Abstract
Tissue-engineered nasal septal cartilage may provide a source of autologous tissue for repair of craniofacial defects. Although advances have been made in manipulating the chondrocyte culture environment for production of neocartilage, consensus on the best oxygen tension for in vitro growth of tissue-engineered cartilage has not been reached. The objective of this study was to determine whether in vitro oxygen tension influences chondrocyte expansion and redifferentiation. Proliferation of chondrocytes from 12 patients expanded in monolayer under hypoxic (5% or 10%) or normoxic (21%) oxygen tension was compared over 14 days of culture. The highest performing oxygen level was used for further expansion of the monolayer cultures. At confluency, chondrocytes were redifferentiated by encapsulation in alginate beads and cultured for 14 days under hypoxic (5 or 10%) or normoxic (21%) oxygen tension. Biochemical and histological properties were evaluated. Chondrocyte proliferation in monolayer and redifferentiation in alginate beads were supported by all oxygen tensions tested. Chondrocytes in monolayer culture had increased proliferation at normoxic oxygen tension (p = 0.06), as well as greater accumulation of glycosaminoglycan (GAG) during chondrocyte redifferentiation (p < 0.05). Chondrocytes released from beads cultured under all three oxygen levels showed robust accumulation of GAG and type II collagen with a lower degree of type I collagen immunoreactivity. Finally, formation of chondrocyte clusters was associated with decreasing oxygen tension (p < 0.05). Expansion of human septal chondrocytes in monolayer culture was greatest at normoxic oxygen tension. Both normoxic and hypoxic culture of human septal chondrocytes embedded in alginate beads supported robust extracellular matrix deposition. However, GAG accumulation was significantly enhanced under normoxic culture conditions. Chondrocyte cluster formation was associated with hypoxic oxygen tension.
These limitations have encouraged investigators to develop alternatives to cartilage autografts. One approach is to create neocartilage using tissue engineering methods. This approach allows for significant amplification of the chondrocyte population from harvested autologous cartilage, followed by fabrication of the neocartilage graft in the desired size and shape for a particular reconstructive procedure.
Although donor chondrocytes can be obtained from several different anatomic locations, nasal septal cartilage provides an ideal material for tissue engineering. It is easily harvested with minimal donor site morbidity or creation of functional or cosmetic defects. Moreover, native septal cartilage has superior structural rigidity when compared with the more elastic auricular cartilage, making it a preferred autograft material for mechanical support during reconstructive procedures.2,4
Production of appropriate extracellular matrix (ECM) by tissue-engineered chondrocytes is required for the formation of neocartilage tissue with mechanical properties resembling native cartilage. The isolated chondrocyte population is first expanded in monolayer culture causing the chondrocytes to take on a fibroblastic phenotype in a process called dedifferentiation. Dedifferentiated chondrocytes produce less matrix proteoglycans and preferentially synthesize type I rather than type II collagen–the form that is abundant in native hyaline cartilage.5,6 Culture of dedifferentiated human nasal septal chondrocytes after encapsulation in alginate promotes redifferentiation of the chondrocytes. Redifferentiated chondrocytes produce higher levels of type II than type I collagen and accumulate significant quantities of matrix sulfated glycosaminoglycan (sGAG).7–9
Defining the optimal culture environment to promote chondrogenesis in vitro may result in production of clinically useful tissue-engineered cartilage. Aside from being vital for cell metabolism, oxygen also influences developmental processes, including the formation of cartilage.10,11 Although studies have confirmed oxygen's effects on cartilage tissue development, the mechanism by which oxygen influences these processes has not been delineated. Studies using articular chondrocytes, nasal chondrocytes, or chondrocytes derived from mesenchymal stem cells have shown that low oxygen tension either promotes or impedes functional ECM production over standard normoxic oxygen tension.12–16 Evaluating the potential benefit of culturing under low oxygen tension in nasal septal cartilage tissue engineering has been limited.15,16 The objective of the present study was to compare the effect of culturing under different oxygen tensions (5, 10, and 21%) on the morphological, histological, and biochemical properties of engineered human nasal septal chondrocytes.
