Abstract
Between 1995 and 2006, a total of 3280 lycaenid adults belonging to 47 species were collected in order to study their distribution and current status in Bursa, northwestern Turkey. Of these, Tomares nogelii is a newly recorded species for northwestern Turkey. The following lycaenid species had not been seen since the 1860s: Aricia eumedon, Cupido osiris, Kretania eurypilus, Plebeius sephirus, and P. ripartii. The endemic water dock plant species Rumex olympicus was recorded as a new host for the larvae of Lycaena dispar. Both Aricia hyacinthus and the endemic Polyommatus ossmar olympicus are under threat of extinction. In contrast, the status of some Polyommatus species changed from local to widespread. The highest number of lycaenid species was recorded in July with a total of 40 species per month. Widespread lycaenid species were generally caught at altitudes higher than 1000 m. Altitudinal distribution and phenology of lycaenid species as well as their new host plants found are discussed.
Introduction
Lycaenids, also known as gossamer-winged butterflies because of their iridescent wings, are small-sized butterflies (< 5 cm). Lycaenidae comprises more than a third of the world's Papilionoidea with over 6000 different species (Robbins 1982; Ackery et al. 1999). Although both Africa (Ghana) and South America (Colombia) support a rich lycaenid fauna, the distribution of many lycaenid species are limited by their specialised habitat preferences (Legg 1978; Fiedler 1996). This characteristic makes them very vulnerable to habitat loss caused by human disturbance to the environment for agricultural and developmental purposes.
In fact, the loss of suitable habitat can be considered the most imminent threat to butterfly species' persistence (van Swaay and Warren 1999). Because of their habitat loss, many lycaenid species are listed as threatened in Red Data Books in many European countries (van Swaay and Warren 1999). A butterfly habitat includes not only larval hostplants and breeding resources, but also sites for roosting, hibernation and mate location outside the hostplant areas (Dennis 2004). For example, lycaenids such as Plebeius argus (L., 1758) and Polyommatus icarus (Rottemburg, 1775) are known to roost and mate on taller vegetation substrates rather than their host-plants (Emmet and Heath 1990; Dennis 2004). Dennis (2004) also noted the increasing importance of shrubs for lycaenid butterflies as the season progresses.
First reports on the lycaenid fauna of Turkey date back to the 1830s when Albert Kindermann started a comprehensive survey on Turkish Rhopalocera covering the provinces of Istanbul and Bursa (Hesselbarth et al. 1995). Later, many European entomologists carried out seasonal butterfly surveys in different parts of the country (Zeller 1847; Mann 1862, 1864; Oberthur 1872; Staudinger 1878; Fountaine 1904; Graves 1911, 1912; Wagner 1929; De Lattin 1950; Higgins 1966; Betti 1989; Carbonell and Brevignon 1983; Carbonell 1992; Carbonell 2003). Turkish entomologists initiated their work on the local butterfly fauna from the early 1960s (Kansu 1961, 1963; Oktem 1962; Sengun and Guneyi 1968; Guneyi and Kirmiz 1971; Guneyi and Uyar 1972; Kocak 1975, 1976, 1989; Avci and Ozbek 1996; Akbulut et al. 2003). However, the Bursa region has been largely neglected in recent decades by collectors and researchers in favour of the central and eastern parts of Turkey where the chance to find undescribed Lycaenid taxa is believed to be greater.
There are 161 lycaenid species in Turkey according to the checklist of Kocak and Kemal (2006), but the total number of lycaenid species has recently increased to 164 based on the accounts of the Centre for Entomological Studies Ankara (CESA unpublished). Of these species, 54 have been recorded from the Bursa province of northwestern Turkey (Hesselbarth et al. 1995). However, some species were only recorded in 1851 and 1863 by Mann and no specimens were collected after then (Mann 1862, 1864). It has been more than 100 years since some lycaenid species were last seen.
The objectives of this study were: 1) to investigate the changes in the distribution of lycaenid fauna in Bursa province of northwestern Turkey, 2) to assess their current geographical status (widespread or local) in order to identify sites where lycaenid conservation is necessary, and 3) to provide information about the ecological characteristics of the recorded species.
