Abstract
Spiders are one of the most numerous groups of terrestrial predators and these are found in diverse environments such as agroecosystems and nearby areas. Research on spider diversity in agroecosystems is important for following changes in fauna brought about by management. This work evaluated spider richness, abundance, and species composition similarity between a rice agroecosystem and adjacent environments at different development stages of the crop. The study area was Estação Experimental do Arroz, in Cachoeirinha, Rio Grande do Sul state, south Brazil. A sweep net was used with 50 passes per transect as a sample; transects were set in a rice field, in a grassland (a former rice field), and in a nearby forest edge. We sampled 2,717 spiders; 78.7% were young and most adults were females (1.22:1). Representatives of 15 families were collected, the most dominant being Oxyopidae (n = 753) and Araneidae (n = 371). Representatives of 85 morphospecies (adults) were sampled, with the most abundant being Oxyopes salticus (n = 120) and Alpaida veniliae (n=62). Most species were found on the forest edge (62), followed by rice crop (38) and grassland (26). There were significant differences in spider species composition among environments and rice culture periods. Only eight morphospecies were common to all areas; forest edge had the higher number of exclusive species (42). Rice had a typical composition of species, probably due to the ecological selectivity of the spiders. Diversity was higher in the forest edge, suggesting this environment as an important refuge for the fauna living in areas with high anthropogenic disturbance as agroecosystems.
Introduction
Spiders are cosmopolitan terrestrial predators and although they are usually abundant (Turnbull 1973; Nyffeler and Benz 1987; Wise 1993), they are also little studied in environments like agroecosystems and nearby lands. In Brazil, there is only one study on the spider fauna in rice plantations (Corseuil et al. 1994b) even though this culture predominates in large extensions of southern Brazil. Other studies have been carried on in South and North America (Woods and Harrel 1976; Heiss 1984; Heiss and Meisch 1985; Oraze et al. 1988; Young and Edwards 1990; Bastidas et al. 1993, 1994a, b; Medina 1994), however none includes areas besides the agroecosystem itself.
Most of the works on the spider fauna of rice plantations were made in Asia (Pathak and Saha 1998; Ambalagan and Narayanasamy 1999; Bambaradeniya and Edirisinghe 2001; Lee and Kim 2001; Bambaradeniya et al. 2004; Patel et al. 2004; Vijaykumar 2004) but most of them were limited to faunal lists, in some cases investigating only the dominant species.
Research on spider diversity in agroecosystems is highly valuable; both to observe the effect of such predators have on herbivorous pests (Maloney et al. 2003) and to understand how profound changes on the environment affect spider colonisation (Öberg 2007). Thus, it is relevant to evaluate the spider fauna in the agroecosystem surroundings as done in a few cases for the rice culture (Murata 1995; Barrion 1999; Liu et al. 2003). Another potentially important factor is agroecosystem change along plant development, since environmental heterogeneity may be increased by plant growth. Rypstra et al. (1999) stated that spider assemblage density and diversity are intimately related to environmental structural complexity, which may be increasing as plants become larger and more complex.
According to Lewinsohn and Prado (2002) in Brazil ecosystems, created by human activity, there is a serious lack of faunal inventories for most taxa, which could be analysed regarding biodiversity loss or more specific changes. Thus, this work aims to evaluate spider diversity (species richness, abundance and species composition similarity) between the rice agroecosystem and adjacent areas and also along the growth period of the rice plants.
Material and Methods
Study area
Sampling was carried on in the Estação Experimental do Arroz (EEA, Rice Experimental Station) (50°58′21″W; 29°55'30'S) of Instituto Rio Grandense do Arroz (IRGA, Rio Grande Rice Institute), in the municipality of Cachoeirinha, Rio Grande do Sul state, southern Brazil (Fig. 1). The area is situated on the margins of Gravataí river, with an area of 172 ha (IRGA 2004). Cachoeirinha belongs to the Central Depression region of Rio Grande do Sul state (Teixeira et al. 1986), a region responsible for 14,53% of the total rice production in this state (IRGA 2004).
