Abstract
Background
Nonallergic rhinitis with eosinophilic syndrome (NARES) is persistent, without atopy, but with ≥25% nasal eosinophilia. Hypereosinophilia seems to contribute to nasal mucosa dysfunction.
Objectives
This analytical case-control study aimed at assessing the presence and severity of nonspecific nasal hyperactivity and at finding out whether eosinophilia may be correlated with the respiratory and mucociliary clearance functions.
Materials
The symptom score was assessed in 38 patients and 15 controls whose nasal smear was also tested for eosinophils and mucociliary transport (MCT). Nonspecific nasal provocation tests (NSNPT) with histamine were also carried out, and total nasal resistance (TNR) was determined.
Results
The symptom score of NARES after NSNPT were not significantly different from the control group, and there was poor or no correlation among the single symptoms and the differences studied for every nasal reactivity class. This correlation improved when using the composite symptom score. The most severe eosinophilia was observed in high reactivity groups, and it was correlated with an increase in TNR. MCT worsened as eosinophilia and nasal reactivity increased. Unlike controls, a significant correlation was observed between the increase in MCT and TNR.
Conclusions
In NARES, nonspecific nasal hyperreactivity is the result of epithelial damage produced by eosinophilic inflammation, which causes MCT slow down, an increase in TNR, and nasal reactivity classes, with possible impact on classification, prognosis, and treatment control.
Materials and Methods
Study Design
Patients with NARES and healthy adult controls were screened at the Rhinology and Immuno-allergy Unit of the Sense Organs Department, Otolaryngology Section of “Umberto I” General Hospital between March 2010 and June 2013. Six weeks after screening, at the first checkup, a prick test was performed, the symptom score was assessed as well as nasal cytology and mucociliary transport (MCT). A week later, at the second checkup, an NSNPT with histamine and rhinomanometric evaluation of the response was carried out, and the evoked symptom score was recorded. The study's protocol was approved by the ethics committee, and all the people enrolled were asked to sign an informed consent form. All aspects of the study complied with the provisions of the Declaration of Helsinki.
Subjects Enrolled
Forty patients with NARES (18 men, 22 women; mean age, 50.7 years) and 15 healthy volunteers (7 men, 8 women; mean age, 39.4 years) were selected and enrolled in this study. The clinical history of the patients with NARES showed persistent symptoms, including sneezing, aqueous rhinorrhea, nasal and palatal pruritus, and nasal congestion that dated back to 2–5 years earlier, with an on-again-off-again pattern; the controls, instead, had a negative clinical history. The two groups underwent a skin-prick test, a radioallergosorbent test (RAST) to detect specific Immunoglobulin E antibodies to the most common allergens in the serum, and, if necessary, a specific nasal provocation test (SNPT) to ascertain the absence of any atopy. None of them were on any medical treatment with drugs that may cause nasal hypo- or hyperreactivity (which included tricyclic antidepressants, antihypertensive drugs such as clonidine and reserpine, decongestants, etc.) or with nonsteroidal anti-inflammatory drugs. Antiallergenic drugs were discontinued for a 7-week period before the second checkup. Pregnancy and breast-feeding were exclusion criteria, as were concomitant diseases. They all showed an unmistakably negative clinical history of asthma. Rigid fiberoptic nasal endoscopy (LUT, Denzlingen, Germany) was carried out on all the patients and did not show any nasal polyps, any nasal septum deviation, or any other significant alteration. None of the patients enrolled had infectious rhinitis in the 2 weeks before the study. Exclusion criteria also included smoking and exposure to toxic powders or substances. Sinus radiography, which was performed as an ordinary diagnostic procedure, excluded any radiologic and clinical involvement of the paranasal sinuses. Hence, the patients with NARES were still sthenic, that is, in a phase with full nasal symptoms, with frequent “flares,” but showing no anosmia or sinusitis.
Skin-Prick Test
The allergy status was assessed in the two groups based on their sensitization to the most common categories of allergens by means of a skin-prick test. The test was considered as positive for sensitization when it revealed a ≥3 mm-diameter wheal. The allergens panel (Stallergenes, Milan, Italy) included Dermatophagoides pteronyssinus, Parietaria officinalis, Alternaria tenuis, Aspergillus fumigatus, graminacee mix, dog, cat, composite mix, olea, cypress, and hazel.
