Abstract
Among a series of exotic pests that have been reported in Chile, Xanthogaleruca luteola (Coleoptera: Chrysomelidae), a defoliating insect of elms (Ulmus spp.), is causing important damage in urban trees in central Chile. The insecticidal capacity of water and ethanol extracts obtained from new and mature leaves of Cestrum parqui (Solanaceae) was evaluated at various concentrations on X. luteola adults in the laboratory. Bioassays with a completely randomized design were used to analyze the effects of each extract. Also, the mean lethal concentration (LC50) was determined for both mature leaf states and solvents. A proximal analysis was done to determine the physical and chemical differences between the maturity stages. Proximal analysis of the powder of both leaf stages revealed differences (P < 0.01) in the content of humidity, ashes, lipids, and proteins, but not in the raw fiber content nor the non-nitrogenous extract. On the third day after exposure, the order of decreasing effectiveness (LC50) of the insecticide extracts was: new-ethanol (2.5 % w/v), new-water (3.8 % w/v), mature-ethanol (6.7 % w/v) and mature-water (9.2 % w/v). Our results suggest that C. parqui leaf extracts may have potential use as a bioinsecticide in integrated pest management plans for the control of X. luteola adults, however, tests at a larger scale are necessary to confirm our results.
Introduction
A series of pests have been detected in plantations and urban trees, having a direct effect on the economy and recreational areas in central Chile. Such is the case of the elm trees (Ulmus; Ulmaceae) suffering from defoliation by the elm leaf beetle, Xanthogaleruca luteola Müller, 1766 (Coleoptera: Chrysomelidae) for which there is no natural and efficient treatment available (Huerta et al. 2011). This serious pest occurs throughout Europe, North Africa, Asia, Australia, and temperate areas in North and South America (Romanyk and Cadahia 2002; Borowiec and Sekerka 2010; Lefoe et al. 2014). The biology and lifecycle appear in Romanyk and Cadahia (2002), Folcia et al. (2005), Huerta et al. (2011), Dreistadt et al. (2014), and Rodrigo et al. (2019). Both the adult and larvae feed on the parenchyma of leaves, without consuming the veins, and cause severe damage to trees. If the damage is severe and occurs several years in a row, the trees develop deformed canopies and suffer vigor loss, physiological disorders, and reduced photosynthesis, which makes the trees susceptible to the Dutch elm disease (Romanyk and Cadahia 2002). Defoliation also reduces tree shade in summer and the aesthetical values of elms in the urban area (Dreistadt et al. 2014).
Synthetic insecticides have been tested against X. luteola (Lawson and Dahlsten 2003), but these types of compounds are harmful to humans and environmental health. They have also been shown to be toxic to non-target organisms and responsible for the development of resistance in pest populations (Isman 2006; Kumar 2012), being prohibited or restricted in many areas. In this context, arboriculture is called to provide or solutions to increase tree yields while preserving natural resources and the environment. An alternative is the use of environmentally safe techniques such as bio-insecticides (Suthisut et al. 2011). There is a trend towards the elaboration of natural insecticides based on the active components of certain plants (Tavares et al. 2009). The interest in compounds of botanical origin for pest control is based on their efficacy, availability, degradability, physiological activity, relatively low toxicity, and persistence, and, in general, lower risk to the environment (Isman 2006).
Plant insecticides are secondary metabolites produced naturally for protection against pests. They exhibit a range of biological activities, acting as repellents, insecticides, fungicides, nematicides, and bactericides (Isman 2006). On various insect species, botanical pesticides inhibit food ingestion, growth at various stages of development, and egg-laying (Regnault-Roger et al. 2012; Pavela 2011).
