Abstract
To understand and manage viruses causing citrus leprosis it is necessary to identify their alternate host plants and know their distributions. Currently, the citrus leprosis disease is associated with several viruses of the genera Cilevirus and Dichoravirus. In Colombia, symptoms of citrus leprosis have been related to citrus leprosis virus C (CiLV-C - Cilevirus leprosis) and citrus leprosis virus C2 (CiLV-C2 - Cilevirus colombiaense). To detect leprosis-associated viruses in plants of the family Orchidaceae and identify the possible associated vectors, inspections and samplings were carried out on garden plants and orchid crops, and molecular detection of viruses and the taxonomic identification of mites were accomplished. As a result, Brevipalpus californicus s. 1., B. phoenicis s.s., and B. essigi (Prostigmata: Tenuipalpidae) are recorded for the first time on orchid plants from Colombia. A taxonomic key is provided to identify mites of the family Tenuipalpidae on orchids worldwide. The implications of these mites for the phytosanitary protection in citrus crops of Colombia are discussed.
Introduction
Colombia has the largest number of orchids known, with 4,270 species, and, 1,572 endemics of about 30,000 estimated in the world (Ministerio del Medio Ambiente [Minambiente], 2019). Orchids are highly attractive as ornamental plants, and the global trade associated with them is estimated at around 6 billion dollars (Anderson, 2018). In Colombia, the leading orchid growers have traditionally been carried out by associations and collectors who grow them mainly as a pastime (Guzman-Pinilla, 2016). However, over the last two decades, the production and commercialization process has been optimized. The national and international orchid trade in Colombia, although small, has increased considerably during this period (Guzman-Pinilla, 2016; Jiménez, 2018).
Despite the potential value of some Colombian species of orchids as an export crop, the study of phytosanitary protocols has not developed along with production. Two red flat mites in particular, B. californicus s.l. and Tenuipalpus pacificus Baker, have been considered cosmopolitan pests on orchids (Labanowski & Soika, 2011; Ochoa et al., 1994). Of the twelve species of Tenuipalpidae mites known on orchids in the neotropics (Castro et al., 2022), only two have been reported from Colombia (Mesa & Valencia, 2013). There is a notable research gap regarding this group of important plant-feeding mites.
Red flat mites of the genus Brevipalpus (Acariformes: Tenuipalpidae) can cause damage to orchids by feeding (Childers et al., 2003) and are also associated with virus transmission (de Lillo et al., 2021; Rodrigues et al., 2008). Among the viruses that affect orchids that are transmitted by mites of the genus Brevipalpus, members of the genus Cilevirus have been reported (Freitas-Astua et al. 1999; Kitajima et al., 2010). The most recognized of these viruses is OFV (Orchid Fleck Virus), of which there are two types: nuclear and cytoplasmic, as well as CiLV, which causes the disease called Citrus Leprosis (Kubo et al., 2009). Viruses causing citrus leprosis belong to several species distributed in two families of viruses, the genus Cilevirus, family Kitaviridae and the genus Dichorhavirus, family Rhabdoviridae where it is hypothesized that a convergent evolution with the same group of mite vectors resulted in similar symptomatology on plants (Freitas-Astua et al., 2018). Those viruses could be transmitted to some families of wild plants, ornamentals, or crops (Dietzgen et al., 2018; Garita et al., 2014; Kitajima et al., 2010; Ramos-González et al., 2023; Roy et al., 2015). In Colombia, citrus leprosis virus C (CiLV-C) was detected for the first time in 2006 (León et al., 2006), and a Dichorhavirus affecting citrus, orchid fleck virus-citrus (OFV-C) in 2014 (Roy et al., 2014). In Colombia, citrus leprosis is known to be transmitted primarily by B. yothersi (Beard et al., 2015; León, 2012; Roy et al., 2015). Citrus leprosis virus C2 (CiLV-C2, Cilevirus colombiaense) was reported in Colombia for the first time in 2015 (Roy et al., 2015) whereas CiLV-C2H strain from Hibiscus in 2018 (Roy et al., 2018).
Recently, in South Africa the presence of orchid fleck virus in fruits, leaves and branches of Valencia and Navel orange trees Citrus sinensis was reported; these infected plants were linked to the presence of orchids belonging to the genus Brassia on which infestations with B. californicus s.l. were also observed on both hosts, suggesting that the orchid was the possible link with the infection of citrus plants (Cook et al., 2019).