Sampling
Three environments were sampled: rice, grassland adjacent to rice and nearby forest edges. Sampling occurred from October 20, 2004 to June 06, 2005 with an average sampling interval of 11.3 days, covering different stages of the rice development. Overall there were 17 samples across time, the three first before the rice was sown, from the 4th to the 14th sample during rice development and the last three after rice was harvested. The rice culture was sampled from an area of 100 m X 50 m, divided in two subareas. There was a barrier separating the subareas, a small irrigation canal 2 m wide. The culture system was conventional and used the BR IRGA 410 cultivar. Only herbicides were applied (FACET 750 PM, 300 g per ha; STAM 480, 4 L per ha; Sirius 250 SC, 60 mL per ha). None other phytosanitary products were used to allow arthropods to invade and/or establish themselves in the areas. The distance between the grassland and the rice culture was approximately 10 m. The first had not been sown with rice for more than a year, with spontaneous vegetation arising, grasses and herbs characteristic of secondary grasslands around the area. The forest edges were approximately 80 m far from the sampled rice area, accompanying nearby rivers and brooks. These are remnants of the original vegetation, narrow strips of preserved forests (15 m tall on average) composed by a range of species, especially Myrtaceae and Fabaceae. Two transects were sampled on each area on each sampling date (the rice area was divided in two areas, thus there were four transects in rice areas); transects were positioned at least 20 m distant from each other.
Geographic location (left) and representative pictures (right) of the sampling areas, municipality of Cachoeirinha, Rio Grande do Sul state, southern Brazil.
Sampling method
Sampling involved sweeping nets with a 35 cm diameter opening. On each transect 50 pendular net swings were standardised as a sampling unit. All sampling occurred in the morning between 8:00 h and 11:00, to minimise spider migration to the lower vegetation stratum due to high temperatures at noon (Dumas et al. 1964). All identified material was deposited in the Museu de Ciências Naturais of Fundação Zoobotânica do Rio Grande do Sul (MCN/FZB, curator: Erica H. Buckup), where specimens exemplars were catalogued and included in the spider collection except for some Thomisidae tombed in Museu de Ciências e Tecnologia of Pontifícia Universidade Católica do Rio Grande do Sul (MCTP, curator: Arno A. Lise). The zoological nomenclature followed Platnick (2008). All spiders were determined to family and adults were separated in morphospecies when needed.
Data analysis
Overall spider family and species richness for each environment were compared using sample-based rarefaction with EstimateS 8.0 (Colwell 2005). To test for conjunct differences in sample species richness and abundance among environments and among sampling periods (before rice sowing, during rice development and after harvesting), a MANOVA test was employed (with SPSS®13.0). This would allow us to disentangle the influence that both species richness and abundance suffers from the factors tested. To illustrate patterns in arachnofauna species composition among environments and sampling periods a cluster analyses (UPGMA) with Jaccard (qualitative) and Morisita (quantitative) similarity indexes were used. To test for statistical differences in such patterns, we used two one-way ANOSIM (with Bonferroni correction for repeated tests), one for environment and one for sampling period. The latter analyses were done with PAST (Paleontological Statistics - 1.79, Hammer and Harper 2005). Parametric correlation tests were done to compare spider richness and abundance to average temperature and rainfall, available from the FEPAGRO (Fundação Estadual de Pesquisa Agropecuária) meteorological station. Spiders were classified in guilds according to their hunting strategy, following Uetz et al. (1999) and Höfer and Brescovit (2001). Guild proportions for each environment were tested with a heterogeneity G-test.
Results and Discussion
A total of 2717 spiders were obtained, distributed among 15 families; 2138 were young individuals and a minority adults (579); among the latter, females (318) were more numerous than males (261). Overall 85 morphospecies were identified among the adults. The first two sections below analyse spider abundance based on overall numbers, in other words, including adults and young.
