitory synapses often appear to be symmetrical, which means the postsynaptic density (PSD) is relatively thin as can be observed in electron microscopy (EM) images [15–18].
Synapses are highly dynamic. Changes in synaptic connections, often referred to as “synaptic plasticity”, are considered the cellular basis of learning and memory [19, 20]. The idea that plasticity is linked to experience was first raised by the American philosopher and psychologist, William James, in his classic Principles of Psychology [21], even before the term “synapse” was coined. James proposed that “the phenomena of habit in living beings are due to the plasticity of the organic materials of which their bodies are composed”, indicating that physical changes of certain parts of the body underlie experience. He also suggested that the plasticity is activity-dependent. In 1893, the Italian neuropsychiatrist Eugenio Tanzi proposed that learning could facilitate signal transduction between neurons [22]. Building upon Tanzi's idea, Cajal suggested in 1894 that learning results from changes in neuronal connections [23]. By early 1900s, the idea that synaptic plasticity is a crucial mechanism for learning and memory has already been accepted in psychology textbooks [24, 25]. However, it was not until the Canadian neuropsychologist Donald Hebb published The Organization of Behavior [26] that the concept of plasticity being the neurobiological basis of learning and memory became widely acknowledged. In this book Hebb stated that “the persistence or repetition of a reverberatory activity (or “trace”) tends to induce lasting cellular changes that add to its stability. When an axon of cell A is near enough to excite a cell B and repeatedly or persistently takes part in firing it, some growth process or metabolic change takes place in one or both cells such that A's efficiency, as one of the cells firing B, is increased.” Often summarized as “fire together, wire together”, Hebb's rule remains the leitmotif of research on synaptic plasticity for decades.
The first paper showing experimental evidence for synaptic plasticity was published in 1973 [27]. By repeatedly stimulating the perforant path fibers to the dentate gyrus of the hippocampal formation in rabbits, Timothy Bliss and Terje Lømo found that the stimulation could cause a long-lasting potentiation of the population response of granule cells in the dentate gyrus. They named this effect “long-lasting potentiation”, which was later changed into “long-term potentiation (LTP)”. A similar phenomenon, long-term depression (LTD), was first discovered by Masao Ito in 1982 [28, 29]. In LTD, conjunctive low-frequency stimulation of the parallel fibers and climbing fibers in the cerebellar cortex leads to a long-lasting depression in the response of Purkinje cells. Later LTD was also reported in the cerebral cortex and hippocampus [30–32]. Thus, synaptic strength can be modified bi-directionally under different experimental conditions: high-frequency stimulations potentiate synapses, and low-frequency stimulations weaken them.
In 1983, Levy and Steward found that when a weak and a strong input from the entorhinal cortex to the dentate gyrus are activated together, the temporal order of activation is crucial for the type of ensuing synaptic plasticity: LTP is induced when the strong input is activated concurrently with the weak input or following it by no more than 20 ms, while LTD is induced when the temporal order was reversed [33]. This is the first work to show that relative timing is crucial in LTP and LTD induction. More work in the 1990s has further addressed the importance of the temporal order of pre- and postsynaptic spiking, and has characterized the “critical window” for spike timing to be on the order of tens of milliseconds [34–39]. This type of synaptic plasticity is now referred to as spike timing-dependent plasticity (STDP). It is often considered more physiological than the traditional type of LTP induced by high-frequency presynaptic stimulation, since the 100-Hz stimulation exceeds the maximum firing rate of most neurons and could rarely happen in the living brain.