Olmedo-Velarde et al. (2021) reported the presence of OFV in rough lemon Citrus jambhiri and mandarin orange (Citrus reticulata) in the United States, being the first appearance of a citrus leprosis-related virus since it disappeared from Florida in the 1960s. Padmanabhan et al. (2022), using High Throughput sequencing (HTS) protocols for the detection of Cilevirus, found in Colombia positive infections for CiLV-C2, in Citrus sinensis, Swinglea glutinosa, and Hibiscus rosa-sinensis associated with Brevipalpus mites. Also, the presence of CiLV-C2 in a sample of non-citrus plants (León et al., 2023), confirmed the presence of this virus in 52 species from 22 plant families, including Orchidaceae. These recent reports highlight the increasingly complicated relationship between mites, viruses and host plants which represent a high risk to the global citrus industry with many questions still unanswered. The objective of this work was to identify mites of the Tenuipalpidae family on orchids sampled for molecular analysis of viruses of the species Cilevirus colombiaense in Colombia.
Materials and Methods
Plant and Mite Sampling
In 2020 and 2021 two orchid crops were visited. These spaces are not used for commercial purposes through the production of ornamental plants or cut flowers but are relatively small spaces about 200 square meters, which are used for the conservation and research of regional orchid biodiversity, that is, with academic and tourist purposes by members of the Santander Orchidology Society. All plants in these crops were inspected. Complementary collections were carried out in gardens of the urban and rural areas of the municipalities of Charalá, Girón, and Bucaramanga in the department of Santander and the city of Sandona in the department of Nariño (Table 1). Collections were not planned according to seasonality. Plants with virus symptoms transmitted by red flat mites of the genus Brevipalpus, which are characterized by chlorotic or necrotic lesions or ring spots on leaves and stems, were searched (Kubo et al., 2009). Leaves with symptoms of damage by mites were also collected in plastic containers with 70 % ethanol; whole leaves were collected and examined. These samples were taken to the laboratory of La Suiza Research Center of the Colombian Agricultural Research Corporation Agrosavia.
Tenuipalpid mites were carefully collected under a stereo microscope and mounted on Hoyer's medium for identification. These mites were not analyzed with molecular techniques to detect leprosis viruses, due to the low number of specimens collected in each sample. The plant material was selected, carefully packaged, and coded to be sent to the Corporación Colombiana de Investigación Agropecuaria – Agrosavia, La Libertad research center, where molecular diagnoses for viruses of the species Cilevirus colombiaense were performed. Samples of vegetal material collected in Colombia were also sent to the Maryland National Plant Germplasm Inspection Station USDA, APHIS, PPQ. The methodology and results are detailed by León et al. (2023).
Taxonomic Identification of Tenuipalpid Mites
The interactive key to red flat mites of the world was consulted (Beard et al., 2012); also, the key to the B. phoenicis species complex (Beard et al., 2015) and other taxonomic literature (Baker, 1949; Baker & Tuttle, 1987; Castro et al., 2016; Mairena & Ochoa, 1989). The dichotomous taxonomic key for the identification of tenuipalpid mites in orchids was elaborated and modified from Baker (1949), Baker and Tuttle (1987), Castro et al. (2016) and, Mairena and Ochoa (1989). All the arthropod and plant materials were collected under the collection framework permit 1466 of 2014 granted by the ANLA to the Colombian Agricultural Research Corporation. This is the license required by Colombian legislation for the collection of animal and plant materials. The mites were deposited in the “Luis María Murillo” National Taxonomic Collection of Insects (CTNI).
Sampling locations for tenuipalpid mites on orchids
Results and Discussion
Taxonomic Identification of Tenuipalpid Mites
Five species of tenuipalpid mites were found (Table 2), in addition to Brevipalpus obovatus previously reported by Mesa and Valencia (2013) in Colombia. This is the first report of Brevipalpus californicus s.l and B. phoenicis s.s. for Colombia. Mesa and Valencia (2013) reported B. phoenicis on 44 plants, including two orchids, from 15 locations in the department of Valle del Cauca, Colombia. However, this material should be re-examined based on the revision by Beard et al. (2015). Brevipalpus essigi represents the first record for South America since this species has only been reported for Greece, India, New Zealand, Mexico, and North America (Baker, 1949; Baker & Tuttle, 1987; Castro et al., 2022; Nagrare & Barman, 2009). The red flat mite, B. essigi, is also polyphagous like other species reported in this document (Baker & Tuttle, 1987; Hatzinikolis, 1986). In Colombia, it was found on hybrid orchids of Miltoniopsis sp., and although B. essigi has previously been reported on Orchidaceae, it has only been mentioned on Dendrobium nobile plants (Nagrare & Barman, 2009). This mite species has not been reported to cause economically significant damage to crops of agricultural importance or as a vector of the leprosis virus (Childers & Rodrigues, 2011). Therefore, it may have been undetected for a long time. However, a recent introduction through the importation of plant material from one of its hosts, such as chrysanthemums and orchids, cannot be ruled out.