Materials and Methods
Chondrocyte Isolation
The study used remnant human septal specimens removed during routine surgery at the University of California, San Diego, Medical Center or San Diego Veterans Affairs Medical Center. Approval was granted by the Human Subjects Committee of the Veterans Administration San Diego Health Care System and University of California, San Diego, Human Research Protection Programs.
Chondrocytes from 12 patients (5 men and 7 women; mean age, 32.8 years) were isolated from the cartilage as reported previously. 17
Expansion of Chondrocytes
For each patient, isolated chondrocytes were seeded at 3000 cells/cm2 into 24-well culture plates (Corning Costar, Corning, NY) and incubated in a humidified 37°C incubator with 5% carbon dioxide (CO2) under hypoxic (5 or 10%) or normoxic (21%) oxygen tension.
Two wells from each plate under different oxygen tension were digested with 0.5 mg/mL of proteinase-K for 24 hours at 37°C on days 2, 4, 6, 8, 10, 12, and 14. Released chondrocytes were collected for analysis of cell number.
Chondrocytes from each sample were seeded at a density of 5000 cells/cm2 in culture flasks (Corning Costar) and incubated in a humidified 37°C incubator with 5% CO2 and 21% oxygen for 2 weeks.
Culture medium was supplemented with 2% human serum, 1 ng/mL of transforming growth factor β1, 5 ng/mL of fibroblast growth factor-2, and 10 ng/mL of platelet-derived growth factor-BB, and changed every 2 days. Medium was allowed to equilibrate in the incubator overnight before each media change. The effectiveness of this equilibration time was verified using an OxyLab pO2 monitor (Oxford Optronix, Ltd., Oxford, U.K.).
Encapsulation and Culture in Alginate
Expanded cells were released from monolayer and resuspended in alginate as described previously. 17 Alginate beads were seeded at 5 beads/well in 24-well culture plates (Corning Costar) in medium supplemented with 2% human serum, 100 ng/mL of bone morphogenic protein-14, and 200 ng/mL of insulinlike growth factor-1 and cultured at 37°C with 5% CO2 and either hypoxic (5 or 10%) or normoxic (21%) oxygen tension. The media was changed every other day.
Beads from each patient cultured under different oxygen tensions were depolymerized with 55 mM of sodium citrate, 30 mM of EDTA, and 0.15 mM of NaCl on days 2, 4, 6, 8, 10, 12, and 14. Cell pellets were digested in 0.5 mg/mL of proteinase K in 100 mM of phosphate and 5 mM of EDTA (phosphated buffered EDTA, pH 7.1) at 60°C overnight before biochemical analysis.
Quantitative Assays for DNA, sGAG, and Total Collagen
DNA levels were measured using the PicoGreen DNA content determination assay as described in a previous report.17,18,19 DNA content was normalized per bead assayed.
The sGAG content was determined, as reported previously, using portions of the proteinase-K digests and the dimethyl-methylene blue reaction. 20 sGAG content was normalized per bead assayed.
The quantity of hydroxyproline in the beads was determined as described previously using the chloramine T reagent/p-dimethylaminobenzaldehyde assay. 21 Hydroxyproline content was converted to collagen content using a mass ratio of collagen to hydroxyproline of 7.1. 22 Total collagen was normalized to DNA content.
Histology
After depolymerization of the alginate beads, cells from two patients were pelleted, rinsed with phosphate-buffered saline, resuspended in phosphate-buffered saline, placed on poly-L lysine-coated slides (Polysciences, Inc., Warrington, PA) and allowed to dry overnight. Histochemical localization of GAG was performed as previously described. 23
Single and clustered chondrocyte cell numbers was determined by counting eight random fields of view of for each oxygen tension assayed.
Immunohistochemistry was performed with the Vectastain Elite ABC kit (Vector Laboratories, Burlingame, CA) following the manufacturer's instructions. Cells from two patients were prepared as described previously and probed with one of three antibodies: anti-type I collagen (7 μg/mL; no. ab6308; Abcam, Burlingame, CA), anti-type II collagen (0.5 μg/mL; no. 7005; Chrondrex, Inc., Redmond, WA), or a mouse nonspecific IgG (7 μg/mL; no. ab37355, Abcam) used as a negative control. Sections were counterstained with methyl green nuclear stain (Vector Laboratories). Samples were documented by photomicroscopy.