Material and Methods
During 1995-2006, lycaenid adults were collected from March until late October in Bursa province of northwestern Turkey. Using a sweep net, specimens were caught from 81 localities in 16 counties (Fig. 1). Localities were chosen depending on the ecosystem diversity and altitudinal variation. Altitudes, route tracks and directions of localities were measured with Magellan Sportrak Pro GPS (Thales Navigation, CA, USA).
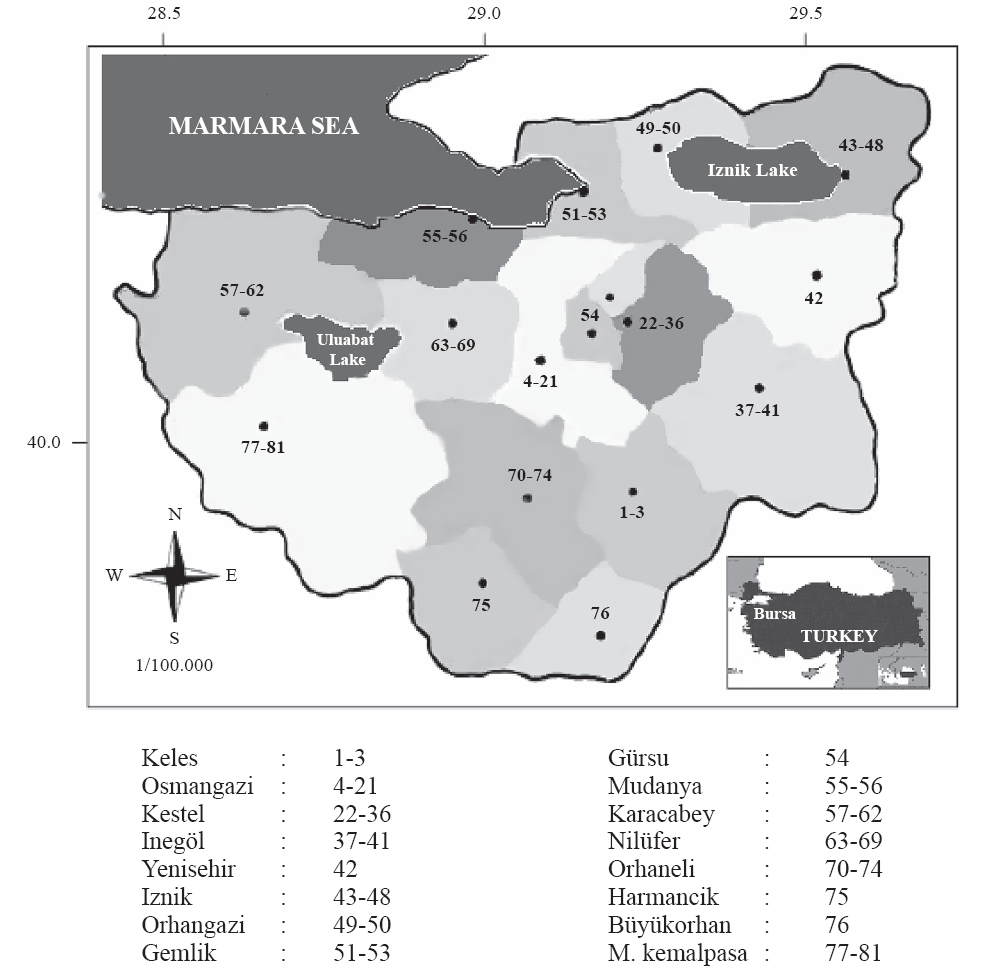
Map of Bursa, northwestern Turkey. Numbers within regions indicate the specific localities where the lycaenid species were collected.
The occurrence of each lycaenid species is determined by recording the total number of localities in which it occurs among all 81 localities visited. Species status was determined according to distribution in Bursa Province and presence in the localities. Species that were found in more than 20% of the examined localities were considered widespread. Subspecies’ names, if determined, were also reported.
