Abstract
Anastrepha manizaliensis Norrbom & Korytkowski, new species, is described from Colombia. It breeds in fruit of Juglans neotropica Diels (Juglandaceae), commonly known in Colombia as “cedro negro”. The new species was previously confused with Mexican fruit fly, Anastrepha ludens (Loew), a major pest of citrus and mango, leading to quarantine problems, but true A. ludens does not occur in Colombia.
Introduction
Anastrepha Schiner is the most diverse genus of Tephritidae in the American tropics and subtropics, with more than 200 species (Norrbom et al. 1999 a, b). It is also the most economically important fruit fly genus in this region, including a number of major fruit pests. Despite its importance, however, many species remain undescribed and the native host plant relationships are poorly known. In this paper, we describe a new species from Colombia that was previously confused with A. ludens (Loew), the Mexican fruit fly, a major pest of citrus, mango, and other crops from the Rio Grande Valley to Panama.
Materials and Methods
Morphological terminology follows White et al. (1999). Examined specimens are deposited in the Colección Taxonómica Nacional de Insectos “Luis María Murillo”, Tibaitatá (CTNI) and the National Museum of Natural History, Smithsonian Institution (USNM).
Terminalia measurements and ratios in Anastrepha manizaliensis and related species
measurements in mm
Anastrepha manizaliensis Norrbom & Korytkowski, new species
Figs. 1-2
Recognition
This species belongs to the fraterculus species group and is most similar to A. ludens (Loew), A. distincta Greene, A. minensis Lima, and especially A. schultzi Blanchard and A. inca Stone, which also have relatively long female terminalia (see Table 1). In the key of Steyskal (1977), most specimens will key to A. ludens (p. 10, 30). Anastrepha manizaliensis differs from A. ludens in having shorter terminalia (compare oviscape length, oviscape/mesonotum ratio, and aculeus length in Table 1) but a longer and broader aculeus tip. It differs from A. distincta by its longer and broader aculeus tip and frequently in the markings of the subscutellum and mediotergite (in A. manizaliensis the subscutellum is always dark brown laterally, but the mediotergite is usually not, whereas in most A. distincta only the mediotergite is brown laterally). Anastrepha manizaliensis also differs from A. minensis in having the subscutellum brown laterally, and by its fewer hook-like dorsobasal denticles on the eversible membrane (more than 100 in A. minensis). It differs from A. schultzi and A. inca, in which the subscutellum and mediotergite are both always dark brown laterally, in having the aculeus tip more extensively serrate. Additionally, in A. manizaliensis the serrations are distinct, whereas in A. schultzi they are weak if present.
Description
Mostly yellow to orange. Setae dark brown to black. Body length, female 8-11 mm, male 6,0-7,5 mm. Head: Yellow to orange except ocellar tubercle brown. Facial carina, in profile, concave. 4-6 frontal setae. 