Statistical Analysis
Analyses were performed using Systat 10.2 (Systat Software, Chicago, IL). Means are presented ±SD. Differences in cell number, DNA content/bead, and GAG/bead were assessed using multivariate repeated measures analysis of variance (ANOVA) with incubation time as a repeated factor followed by post hoc Bonferroni test when significance was detected (p < 0.05). Differences in total collagen/DNA were assessed using a one-way ANOVA. If the ANOVA identified an overall significant effect, post hoc Tukey's honestly significant difference tests were used to verify significant differences between culture conditions. Linear regression analysis was used to analyze the relationship between chondrocyte cluster formation and oxygen tension. A difference was considered significant when p ≤ 0.05.
Results
Chondrocytes were isolated and expanded in monolayer culture under hypoxic (5 or 10%) or normoxic (21%) oxygen tensions. Expansion of human nasal septal chondrocytes in monolayer culture under these oxygen tensions did not result in gross morphological differences (Fig. 1). Cell proliferation tended to increase with higher oxygen tension (p = 0.06; Fig. 2). Based on these observations, the normoxic oxygen tension was applied for monolayer cell expansion before alginate encapsulation to promote redifferentiation of the chondrocytic phenotype.

Light micrographs of human nasal septal chondrocytes cultured as monolayers for 8 days under (A) 5%, (B) 10%, or (C) 21% oxygen tension. Chondrocytes exhibit the elongated morphology typical of dedifferentiated chondrocytes.
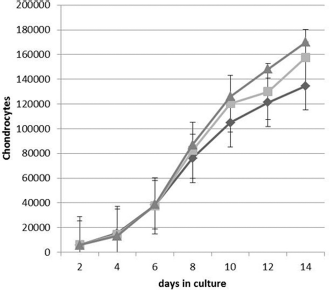
The effect of oxygen tension on chondrocyte proliferation. Human nasal septal chondrocytes from 12 patients were cultured as monolayers for 14 days under (diamond) 5, (square) 10, or (triangle) 21% oxygen tension. There was a trend toward increased cell proliferation when chondrocytes were cultured under normoxic (21%) culture conditions (p = 0.06; mean ± SD).
Chondrocytes encapsulated in alginate beads were harvested every 2 days over the 14-day culture period. At the end of the culture period, chondrocytes were released from the alginate beads and subjected to biochemical and histological analyses. Biochemical testing showed that the amount of DNA per bead did not change significantly over the 14-day culture period and was not significantly different under the three oxygen tensions tested (Fig. 3 A). Moreover, all three oxygen tensions were able to support chondrocytic redifferentiation as measured by accumulation of sGAG per bead over time (Fig. 3 B). There was a significant increase in the amount of sGAG per bead with increasing oxygen tension (p < 0.05; Fig. 3 B). In addition, the accumulation of total collagen per DNA was similar at day 14 for the three oxygen tensions tested (p > 0.05; Fig. 4).
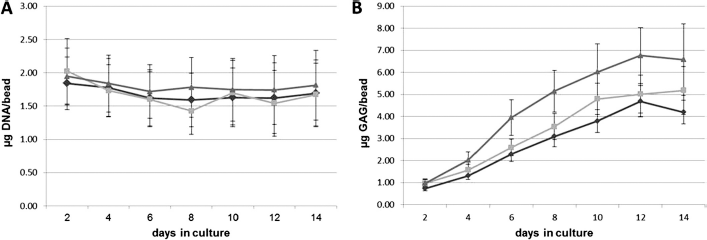
The effect of oxygen tension on DNA and sulfated glycosaminoglycan (sGAG) accumulation during chondrocyte redifferentiation. After monolayer expansion under normoxic culture conditions, human nasal septal chondrocytes from 12 patients were redifferentiated by encapsulation and culture in alginate beads for 14 days under (diamond) 5, (square) 10, or (triangle) 21% oxygen tension. Over the 14-day culture period, chondrocyte DNA per bead did not vary significantly and remained similar under the three oxygen tensions tested. In contrast, the amount of sGAG per bead increased with increasing oxygen tension (p < 0.05; mean ± SD).