LTP can be divided into two phases: the protein synthesis-independent early LTP (E-LTP), and the protein synthesis-dependent late LTP (L-LTP). Synaptic proteins are synthesized and transported into the potentiated synapses during L-LTP, leading to enlargement of spines [40–42] or formation of new spines [43]. Long-term synaptic remodeling can be visualized in vivo using two-photon (2P) laser scanning microscopy [44, 45]. Because of its low phototoxicity and capability to penetrate deep into scattering tissue, 2P microscopy is especially suitable for imaging the brain in living animals. The principle and application of two-photon microscopy has been reviewed extensively [46, 47]. With fluorescent labeling using transgenic mice [48], in utero electroporation [49, 50], or virus infection [51], neuronal structures can be imaged in vivo for an extended period of time, and synaptic plasticity can be observed. Serial-section EM of dendritic segments previously imaged in vivo reveals that the emergence and retraction of spines is associated with the formation and elimination of synapses, respectively [52], directly connecting spine structure with synaptic function. Transient new spines often lack synapses, whereas persistent new spines always have synapses [13]. In this review we will focus on in vivo studies of structural changes of synapses in the mouse cortex, including enlargement and shrinkage of synaptic structures, as well as formation and elimination of synapses.
Structural plasticity of synapses associated with development, sensory experience, and learning
Basal level synaptic turnover at different developmental stages
Synapses are considered the basic elements of memory storage [53]. Long-term memory can be incredibly stable, lasting for years or even a lifetime. However, the neural substrate of memory, the synapses, are quite dynamic. In the adult brain, although dendritic arbors of pyramidal neurons (PyrNs) are highly stable, structural plasticity of dendritic spines is commonly observed across different cortical regions [52,54–56]. The spine turnover rate, i.e., the percentage of newly formed and eliminated spines, is lower in the adult brain than in the developing brain [55], indicating that synapses are more stable in adult than in juvenile brains. The basal spine turnover rate in the adult cortex varies with anatomical location and cell type [55–57]. Spines on layer (L) 2/3 PyrNs and spines on L5 PyrNs have different turnover rates [56, 58]. Inhibitory synapses are also highly dynamic [59–62]. Unlike PyrNs, whose dendritic arborization stays the same throughout adulthood, dendritic branches of interneurons in the adult cortex may extend, retract, or even be added over a period of months [59, 60]. Axons also undergo constant structural remodeling, including elongation and retraction of axon branches, and formation and elimination of axonal boutons [57, 63, 64]. New boutons make synapses; thus bouton dynamics also represent synaptic plasticity [63, 64].
Aging affects both axonal bouton and spine turnover, as well as dendritic arbor morphology. Dendritic spines and en passant boutons have higher density and turnover rates but smaller sizes in old mice than in young adult mice. Large boutons are generally stable in young adult mice but highly dynamic in the aged cortex [65, 66]. Because the size of a synapse positively correlates with its strength [12, 67], these results suggest that in the aging brain, synapses are weaker and less stable, which may be related to aging-related cognitive impairments. There is no substantial difference in inhibitory interneuron number, relative proportion of different subtypes of interneurons, or number of excitatory and inhibitory synapses made onto L2/3 and L5 PyrNs of aged mice compared with younger adults, but dendritic arbors of interneurons are less complex in aged mice, due to progressive dendritic branch retractions [68]. The mechanism and function of the basal level synaptic turnover is not yet clear [69]. The stability of memory and the dynamism of synapses remain to be reconciled.
Structural plasticity associated with sensory experiences
Sensory experiences influence synaptic plasticity. In 1970s, David Hubel and Torsten Wiesel discovered that deprivation of visual input into one eye during a developmental period especially sensitive to the sensory experience, termed “the critical period”, could dramatically change the neuronal connectivity in the visual cortex of cats. Monocular deprivation (MD) and binocular deprivation (BD), in which visual input into one or both eyes is deprived respectively, have since become classical models for studying experience-dependent plasticity in the visual cortex. Using time-lapse in vivo 2P imaging, studies show that both MD and BD affect synaptic rewiring. The motility rate of spines on apical dendrites of L5 PyrNs in the primary visual cortex (V1) is high during the critical period, but declines quickly afterwards [70]. Both BD and MD increase spine motility rate at the peak of the critical period, consistent with the idea that sensory deprivation shifts spine dynamics towards an immature state and delays the critical period [71, 72]. MD increases spine elimination during the critical period, and this effect persists for 2–4 days after binocular recovery [73]. Brief light exposure after prolonged BD results in a rapid and transient elevation of spine dynamics, suggesting that visual experience could reorganize cortical circuitry quickly [74].
