Abstract
Critical aspects of formulating allergy immunotherapy vaccines include the selection, total number, and proportions of each allergen component in therapeutic mixtures. The immunotherapy prescription, determined by a medical provider, details the dosing and schedule for treatment as well as the specific composition of the treatment vials. Allergen extracts are composed of many components such as proteins, glycoproteins, and proteases. Some components in allergen extracts are cross-reactive, meaning that treatment with an extract from one species may confer partial protection against a triggering allergen from another species. Conversely, some allergen extracts are incompatible with other extracts when combined in a mixture for treatment, resulting in lowered therapeutic potential for the patient. Therefore, knowledge of allergen extract cross-reactivities and incompatibilities guides the preparation of subcutaneous immunotherapy prescriptions. In a clinical setting, an understanding of what can and can not be mixed is one critical element in improving treatment outcomes.
Through carefully controlled dosing with allergens, patients are desensitized and are better able to tolerate subsequent exposure. SCIT is used to treat allergic asthma and allergic rhinitis/conjunctivitis in patients whose symptoms are not well controlled by medications or who do not want to use medications to treat symptoms long term. 1 Patients with asthma must have symptoms under control before starting allergy testing or immunotherapy. 1 The efficacy of allergy immunotherapy is dependent on delivering an adequate dose of relevant allergens throughout the course of 3–5 years of treatment. 1 Unlike most pharmacy-based sterile products, allergy immunotherapy prescriptions are often prepared in offices by trained health care personnel.
The preparation of an allergy immunotherapy prescription is a critical step and involves the careful dilution and mixing of individual extracts. As in many areas of medicine, there are guidelines for the preparation of allergy immunotherapy treatments. Guidelines have been published both by the U.S. Pharmacopia (USP) 797 4 and by a collaboration (the Joint Task Force) between the American Academy of Allergy, Asthma, and Immunology (six members), the American College of Allergy, Asthma, and Immunology (six members), and the Joint Council of Allergy, Asthma, and Immunology (one member). The American Academy of Otolaryngic Allergy was invited to review and later endorse the most recent update to the Joint Task Force's guidelines, which were published in 2011. 1 The goal of these guidelines is to ensure sterile mixing, optimize the stability and potency of treatment vials, and decrease practice variance. All guidelines emphasize aseptic technique, handwashing, storage temperatures, use of personal protective equipment, expiration dating, and patient identification on labels and require that staff preparing treatment vials pass a written examination and an annual media-fill test.1,4 The Joint Commission accreditation organization has adopted the USP 797 guidelines and most hospitals require full compliance with these standards. If an allergy practice is not hospital based, then the physician must determine 1 which guidelines1,4 will be followed to meet the needs of the practice.
Preparation of Allergy Immunotherapy Prescriptions
Extracts used for SCIT are complex mixtures. The extracts used for mixing can differ in starting and final concentrations depending on the allergen used.1,5 One recommendation is for each medical practice to develop a manual to support the office staff in maintaining consistent procedures for preparing treatment vials. 5 This streamlines the preparation process and makes it easier to train other staff members. The manual should include a competency assessment for staff members that compound prescriptions (such as the Extract Quiz found at www.jcaaiservices.org) 6 as well as training guidelines and checklists for prescription preparation (such as those found at www.jcaai.org/page/quiz) 7 .
Other recommendations for the compounding of allergy immunotherapy prescriptions are based on the type of extracts used. Extracts are available in two forms: standardized and nonstandardized. Currently, the Food and Drug Administration has approved 19 standardized allergen extracts, including grass pollens, dust mites, cat, short ragweed, and Hymenoptera (an insect order that includes wasps, bees, ants, and other insects) venoms (Table 1).8,9 Nonstandardized extracts have been used for decades and an analysis of the literature has found most of these extracts to be safe and effective. 10 Nonstandardized extracts are used when a standardized extract is not available.
Standardized allergen extracts available in the United States
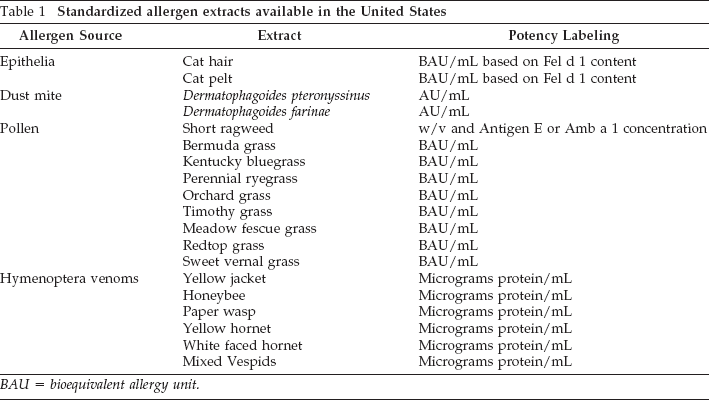
BAU = bioequivalent allergy unit.
Unlike standardized extracts, nonstandardized extracts are made and sold at different concentrations.11,12 This may complicate the dilution process for mixing treatment vials. To retain a clinically relevant concentration for treatment, it is recommended that the volume of each allergen added reflects the recommended dose and that, after the first allergen, subsequent additions only replace diluent. 1 Therefore, it may help the mixing staff if the manual includes a dosing range based on the most recent recommendations from the 2011 Practice Parameters for prescription preparation. 1 Examples of the doses recommended for representative extracts are listed in Table 2. 13
Target immunotherapy doses