Spider abundance
On average there were 19.98 (±5.14 s.e.) spiders per transect (for grassland: 30.86±4.58; for rice: 13.5± 2.26; for forest edge: 22.09 ± 2.38), with only two samples without spiders. Spider abundance differed among environments (MANOVA, Pillai's Trace, F=23.25; P<0.001) as well as among periods (F = 15.18; P = 0.001, Fig. 2). All three environments differed for abundance: grassland had more spiders, followed by forest edge and finally the rice culture.
Habitat complexity is directly related to spider abundance (Rypstra 1986; Greenstone 1984; Döbel et al. 1990; Gunnarsson 1990) and many spiders live strictly in specific environments related to the kind of vegetation present (Foelix 1982). Differences among the three environments are probably connected to such differing physiognomy and its changes. The rice culture is relatively simple, with few substrates for web building and hunting, although its complexity increases with time as the plants grow. The grassland has a richer structure (higher plant species richness). The forest edge may had more spiders (and spider species, below), but the smaller number of individuals compared to grasslands may have to do indirectly with plant productivity (forest edges are shadowed). Furthermore, forest edges sampled with sweeping nets are actually grasslands on which a "rain" of spiders from shrubs and trees falls, so only part of the forest fauna is expected to be actually sampled.
In the forest edge anthropogenic factors such as management, cultivation, soil preparation, and irrigation are reduced and the abundance curve is relatively stable. In the grassland, subject to intermediate anthropisation due to vehicle transit and eventual mowing, the curve fluctuates (Fig. 2). In the rice culture, abundance increases up to the end, when plants reach their full size; after the harvest, abundance decreases rapidly. Also, on both occasions where no spiders were found in a transect this happened in rice areas shortly after sowing. Bambaradeniya and Edirisinghe (2001) demonstrated that abundance along rice development can be very variable; Corseuil et al. (1994b) recorded lower abundances both at the beginning and the end of their sampling period. Ambalagan and Narayanasamy (1999) argue that both spider abundance and richness is linked to the different stages of rice growth. Such pattern of dependence on plant substrate is thus probably universal (Lee and Kim 2001).
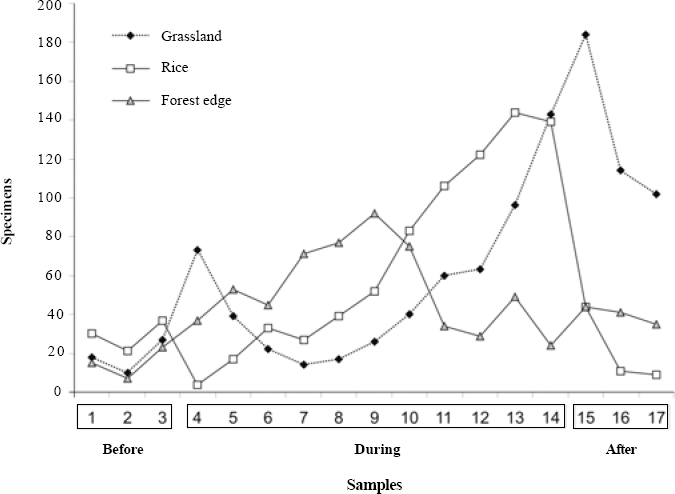
Spider abundance in grassland, rice and forest edge environments for different periods of the rice culture (before sowing, during development and after harvest) between 20-Oct-2004 and 06-June-2005 in Cachoeirinha, Rio Grande do Sul state, southern Brazil.
Spider families
Of the 15 spider families recorded (Fig. 3), Oxyopidae, Araneidae, and Tetragnathidae, summed up more than half of all spiders (Table 1). Oxyopidae predominates in the grassland and forest edge. In the rice areas Araneidae was dominant. The number of families was nearly the same in the three environments, but family richness was statistically different according to the sample-based rarefaction between grassland (12±1.32; 95% CI) families and the forest edge (14° 1.87; 95% CI) and rice area (14±0.00; 95% CI).