Sensory deprivation affects spine dynamics not only during development but also in adulthood. MD between postnatal day (P) 61 to P116 significantly increases spine formation, but not elimination, of L5 PyrNs in the binocular zone of V1 in adult mice, without changing the spine dynamics of L2/3 PyrNs. Spines formed during this MD persists long after binocular vision is restored, and spine density also remains elevated. If a second MD is imposed 16–20 days after the first MD, spine number does not increase, but the spines formed during the first MD become enlarged, possibly contributing to the faster functional shift induced by the second MD. The maintenance and enlargement of the spines formed during the first MD may represent a structural memory trace for the functional shift of visual cortical responses induced by sensory experience [75, 76].
Although MD during adulthood does not affect spine dynamics of L2/3 PyrNs [76], it highly modulates the dynamics of inhibitory synapses on the dendritic shafts of L2/3 PyrNs [77]. Inhibitory synapses often disappear and reappear at the same location in close vicinity to dynamic excitatory synapses, and the recurrence rate of inhibitory synapses is upregulated by MD [78]. The elimination rate of inhibitory synapses is higher during MD, resulting in a significant inhibitory synapse loss and weaker inhibition. MD changes the structural dynamics of both excitatory and inhibitory synapses, but the two types of synapses have different functions: excitatory synapses likely represent memory trace for the sensory experience at the synaptic level, whereas the inhibitory synapses modulate the strength and dynamics of excitatory synapses, reflecting the circuit adaptation according to the sensory experience. The impact of MD during adulthood on excitatory and inhibitory synapses outlasts the sensory experience itself, indicating a long-lasting influence of sensory modification on neural circuit.
In addition to changing the dynamics of postsynaptic structures, sensory deprivation also affects the structural plasticity of presynaptic boutons in the visual cortex. MD shifts the size of excitatory axonal boutons in L2/3 of adult mouse visual cortex towards the middle, leading to a reduced range of bouton sizes, which may indicate greater potential for bidirectional structural and functional plasticity, consistent with the idea that sensory deprivation shifts the cortical circuit towards a more plastic state [79].
Another commonly used modality for studying sensory deprivation is the somatosensory system, namely the whisker barrel system in rodents. Dendritic spines and filopodia in the barrel cortex are highly dynamic during development. In juvenile rats, sensory deprivation by whisker trimming reduces spine/filopodia motility in deprived regions of the barrel cortex without changing the density, length, or shape of spines and filopodia [80]. In mice, long-term sensory deprivation results in higher spine and bouton density [81–83]. Restoring sensory experience after adolescence accelerates spine elimination [84]. Pruning of excessive synapses is a key developmental process, and it depends highly on sensory experience. In both visual and somatosensory systems, deprivation of sensory input delays or obstructs the maturation of cortical circuits.
Introducing novel sensory experience by trimming alternate whiskers increases spine turnover rates, stabilizes newly formed, often small-sized spines, and destabilizes large, persistent spines of L5 PyrNs in the mouse barrel cortex [52, 85]. Spine stabilization is most pronounced at the border of spared and deprived barrel columns, and depends on alpha Ca2+/calmodulin-dependent protein kinase II (alpha-CaMKII) autophosphorylation [86].
Structural plasticity associated with learning
In the adult brain, learning effectively modifies synaptic structures in both sensory and higher-order cortical regions. The most commonly used learning paradigms are motor skill learning and associative fear learning.
Motor learning rapidly increases the formation and elimination of spines of L5 PyrNs in the mouse primary motor cortex (M1), leading to a transient increase in spine number, which over days returns to the baseline [87, 88]. New spines formed during motor learning are preferentially stabilized, and the number of new spines directly correlates with the behavioral performance [87, 88]. New spines tend to emerge in clusters, and the clustered new spines are more likely to persist long after training ends than their non-clustered counterparts [89]. Spine clustering has also been observed in contextual fear learning [90]. Retraining with the same motor task does not trigger an additional increase in spine dynamics, but a different motor task again induces an increase in spine formation and elimination [88], suggesting that different motor behaviors are stored in different ensembles of synapses. Using a synaptic opto-probe, AS-PaRac1 (activated synapse targeting photoactivatable Rac1), Hayashi-Takagi and colleagues showed that using light to specifically shrink newly potentiated spines associated with a motor task can disrupt the motor behavior without affecting other motor tasks [91], further confirming that specific motor behaviors are controlled by corresponding sets of synapses.