Source: Refs. 1 and 13.
AgE = Antigen E or Amb a 1; AP = acetone precipitated; AU = allergy unit; BAU = bioequivalent allergy unit; Can f 1 = canis familiaris allergen 1; HTD = highest tolerated dose.
Cross-Reactive Properties of Allergens
It is important to note that allergens from the same family may cross-react, i.e., the immune system may not fully differentiate them. 14 Although it has been suggested that using a representative cross-reactive allergen can substitute for others in the same family, a growing body of data show that the immune response to related allergens is a mixture of specific and cross-reactive T-cell and antibody responses.15–17 Treatment with the specific allergens causing the clinical problem may be the most effective approach to inducing tolerance.15,18
Keeping cross-reactivity in mind is critical to the safety of immunotherapy prescriptions. If partially cross-reactive binding sites are contributed by multiple allergens in the treatment vial, the concentration of the cross-reactive segments of the allergen may be greater than the target dose and could induce a side effect that might be uncomfortable for the patient and slow their course of treatment.
When choosing extracts, it is important to know that the genetic relationships behind cross-reactivities can vary for extracts from different categories of source materials.19,20 Thus, when choosing representative allergens, it is better to rely on established and published evidence of cross-reactivity without making assumptions across general classifications (i.e., the same genus or family) that may not be accurate. 21 It is also important that skin-test panels include relevant cross-reactive allergens from geographic regions that would be therapeutically appropriate for the patient.21,22
There are many examples of cross-reactive allergens. For example, temperate pasture grasses of the Pooideae subfamily are partially cross-reactive.11,21,23,24 Examples of cross-reactive trees,25,26 weeds,25,26 and grasses 27 are listed in Tables 3–5, respectively. A common approach to addressing cross-reactive allergens is to use mixes. For prescriptions that include dust mites, either an extract containing the two most common species, Dermatophagoides pteronyssinus and Dermatophagoides farinae, or the individual extracts is recommended because although the two species are similar, they are sufficiently different that both are necessary for effective treatment. 1 The same is true for cockroaches. A mixture of both German and American cockroach species or the individual extract is recommended in treatment vials.1,28 The use of mixes can both help to reduce the safety risk of cross-reactive allergens and address the limitations of cross-reactivity when treating to achieve tolerance.1,30
Cross-reactive trees

Source: Refs. 19 and 20.
Cross-reactive weeds

Source: Refs. 19 and 20.
Cross-reactive grasses

Source: Refs. 19 and 20.
Allergen Incompatibility in Mixing Prescriptions
Because extracts are mixed to produce treatment vials, research has focused on the stability and potency of individual extracts after mixing. It has been found that some mixtures of extracts are incompatible; i.e., mixing particular allergen extracts together lowers the stability and potency of one or more of the allergens. 29 Some allergenic products used for immunotherapy naturally include proteases. Proteases are enzymes that degrade other proteins and are the reason for allergen incompatibility. For example, when two extracts are mixed, proteases from one extract can degrade allergens from the other extract and can compromise the potency of both extracts in the resulting treatment vial. 30 Research has identified allergen extracts that are particularly rich in proteases and compounding recommendations have been developed to maintain potency of allergy immunotherapy treatment vials.
The allergenic products with the highest protease activity are insect and fungal (mold) extracts. As an example, studies have shown that the potencies of representative grass and tree pollens, as well as cat, dog, ragweed, and other weed extracts, are reduced when these extracts are mixed with mold or fungal extracts (Alternaria, Aspergillus, and Penicillium).1,5,29–31 Certain mixtures of fungi and insects are also incompatible. For example, Alternaria extract did not retain potency when mixed with insect extracts and German cockroach extract did not retain potency when mixed with fungal extracts. 31 Dust mite extracts manufactured in the United States can be successfully mixed with grass pollens because they are more highly purified than European dust-mite extracts but may be unstable when mixed with molds.19,28 Recommendations on considering incompatibility when mixing treatment vials are based on ongoing research and summarized in Fig. 1.1,19,28–33