Each lycaenid specimens was collected from its harboring plant. Living specimens were killed with ethyl acetate after being captured and they were brought back to the laboratory for identification. Adults were mounted according to standard entomological procedures and stored as vouchers in the Plant Protection Department collection at Uludag University. Species were identified by Prof. Bahattin Kovanci according to the descriptions of Carter (1982), Higgins et al. (1991), and Hesselbarth et al. (1995). The scientific names of lycaenid species used in the annotated checklist was mainly based on the nomenclature of De Prins (2004) and Kocak and Kemal (2006). Both subspecies names and names of species that were not present in Europe were adopted from Hesselbarth et al. (1995).
Results
A total of 3280 lycaenid adults, representing 47 species were caught. Polyommatus icarus and Lycaena phlaeas (L., 1761) were the most widespread species in northwestern Turkey, followed by Phebeius argus, Aricia agestis (Denis and Schiffermüller, 1775) and A. anteros (Freyer, 1838) (Table 1).
Occurrence, current status, altitudinal range, and flight period of lycaenid species in Bursa province, northwestern Turkey, between 1995 and 2006.
The total number of localities in which particular lycaenid species occurs among all 81 localities visited.
W = Widespread, L = local.
Adults of a few species such as Lycaena dispar (Haworth, 1802) emerged in April while most species appeared in May (Table 1). An increasing amount of adult flight activity during the summer months was observed. The highest number of lycaenid species was recorded in July with a total of 40 species per month. While the capture of some species continued until October, some were caught only in specific times. For example, adults of Lycaena alciphron (Rottemburg, 1775), Lycaena virgaureae (L., 1758) and Satyrium acaciae (Fabricius, 1787) were collected mainly in the summer whereas Callophrys rubi (L., 1758) adults occurred only in the spring.
Plebeius argus appeared to have two generations in north-western Turkey, the first from May to June and the second from July to August. Aricia agestis had two generations at high altitudes, the first from April to June and the second from July to August but a third generation may occur at low altitudes from August to October. A. anteros may also complete two or three generations from April to September depending on the altitude.
As far as habitat altitude is concerned, P. icarus had the greatest altitudinal range (15-2400 m) among all lycaenid species (Table 1). Widespread lycaenid species were generally distributed at altitudes higher than 1000 m. The species richness increased until 1500 m. Some species such as A. agestis, L. phlaeas, and Polyommatus icarus were eurytopic and found at altitudes of up to 2000, 2200, and 2400 m above sea level, respectively. Plebeius argus and Polyommatus bellargus (Rottemburg, 1775) varied in their altitudinal range between 300-2000 and 125-2200 m, respectively. Some lycaenids such as Agriades pyrenaica (Boisduval, 1840) and Aricia hyacinthus (Herrich-Schäffer, 1847) were restricted to the subalpine and alpine zones.
Detailed information on the distribution of local lycaenid species according to years and localities, and number of males and females caught at particular dates is presented in Table 2.
Temporal distribution of local lycaenid species according to years and localities, and number of males and females caught at particular dates in Bursa province, northwestern Turkey between 1995 and 2006.
Locality numbers are noted in parentheses.
Discussion
During the 12-year survey in the Bursa province of northwestern Turkey, a total of 47 lycaenid species were collected of which 12 were widespread and 35 were more restricted species in their distribution (Table 1). The latter include A. hyacinthus, Lycaena candens (Herrich-Schäffer, 1844), Pseudophilotes bavius (Eversmann, 1832), Polyommatus semiargus (Rottemburg, 1775), Polyommatus cornelia (Gerhard, 1851), Polyommatus ossmar olympicus (Gerhard, 1853), Polyommatus iphigenia (Herrich-Schäffer, 1847), Polyommatus menalcas (Freyer, 1837) and Tomares nogelii (Herrich-Schäffer, 1851). Aricia eumedon (Esper, 1780), Cupido osiris (Meigen, 1829), Kretania eurypilus (Freyer, 1851), Plebeius sephirus (Frivaldzky, 1835), and Polyommatus ripartii (Freyer, 1830) had not been seen since 1860s (Mann 1862, 1864). In addition, the last reported sightings of Chilades trochylus (Freyer, 1845), Polyommatus semiargus (Rottemburg, 1775), P. bellargus, S. acaciae and Satyrium spini (Denis and Schiffermüller, 1775) in the area came from the early 1900s (Hesselbarth et al. 1995). Until this study, all these species were regarded as extinct according to the IUCN Red List of Threatened Butterflies because it had been more than 50 years since they were recorded in wild (Wells et al. 1983). These results highlight the importance of detailed sampling efforts to clear up the risk status of some species.