The South American shrub Cestrum parqui L'Heritier (Solanaceae), also known as green cestrum or willow-leaved jessamine, occurs in Chile from Coquimbo through Osorno (Hoffmann 1982). The toxicity of the plant is concentrated in the raw saponic extract (Barbouche et al. 2001). This plant and its saponins (defense substance) have shown anti-feeding, repulsive or insecticidal activities, or malformations on various insect orders such as Orthoptera Schistocerca gregaria (Forsskål, 1775) (Acrididae) (Barbouche et al. 2001), Lepidoptera Spodoptera littoralis (Boisduval, 1833) (Noctuidae), Helicoverpa armigera (Hübner, 1805) (Noctuidae) and Pieris brassicae (Linnaeus, 1758) (Pieridae) (Chaieb et al. 2001; 2007) and Diptera Ceratitis capitata (Wiedemann, 1824) (Tephritidae) (Zapata et al. 2006) and Culex pipiens (Linnaeus, 1758) (Culicidae) (Chaieb et al. 2009). However, its effectiveness on Coleoptera, as far as we know, has been proven only three in species (Tenebrionidae: Tribolium castaneum Herbst, 1797) (Rafael et al. 2000), (Tenebrionidae: Tribolium confusum Jac. 1863) (Chaieb et al. 2006), and (Curculionidae: Hylurgus ligniperda Fabricius, 1787) (Huanquilef et al. 2020) so this is the first report on a Chrysomelidae.
The aim of this work was i) to physically and chemically characterize new and mature leaves of C. parqui, and ii) to evaluate the insecticidal activity of new and mature C. parqui leaf extracts on X. luteola adults to contribute to integrated pest management.
Materials and methods
During the summer, 1 kg foliage from C. parqui shrubs was collected randomly from the canopy at 1.5 m from the soil, from 10 trees with the trunk of at least 10 cm in diameter to ensure diversity in Punitaqui (30°54'S 71°16”W), Coquimbo Region, Chile. The leaves were cut from the stems (which were discarded) and separated into two groups, new (light green and tender) and mature (darker green and harder). Then, the leaves were washed with distilled water and dried in a forced-air oven (Memmert, Schwabach, Germany) at 37 °C for 60 h at the Chemistry Laboratory, in the Department of Agroindustry and Enology, Faculty of Agronomic Sciences, University of Chile, Santiago, Chile. Larvae of X. luteola were collected from adult Ulmus minor trees, not chemically treated, in Maipú (33°32'S 70°46′W), Santiago. In the Forest Entomology Laboratory, Faculty of Forestry and Nature Conservation Sciences, University of Chile, they were set on Petri dishes lined with filter paper humidified with water. They were fed with fresh elm trees until pupation then, they were provided humidity and covered with more leaves. All insects were maintained at 20 ± 3 °C and 60 ± 4 % RH. The emerging adults were fed on fresh leaves and were used in the bioassays.
The leaves were ground separately in a mechanical mill with a graduated sieve (No 60 mesh) into 0.25 mm particles and then stored in labeled glass vials. The resulting powder was used to determine the proximal analysis and color and the extract preparing by bioassays. 10 g (each) of these powders were analyzed to determine their physical and chemical characteristics (Association of Official Analytical Chemists 1984): humidity thorough drying in a forced air heater at 105 °C until constant weight; lipids by Soxhlet extraction with petroleum ether; raw fiber using acidic and basic hydrolysis; ashes from muffle incineration at 550 °C; proteins by the Micro-Kjeldahl method; and non-nitrogenous extract (by difference from the other contents). The color was determined for both leaf maturity stages with a colorimeter (CR 300 Minolta, New York, USA) through the CIELab method using three values: L*, the difference between light (where L* = 100) and dark (where L* = 0); a*, the difference between green (- a*) and red (+ a*); and b*, the difference between yellow (+ b*) and blue (- b*) (Ngo et al. 2007). All the analyses were done three times. For leaf maturity stages an one-way ANOVA was conducted for each determination of proximal analysis followed by the Tukey test (P < 0.01) when significant differences occurred between leaf maturity stages.