Mixing recommendations for subcutaneous allergen extract immunotherapy: extract compatibilities after storage for 1–3 months at 2–8°C in (A) 0–10% glycerin, (B) 25% glycerin, or (C) 50% glycerin. In this two-way chart, the combination is designated as unstable if either of the components have reduced potency on storage. Mixtures are designated as compatible (green), partially unstable (yellow), or not recommended (red) according to research presented by Cox et al. 1 , Esch 19 , Grier et al., 33 and Esch and Grier. 34 © GREER 2015. Reprinted with permission from GREER Laboratories, Inc, Lenoir, NC.
Mixing compatibility charts, such as shown in Fig. 1, can be a very practical resource in a busy practice but, to accurately use them, it is important to understand the complex information they portray. Variables that impact the data in these charts include the extract type and source(s), whether the stability portrayed represents a single extract or both components in the mixture (also called one-way or two-way charts, respectively), the length of time the mixtures have been stored, the storage temperature, and the percent of glycerin in the mixtures. Figure 1 is a two-way chart showing stability of both components in a mixture stored for between 1 and 3 months at three different glycerin concentrations. For purposes of comparison, the chart published by Grier et al. 29 is a one-way chart showing the stability of single extracts after storage with a second extract for 12 months. Not surprisingly, the results are quite different.
Because of extract incompatibility, guidelines suggest that when incompatible extracts are required for a patient, accurate dosing and potency of allergy immunotherapy treatment vials be maintained by compounding two or more separate treatment vials. 1 Some studies have shown that higher concentrations of glycerin reduce allergen incompatibility and help to retain potency but lower concentrations are less painful to patients. 1 Storage time can also affect potency. For example, when a combination of Alternaria and Penicillium was studied, potency in 10% glycerin decreased to ~80% after 12 months of storage but was maintained in 25% glycerin. 13 Although studies have begun to characterize the complex relationship between multiple allergens, glycerin concentration, and storage time, currently, a relatively small number of allergens have been investigated. Phenol, a bacteriostatic preservative, is added to help prevent the growth of microorganisms. 1 This information should be integrated into allergy practices manuals for prescription preparation. Allergen extract incompatibility is important to remember when compounding treatment vials so that potency is retained and an effective immunotherapy treatment is delivered to the patient.
High and Low Sensitivities to Allergens
Allergy testing reveals how allergic a patient is to each allergen in the treatment vial. The allergens to which a patient is the most sensitive are most likely to cause an adverse reaction during advancement to the maintenance level. For this reason, some clinicians choose to combine allergens based on each patient's sensitivity levels; this allows them to manage the advancement of dose for the “high-sensitivity” allergens separately from the advancement of dose for the “low-sensitivity” allergens. 24
Extract Integrity
Other issues can affect the quality of allergenic extracts. Over time, allergenic extracts, particularly aqueous extracts and extracts at high concentrations, may precipitate out of solution. There are few published studies that carefully characterize the nature and quantity of these precipitates. One unpublished reference suggests that a precipitated extract had no significant change in potency. 34 It has also been suggested that, in theory, injecting precipitates could result in patients developing granulomas. 34
Some extracts, particularly certain pollens and foods, may discolor over time. Generally, this is caused by the oxidation of extracts resulting from the vials having been opened, and thus exposed to oxygen, multiple times. 34
Delamination of glass vials, which can result in glass flakes, 35 is very rare with pharmaceutical-grade glass but can occur. In response to this, the USP has proposed a new chapter discussing methods that manufacturers might use to predict glass delamination. 36
Anytime the mixing staff sees a change in an extract, they should contact the manufacturer for advice on how to proceed. 34
Summary
From the basic process of diluting allergenic products to the properties of each individual extract, care must be taken at each step to ensure an effective immunotherapy treatment. Some key points include
USP 797 and Joint Task Force guidelines should be used for preparation of SCIT treatment vials. Both include recommendations on aseptic technique, handwashing, storage temperatures, use of personal protective equipment, expiration dating, patient identification on labels, and require that staff preparing treatment vials pass both a written examination and annual media-fill test. Develop a manual for the practice to promote consistency with the previous recommendations as well as to highlight information on cross-reactivity and allergen incompatibility. Knowledge of cross-reactive allergens will simplify compounding SCIT treatment vials—use one cross-reactive representative extract or a mix of cross-reactive allergens. Keep allergen extract incompatibility in mind when compounding treatment vials to maintain potency and efficacy for patients. For some patients, separation of allergens into separate vials based on high and low sensitivities may avoid problems related to adverse reactions that are sometimes caused by highly sensitive allergens and that may be encountered during advancement of dose.
Compounding allergy immunotherapy treatment vials is a complicated process that when done appropriately, will optimize the treatment outcome. Keeping the basic principles of cross-reactivity and incompatibility in mind when compounding allergy immunotherapy prescriptions offers the potential for a safer and more effective approach to providing this important, disease-modifying 2 therapy.
Footnotes
Acknowledgments
The authors thank Sarah B. Guttenplan, Ph.D., for writing support of the article, and Thomas Grier, Ph.D., for advice and guidance.