Linyphiidae (17 spp.), Theridiidae (16 spp.) and Salticidae (13 spp.) had the largest number of morphospecies; however, the first is only the seventh in abundance. Linyphiidae occurred in all environments, but mainly in rice. This family appears to be adapted to frequently perturbed areas, especially agricultural lands (Thorbek et al. 2004; Öberg 2007). In studies of South American agroecosystems, Linyphiidae appears poor in species compared to other families (Aguilar 1989; Corseuil et al. 1994a); however in Europe it is represented by many morphospecies (Pommeresche 2002, 2004; Pekár and Kocoukek 2004). 15 of the 16 morphospecies of Theridiidae were found in the forest edge. Some papers on spiders in rice captured with sweeping nets do not even record this family (Oraze et al., 1988; Corseuil et al., 1994b). Bambaradeniya and Edirisinghe (2001), record many Theridiidae morphospecies in rice cultures for Sri Lanka and Sebastian et al. (2005) also surveyed many species of this family in India.
Spider species richness
Of the adult individuals, 85 morphospecies were determined (Table 2); 52 were exclusive to at least one of the environments. The most abundant morphospecies were Oxyopes salticus Hentz, 1845, Alpaida veniliae (Keyserling, 1865), Misumenops pallidus (Keyserling, 1880), Tetragnatha aff. jaculator, Cheiracanthium inclusum (Hentz, 1847) and Ashtabula sp. 1; these species totalise 51.46% of the adult spiders. Most morphospecies (68) did not reach 1% of the total.
The forest edge had significantly more species (sample-based rarefaction, S = 62±12.42; 95% CI), than the rice culture (38 ± 9.10; 95% CI) and grassland (26 ± 9.38; 95% CI). The period during rice development also has a higher total richness than before and after, discounting the differing sampling effort (Fig. 4). As predicted, the less disturbed and more heterogeneous environment had higher spider richness than the others.
Comparing environments for average species richness show significant differences again (MANOVA Pillai's Trace, F = 7.45; P = 0.001), but between grassland and rice; among sampling periods there are also significant differences (F = 6.34; P=0.002), between periods before and after the rice was sown. Each environment sampled here presents a different pattern of species richness across time, perhaps because of this differential resource availability and temporal dynamics, both in terms of quantity and quality of preys, and also of substrate or refuge for spiders.
Whitmore et al. (2002) state that increasing disturbance levels lead to decreasing spider richness. This is true for the sample-rarefied richness, in which forest edge had higher richness than the disturbed grassland and rice culture, but it is not so for average species richness, in which the intermediate disturbance environment holds more species per sample. The general answer is that more species inhabit the forest edge overall suggesting a temporal partition of niches, but species that inhabit the grassland appear more frequently; although almost always the same species are found, suggesting that constant disturbance keeps the environment at the same point in the ecological succession.
Comparison among guilds
There were significant differences among areas for spider feeding guild proportions (G = 218.4; d.f. = 6; P<0.001). Overall ambush hunters were predominant (49.46%) (Fig. 5). For the grassland, they were better represented than in the other environments, due to the large number of Oxyopidae found. Orbicular web builders need open spaces to construct their webs and capture flying insects (Blackledge et al. 2003) and thus were expected to be more abundant in rice than in grassland. Muma and Muma (1949) already suggest the absence of support structures for webs (shrubs and trees) are responsible for the rarity of web-building spiders in grasslands. Running hunters are rarer in the grassland, probably due to low numbers of Anyphaenidae and Miturgidae. In grassland 70% of the spiders were hunters of either ambusher or running kind (Fig. 5), and as Jocqué (1984) mentions, high temperatures are detrimental to web building spiders, there is a suggestion that abiotic factors such as insolation can also affect predatory strategies apart from absence of web support structures.
Number of spider individuals per family and functional group (guilds) found in grassland, rice and forest edge, in Cachoeirinha, Rio Grande do Sul state, southern Brazil. ORB: Orb weavers; SPW: Space web in sheets; HAM: Hunting - Ambushers; HRU: Hunting - Runners.