The leaf powders were mixed in the Chemistry Laboratory with water or 96 % ethanol (Merck, Darmstadt, Germany), until obtaining the highest concentrations possible, 1/10 for water, and 1/6 for ethanol. The solutions were mixed 18 h in a magnetic stirrer (Heidolph, MR 3001K, Schwabach, Germany), at 37 °C the first hour, then filtered through a Whatman No 1 filter paper, centrifuged (HN-S Centrifuge, International Equipment Co., Philadelphia, USA) 15 min at 1,500 rpm and re-filtered to obtain the base extracts. The soluble solids in the base extracts were determined by weight differences, oven-drying aliquots 1 h at 100 °C. These levels were used to prepare the concentrations to be used in the bioassays by dilution. Both extracts (aqueous and ethanolic) from the new and mature leaves were tested at 0.5, 1.7, 3.1, and 5.5% w/v. The bioassays for both leaf powders were separate, also considering controls (only solvents), all treatments with three replicates. The experiment units were Petri dishes lined with slightly wet filter paper at the bottom and a fresh and washed elm leaf, with the petiole wrapped in wet cotton, set with five X. luteola adults. The leaves were immersed for 1 min in the respective treatment, let dry at room temperature, and set on the humidified filter paper-lined Petri dishes with the insects. The control was not immersed in C. parqui extracts, only solvents (water or ethanol). Daily and total mortality were determined until mortality stabilized, on day 8. The results were normalized by Bliss. After the evaluation, data were submitted to a factorial design (5 x 4) ANOVA, where factor A was the concentration and factor B, the extract effect. When significant differences occurred between treatments, they were separated using the Tukey test (P ≤ 0.05). The results were examined using mathematical functions to identify the best fit to obtain the LC50 (lethal concentration to kill 50 % of the individuals) through the Probit method (Robertson et al. 1984), using chi-square to measure the fit to the model. All statistical analysis using the InfoStat (2009) software.
Results and discussion
Physical and chemical characterization of the leaves from C. parqui
The initial water content of the fresh new and mature leaves was statistically similar, both near 77%. However, the dry weight was 40 % greater in the mature leaves (Table 1). Chiffelle et al. (2011b) on new and mature Peumus boldus Molina (Monimiaceae) leaves reported contents lower, 65 and 58 %, respectively. Similarly, Chiffelle et al. (2009) reported very close humidity levels for new and mature Melia azedarach L. (Meliaceae) leaves, 61 and 57 %, respectively.
Initial humidity contents and dry weight of new and mature leaves, and chemical parameters of dry powdered leaves (± SD) of Cestrum parqui.
Means in a line with different letters are significantly different (Tukey test, P≤ 0.01).
Non-nitrogenous extract was obtained by weight difference.
The proximal analysis (Table 1) of the powdered leaves yielded significant differences between the development stages, except raw fiber and non-nitrogenous extract. Our results were similar to those in Chiffelle et al. (2009) and Chiffelle et al. (2011b) for M. azedarach and P. boldus powdered leaves, respectively. The ash contents were significantly greater in the mature leaves (4 %), coinciding with the results by Chiffelle et al. (2011b) in P. boldus leaves. Proteins were significantly greater in the new leaves, as also found in M. azedarach by Chiffelle et al. (2009), but with a near 9 % difference instead of the 0.5 % in the second leaves (mature). The lipids differed significantly in the powdered new and mature leaves and were slightly greater in the last ones. Likewise, Chiffelle et al. (2011b) also observed, but only as a trend, an increased lipid content with maturity in P. boldus. The content of raw fiber did not vary between leaf maturity levels, although a slight decrease occurred with age, similarly to what other authors mentioned before. Several reports have identified chemical components from C. parqui (Baqai et al. 2001) but only a few aimed at their insecticidal effect, pointing mainly to saponins (Chaieb et al. 2007). Although some saponins are soluble in water (Guclu-Ustundag and Mazza 2007), extractions from plants usually use ethanol (Segura et al. 2020), which probably explains our better results with the ethanolic extracts.
The luminance (L*) of the powder from new leaves was significantly greater and with a lesser green tone (a*) than those from mature leaves. The yellow level (b*) was similar (Table 2). Luminance and color are important for differences in the leaf maturity stages and thereby facilitate their classification and subsequent standardization of the vegetal material.
Color means ± SD of the powder from new and matures leaves from Cestrum parqui.
aL*, Luminance; a*, range from green to red; b*, range from blue to yellow. Means in a line with different letters are significantly different (Tukey test, P≤ 0.05).
