Abstract
The aim of the current study was to investigate the operation skills and complications associated with skull defect repair using biomimetic bone (nano-hap/collagen composites). Clinical data from 45 patients with skull defects who underwent cranioplasty with biomimetic bone from January 2014 to January 2017 were retrospectively analyzed. All patients participated in follow-up visits from 8 months to 3 years postoperatively. Cranioplasties were successful in all 45 patients, but 17 patients (20 cases) presented various complications, including subcutaneous hydrops (14 cases), biomimetic bone fixation loosening (4 cases), wound indolence (1 case), and biomimetic bone fragmentation (1 case). Cranioplasty complications in surgeries using biomimetic bone are more common in children than in adults. Understanding the physicochemical properties of biomimetic bone and the normal developmental process in children, as well as mastering the appropriate surgical skills and implementing the effective preventive measures are all ways to reduce and control the incidence of complications associated with biomimetic bone cranioplasty.
Introduction
Skull defects are often the result of decompressive craniectomy following head trauma or brain disease. Early cranioplasty following craniectomy not only improves the craniofacial appearance, but also accelerates the recovery of cerebral function. Current allograft bone repair materials have progressed considerably from traditional repair materials. Where metals and polymers were historically used, medically engineered tissue is now the focus. However, there are many unknown factors and cranioplasty risks associated with the introduction of these new materials. Since 2014, our department has used biomimetic bone constructed by three-dimensional printing to repair skull defects in 45 patients. Furthermore, we have collected clinical outcomes from these cases in order to analyze postoperative complications, and suggest ways to avoid their occurrence.
Experimental details
General information
Forty-five patients with skull defects were chosen. Twenty-six patients were male, 19 were female, and their ages ranged from 1 year 2 months to 24 years old. Brain injury resulted in 39 craniectomy cases, including 32 closed brain injury cases (hematoma and other causes of intracranial hypertension), 7 open brain injury cases (3 falling injury, 3 traffic accident injury, and 1 chop injury case). Five craniectomy cases were non-traumatic, including 2 ruptured intracranial aneurysms, cerebral hernia after brain tumor resection (1 case), skull tumor (1 case), and unexplained intracranial hemorrhage (1 case). The last case was a result of a growing skull fracture. The average follow-up time after craniectomy was 1 year and 9 months, with the earliest follow up at 2 months, and the latest, 46 months.
Position and size of skull defects
Unilateral skull defects were present in 42 cases (23 left sided, 19 right sided), and included frontal-temporal-parietal (29 cases), frontotemporal (8 cases), frontal (3 cases), and occipital (2 cases) skull defects. Bilateral skull defects were present in 3 cases, 2 of which were bone bridge defects in the middle route of the skull, and 1 of which was a bilateral frontal bone defect crossing the midline. Incisions in 33 of these cases healed well before operation, 9 cases had preoperative scars that were still healing and there were 3 cases of wound indolence due to escharosis. Defect size ranged from 28 cm2 to 126 cm2, with an average size of 72 cm2.
Preoperative assessment
Symptoms of contralateral hemiplegia and abnormal MRI with encephalomalacia were seen in 35 cases. These cases also displayed declined limb myodynamia, increased or reduced muscle tension, and poor coordinated dynamic function. Six cases had severe meningoencephalocele, and 4 cases were accompanied with hydrocephalus. All hydrocephalus cases were relieved by ventriculoperitoneal shunts (B.braun VPS) with an adjustable voltage shunt ventricle. Preoperative seizure symptoms and long-term antiepileptic drug use were found in 5 cases, 3 cases had scalp hydrops around the defects, and 1 case had serious psychiatric symptoms without cranioplasty.
Repair materials
Three-dimensional CT scanning and printing was used to make the operation plan for skull shape reconstruction using biomimetic bone. Proportion of biomimetic bone ingredients and bone properties varied according to age group. In older patients, the proportion of nano-hydroxyapatite and biomimetic bone flap hardness was greater. Doctors and technologists communicated the appropriate ingredient proportion during the process of biomimetic bone flap manufacturing.