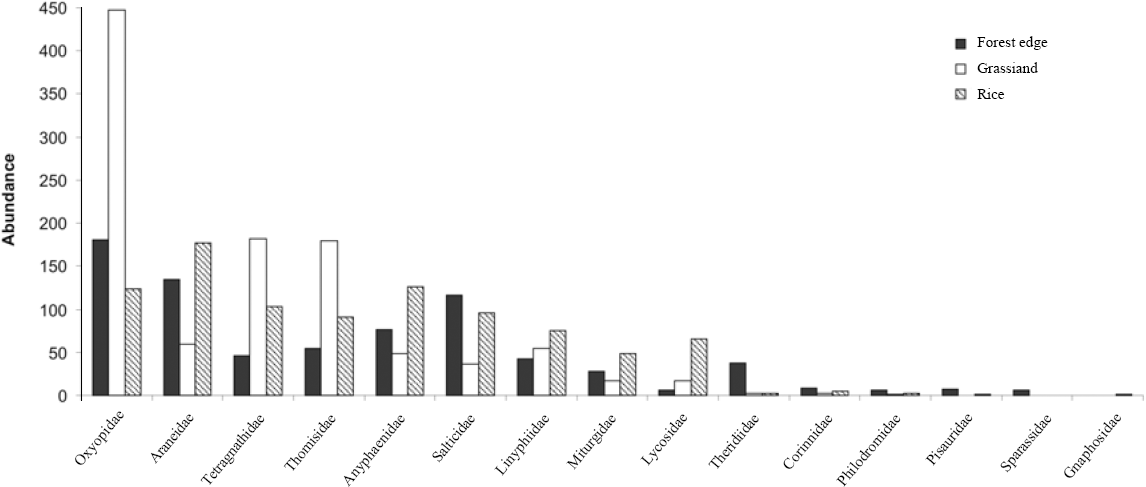
Abundance for the spider families in grassland, rice and forest edge in Cachoeirinha, Rio Grande do Sul state, southern Brazil.
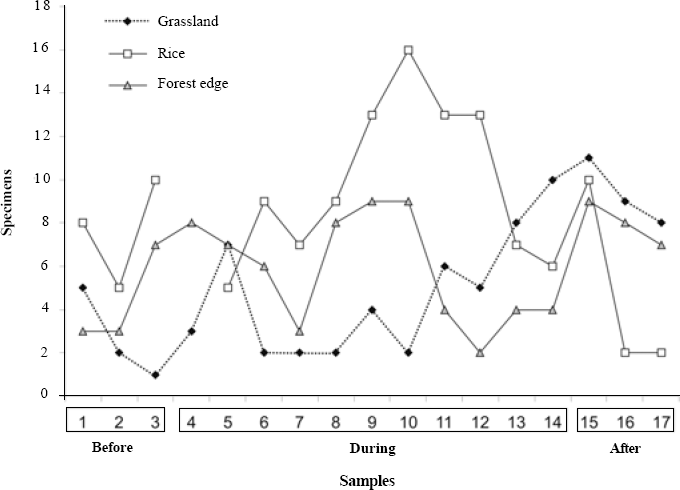
Adult spider species richness in the grassland, rice and forest edge environments, in different periods of the rice culture (before sowing, during development and after harvest) in Cachoeirinha, Rio Grande do Sul state, southern Brazil.
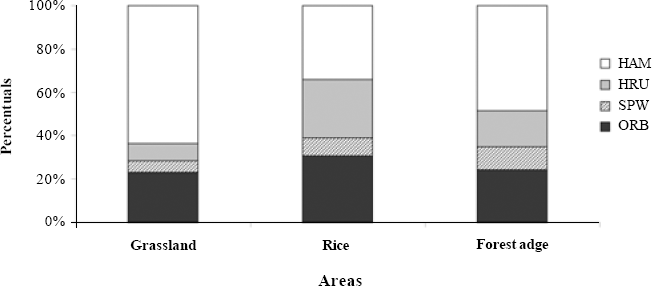
Spiders feeding guilds for the grassland, rice and forest edge environments in Cachoeirinha, Rio Grande do Sul state, southern Brazil ORB: Orb weavers; SPW: Space web sheet builders; HAM: Hunting – Ambushers; HRU: Hunting - Runners.