Nasal Cytology Test and Staining
For all the patients, nasal smear was collected by scraping the medial surface of the inferior turbinate. The material collected was spread on a glass slide and fixed with cytologic fixative. The material was then stained with May-Grünwald-Giemsa stain (C. Erba, Milan, Italy); then it was mounted, cover slipped, and observed under an optical microscope (BM100FL, Seben, Berlin, Germany). The eosinophil count was performed on 10 immersion microscope fields (×1000) and was expressed as a percentage over the cell count total. NARES was diagnosed when there were ≥25% eosinophils.
Mucociliary Transport
A small amount of Edicol Orange (Edex, Newcastle, Australia) powder was spread on the anterior section of the inferior turbinate by means of a swab. Said powder was anteroposteriorly transported by the cilia of the nasal epithelium toward the nasopharynx, and the time required to cover such distance was the mucociliary clearance time. Edicol Orange left reddish traces along the way, therefore, it was possible to highlight with the staining agent collected by means of a swab positioned behind the soft palate. With this technique, the mucociliary clearance time for healthy patients was 15–18 minutes.
NSNPT with Histamine
Endonasal administration of a solution that contained 2.5 mg of histamine phosphate per milliliter (Sigma Co, St. Louis, MO) was performed in both nasal cavities of each subject, with a predosed spray in a volume of 0.1 mL to give a total dose of 0.5 mg. 9 This dose was the minimum provocation histamine quantity that allowed a 100% increase in total nasal resistance (TNR) (expressed as Pa × sec × cm−3) and determined, based on regression coefficients, the NSNPT response magnitude according to four nasal reactivity classes, including low (TNR between 0.51 and 1.1 Pa × sec × cm−3), medium (TNR between 1.11 and 2.5 Pa × sec × cm−3), high (TNR between 2.51 and 4.7 Pa × sec × cm−3), and very high (>TNR 4.7 Pa × sec × cm−3). TNR values were matched with histamine doses by using the following exponential equation: ln(TNR) = a + bH, in which “a” and “b” were generic regression coefficients, TNR was the nasal resistance response, and “H” was the independent variable, that is, the quantity of histamine inhaled (0.2–0.8 mg). Regression coefficient “b” designed curves that represented the borders of the areas that encompassed the nasal reactivity classes. 9 Active anterior rhinomanometry using Version 5 of the Jaeger Master-Scope (Carefusion, Yorba Linda, California) was performed 5 minutes before and 5 minutes after the NSNPT with histamine, and TNR was determined at a 150 Pa based on the following calculation: TNR = (right NR × left NR)/(right NR × left NR). A hundred TNR increase was chosen as the basic value because such an increase cannot be due to the variations of a normal nasal cycle because other studies showed important increases already starting from 75–100%, although without a proper standardization9,10 and because excellent correlation has been observed between a minimum 100% TNR increase and the SNPT. Such an increase was the clinical expression of NSNH.
Symptom Score
The following symptom score 11 was adopted: 1 point, 3–4 sneezes; 3 points, ≥5 sneezes; 1 point, anterior rhinorrhea; 1 point, posterior rhinorrhea; 1 point, nasal breathing problems; 2 points, one blocked nostril; 3 points, both blocked nostrils; 1 point, nose pruritus; 1 point, palatal or ear pruritus; 1 point, conjunctivitis. The overall symptom score was deemed positive if it was ≥5.
Statistics
The VassarStats software (Vassar College, New York, NY) was used to carry out the statistical analysis. Depending on the type of data distribution and on the differences between the two groups, the nonparametric Mann-Whitney U-test or the parametric Student t-test was performed. Linear regression analyses were calculated with a view to determining the existing correlations of all parameters examined, and p < 0.05 values were deemed statistically significant.
Results
Two patients in the NARES group (5% of cases) were prick test results positive for D. pteronyssinus (+++) and P. officinalis (++), which also was confirmed by RAST. Another patient, who showed an uncertain skin response for the first allergen and was RAST results negative, underwent the SNPT and had a negative result after SNPT. The first two patients were excluded from the study.