Operation procedure
General anesthesia with intubation was conducted in all patients. 0.2% lidocaine and 0.1% adrenalin were injected subcutaneously to clarify the subcutaneous levels and reduce bleeding and postoperative pain. The previous scar incision on the skin was cut to create a free skin flap, as was the underlying subcutaneous fat and galea aponeurotica. The biomimetic bone was placed under the temporalis myofascial flap when the temporal muscle had abundant blood supply. Otherwise, enough space was kept on the lower biomimetic bone edge for outside temporal muscle cranioplasty. The soft tissue was separated by monopolar electrical cautery around the bone window. All bone fragments were removed using a rongeur, and dural closure and reconstruction was verified. The dura was routinely suspended and skull shape was secured using thread through holes with 10-suture fixation (double 7-suture fixation after 2016) to secure the bone flap. Knots in the biomimetic bone flap holes were positioned away from the previous incision. Polyether-etherketone (PEEK) skull lockers were used in some cases beginning in 2016. Absence of muscle incarceration and ischemia were verified. All tissue layers were sutured and the incision was closed with or without external drainage depending on the presence of cerebrospinal fluid (CSF) leakage and bleeding.
Results
All procedures were successful, and after craniotomy CT imaging confirmed proper matching of the biomimetic bone flap with the curve of the original bone window margin. The overall shape was satisfactory, and there was no intraoperative bleeding, epilepsy, infection, or other serious complications during any perioperative period.
Fourteen patients developed subcutaneous hydrops within 2 weeks of operation. Among them, 8 patients were only given syringe suction and pressurized bandage, 4 were treated with subcutaneous puncture, continuous drainage and compression, and 1 was given continuous lumbar puncture to eliminate hydrops. In one case, all treatments failed to solve subcutaneous tissue intractable hydrops. Craniotomy was again performed, and the subcutaneous fluid disappeared after the dura was repaired.
Follow-up in 4 children (earliest 3 and latest 20 months post-operatively) revealed that the biomimetic bone flap was loose. The edge was tilted or the entire flap was protruding, and bone window pulsation was accompanied by decubitus or crying. In 2 of these cases, a PEEK skull lock was added in a subsequent operation. In the other 2 cases, there was no obvious pressure, and the family's desire to re-operate was not strong, so observation without intervention continued. One of these cases was discharged after one month with line response and exposure, which slowly healed after surgical removal of the subcutaneous line incision. After two months, however, there was similar reproduction with scar formation, subcutaneous fistula varus, and cracking. In order to avoid infection, the biomimetic bone flap was removed and the wound appeared to heal. Fourteen months after surgery, one child, whose CT showed signs of biomimetic bone flap fragmentation, was not treated again due to their previous trauma. Ten months after this CT review, the biomimetic bone flap completely broke, was surgically removed, and a new custom body bone flap was inserted. One-year followed up indicated no further abnormalities.
In accordance with the type and number of complications in our biomimetic bone operations, which included 2 cases with complications from other hospitals, postoperative complications were divided into 3 stages according to the time of occurrence: perioperative surgical period (within 7 days) of surgery, short-term postoperative (within 1 month after surgery), and long-term postoperative (more than 1 month after surgery) (Table 1).
Complications of cranioplasty using biomimetic bone.
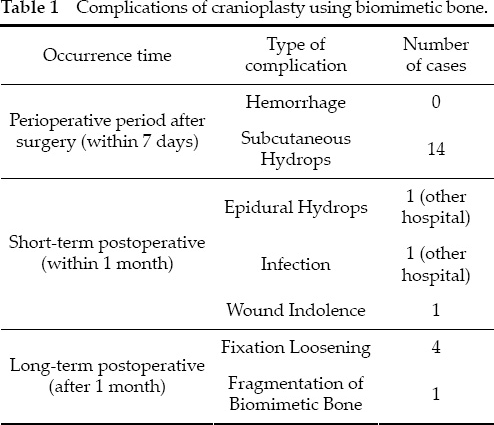
Complications of cranioplasty using biomimetic bone.

Metal, polymer, and engineered tissue are commonly used for cranioplasty. Because of its histocompatibility, reasonable price, and easy installation, titanium mesh is the most commonly used material. Polymer materials such as PEEK have similar plasticity and tissue compatibility, but the expensive price limits its use. Some scholars use autologous bone destruction or subcutaneous preservation in liquid nitrogen after live/subgaleal burial preservation, but these techniques often result in repair, bone window inversion, and collapse. As such, this method is now seldom used. Organic glass, silica gel, bone cement, and other materials are also rarely used nowadays. Therefore, biologically engineered tissue with biological activity is the focus of current research [1, 2].