In the rice culture ambushers were also more common; however, orbicular web builders had a higher proportion, mainly due to Araneidae and Tetragnathidae. These were also the most abundant for Sebastian et al. (2005) and Corseuil et al. (1994b) in rice. These families could be rather common in rice presumably because rice plants are good spots for web building and also given the high number of prey occurring on agroecosystems.
Bambaradeniya and Edirisinghe (2001) observed orbicular web builders feeding on insect pests of rice in Sri Lanka. The most abundant guild in that case was the irregular web builders, mainly due to Theridiidae and Linyphiidae. In Asia Theridiidae are common in rice whilst here it has been found exclusively for the forest edge.
In the forest edge, ambushers were proportionally more abundant than in rice. Differently from the grassland, irregular web builders were recorded more commonly, due to the quantity of Theridiidae found, more than 90% of the individuals. They may be better adapted to areas with lower disturbance, possibly due to web format and construction site, being observed between short shrubs in the forest edge. According to Whitmore et al. (2002) there are indications that vegetation structure influences spider diversity.
Abiotic factors influencing abundance and richness
According to Rosenzweig (1995), the main parameters compared here, richness and abundance can be affected by diverse factors such as: seasonality, spatial heterogeneity, competition, predation, habitat type, environmental stability and productivity. Correlation between abiotic data (temperature and rainfall) and either abundance or richness reveals significance only between temperature and abundance for forest edge (rβ = 0.31; P=0.02). During the sampling period (2004-2005), Rio Grande do Sul state passed through an intense dry period with variable rains, which could have influenced the results. Vijaykumar (2004) also did not found significance for correlations between temperature and humidity for rice spiders in India.
Faunal similarity
Clustering transects using the Jaccard qualitative index (presence-absence, Fig. 6), reveals forest edge as a significantly separate group (R=0.218,P=0.014), although with low similarity within (19.67%). For Morisita's quantitative index (Fig. 7) rice sites clustered separately from grassland and forest edge, but significantly different assemblages occur for each environment (R=0.302,P=0.001, all pairwise comparisons P<0.05). This occurs especially due to Alpaida veniliae having a disparately high abundance in rice being absent in the forest edge and rare in the grassland (SIMPER analysis: 25.42%); and both grassland and forest edge being strongly influenced by the dominant Oxyopes salticus (SIMPER analysis: 17.19%). This pattern conforms to expectation, forest edge having the most distinct arachnofauna and rice having also a typical fauna, possibly due to spider environmental selectivity.
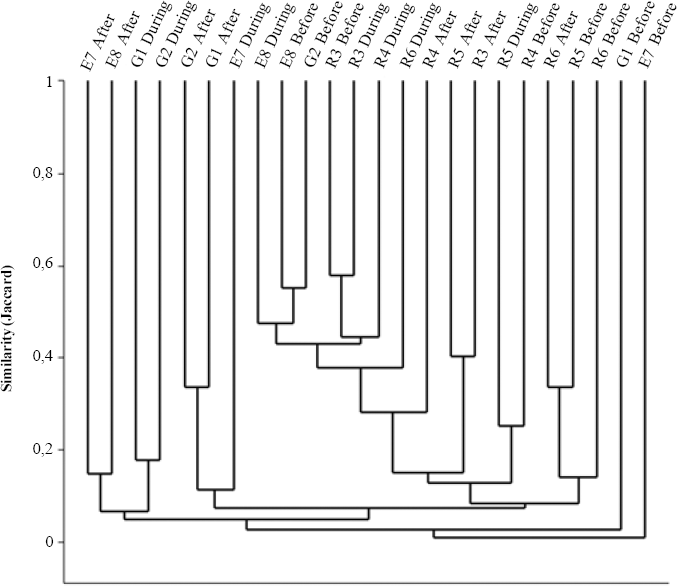
Cluster analysis (UPGMA) using the Jaccard index grouping grassland, rice and forest edge environments and rice period (before rice sowing, during rice development and after rice harvest) for spider morphospecies in Cachoeirinha, Rio Grande do Sul state, southern Brazil (G, grassland; R, rice; E, forest edge).