Mortality of Xanthogaleruca luteola adults with the extracts
In this study, the level of mean mortality of X. luteola adults was directly related to the concentration applied of C. parqui leaf extract. Both the water and ethanol extracts caused greater mean mortality when using new leaves. The treatments with the ethanol extract from new leaves caused a greater mean mortality than the water extract. Concentration (F4.12= 183.1; P < 0.05) and extract effect (F3.12 = 52.4; P < 0.05) factors were significant. Also, the ethanol extracts from new leaves caused the greatest average mortality (over 20%) compared with mature leaf ethanol extracts at the same concentration. Mean mortality near 73% occurred with the water extract from new leaves at just 5.5 % w/v concentration with the greatest concentration used, while with the same solvent from mature leaves, at the greatest concentration (5.5 % w/v), mortality did not exceed 51 %. The lowest mortality effects were achieved with the lowest concentrations in both stages of maturity (Table 3).
Mean mortality (% ± SD) of Xanthogaleruca luteola adults by the effect of the ethanol and water extracts from new and mature Cestrum parqui leaves.
Different small letters in a column and capital letters in a row indicate significant differences in concentrations and extracts, respectively, according to Tukey tests (P ≤ 0.05) at day 4 after expositure.
Cumulative mortality of X. luteola adults was gradual in new leaf extracts and reached 100% with the highest concentration (5.5 % w/v) in water (8 d) (Fig. 1A) and in ethanol (6 d) (Fig. 1B). The mortality of water and ethanol from new leaf extracts had major insecticidal capacity, which indicates a faster effect than that of the water mature extracts (Fig. 2).