Biomimetic bone material (nanophase hydroxyapatite/collagen composite bone) mimics the fine structures of natural bone components. This mimetic property is highly advantageous as it allows for the formation of new blood vessels, bone tubes, and other fine structure of the bone tissue network channels, and allows for exchange and transport of nutrients and metabolic waste, so as to participate in normal metabolic processes [3]. Research shows that after biomimetic bone implantation, coordination with osteoclasts and autologous tissue repair at the surgical interface is a dynamic process of alternating osteolysis and calcium deposition, and eventual “creeping substitution” for autologous bone tissue is achieved [4].
In personalized customization, the proportion of biomimetic bone components varies according to patient age. According to the author's own experience, a larger proportion of collagen in biomimetic bone is intended for children under 6 years of age (Fig. 1a). This biomimetic is smooth and white, and its durability and toughness can be felt with a rongeur. In the case of grinding mill brick heating, the melting point was also lower, which better mimics the toughness of bone in infants and young children. Biomimetic bone occupied mostly by hydroxyapatite is applied in children above 6 years of age, or in adult patients (Fig. 1b). The specific composite proportion is determined according to the children's skull development standard. The surface of this material is rough and yellow, and trimming with a rongeur allows the high hardness and fragility to be detected. Furthermore, this material is not easy to polish with a file and drill grinder. Therefore, with hydroxyapatite-dominant biomimetic, careful pruning should be conducted to prevent biomimetic bone cracking.
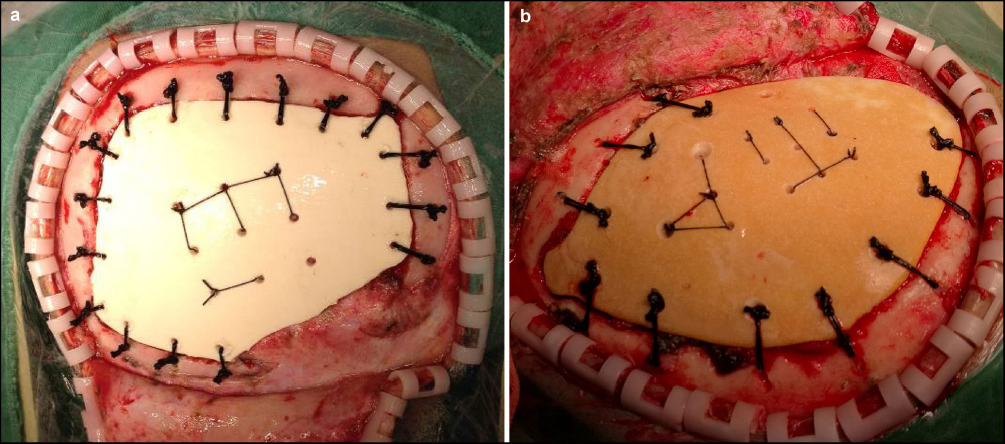
Biomimetic bone appearance is smooth and white in children under 6 years of age (a), and rough and yellow in individuals above 6 years of age (b).
In the week after cranioplasty, the most common complication was bleeding. Hill et al. reported a 1.1% incidence of brain bleeding after cranioplasty [5]. However, there were no postoperative bleeding cases in our hospital or in other hospitals. Instead, subcutaneous hydrops was more common. Schuss et al. reported a 1.1% incidence of scalp hydrops after repair [6]. Wachter et al. reported a 2.2% incidence of hydrocele after repair [7]. In our hospital, 14 cases of subcutaneous hydrops occurred in 45 cases, and the incidence was 31.1%, much higher than that reported previously. Reasons for this incidence include: (1) The biomimetic bone flap was composed of allograft, whose biocompatibility is general and surface is relatively smooth. These characteristics make it difficult for subcutaneous granulation tissue attachment. (2) There was intraoperative dural damage and CSF leakage, and sutures were not tight. (3) The number of holes in the bio-mimetic bone flap were fewer, and their diameter was smaller. The adhesion between flap and external dura granulation is therefore difficult. (4) Under the biomimetic bone flap, dura mater suspension is not tight, a cavity is created, and blood/fluids can easily seep out. (5) Subcutaneous hemostasis is not complete, and the bandage was not tight.