The nasal response to NSNPT with histamine in the two groups, which was measured based on the symptom score, is shown in Fig. 1. The data are expressed as median values with 75% confidence intervals. The comparison of data referred to the symptom score obtained after and before the NSNPT with histamine has not allowed differentiation of the NARES from the control groups. Only the symptom of sneezing reached a reduced statistical significance (p = 0.02). In the NARES, the index of correlation between the score of single symptoms and the TNR, MCT, and the eosinophilia for each reactivity nasal class did not reach, or reached only for some symptoms, low levels of correlation, which, however, improved when using the composite symptom score (r, from 0.75 to 0.87) (Table 1). The median values with 75% confidence intervals for the nasal obstruction symptom score after NSNPT with histamine in the NARES group are shown in Fig. 2. Such values were compared with those of TNRs and then were matched with the nasal reactivity classes obtained after nasal provocation, thus showing that 39% of patient showed very high reactivity, 37% showed high reactivity, 21% showed medium reactivity, and 3% showed low reactivity. The differences among the nasal reactivity classes so obtained were significant, that is, p = 0.0001 between strong and consistent reactivity and p = 0.002 between consistent and moderate reactivity. As to the controls, 20% of them showed low reactivity and 80% did not show any reactivity after NSNPT.
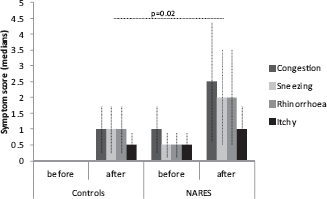
Congestion, sneezing, rhinorrhea, and pruritus symptom scores for controls and patients with NARES, before and after NSNPT with histamine. Data are presented as medians with 75% confidence intervals.
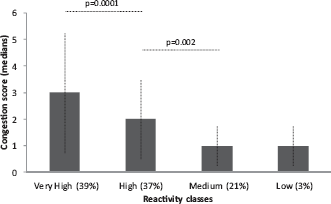
Reactivity classes for the nasal obstruction symptom in patients with NARES after NSNPT with histamine. Data are presented as medians with 75% confidence intervals. The percentage of patients observed in each nasal reactivity classes is reported.
Index of correlation for nasal reactivity class between Eos, MCT, TNR, and CSS in the NARES group

Eos = nasal eosinophilia; CSS = composite symptom score; MCT = mucociliary transport; TNR = total nasal resistance.
TNR values after NSNPT with histamine in the NARES group are shown in Fig. 3. The pretest average (SD) values were 0.22 ± 0.03 Pa × sec × cm−3 for the controls and 0.65 ± 0.27 Pa × sec × cm−3 for the NARES group (p < 0.0005). The average (SD) values after NSNPT were 4.28 ± 2.31 Pa × sec × cm−3 for the NARES group, and 0.31 ± 0.15 Pa × sec × cm−3 for the control group, and a comparison of the two groups showed significant results (p < 0.0005). In fact, after NSNPT, 20% of the controls showed an increase in TNR average value (0.57 ± 0.02 Pa × sec × cm−3), that is, low reactivity, whereas 39% of the patients with NARES were in the very high reactivity class (average, 6.72 ± 1.32 Pa × sec × cm−3), 37% showed high reactivity (average, 3.40 ± 0.49 Pa × sec × cm−3), 21% showed medium reactivity (average, 1.7 ± 0.44 Pa × sec × cm−3), and 3% showed low reactivity (Table 2). These percentages of the subjects were similar to those mentioned above for the nasal obstruction symptom after NSNPT. In the NARES group, the mean percentage of eosinophils in the nasal smear was 45 ± 9%. The mean eosinophilia values matched with the reactivity classes were 54 ± 4% for the very high class, 45 ± 2% for the high class, 32 ± 5% for the medium class, and 25% for the low class (Table 2). The linear regression analyses of the relationship between the TNR increase after NSNPT and the eosinophils counted in the nasal smear of patients of NARES (Fig. 4) showed remarkable correlation between the two variables (R 2 = 0.82; p = 0.001). Conversely, there were no eosinophils in the nasal smear of the control group.

TNR values and nasal reactivity classes after NSNPT with histamine in patients with NARES. Horizontally, there are the following reactivity classes: very high (VH), high (H), medium (M), and low (L).