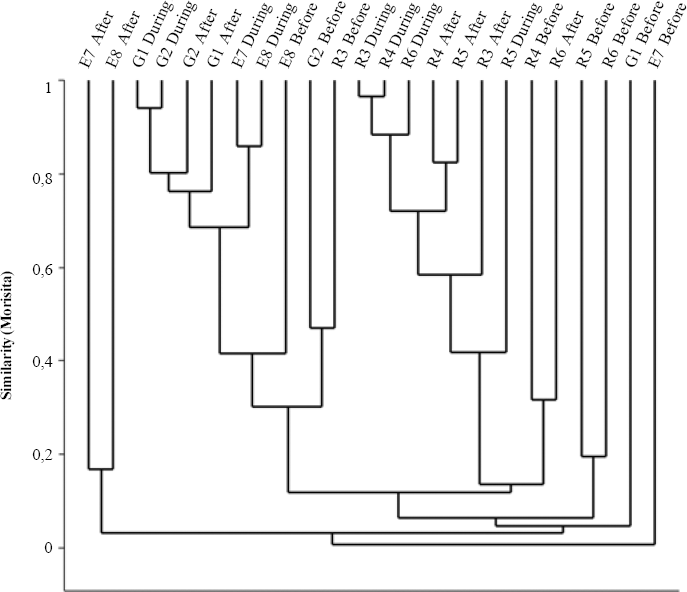
Cluster analysis (UPGMA) using the Morisita index grouping of grassland, rice and forest edge environments and rice period (before rice sowing, during rice development and after rice harvest) for spider morphospecies in Cachoeirinha, Rio Grande do Sul state, southern Brazil (G, grassland; R, rice; E, forest edge).
The three rice culture stages (Figs. 6 and 7) also had significantly different spider faunas for both Jaccard (R=0.263, P<0.0001) and Morisita (R=0.179, P=0.005) indexes. Jaccard clusters periods after and during for grassland and rice, whilst forest edge sampling units has a spread out distribution; each environment is significantly distinct from the others. For Morisita, periods during and after sown do not differ in species composition (P = 0.87), only the period before does.
List of spider morphospecies and number of individuals (adults) for grassland, rice and forest edge environments in Cachoeirinha, Rio Grande do Sul state, southern Brazil.
Nearby areas as refuges
Higher spider diversity in natural or better preserved environments compared to nearby agroecosystems is also found for wheat, maize, other cereals and soybean (Nyffeler and Benz 1987; Liljesthröm et al. 2002). Areas on the margins of cultured lands, having vegetation not only differing from that of the agroecosystems, and sometimes being more complex and usually less managed, could work as refuges in times of disturbance, forcing spiders to disperse. This question needs to be addressed in detail to better evaluate the prospect of using spiders as biological control agents in agroecosystems.
The importance of nearby areas for agroecosystems is recognised and demonstrated here, a high species richness recorded for the forest edge, and similar faunal composition between rice and a nearby disturbed grassland area. Spiders can move among close by environments, invading areas changed by human management, or leaving them when disturbance is too high. Unmanaged or little managed areas can serve as refuges for spiders, predators potentially interesting for biological control programs.
Suggestions for future studies could include longer periods, both for agroecosystems and adjacent areas, but also using varied methods so as to cover a wider portion of the fauna present. More detailed observations such as capturing parachuting spiders and trapping spiders running into the cultured area are also possible suggestions. Sampling at increasing distances from the cultured land could also provide clues to the spatial scale of the spider dispersing process.
Footnotes
Acknowledgements
To CNPq and CAPES for scholarships to E. N. L. Rodrigues and M. Mendonça Jr., respectively. To Erica H. Buckup for access to MCN spider material and bibliography. To Jaime Vargas de Oliveira (IRGA) for help in the EEA. To Maria Aparecida de L. Marques (MCN) for helping determining Theridiidae and Arno A. Lise (PUCRS) for help with Thomisidae. We are grateful to two anonymous reviewers for important suggestions.