The aforementioned experiments were all done on L5 PyrNs in M1. Studies on L2/3 PyrNs in M1 show that motor learning induces spine reorganization on the distal branches of their apical dendrites in L1 but not on the basal dendrites. Coincidentally, somatostatin-expressing inhibitory neurons, which mainly inhibit distal dendrites, show a rapid decrease in axonal boutons, whereas parvalbumin-expressing inhibitory neurons, which mainly inhibit perisomatic regions of pyramidal neurons, exhibited a gradual increase in axonal boutons [81, 92]. However, another study showed that spine dynamics on L2/3 PyrNs are not affected by motor learning [58]. The difference may be due to the different motor tasks used in these studies.
A specific type of motor learning is the bird song learning of zebra finches. This learning process is also associated with spine dynamics. Song imitation increases spine turnover rate in the forebrain nucleus HVC in young birds, and song tutoring causes rapid spine stabilization and enlargement in HVC. Interestingly, higher spine turnover rate before the bird first experienced tutor song correlates with a greater capacity for subsequent song imitation [93]. Similarly, spine dynamics in mouse retrosplenial cortex before contextual fear learning predicts future learning and memory performance [90].
Associative learning also induces changes in spine dynamics in the cortex. In the barrel cortex, the tactile trace eyeblink conditioning causes a substantial learning-related spine elimination in L5 PyrNs in the principal barrel column but not in other columns [94]. Auditory-cued fear conditioning leads to transient [95] and long-lasting [96] changes in spine dynamics of L5 PyrNs in the auditory cortex. The long-term increase in spine formation is pathway-specific, namely, spines receiving input from the lateral amygdala show higher formation rate than other spines. Auditory fear conditioning also leads to bouton formation in lateral amygdala neurons projecting to auditory cortex [96]. Fear extinction by repeated presentation of the same auditory cue preferentially eliminates new spines in a cue-specific manner [97]. In the mouse frontal association cortex, fear conditioning increases the rate of spine elimination, while fear extinction increases the rate of spine formation [98]. Associative learning selectively influences the synaptic dynamics of corresponding sensory cortical regions, and with new tools to label specific pathways, the synaptic plasticity is found to be not just region-specific, but also pathway-specific. Memory may be coded in specific sets of synapses, and these synapse ensembles may reside in selected neural pathways.
Synaptic remodeling associated with lesion and neurological diseases
Synaptic dynamics during stroke and stroke recovery
Besides sensory experience and learning, pathological conditions can also lead to structural plasticity. In ischemic stroke, blood supply to the brain tissue is blocked, which causes behavioral dysfunctions. With time, a partial recovery often occurs. The neural basis of the behavioral deficits and the recovery remains a mystery. Functional magnetic resonance imaging (fMRI) is commonly used to study the neural mechanism underlying stroke-related behaviors [99, 100], but its temporal and spatial resolution are limited. Longitudinal 2P microscopy allows a closer look at the neuronal structures at different time points after stroke, providing direct evidence for the neural basis of the long-term behavioral changes. After stroke, peri-infarct dendrites become exceptionally plastic, exhibiting long-lasting increases in spine turnover, dendritic tip elongation away from the infarct region, and dendritic tip retraction and shortening near the infarct region [101–103]. But no evidence of addition of new dendritic branches was observed after as long as 3 months after the stroke [104]. Moderate ischemia (approximately 50% blood supply remains) does not significantly affect spine structure within a few hours, while severe ischemia (less than 10% blood supply remains) can cause a rapid loss of spine and apical dendrite structure within as little as 10 min, during the passage of a propagating wave of ischemic depolarization. The depolarization after stroke also temporally coincides with dendritic beading, appearing as rounded expansions extending beyond the diameter of the parent dendrite [105]. If the blood flow is restored within 20–60 min, most spines and dendrites can regain their original structures [106, 107]. In most cases, stroke causes long-lasting severe damage to the neuronal structures in the brain within minutes; unless the blood flow is restored quickly, the damage is irreversible.