Material Examined
1. With one to three segments on the palps ……………………………………………………………………………………… 2
1’. With four segments on the palps…………………………………………………………………………………….. 6
2. H2 elongated, with two pairs of anal setae, pregenital setae, and dorsal setae variable …………………………………………………………………………………….. 3
2’. H2 not elongated, with a pair of anal setae, pregenital setae separated by less than the width of the genital plate, dorsal seta on femur II shorter or not much longer than this segment.
3. Dorsum with a pair of lateral projections associated with seta C3…………………………………………………………………………4
3’. Dorsum without lateral projections associated with seta C3 ………………………………………………………………………………………………………5
4. Central prodorsum with a sculpture of rhomboid to hexagonal cells like a honeycomb.
4’. Central prodorsum with rough and striated sculpture.
5. With two pairs of IC3 and one pair of IC4. Ventral lateral cuticle anterior to coxa III and lateral to setae 3a1 and 3a2 with characteristic pebble-like pattern
5’. With one pair of IC3 and two pairs of IC4. Ventral lateral cuticle without characteristic pebble-like pattern
6. Setae f2 present (Figure 1A). …………………………………………………………………………………….. …7
6’. Setae f2 absent …………………………………………………………………………………….. 10
7. Two solenidia in tarsus II (Figure 1B). Spermatheca circular, with an internal bubble (Figure 1A and 1C)……………………
7’. One solenidion in tarsus II. Spermatheca with variable shape and never with an internal bubble …………………………………………………………………………………….. …8
8. The two pairs of ventral hysterosoma setae of the same size …………………………………………………………………………………………………………………….
8’. Pair of anterior ventral hysterosoma setae notably shorter than posterior pair ……………………………………………………………………………………………………… …….9
9. Reticulate pattern throughout the ventral plate (Figure 1D). Propodosoma and hysterosoma with reticulations evenly distributed over the entire area (Figure 1E) Propodosoma with dorsal setae strongly serrate.
9’. Reticulate pattern of the ventral plate is present only laterally. Propodosoma and hysterosoma with reticulations are not present over the entire area. Propodosoma with dorsal setae slightly serrate.
10. One solenidium on tarsus II………………………………………………………
10’. Two solenidia on tarsus II …………………………………………………………………………………….. 11
11. Dorsal seta on femurogenu setiform, cuticle on dorsal opisthosoma between setae e1-e1 and h1-h1 with V-shaped folds, oval spermatheca vesicle with a marked distal duct. Ventral plate cuticle uniformly verrucose, with small, rounded “warts” that are sometimes weakly impressed centrally..
11’. Dorsal seta on femurogenu broad and flat (Figure 1F), cuticle on dorsal opisthosoma between setae el-el and h1-hl with few transverse folds (Figure 1G), spermatheca vesicle not visible, duct sometimes visible. Ventral plate cuticle usually with some “warts” laterally that are fused medially to form transverse bands
Lesions typically caused by leprosis virus (Figures 2a and 2b), coincide with the presence of B. yothersi. The red flat mite B. yothersi is polyphagous and confirmed as the main vector of the leprosis virus in Colombia (León et al., 2006; Roy et al., 2015). Brevipalpus yothersi has been reported as a frequent carrier of CiLV-C and OFV in citrus crops (Beltran-Beltran et al., 2020; Olmedo-Velarde et al., 2021). Considering that these viruses can produce symptoms in orchids, it could be possible that B. yothersi is the vector of Cilevirus colombiaense virus in orchids from Colombia. However, it is necessary to include the molecular analysis and experimental transmission assays of CiLV-CH2 in B. yothersi affecting orchids in future research to confirm the vector-virus-host relationship in Orchidaceae.
Brevipalpus californicus s.l., which can be the carrier of CiLV-C and OFV (Beltran -Beltran et al., 2020; Roy et al., 2015), is now accepted as a complex of seven species (Tassi et al., 2022). (Beard et al., 2012). Given this development, it is necessary to verify the transmission capacity of the leprosis viruses by each member of this mite complex.