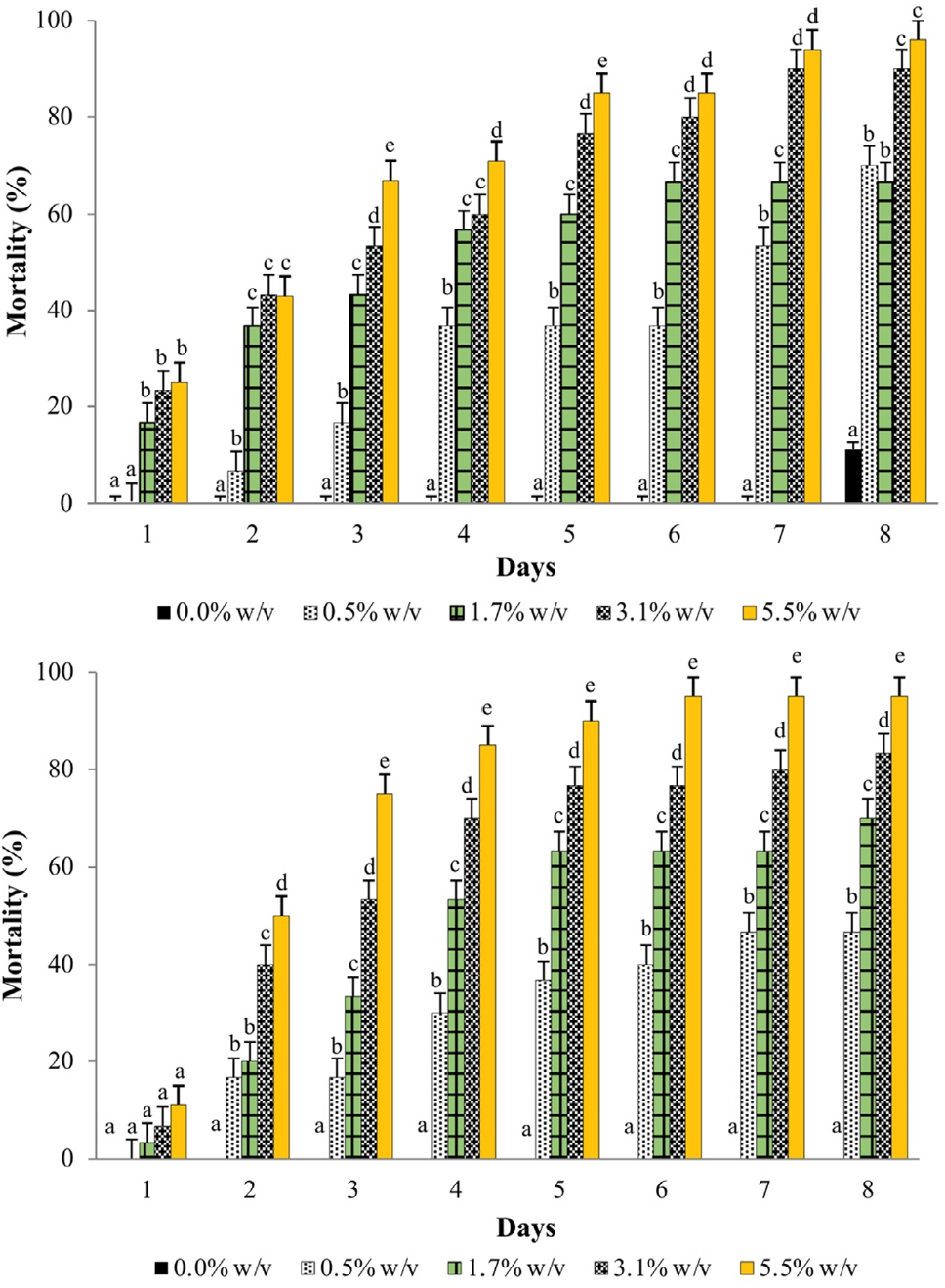
Cumulative mean mortality of Xanthogaleruca luteola adults exposed to water (top), and ethanol (bottom) extracts from new Cestrum parqui leaves at several concentrations, during 8 d evaluation. Means with different letters horizontally were different (Tukey, P ≤ 0.05).
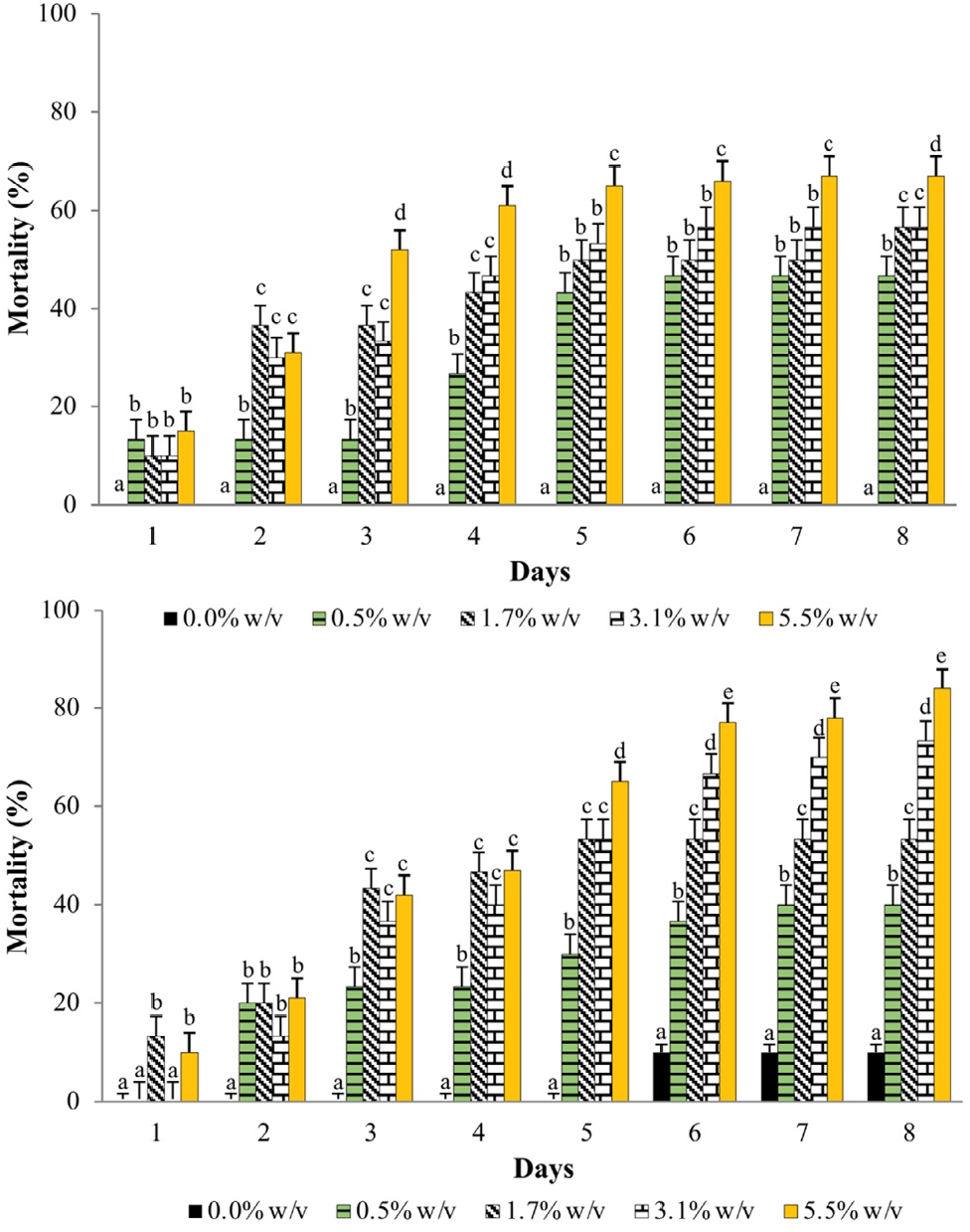
Cumulative mean mortality of Xanthogaleruca luteola adults exposed to water (top), and ethanol (bottom) extracts from mature Cestrum parqui leaves at several concentrations, during 8 d evaluation. Means with different letters horizontally were different (Tukey, P ≤ 0.05).
This response at a lower concentration demonstrates a greater insecticide of C. parqui leaf powder when using ethanol as solvent. The effectiveness of the extracts was related to their concentration, mainly dependent on the leaf maturity stage used.
With other pests, C. capitata adults, various concentrations of extract from C. parqui leaves were evaluated. The extracts obtained with organic solvents (hexane, acetone, and methanol) were innocuous, except for the mixture of methanol and water (80:20), which killed 12.5 % of adults. Water and lyophilized water extracts increased adult mortality, mainly with water extracts at 2 % from day 2 and on. The toxicity of the other concentrations increased from day 3 until the end of the assay, and at 2 % reached 100 % mortality on