2P imaging has provided direct evidence against some of the earlier hypotheses regarding the recovery from stroke-induced behavioral deficits. It has been proposed in the past that massive plasticity in the cortical region contralateral to the lesion (contralesional cortex) contributes to functional recovery after stroke. However, no de novo growth of dendrites or change in the density or turnover of spines on apical dendrites of L5 neurons in the contralateral cortex has been detected for up to 4 weeks after stroke. Therefore, the contralesional cortex may not contribute to functional recovery [108].
Altered synaptic dynamics in neurological disorders
Compared to the relatively mild neuronal morphology changes caused by normal aging, Alzheimer's disease (AD) model mice exhibit more severe synaptic and neuronal structure abnormalities. In a series of transgenic AD mouse models with plaque formation caused by overexpression or mutation of the amyloid precursor protein (APP), dendritic spine loss and dendritic shaft atrophy are observed near or even far away from the plagues, leading to permanent disruption of neuronal connections all over the cerebral cortex [109, 110]. Spine plasticity is also impaired but can be rescued by anti-inflammatory treatment [111]. Surprisingly, there is a twofold increase of spine density in the cerebral cortex of adult mice deficient for APP [112]. BACE1 (beta site amyloid precursor protein cleaving enzyme 1) is the rate-limiting protease in amyloid β production. Chronic inhibition of BACE1 activity causes spine loss by reducing formation of new spines, as well as reduced miniature excitatory postsynaptic currents and LTP. These neuronal structural and functional changes are associated with cognitive deficits [113–115]. The impairment of spine morphology and synaptic plasticity in AD model mice is consistent with their behavioral defects. Drugs that restore spine morphology and plasticity may also rescue the behavioral phenotypes, and spine dynamics could be a readout for assessing their effectiveness.
Dendritic spine abnormalities have also been reported in other neurological disease models. The Angelman syndrome is a neurodevelopmental disorder caused by the loss of the maternally inherited UBE3A allele. In the visual cortex of one-month-old mice lacking UBE3A protein, spine density is decreased due to greater elimination; this effect is not observed in dark-reared mice. Therefore the changes in spine density and dynamics are experience-dependent [116]. A mouse model of Huntington's disease exhibits a steady decrease in the density and survival of dendritic spines in the cortex, despite an increase in spine formation: the newly formed spines are not properly stabilized [117]. In a mouse model of fragile X syndrome, the Fmr1 knock-out (KO) mice, the stabilization of new spines is also impaired. But these transgenic mice have an increased spine density and significantly higher spine turnover rate and show a developmental delay in spine maturation. The Fmr1 KO mice also have defects in experience-dependent and learning-associated spine dynamics, coincident with impairment in motor-skill learning [118–121]. Motor skill learning and learning-induced spine turnover are also impaired in dopamine-depleted Parkinson's Disease mouse models [122]. In two autism spectrum disorder mouse models, patDp/+ mice and NLG R451C mice, PSD-95-positive spines show higher turnover, but gephyrin-positive spines are unaffected [123]. For the Rett syndrome model, young transgenic mice that overexpress the human MECP2 gene initially have higher spine density in terminal dendritic branches, but after postnatal week 12, spine density falls below control levels because of the high baseline spine turnover [124]. CDKL5 (cyclin-dependent kinase-like 5) is mutated in many severe neurodevelopmental disorders, including atypical Rett syndrome. Adult male cdkl5 KO (cdkl5-/y) mice show a significant reduction in spine density and impaired LTP. In juvenile mutants, short-term spine elimination, but not formation, is dramatically increased [125].
Multiple-synaptic boutons and spines
Textbooks always portray the synapse as a pair of presynaptic and postsynaptic elements (Fig. 1A). But in real life, synapses do not always form one-to-one connections. There are synapses with multiple contacts (Figs. 1B and 1C), and these specialized synapses are potentially associated with specific functions.