The effect of oxygen tension on accumulation of collagen during chondrocyte redifferentiation. After 14 days of redifferentiation in alginate beads, the amount of total collagen per DNA was similar between the three oxygen tensions tested (p > 0.05; mean ± SD).
Chondrocytes from two patients were analyzed histologically at day 14. Chondrocytes differentiated under hypoxic and normoxic oxygen tensions developed substantial ECM (Fig. 5). Alcian blue staining showed robust GAG accumulation under all three oxygen tensions tested (Fig. 5, A-C). Moreover, chondrocytes differentiated under hypoxic and normoxic oxygen tensions exhibited positive immunohistochemical staining for collagen (Fig. 5. D-I) when compared with the nonspecific IgG control antibody (Fig. 5, J-L). As expected for redifferentiated chondrocytes, collagen immunohistochemistry revealed strong staining for type II collagen and weaker staining for type I collagen under all three oxygen tensions.

Chondrocytes redifferentiated under hypoxic (5 and 10%) and normoxic (21%) oxygen tensions developed substantial extracellular matrix (ECM) after 14 days of redifferentiation by encapsulation and culture in alginate beads. Chondrocytes showed characteristics of differentiated chondrocytes including (A–C) robust accumulation of glycosaminoglycan (GAG) detected by Alcian blue histochemistry as well as (D–F) low levels of type I collagen immunohistochemical staining and (G–I) strong type II collagen immunohistochemistry) with minimal immunohistochemical staining (J–L) with a nonspecific IgG.
Additionally, linear regression showed a significant association between the number of observed chondrocyte clusters and decreasing oxygen tension (Fig. 6; p < 0.05).

Redifferentiation of human nasal septal chondrocytes in alginate beads under decreased oxygen tension resulted in an increased number of chondrocyte clusters. Linear regression analysis revealed a positive correlation between the number of chondrocyte cell clusters formed and decreasing oxygen tension. Regression line and corresponding R 2 and p values are shown.
Discussion
Historically, the response of articular and septal chondrocytes and chondrocytes derived from mesenchymal stem cells to culture under low oxygen tension has been variable.12–17 However, before this study, evaluation of hypoxic cell culture conditions on the redifferentiation of human nasal septal chondrocytes cultured in alginate beads has not been reported. In this study, we successfully showed that human nasal septal chondrocytes can be expanded in monolayer culture and induced to redifferentiate ECM successfully when cultured under normoxic (21%) or hypoxic (5 and 10%) oxygen tensions. Culture under normoxic oxygen levels tended to enhance the proliferation of chondrocytes when cultured as monolayers.
Chondrocyte redifferentiation was supported by culture of nasal septal chondrocytes in alginate beads under normoxic or hypoxic oxygen tensions. Under all oxygen tension conditions tested, nasal septal chondrocytes developed phenotypes typical for chondrocytes that generate hyaline cartilage including a rounded morphology, formation of chondrocyte clusters, production of sGAG, type II collagen, and a reduced expression of type I collagen.
The accumulation of DNA and the final amount of total collagen did not differ significantly when chondrocytes were cultured under different oxygen tensions. However, culture under normoxic oxygen tension resulted in significantly increased sGAG accumulation.
Although oxygen tension did not significantly impact the accumulation of nasal septal chondrocyte DNA in alginate bead cultures, the formation of chondrocyte clusters was positively associated with culture under hypoxic oxygen tensions. A potential explanation for this observation is that increased production of sGAG at higher oxygen tensions acts to enhance the separation of cells resulting in a smaller number of clustered cells. Alternatively, it is also possible that more chondrocyte clusters form under hypoxic culture conditions as a stress response to growth under low oxygen tension.