day 6 (Zapata et al. 2006). These results differ from those herein on X. luteola, as with the water extracts from new leaves at 1.7% w/v, only 62 % mortality occurred on day 6 (Fig. 1A). Saponins have been identified as insecticidal compounds obtained from C. parqui (Chaieb et al. 2007).
The results of mortality of X. luteola adults obtained herein were smaller than those found by Huerta et al. (2010) using leaf (both new and mature) extracts from Schinus molle L. (Anacardiaceae), who reported 100 % mortality with the ethanol extract at 4.7% w/v. At the greatest concentration (5.6 % w/v) of the water extract, they found 28 % average mortality on day 8, a result inferior to those herein. Chiffelle et al. (2013) evaluated the mortality of X. luteola larvae obtained 89 % mortality with ethanol extract at 4.3 % w/v using new leaves of S. molle at 12 d, being similar to those obtained in the present study.
The insecticidal effect of extracts from immature fruits of M. azedarach on X. luteola was evaluated, and obtained 86 % cumulative mortality with the ethanolic extract at 2.4 % w/v at day 8 (Chiffelle et al. 2011a).
Extracts from new and mature P. boldus leaves were used and obtained over 40% cumulative mortality with the ethanol extract from new leaves at 2.9% w/v at day 3. With ethanol extracts from both stages of leaf development, 70% mortality was exceeded at concentrations above 3.6 % w/v on the 3rd day (Chiffelle et al. 2011b). Those results demonstrate a greater insecticidal effect for ethanol extracts in both plant species, which coincides with our study.