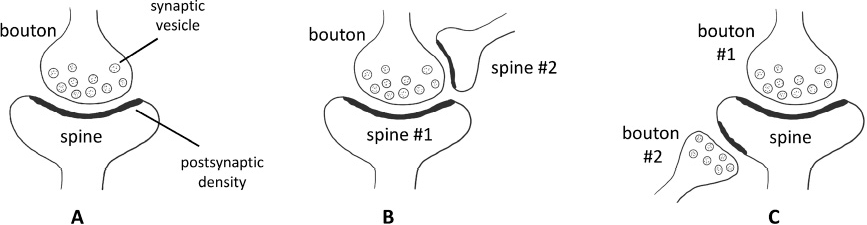
Single-contact and multi-contact synapses. (A) Single-contact synapse formed by one presynaptic bouton and one postsynaptic spine. (B) Multi-contact synapse formed by one multi-synaptic bouton and two spines. (C) Multi-contact synapse formed by one multi-synaptic spine and two boutons.
Multiple-synaptic boutons (MSB, Fig. 1B) are boutons that make synaptic contacts with more than one spine, making a one-to-many connection, and multiple-synaptic spines (MSS, Fig. 1C) are spines that receive inputs from multiple boutons, making a many-to-one connection. MSB was first seen in the cat cerebral cortex half a century ago [126], and has since been observed in multiple organisms, including mice, rats, rabbits, cats, and non-human primates [96,127–131]. The proportion of MSB in all boutons varies among different brain regions. In the adult monkey brain, MSBs comprise as many as 40% of all boutons in the hippocampal dentate gyrus, but only 10% in the dorsolateral prefrontal cortex [132]. In rat hippocampal CA1 PyrNs, most MSBs are composed of small synapses with low NMDAR and AMPAR expression, or one large synapse and several small synapses [133]. These MSBs are unlikely to generate depolarization simultaneously in more than one postsynaptic spine because of the small size of the synapses, so they may represent a transient state of competition between newly formed small synapses and existing synapses.
There is also evidence suggesting that MSB may not always represent a transient state. In rodent hippocampus and cerebellum, LTP or motor skill learning can lead to the growth of postsynaptic spines onto pre-existing synapses on the same dendrite, forming MSB contacting spines on the same dendrite, possibly generating a stronger connection between the pre- and post-synaptic neurons [127, 134]. Spines in one MSB could also come from different cells. In stratum radiatum and their target CA1 cells, at least some of the spines that contact the same MSB belong to different cells [135]. This form of connection may represent an efficient form of signal transduction between multiple neurons.
MSB is related to various forms of plasticity-inducing phenomena, such as manipulation of the sensory experience, brain lesion and learning. Sensory deprivation by neonatal enucleation in rats produces a significant reduction in the proportion of MSB in the visual cortex [136]. After long-term monocular deprivation, non-deprived geniculocortical axons in cat visual cortex form more MSBs than either normal or deprived axons [130]. Enriched environment increases MSB proportion in the visual cortex of rats without changing the total synapse number [137]. Therefore, MSB formation seems to depend highly on experience, as the number of synapses per bouton in sensory cortices increases with sensory experience.
Lesion has a profound impact on the formation of MSBs. In adult rats, lesioning the ipsilateral entorhinal cortex increases MSB formation in the dentate gyrus [128] and lesioning the ipsilateral sensorimotor cortex increases the number of MSBs in the contralateral motor cortex [138]. Frontal cortex and substantia nigra lesions increase the percentage of MSBs in the dorsolateral striatum [139]. Motor learning could also increase the number of MSBs, but not single-contact synapses [138]. Trace eyeblink conditioning increases the number of MSBs in the rabbit hippocampus [129], and auditory fear conditioning induces formation of spines onto existing synapses in mouse auditory cortex, possibly leading to formation of MSBs [96].
MSB is also related to estrogen levels. Estrogen treatment increases MSB formation in the hippocampus of ovariectomized rats and monkeys [132, 140], and the proportion of MSBs in the monkey hippocampus correlates with the delayed nonmatching-to-sample (DNMS) working memory task [131, 132].