It is noteworthy that although citrus leprosis-causing viruses are present in orchids, symptoms have not been reported on citrus plants by farmers or extension workers even though periodic monitoring of the phytosanitary status of citrus crops is carried out by trained personnel from the Colombian Agricultural Institute (ICA), looking for symptoms of leprosis, HLB, and other pests and quarantine diseases.
Citrus leprosis-causing viruses have had little impact in the Santander citrus growing region, possibly due to several factors influencing their survival and dispersal. Firstly, there are differences between citrus species and cultivars that vary in susceptibility to leprosis (Freitas-Astua et al., 2008). The predominant citrus crops in the Santander region are Tahiti lime and tangerine, which are less susceptible than oranges (Childers & Rodrigues, 2011). For example, CiLV-C is more important and causes more damage in the state of Sao Paulo, which produces 80% of the oranges in Brazil (Bastianel et al., 2010). Secondly, regarding orange production, the most susceptible citrus species is generally found in agroforestry systems with coffee and cacao but not in monoculture, making virus dispersion more difficult. Thirdly, the citrus industry in Santander, Colombia, is mainly based on small producers with small areas that are less than four hectares; these relatively small plots are mixed in a matrix with other crops, fallow land, and natural areas. Bastianel et al. (2010) mentioned that even though citrus leprosis is present in some regions of Brazil, it does not cause significant damage because it is not farmed in large monocultures and its environmentally friendly management favors a reasonable natural mite control. Another factor could be that a large proportion of the crops, especially tangerine, are grown in areas with good humidity and rainfall above 2000 mm per year which has an inversely proportional relationship with mite proliferation (Solano et al., 2008).
Other factors related to the biology and ecology of the mite to prevent the proliferation of citrus leprosis are: (i) the acquisition of the virus occurs in parts of the plant with lesions because the virus is not systemic (Locali et al., 2004); (ii) there is no transovarial transmission of the leprosis virus (Boaretto et al., 1993) and (iii) reduction in the reproductive potential of mites fed on leprosis-affected tissue has been recorded (Salinas-Vargas et al., 2019). In Brazil, Bastianel et al. (2010) reported that the disease is found in several citrus-productive regions in low incidence and severity and that farmers only become concerned when the damage becomes visible and appears to spread rapidly.
Tenuipalpid mites and virus detection in orchid plant material.
The results of molecular analyzes of plant material are taken from León et al. (2023)