2 orbital setae (in all 37 specimens). Ocellar seta weak, short to minute, less than 2 times as long as ocellar tubercle. Antenna extended 0,72-0,87 distance to ventral facial margin. Arista short pubescent. Thorax (Fig. 1): Mostly orange, postpronotal lobe, scutellum (except sometimes extreme base of dorsum, extending less than halfway to level of basal scutellar seta), scutal vittae and dorsal margin of anepisternum white; medial scutal vitta sometimes less differentiated that other white areas, very slender, slightly broadened and rounded posteriorly, but not extended laterally beyond acrostichal seta. Scuto-scutellar suture medially with brown mark, usually dark, and always much broader than long. Subscutellum with dark brown mark on lateral 1/5-1/3 (Figs. 2B-C). Mediotergite usually entirely orange, rarely (1 male and 1 female of 37 specimens) with lateral mark from subscutellum extended along dorsal half of lateral margin. Mesonotum 2,57-3,65 mm long, entirely microtrichose. Scutal setulae yellow medially, brown laterally. Chaetotaxy as usual for genus, katepisternal seta moderately developed, usually longer but weaker than postocellar seta, pale to moderate brown. Wing (Fig. 2A): Length 6,3-8,1 mm, width 2,4-3,3 mm, ratio 2,37-2,63. Cell c 1,20-1,49 times as long as pterostigma. Apex of vein R1 at 0,52-0,56 wing length. Vein R2+3 nearly straight. Vein M moderately curved apically; cell r4+5 0,83-1,05 times as wide at apex as at level of DM-Cu. Crossvein R-M at 0,67-0,71 length of cell dm. Distal lobe of cell bcu moderately long, bcu 1,42-1,66 times as long as its anterior margin. Pattern mostly orange to yellow brown; most margins of bands, most of pterostigma, distal part of S-band, and posterior parts of V-band brown. C- and S- bands usually narrowly connected (in 32 of 39 specimens) or narrowly separated along vein R4+5 (if bands separated, hyaline area distinctly narrowed). S-band with distal section relatively narrow, at apex of vein R2+3 0,53-0,50 times width of cell r2+3; not extended to apex of vein M. V-band separated from S-band; usually complete, distal arm rarely narrowly isolated (5 of 37 specimens); proximal arm extended basally along posterior wing margin almost to vein A1+Cu2 but not connected to base of S-band. Microtrichose except cell bc, much of cell c (except anterior margin), extreme base and usually part(s) of subapical hyaline area of br, most of bm, part of basal hyaline area in dm, anterior and posterior areas in bcu, most of alula, and usually small basal area in cu1 and small anterobasal area in a1+cu2. Abdomen: Tergites yellow to orange with posterior margins paler. Male terminalia: Similar to A. ludens. Lateral surstylus in posterior view elongate triangular; medial margin straight to slightly concave; apex often truncate. Proctiger with lateral fold separating sclerotized areas. Phallus 4,5-4,7 mm long; 1,30-1,55 times as long as mesonotum. Glans 0,6 mm long. Female terminalia: Oviscape (Fig. 1) 2,65-3,40 mm long, 0,86-1,05 times as long as mesonotum, entirely yellow to orange; spiracle at basal 0,29-0,35. Eversible membrane (Fig. 2D) with 40-60 long, slender, hook-like dorsobasal scales in triangular pattern. Aculeus (Fig. 2E) 2,7-3,3 mm long; base 0,27-0,31 mm wide; shaft 0,13-0,16 mm wide at midlength; tip (Fig. 2F) 0,50-0,66 mm long, 0,16-0,19 mm wide, 2,94-3,56 times as long as wide, distal 0,39-0,49 finely but distinctly serrate; tip gradually tapered except for slight expansion at base of serrate part. Spermathecae (Fig. 2G) elongate teardrop-shaped.

Anastrepha manizaliensis, dorsal habitus, female.
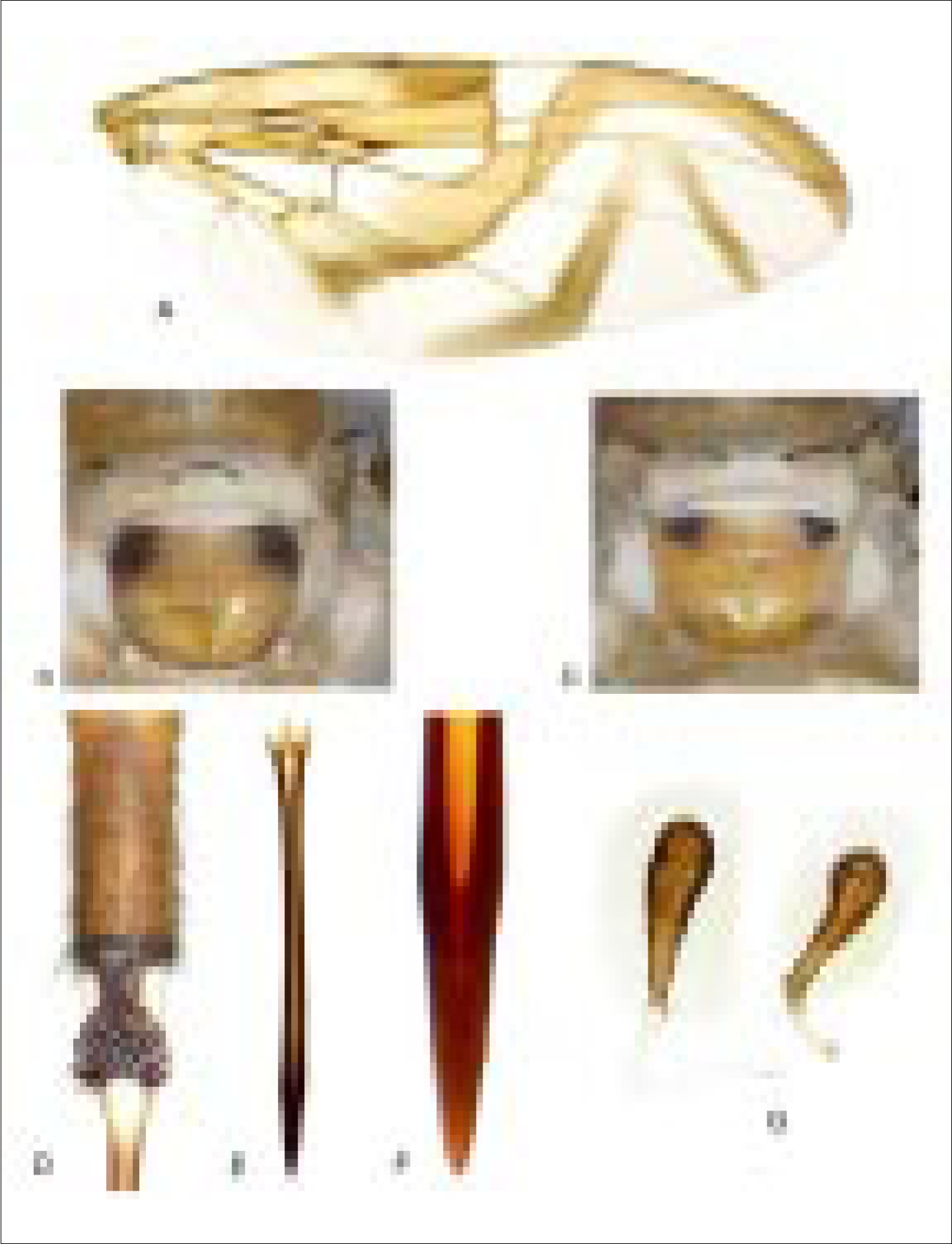
Anastrepha manizaliensis. A, wing; B-C, subscutellum and mediotergite; D, eversible membrane; E, aculeus; F, aculeus tip; G, spermathecae (2 of 3 shown).