In almost all patients with subcutaneous hydrops, hydrops was from the bottom of the temporal bone joint puncture point, which was also the treatment point. In addition to the temporal base position being low, temporal muscle activity and repeated bone edge friction may have also led to fluid leakage. Therefore, the authors suggest that the external reduction of the temporalis muscle bone flap may be helpful to avoid hydrops (Fig. 2a). However, if temporalis muscle atrophy exceeds 3/4, there is no need to isolate the temporalis muscle, which can be repaired (Fig. 2b). This operation must, however, be done in accordance with temporal muscle thickness to judge the remaining space. In these procedures, the side was polished with a file, generally not more than 1.5 cm, to prevent temporal muscle incarceration. After using a subcutaneous drainage tube combined with postoperative compression bandages, the flap was closed to the outer surface of the biomimetic bone, leaving no dead space. The overall effect was good.
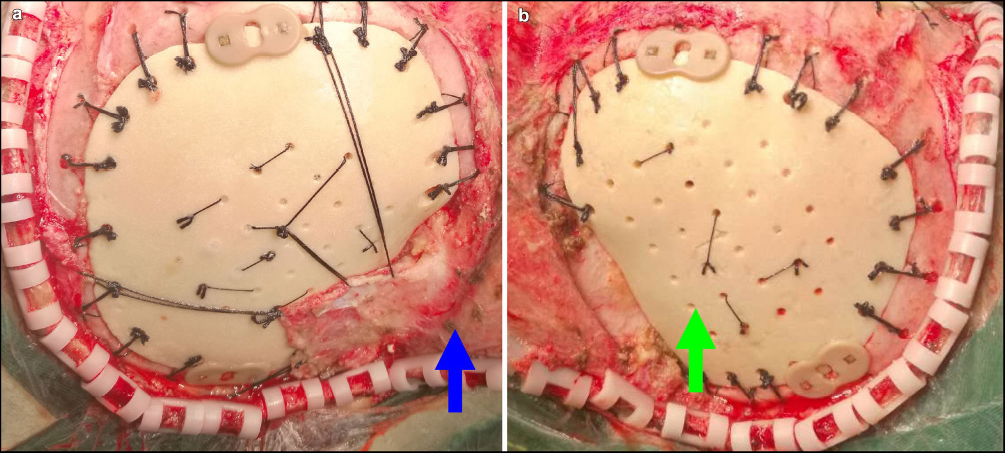
The temporalis muscle on the biomimetic bone flap is indicated by a blue arrow (a). The temporalis muscle under the biomimetic bone flap is indicated by a green arrow (b).
As for patients with subcutaneous hydrops after removal of the drainage tube, the procedure is as follows: After syringe suction, add pressure to the bandage. Induce a subcutaneous puncture for continuous external drainage and add additional pressure to the bandage. Continue lumbar puncture catheter drainage and pressure maintenance. Perform craniotomy to again repair the dura. Each step should not exceed 7 to 10 days to avoid creation of a pseudomembrane by the protein-rich fluid on the tissue surface. This causes the skin flaps to laminate with the outer surface of the biomimetic bone with more difficultly. One patient with refractory dropsy in this group had no obvious dural leaks after subsequent craniotomy to remove the biomimetic bone flap. A thin gelatin sponge can be placed in the epidural space to strengthen the suspension of the dura. The smooth pseudomembrane in the epidural space and under the flap can then be removed. The subcutaneous drainage tube can be left for 7 days after operation, and intravenous mannitol drip should be administered reduce intracranial pressure. After 3 weeks, the flap and biomimetic bone should fit tightly. To improve this plan, the author thought that as long as the outer surface of the biomimetic bone flap was made rough (like ground glass), the problem of misalignment can be solved easily, obviously reducing subcutaneous hydrops.
Postoperative epidural hydrops (Fig. 3) and postoperative infection (Fig. 4) are both serious postoperative complications that require secondary surgical treatment. Although no patients in this group had the situation shown above, in April and November 2016, this hospital accepted 1 case of chronic epidural hydrops and 1 case of postoperative infection after biomimetic bone surgery from another hospital. The patient with epidural hydrops 3 months postoperatively had severe symptoms of contralateral hemiplegia. Surgery was again performed, the scalp was opened to peel the granuloma from the skin flap. A large amount of transparent light-yellow liquid (mainly CSF) gushed out. One CSF leakage point was clearly seen during the surgery. Possible reasons for epidural fluid formation are considered as follows: (1) Dural repair is not tight. (2) Unidirectional CSF leakage exports fluid to the epidural space. (3) The suspension of the dura is not strict, and avulsion of the suspension lines may have occurred. Postoperative infection is mainly caused by improper aseptic techniques during operation. The infected patient from another hospital had repeated surgical wound scab cracking with periodic yellow liquid outflow for nearly 8 months postoperatively, which formed a sinus tract and severe skin infection. After entering our hospital, the implanted biomimetic bone flap was removed, infected soft tissue was cleaned, and inflammatory bone tissue dissection was expanded.