A. hyacinthus is present only in western Anatolia and was previously recorded at an altitudinal range of 1150-2350 m from eight provinces of Turkey including Bursa (Hesselbarth et al. 1995). In this study, this species was only found at four localities at altitudes in the range of 1900-2400 m. The whole area containing the endemic larval food plant Erodium olympicum was covered with alpine grasslands used for sheep grazing. Likewise, the Ketenlik plateau of Sogukpinar village, which harbours the endemic P. ossmar olympicus, is under moderate to high grazing pressure. The larval host plant of this endemic lycaenid, Coronilla varia subsp. varia L., is grazed by sheep as well. Therefore, both A. hyacinthus and P. ossmar olympicus are believed to be under threat of extinction.
T. nogelii is found in Armenia, Lebanon, Romania, Syria, Palestine, Turkey, and Ukraine (Tuzov et al. 2000). It is now recorded in northwestern Turkey. The members of this species were caught only in the subalpine zone of Mt. Uludag, which contains dry, stony slopes and hot ravines. The predominant plants in the habitat were Astragalus angustifolius Lam. and Astragalus sibthorpianus Boiss. but no larvae were recovered from these plants. Since this species is monophagous and closely associated with its host plant, further studies can be directed toward potential Astralagus spp. hosts.
L. candens has a limited range which includes the area from the Balkan Peninsula and Turkey to Iran and the Caucasus (Martin and Pullin 2004). It was only caught in the mixed forest zone at an altitude of 1430 m. Unlike L. candens, L. dispar is widely distributed in Europe as far north as southern Finland, and across Asia but its populations are declining in many European countries. It is also a rare species in Turkey and classified in the lower risk, near-threatened status by the IUCN Red List of Threatened Butterflies (Wells et al. 1983). The presence of L. dispar in Turkey has recently been reported by Akbulut et al. (2003). We recorded L. dispar at five localities in small populations. These populations breed in open grassy vegetation where the endemic water dock species, Rumex olympicus Boiss., grows at a high density in sunny areas.
Pseudophilotes bavius is very localised in the Balkans including Greece, Romania, Yugoslavia and the former Yugoslav Republic of Macedonia (van Swaay and Warren 1999). The subspecies P. bavius egea is widespread in southern, central and parts of eastern Anatolia (Hesselbarth et al. 1995) but it is restricted locally in northwestern Anatolia based on our observations. Some adults were collected from Salvia argentea L. plants on which larvae of this species develop (Higgins and Riley 1970).
Polyommatus menalcas, also known as Turkish furry blue, is endemic to Turkey (Balint 1999). It is present in all regions, except the southeast. P. cornelia is another endemic species and found in the rocky slopes of the mountains. Similarly, P. iphigenia, which occurs only in Turkey and the Balkans, prefers open subalpine slopes. Unlike the previous Polyommatus species, P. semiargus extends over a large area from Morocco, Europe, Turkey, Middle East, temperate parts of Asia to Kazakhstan, North China, and Korea (Kudrna 2002). In northwestern Turkey, it is a local species occurring in the Bursa plain where it is vulnerable due to urbanization. Adults were captured between May and July in meadows where hostplants Trifolium repens L. and Trifolium pratense L. are present.