Type data
Holotype female (National Museum of Natural History, Washington, DC (USNM); USNM00213557), COLOMBIA: Caldas: Manizales [5°3′17″N 75°29′37″W, 2.150 m.], emerged 1-Oct-2003 reared ex fruit of cedro negro, Juglans neotropica Diels, ICA. Paratypes: COLOMBIA: Caldas: La Merced, trampa McPhail, Mar-2002, E. Arévalo EA 62, 1f (USNM; USNM00213434); Manizales, emerged 1-Oct-2003 reared ex fruit of cedro negro, Juglans neotropica Diels, ICA, 7m6f (USNM, CTNI; USNM00213554-56, USNM00213558-67); Manizales, trapped Oct-2003, 3m5f (USNM; USNM00213697-704), 2m2f (CTNI); Manizales, McPhail traps, Oct-2003, J. Rodríguez, 5m6f (USNM, CTNI; USNM00213542-52). Cundinamarca: Fusagasugá [4°20′38″N 74°22′4″W, 1.730 m.], 2000, 1f (USNM USNM00213696), 2f (CTNI).
Other specimens examined
COLOMBIA: Cundinamarca: Cachipay [4°42′8″N 74°29′24″W], Finca Mesitas de Santa Inés, trampa en cítricos, Apr-1975, L. Núñez, 1f (CTNI); same, SV577, 1f (USNM; USNM00213641); same, SV571, 1f (USNM; USNM00213461); same, 11-May-1975, SV576, 1f (USNM; USNM00213460).
Biology
Some of the type specimens were reared from fruit of Juglans neotropica Diels (Juglandaceae), commonly known in Colombia as “cedro negro”.
Distribution
This species is known only from the Andean region of Colombia. The collection localities are from middle elevations, up to 2.150 m altitude, similar to the altitudinal range of the only known host plant, Juglans neotropica (Bartholomäus et al. 1990). Because the distribution of J. neotropica extends to Ecuador, Peru, and western Venezuela, it is possible that A. manizaliensis also occurs in those countries.
Comments
The examined specimens from Cachipay in the USNM were tentatively identified as Anastrepha ludens by George Steyskal in 1977, leading to the report of that species occurring in Colombia (Núñez Bueno 1981). This has caused occasional problems for the export of certain fruits, particularly citrus, from the country. Unfortunately, the terminalia of the three female specimens that were retained by Steyskal are missing their terminalia except for the oviscape on one specimen. This prevented the verification of their identity, but one of us (F. Gonzalez) was able to find a female with intact terminalia in the CTNI collection that came from the same series that was sent to Steyskal. Study of that specimen as well as external characters in all of the Cachipay series led us to conclude that those specimens are conspecific with the types of A. manizaliensis, and are not A. ludens. Murillo (1931) also reported specimens from Colombia as possibly A. ludens, but the whereabouts of those specimens are unknown. They may also have been A. manizaliensis. No verified records of A. ludens from Colombia are known to us, and the currently known southern limit of the range of that species is western Panama.
Etymology
The name of this species is an adjective derived from the type locality.
Footnotes
Acknowledgments
We thank Lucrecia Rodriguez, who produced or improved many of the illustrations, Aristóbulo López-Ávila, curator of the Colección Taxonómica Nacional de Insectos “Luis María Murillo”, Tibaitatá (CTNI), and Jorge Iván Rodríguez (Programa de Moscas de la Fruta, Caldas, ICA) whose team collected much of the type series. We are also grateful to Ligia Nuñez (ICA, retired), Manuel Mejia Lago (formerly of APHIS), and John Shaw, U. S. Aggregate (USDA-APHIS) for their support of this work. Roberto Zucchi, Steve Lingafelter, and F. Christian Thompson kindly reviewed the manuscript.