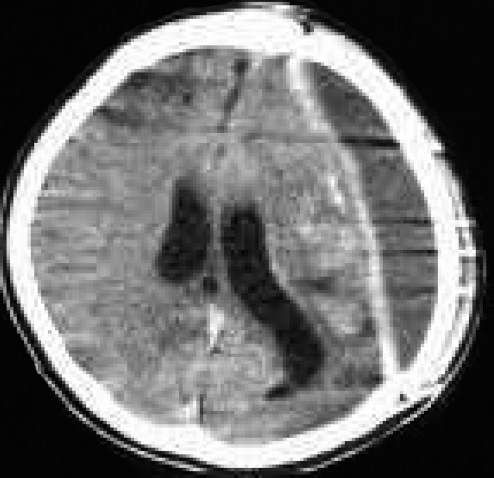
Postoperative CT image: left frontal epidural hydrops.
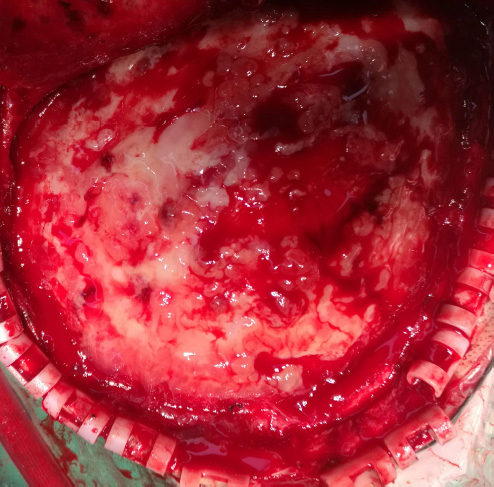
Evacuation of the epidural abscess during re-operation.
In cases native of our hospital, there was one child's case of protracted course of surgical incision with recurring line rejection. Finally, the biomimetic bone flap was removed to prevent infection. The reasons for this complication were are as follows: (1) Air bricks in children with defects after cranial injury necessitate debridement, leading to severe injury and residual poor blood supply to the scalp. (2) Post-traumatic subcutaneous foreign objects (such as gravel) are not completely removed, causing a powerful immune response. (3) Bone flap fixed lines were located just below the surgical incision, affecting wound healing by repeated stimulation of inflammation.
The 4 patients with loose biomimetic bone flaps were children, and included 2 cases of simple frontal and 2 cases of forehead and temporal surgery. Loose flaps appeared between 3 months and 1.5 years postoperatively. The reasons for biomimetic bone flap loosening are more complex, and are mainly due to rapid growth, where the brain grows far faster than the biomimetic bone “extended” speed and autogenous bone “bonding” speed.
The outward force of brain tissue expansion makes the gap between the biomimetic bone material and autogenous bone widen, which does not allow for a completely bonded bone connection. Furthermore, in the initial surgery, the strength and high tension of the double line 10 knotting fixed thread itself formed a shearing action on the autogenous skull of children (Fig. 5), which led to bone atrophy around the hole and a fixed line that nearly “tore up” [8]. A pair of size 7th thread was used to fix a bone flap in 2016. It had a moderate elastic line and the location was positioned away from the incision. At the same time, high pressure in partial patients with frontal lobe hydrocephalus is not resolved. The pulsatile pressure conduction of the brain also supports this hypothesis. Thus, the author suggested that hydrocephalus should be addressed first before skull surgery. Partial necrosis encephalomalacia in patients with meningioma encephalocele should be excised during repair. Ventriculostomy can be done if necessary to promote cerebrospinal fluid drainage. Children with skull defects in the two growth peaks, with largely unaffected brains, and with operations covering the frontotemporal had 2-3 PEEK skull locks added, with two metals fixed on the forehead and temple lifting, or three fixed frontotemporally (Fig. 6). As for patients with fusion problems, having a larger gap between biomimetic and autogenous bone in the second operation, collagen bone paste can be used to fill the gaps (Ao Jing Company). Although its texture is soft, its ability to induce bone health is better than the biomimetic bone flap [9], which can create favorable conditions for future bone fusion.