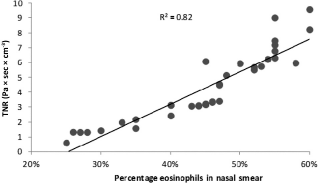
TNR linear regression after NSNPT with histamine versus the eosinophil percentages in nasal smear in the NARES group (R 2 = 0.82; p = 0.001).
Mean (SD) values of TNR before and after NSNPT with histamine, of MCT, and of nasal eosinophilia in patients with NARES and in the controls

MCT = mucociliary transport; TNR = total nasal resistance; NSNPT = nonspecific nasal provocation tests; NARES = nonallergic rhinitis with eosinophilic syndrome.
Comparisons between before and after NSNPT in the NARES.
Comparison between NARES and control groups before NSNPT.
Comparison among nasal reactivity classes.
Very high vs high.
High vs medium.
Very high vs medium
The MCT average (SD) values of the NARES group, expressed in minutes, was higher than that of controls (30 ± 7 minutes versus 14 ± 2 minutes; p = 0.001), and the related mean values matched with the reactivity classes. These values were as follows: 37 ± 4 minutes for the very high class, 27 ± 2 minutes for the high class, 24 ± 3 minutes for the medium class, and 19 minutes for the low class (Table 2). MCT was related to the percentage of eosinophils in the nasal smear of the NARES group (Fig. 5), and linear regression analysis showed a determination coefficient of the two variables, of R 2 = 0.71, with p = 0.001. MCT and TNR responses after NSNPT with histamine in the NARES group (Fig. 6) exhibited a significant linear correlation (R 2 = 0.89; p = 0.001), whereas there was no correlation for the control group (R 2 = 0.05; p = not significant). In regard to the TNR, MCT and nasal eosinophilia (Table 2) in the NARES group were observed with significant differences between before and after NSNPT for all classes of nasal reactivity (p > 0.0005) and for the TNR between NARES and control groups both before and after NSNPT (p < 0.0005). In the end, the correlations among TNR, MCT, and nasal eosinophilia were strong or moderate for every reactivity class (Table 1).

MCT linear regression versus the eosinophil percentages in nasal smear in the NARES group (R 2 = 0.72; p = 0.001).
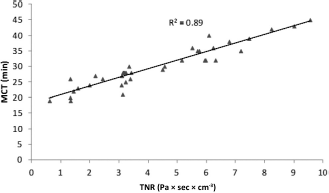
TNR linear regression after NSNPT with histamine versus the MCT of patients with NARES (R 2 = 0.89; p = 0.001).
Discussion
NARES is described as a chronic eosinophilic self-perpetuating inflammation with nonspecific histamine release. It is characterized by increased tryptase in nasal fluid (which also characterizes allergic rhinitis and various forms of nonallergic rhinitis) and eosinophil cationic protein levels, whereas the fluid's Immunoglobulin E level shows no significant increase. The level of eosinophils in the nasal smear, instead, is increased 12 as well as the levels of proinflammatory Th2-type cytokines, IL-6 and IL-17 in the nasal fluid, which seem to be a factor of neutrophil cell infiltration and NARES remodelling processes. 13 Almost all our patients with NARES (95%) did not have clinical histories positive for allergy and allergic sensitization to conventional tests. Only two of the patients (5%) showed sensitization to perennial allergens. Hence, hypereosinophilia of the nasal mucosa proved to be a pathognomonic sign for this disease because it can contribute to its dysfunction.14–16
NSNH evoked by the NSNPT with histamine17,18 was significantly stronger in patients with NARES than in the healthy controls. Generally, nonspecific, autonomic stimuli have a stronger effect on nonallergic rhinitis and, hence, on NARES than they have on healthy subjects or on patients with allergic rhinitis, 1 and strengthen NSNH. Although the nasal response to NSNPT with histamine of patients with persistent allergic rhinitis seems to be related to the symptom score that corresponds to the number of sneezes and to the quantity of nasal secretions, 19 in our study, it was not possible to differentiate the NARES group from the control group based on the symptoms. The composite symptom score 11 can be considered an instrument to verify the nasal reactivity in the patients for the inclusion of a variety of symptoms, which is preferably measured in a more objective and reproducible way from the variation of the TNR. Severe nasal congestion appeared to be the prevailing symptom 20 in the very high and high classes, 9 whereas patients who belonged in the medium and low categories only showed partial obstruction. Most patients with NARES showed very high and high hyperreactivity (76%), whereas medium and low NSNH with no normal reactivity was observed in the others subjects (24%). All control group patients belonged to lower nasal reactivity classes; in particular, 20% of them showed low reactivity and 80% showed normal reactivity. In the NARES group, there was good correspondence between the nasal obstruction symptom score and TNR after the histamine challenge test. The patients with NARES enrolled in our study showed considerable nasal eosinophilia, in line with the definition of this special form of persistent rhinitis, and more or less significant forms of nasal eosinophilia are reported in literature,21–24 but they have never been related to the nasal reactivity classes obtained afterward. The nasal reactivity has been classified as very high, high, medium, and low based on reactivity coefficients. 9