MSS has not been studied as extensively as MSB, being reported mainly in development and under pathological conditions. In developing rat hippocampus, filopodia or stubby spines often have multiple synaptic contacts, which are rarely encountered in the mature brain [141, 142]. PSD-95 up-regulation or overexpression promotes the formation of MSS [143, 144]. MSSs are also seen in severe epilepsy patients [145]. In a few instances, MSS has been found to be associated with learning. Motor learning induces MSS formation in contralesional motor cortex of rats [96, 138], and auditory fear learning induces formation of MSS in the auditory cortex of mice [96, 138]. A recent study using multicolor stimulated emission depletion microscopy (STED) combined with confocal imaging demonstrates that experience-dependent structural plasticity is linked to the addition of unitary synaptic nanomodules to spines, possibly associated with MSS formation [146].
New boutons and spines growing onto existing synapses would lead to formation of MSSs and MSBs. The estradiol-induced new spines form synapses primarily with preexisting boutons in the stratum radiatum of female rats [140]. This form of synapse formation is seen in mice too. In the barrel cortex, new spines preferentially contact boutons with other synapses [13]. In the visual cortex, inhibitory synapses frequently disappear and recur on existing excitatory synapses [78]. In the auditory cortex, new spines almost exclusively grow onto existing boutons, both after fear learning and under basal conditions [96]. Forming new connections on existing synapses saves cellular resources and space and is an efficient way to form new connections or strengthen existing connections.
To change, or not to change, that is the question constantly posed to the synaptic circuit. In the living brain, stability and dynamism of synaptic circuits coexist: dynamic changes are required for learning from experiences, while stability is prerequisite to memory. The brain thus must “know” which connections to retain and which ones to alter in order to satisfy these two antithetic requirements. When the balance is tilted towards either direction, neuropsychiatric disorders and cognitive defects ensue. We are only beginning to understand the mechanisms that enable and maintain such delicate balances.
With the ability to image micrometer-scale synaptic structures in the living brain, 2P microscopy has greatly facilitated the understanding of synaptic structural plasticity associated with sensory experience, learning, and pathological conditions. The molecular mechanisms underlying the dynamics and the fate of spines and boutons are still largely unknown, but have recently attracted more and more attention. Using transgenic animal models, several molecules have been found to play important roles in modulating synaptic dynamics [147–149]. In addition to imaging the structure of synapses, development of ultrasensitive calcium indicators, including small molecule dyes and genetically encoded calcium sensors, allows observation of functional properties of spines and boutons in the living brain [150–159]. The highly sensitive fluorescence resonance energy transfer (FRET)-based kinase sensors can be used for monitoring kinase activity under two-photon fluorescence lifetime imaging microscopy (2pFLIM) [160]. These powerful tools will help unravel the functional and molecular underpinnings of the synaptic dynamics.
It is now possible to image simultaneously the pre- and postsynaptic structures [96], which provides information on circuit-specific plasticity. However, since the width of the synaptic cleft is below the diffraction limit of the 2P microscope, it is hard to determine if an observed pre- and postsynaptic contact is indeed a functioning synapse. The best way to define a synapse is by using the electron microscopy, but it is very difficult to co-localize 2P images with EM images [13, 77, 161, 162], and the data analysis for both 2P and EM images is extremely time-consuming. With recent invention of serial block-face scanning EM [163], focused ion beam/scanning EM [164–167], and automated tape-collecting ultramicrotome [168], combining with automated data analysis methods empowered by machine learning [169–172], it is now more feasible to perform correlated 2P and EM imaging. Together with retrograde labeling and trans-synaptic labeling viral tools [173–176] and whole-brain connectomic mapping methods [177–180], the source of presynaptic input and the target of postsynaptic output will also become identifiable. These methods make it possible to combine brain circuit mapping with animal behavior at a synaptic level.
Footnotes
All contributing authors have no conflict of interests.
Acknowledgements
We thank the following funding agencies (Startup funding from ShanghaiTech University and 2018YFC1005004 to YY, and the National Institute of Health R01MH104227, R01MH109475, R01NS104950 to YZ) to the support of the work.