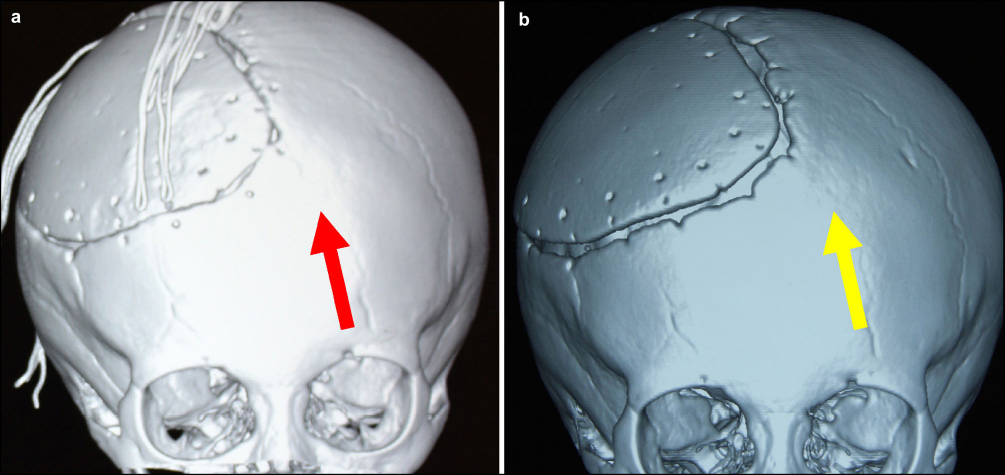
The bone hole/fixed point is undamaged, as indicated by the red arrow. The bone hole/fixed point was “torn up” after 1 year, as indicated by the yellow arrow.
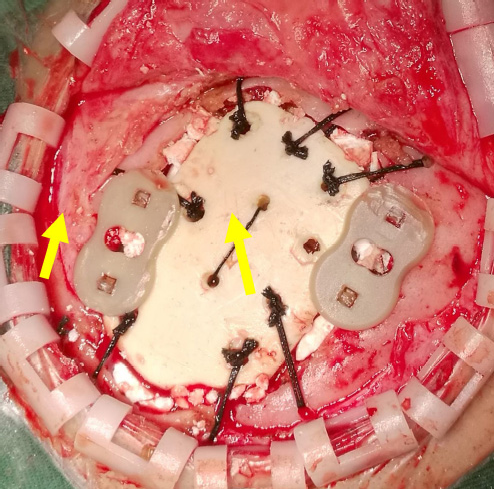
PEEK-skull lock is indicated by yellow arrows.
The cause of biomimetic bone flap fragmentation in one of our cases was not determined (Fig. 7a–7d). Children in our early cases had biomimetic bone material with more collagen and bone flap thickness was relatively thin. After 2015, all custom autogenous calvarial bone flap thickness was 1–2 mm thicker than before. One year after operation, we found a biomimetic bone crack, but because of the possible VRT stress on the bone flap (Fig. 7c) and an unclear description of traumatic history, no further treatment was administered. However, the patient was again reviewed 2 years postoperatively, and surgery was again conducted (Fig. 7e–7f). The reasons for fragmentation are believed to be as follows: (1) The child's biomimetic bone flap had more collagen, and a less hydroxyapatite was in the matrix. After osteoblasts collagen is absorbed, as creeping substitution was not sufficient, the matrix structure collapsed, as it is easily damaged under external force. (2) After initial discharge, the child had subcutaneous hydrops and was not re-administered. The child's father himself aspirated the hydrops, which possibly led to infection. The infection enhanced immunity, and the physical and chemical properties of the biomimetic bone changed. (3) After operation, the child needed to return to the hospital multiple times due to subcutaneous line issues (wire knot silk stitching of CAP shape tendinous film, 1st). The immune system in children is relatively well developed. Thus, changes in the physical and chemical properties of a biomimetic bone flap after rejection should not be excluded.