Defagó et al. (2006), evaluated the insecticidal activity of ethanol extracts from new and mature leaves of M. azedarach and at 2, 5, and 10 % from new leaves obtained of X. luteola adult mortality close to 100 %. Maistrello et al. (2005) used the ethanol extract from Daphne gnidium L. (Thymelaeaceae) leaves on second instar larvae of X. luteola, and at 0.1, 0.2, and 0.3 % w/v recorded 27.3, 70.4, and 73.4 % mortality, respectively. Those results were greater than those obtained herein, a difference that may be due to the extracting method. Whereas they did the extraction from the lipid portion, and we worked with raw extracts. Also, Maistrello et al. (2005) used insect larvae, a stage more sensitive than the adults.
Determination of the LC50
The LC50 obtained with the Probit analysis at the 3d evaluation was smaller with the new C. parqui leaf extracts than with the mature leaf ones, which indicates a greater effectivity of the first, which required a smaller concentration to cause the same mortality (50 %). With both solvents, the extracts from new leaves produced smaller LC50, 2.25 and 3.80 % w/v for ethanol and water, respectively, on X. luteola adults. In order of decreasing insecticidal effectiveness from leaf extracts, at 3 d from application, the LC50 were 2.25, 3.80, 6.67, and 9.16 % w/v for the new-ethanol, new-water, mature-ethanol, and mature-water, respectively (Table 4).
Insecticidal effects of leaf extracts from Cestrum parqui, as indicated by the LC50 on Xanthogaleruca luteola adults.
CI confidence interval.
χ2 values obtained below those tabulated for new and mature leaves for both water and ethanol (χ2 = 7.81, df = 3; P < 0.05), from which the Probit model fits the experimental data.
The LC50 determined for ethanol extracts from new and mature leaves of P. boldus by Chiffelle et al. (2011b) 48 h after beginning the treatment on X. luteola were of 1.2 and 2.3 % w/v, respectively. At the same period, LC50 of 3.7 and 4.0 % were obtained herein, respectively. On day 8, Chiffelle et al. (2019) found LC50 for ethanol extracts from new and mature leaves of P. boldus but on X. luteola larvae, 0.92 and 1.20 % w/v, respectively.
On day 5, Chiffelle et al. (2011a) found that the water extracts of M. azedarach green fruits had a LC50 of 6.55 % w/v on adult X. luteola, similar to that found herein with the same extract.
With concentrations of 5, 10, 20, 40, and 80 % of a methanol extract of Artemisia annua L. (Asteraceae) leaves, Shekari et al. (2008) obtained LC50 of 43.77 and 15.43 % for third-stage larvae and adult of X. luteola, respectively. Herein, both the water and ethanol extracts of C. parqui leaves evaluated in our study are more effective on adult X. luteola.
Finally, the insecticidal effect obtained with extracts from C. parqui leaves was significant, verified when observing the small mortality of the controls. When comparing the extracts from both leaf maturity types, significant differences appeared between them, with greater mortality when using new leaves, which could be explained by the higher concentration of active compounds in this state of maturity of the leaves (Gros et al. 1985).
Conclusions
The two stages maturity in leaves of C. parqui presented significant differences in contents of water, ash, lipids, and proteins. Mortality of X. luteola adults was directly related to the concentration of the extracts in both solvents. The LC50 at the 3rd day of the evaluation was smaller with the ethanol than with the water extract, for both leaf development stages. Given the risk of applying conventional insecticides on elms in streets, C. parqui represents a bioinsecticide source for use in urban tree integrated pest management. Further studies should be conducted on the insecticide properties of C. parqui against X. luteola in open areas, and the larval stage.
Origin and funding
This work was derived from the self-financed memoir of Laura Araya, third author, to apply for the title of Forest Engineer from the Faculty of Forest Sciences and Nature Conservation of the University of Chile.
Author contribution
Amanda Huerta, first author, participated in the design, data processing, analysis and discussion of the information in the manuscript and in the writing of the manuscript.
Italo Chiffelle, second author, participated in the analysis and discussion of the information.
Laura Araya, third author, did the field and laboratory work, participated in the processing of the data and its analysis.
Tomislav Curkovic, fourth author, participated in the analysis and discussion of the information, writing and translation of the manuscript.
Jaime E. Araya, last author, participated in the analysis and discussion of the information and translated the manuscript.