In NARES, the highest nasal eosinophilia values were observed in patients who belonged to the very high and high reactivity classes, whereas milder eosinophilia was shown by the subjects included in the lower medium and low reactivity classes. This statement was further confirmed by the linear regression analysis between the TNR after histamine and eosinophilia variables. As it has already been pointed out for late allergic reactions 16 and for persistent allergic rhinitis, 25 nasal eosinophilia was correlated with nasal obstruction caused by NSNPT with histamine, thus proving that the eosinophilic inflammation was the cause for nasal hyperreactivity in patients with NARES and, therefore, in the relating nasal reactivity classes. Eosinophils are important cells for the physiopathology of many forms of chronic rhinitis, and there is strong evidence that they play a crucial role in inflammation and consequential tissue lesions. 26 The granules of the eosinophils contain toxic basic proteins, most of which are eosinophil cationic proteins. Experimental studies on the eosinophil function revealed the cytotoxic effects on the respiratory epithelium.27,28 Of the patients with NARES enrolled in our study, the mucociliary clearance time showed a progressive and significant increase as the proportion of eosinophils in the nasal smear increased, which indicated ciliostasis and a possible relationship with the nasal reactivity classes.
Neural dysfunction can also contribute to NARES symptoms. 29 Nasal hyperreactivity shows itself with a worsening of the rhinitic symptoms after NSNPT with histamine, and it has been established that it can be caused by disequilibrium in the sympathetic or parasympathetic nerve system and by the antidromic release of some neuropeptides that cause neurogenic inflammation.2,30,31 This hyperreactivity was associated with an increase in the quantity of eosinophils in the smear and results in an increment in nasal resistance. The eosinophils have a cytotoxic effect on the respiratory epithelium by slowing down the clearance time; in fact, we have observed a significant correlation between MCT and TNR obtained after NSNPT with histamine in the NARES group, which was not the case for the controls, thus confirming their role in altering the respiratory function and mucociliary clearance.
Conclusions
NARES is a poorly understood clinical syndrome, with complex pathophysiogenetic implications,32–35 in which the absence of atopy and an increase in nasal eosinophilia was demonstrated. There is a ≥25% of eosinophils over the cell total in the nasal smear, and it is pathognomonic for this disease. Other authors define it as a nasal hyperreactivity syndrome without atopy and with >20% nasal eosinophilia. 36 Eosinophils contribute to the dysfunction of the nasal mucosa. In most cases, NARES showed very high or high NSNH, and the proportion of eosinophils had strong correlations with the nasal reactivity classes. Eosinophils play an important role in causing lesions of the respiratory epithelium, which result in ciliostasis, that is, slowing down of mucociliary clearance, which is correlated with an increase in their proportion in the nasal smear. Ultimately, in NARES, NSNH is the result of the inflammation caused by eosinophils that, by damaging the respiratory epithelium, slow down MCT, cause TNR increase after NSNPT with histamine, and, hence, higher relating nasal reactivity classes. This could have useful applications in NARES, including for classification purposes, based on the response intensity; for prognostic purposes, in that higher reactivity classes have a worse prognosis; and to control the efficacy of available treatments (antileukotriene drugs, corticosteroid, capsaicin, etc.) for NSNH1,37–40 by improving the nasal reactivity classes.
Footnotes
The authors have no conflicts of interest to declare pertaining to this article