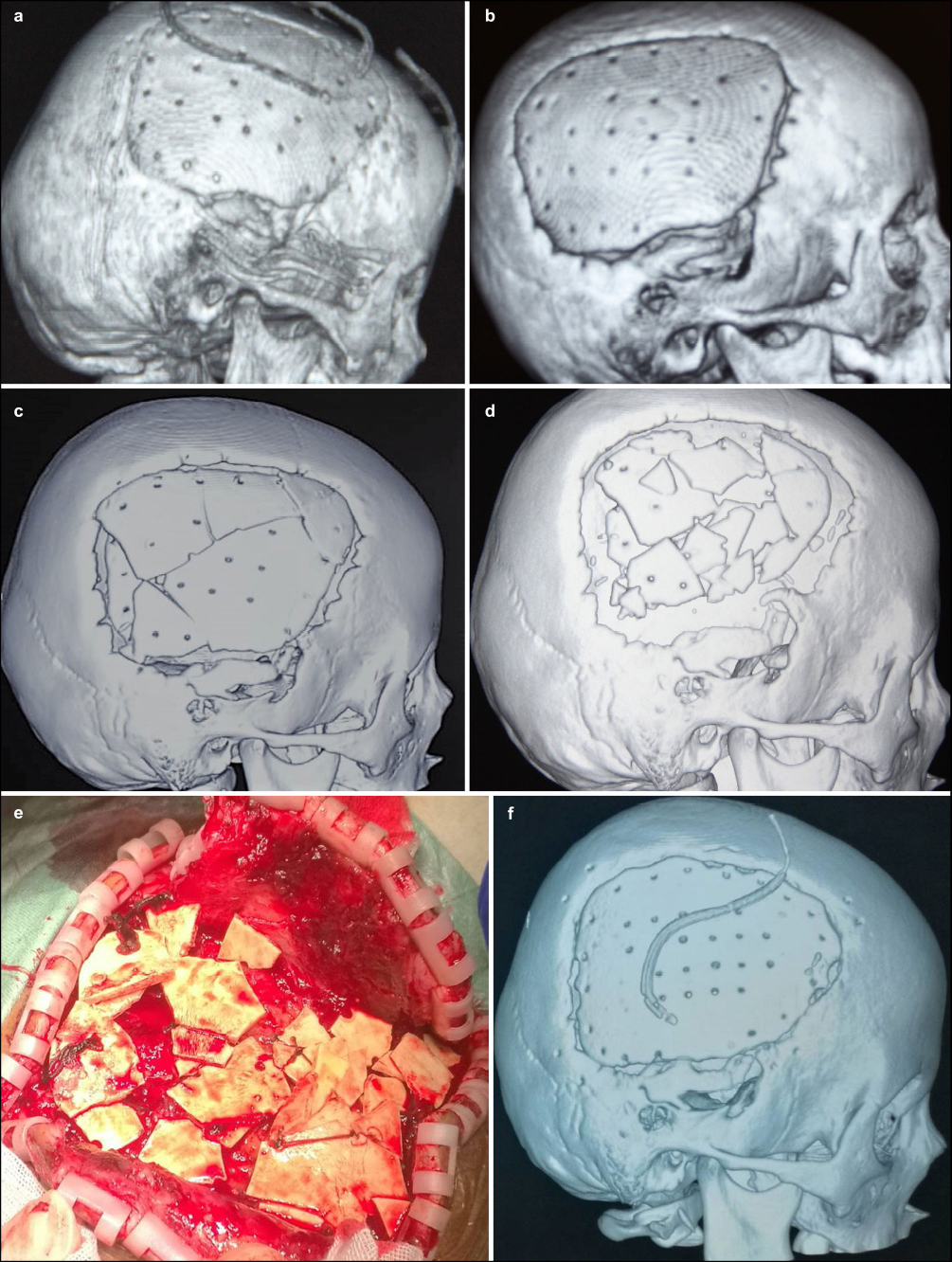
Postoperative VRT image on the day of surgery (a). Follow-up VRT images 6-months (b), 1-year (c), and 2-years (d) post-operatively. (e) Re-operation findings determined the presence of biomimetic bone flap fragmentation. (f) VRT image after re-operation.
With a deep understanding of the physical and chemical characteristics of biomimetic bone, continuous improvement of surgical methods, and improvements in biomimetic bone flap composition, the superiority of biomimetic bone for skull repair has become more obvious. Clinical experiences can be used to avoid the appearance of biomimetic bone complications following cranioplasty surgery. Despite our current findings, however, there are certain complications associated with growth or unknown factors that must still be uncovered.
Footnotes
All contributing authors have no conflict of interests.