Aricia anteros anteros is an Irano-Turanian species distributed locally from Lebanon to North Iran, Caucasus, Turkey, and the Balkan Peninsula (Hesselbarth et al. 1995). Although widespread, this subspecies is restricted to a small territory (e.g. a mountain range). Both larvae and adults were seen on Geranium pusillum L.
Plebeius idas baldur was reported from the mountains of the Balkan Peninsula, Turkey and Caucasia. P. i. baldur adults are monophagous on the Chamaecytisus absinthioides (Janka) Kuzm., which is a Balkan endemic plant, in Bulgaria (Kolev 2005) and the geographical ranges of the plant and the lycaenid coincide significantly. This strict regional monophagy differs from the relatively wide polyphagy on the nominotypical Chamaecytisus absinthioides idas, which suggests that the latter may be a separate species as asserted by Kolev (2005). In this case, there must be another host plant, which remains to be identified in Turkey.
According to Pollard and Yates (1993), the presence of a food plant is essential for the presence of a breeding population of a species at any site. In fact, many widespread butterfly species have higher diversity of larval foodplants than the local and localised butterflies (Hodgson 1993). For example, the plant flora of the study area is rich in Fabaceae, Rhamnaceae and Rosaceae (Rubus spp., Potentilla sp.), which are the common hostplants of many lycaenids such as C. rubi. The main hostplants of S. ilicis larvae are Quercus spp. predominantly found in Mts. Uludag, Katirli and Samanli. In addition, Astragalus spp. is common in Mt. Uludag and provides a suitable habitat for P. daphnis and possibly for T. nogelii. Proactive efforts of growing butterfly food plants near urban areas are recommended to increase their chance of survival in Santiago de Cali, Colombia (Ramírez et al. 2007) but similar actions are also needed for mountain villages in northwestern Turkey to protect the natural habitat of lycaenid butterflies where their food plants thrive.
Both habitat altitude and connectivity are important variables because most lycaenids are small and sedentary species with a small breeding areas (Dennis 1992). Some lycaenids such as Aveexcrenota anna (Druce, 1907) can only be found at hilltopping sites at or near the mountain peak as observed in the Cerro San Antonio (2200 m) near the City of Cali, Colombia (Balint et al. 2006). Similarly, Agriades pyrenaica and Aricia hyacinthus were only captured in the hilltop areas of flat terrain covered with shrubs at or near Mt. Uludag's peak (2345 m). A. pyraenica is known to be strictly confined to its barren habitat containing the larval host plant Androsace villosa L. (Tolman and Lewington 1997). The local species are more or less confined to restricted areas, which provide the specific resources that they require. Individual populations may be large, but nevertheless, when a species becomes restricted in this way to a limited number of more or less isolated localities, it must be regarded as endangered (Pollard and Yates 1993). Since A. eumedon, P. cornelia and C. trochylus were found at only one or two localities, they are considered to be endangered. A. eumedon specimens were collected in June from subalpine grasslands near a public picnic area while C. trochlyus occurrred from June to September in a garrigue shrubland around fruit orchards. In contrast to these rare species, some local species expanded their habitat ranges and their status changed from local to widespread. These species include P. semiargus, P. amandus and P. bellargus.
Development and implementation of management plans for existing rare colonies of lycaenid butterflies such as A. hyacinthus and P. ossmar olympicus are necessary. Special importance should be given to the protection of essential habitats of lycaenids from adverse development within and outside mountainous areas. Suitable secure habitats should be established for self-sustaining viable populations of lycaenids feeding on endemic host plants as in the cases of L. dispar and L. candens larvae on Rumex species. Very little information on the host plants of lycaenids, which is needed for both conservation and ecological studies, from the world has been published. The occurrence of some lycaenid species may be limited by their habitat and altitude preferences so some lycaenid species may become extinct while others can expand their habitat ranges in response to the occurrence of environmental degradation.
Acknowledgments
We would like to thank Dr. Zdravko Kolev (University of Helsinki, Finland) and an anonymous reviewer from the Centre for Entomological Studies Ankara (CESA, Turkey) for the revision of an earlier version of the manuscript. The authors also thank Amy Groome for language editing and proofreading of the final draft.