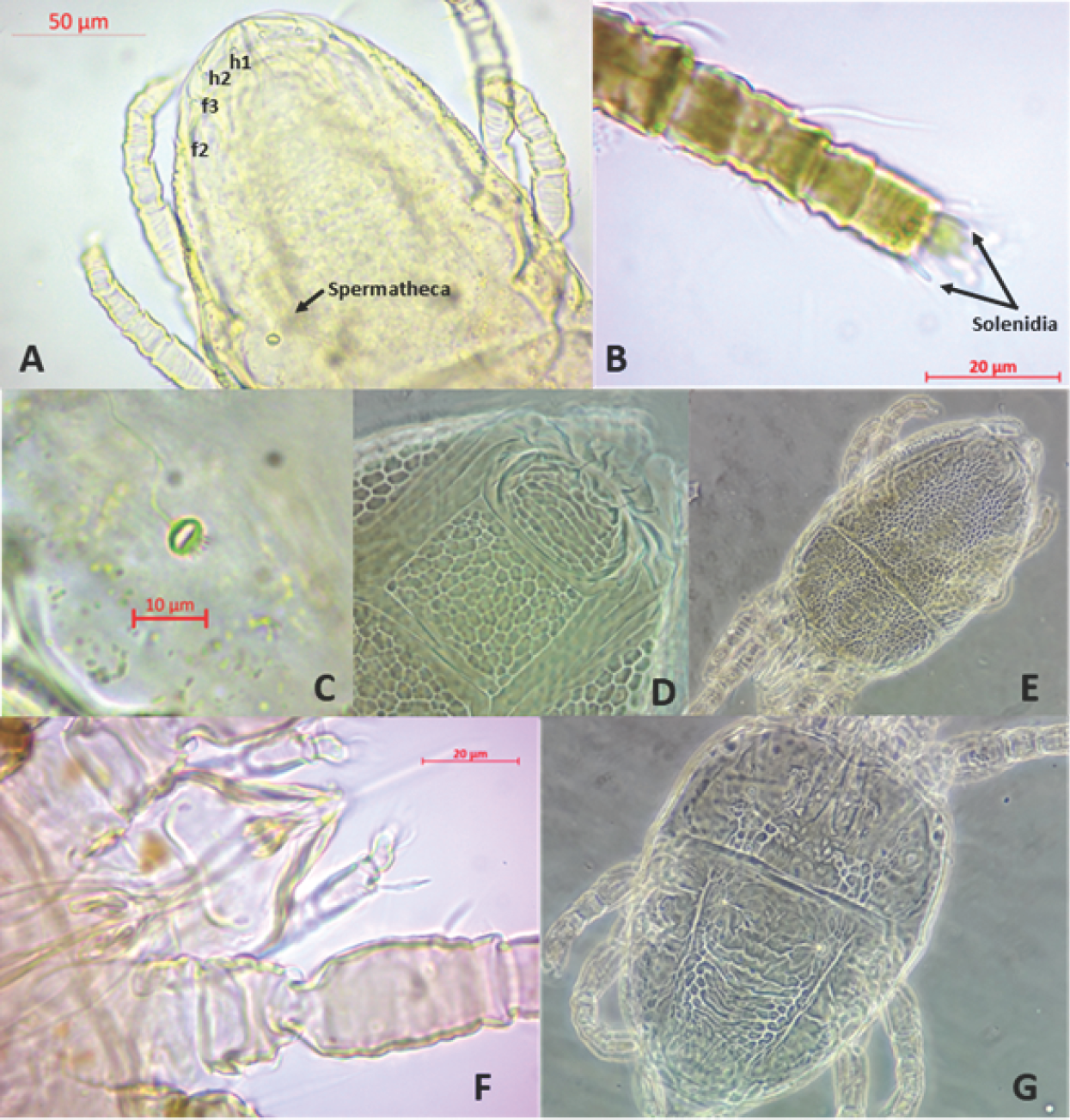
A-C. Brevipalpus californicus s.l. A. Opisthosoma. B. Tarso II. C. Spermatheca. D-E. Brevipalpus essigi. D. Ventral and genital plate. E. Dorsum. F-G. Brevipalpus phoenicis s.s. F. Palps. G. female dorsum. Photos: A-C, F by J. Montes; D, E, G by A. Sierra.

Symptoms and damage of tenuipalpid mites on orchids infected by Cilevirus colombiaense virus. A. Arundina graminifolia. B. Spathoglottis plicata. C. Maxillaria sp. D. Phalaenopsis sp. E. Maxillaria sp. F. Maxillaria desvauxiana. G-H. Ada sp. G. Leaf dorsal side. H. Leaf ventral side. I. Sobralia sp. Photos by J. Montes.
Conclusions
Three new reports of species of tenuipalpid red flat mites of the genus Brevipalpus are recorded for the first time from Colombia. Of the red flat mite species now recorded in Colombia, two species have a high capacity to transmit leprosis virus, namely, B. yothersi and B. californicus s.l. Orchids are a group of host plants of Cilevirus colombiaense virus in Colombia and because these plants are often found near or within citrus groves, they should be monitored for leprosis-causing viruses as well as their mite vectors.
Origin and funding
This work was carried out within the framework of the USDA-Agrosavia agreement, with resources from the agreement: “Basic and strategic research on citrus leprosis virus (CiLVs), a quarantine disease present in Colombia.”
Author contribution
The first author participated in the sampling and taxonomic identification of mites, sampling of plant material, contributed to the analysis of the data and the writing of the final document.
The second author participated in the sampling and taxonomic identification of mites, contributed to the analysis of the data and in the writing of the final document.
The third author stated the objectives of the research, obtained funding, processed the plant material, contributed to the analysis of the data and to the writing of the final document.
The fourth author participated in the taxonomic identification of mites, contributed in the analysis of the data and in the writing of the final document.
Conflict of interest
The authors declare they have no financial interest. This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Footnotes
Acknowledgments
The authors express their gratitude to the orchid growers who allowed the sampling of mites and plant material and to the reviewers of the final manuscript for their comments and suggestions. Thanks also to Verónica Lucía Rodríguez for her advice and comments in the materials and methods section. Special thanks to Ronal Ochoa for his help in the taxonomic identification of the tenuipalpid mites. This work was carried out within the framework of the USDA-Agrosavia agreement, with resources from the agreement: “Basic and strategic research on citrus leprosis virus (CiLVs), a quarantine disease present in Colombia.”