An increase in chondrocyte clusters was observed, although not quantified, when primary bovine articular chondrocytes were cultured under hypoxic conditions. 12 In articular cartilage, an increase in chondrocyte clusters has been noted in injured and diseased tissue.24–29 It has been postulated that formation of large chondrocyte clusters is part of the initial phase in tissue repair, where cell proliferation would be followed by chondrocyte migration and differentiation with formation of new ECM. 30
Culture conditions that promote chondrocyte redifferentiation and enhance chondrogenesis are of great interest for their potential to produce clinically useful tissue-engineered cartilage. Recent studies suggest that oxygen tension is an important culture condition that may be capable of affecting chondrocyte redifferentiation.12–17
Domm et al. 12 expanded bovine articular chondrocytes in monolayer culture under normoxic (21%) oxygen tension and then subcultured in monolayer or encapsulated in alginate beads followed by culture in normoxic or hypoxic (5% oxygen) culture conditions for 2 or 3 weeks. The authors measured cell viability, production and accumulation of collagen, and accumulation of DNA and GAGs and concluded that low oxygen tension induced or promoted redifferentiation of dedifferentiated bovine articular chondrocytes in alginate bead culture and increased type II collagen production in primary bovine articular chondrocytes cultured in alginate beads.
Given that the oxygen level in articular cartilage is between 7 and 10% at the surface and 0.1% near the subchondral bone, Mhanna and colleagues 13 used 1% oxygen as the hypoxic environment in their study of articular chondrocyte differentiation. Type II collagen gene expression was compared in articular chondrocytes encapsulated in alginate beads cultured under normoxic (21%) and hypoxic (1%) conditions. Alginate bead-encapsulated chondrocytes cultured under hypoxic conditions exhibited a 49.5-fold up-regulation in type II collagen gene expression compared with those cultured under normoxic conditions. The in vitro results are in accordance with the oxygen distribution in articular cartilage where the deep and middle zone oxygen concentration is <1% and type II collagen is greatly produced.
The effect of low oxygen tension on the chondrogenic differentiation of mesenchymal stem cells has also been investigated. Malladi et al. 14 induced chondrogenesis in vitro in mouse adipose-derived mesenchymal cells in a three-dimensional micromass culture model under normoxic (21%) and hypoxic (2%) conditions. Type II collagen immunohistochemistry, histochemical staining for GAG and quantitation of DNA and sGAG were evaluated at various time points over 12 days of culture. Although micromass cultures redifferentiated under hypoxic oxygen tension proliferated at a greater rate through the culture period, a marked decrease in the expression of type II collagen and GAG was observed when these cells were redifferentiated under low oxygen tension.
In contrast, monolayer culture of human and rabbit adipose-derived mesenchymal stem cells for 21 days in chondrogenic medium under low oxygen tension (5% oxygen) resulted in enhanced expression of type II collagen and aggrecan when compared with cells cultured under normoxic conditions. 15
Studies investigating the effect of low oxygen tension on human nasal septal chondrocyte differentiation have been limited and have resulted in differing conclusions as to the value of hypoxic culture conditions. In a study by Malda et al., 16 human septal chondrocytes were isolated and cultured in monolayer under normoxic conditions. The cells were then transferred to a bioreactor in pellet form and cultured for 21 days at 100 (21% oxygen), 25 (5.25% oxygen), or 5% (1% oxygen) dissolved oxygen (DO). The cultures were assessed histochemically for GAG and immunolocalization of a fibroblast membrane marker (11-fibrau) and collagen types I, II, and IX. Additionally, DNA and GAG content were determined biochemically. In this study, culturing chondrocytes in 25 and 5% DO resulted in abundant expression of type II collagen with no detectable expression of type I collagen. In addition, these pellets had significant GAG content with the 5% DO group showing the highest GAG content. This study concluded that low oxygen tension stimulates the redifferentiation of dedifferentiated adult human nasal septal chondrocytes.
In contrast, Scotti et al. 17 expanded human nasal septal and articular chondrocytes in monolayer culture and then cultured them as pellets for 1 or 2 weeks under normoxic (19% oxygen) or hypoxic (5% oxygen) conditions on a three-dimensional orbital shaker. At the end of the culture period, the pellets were analyzed for the accumulation of GAG histochemically, GAG and DNA biochemically, and type II collagen immunohistochemically. The authors found that hypoxic culture conditions enhanced GAG and type II collagen accumulation in articular but not in septal chondrocytes. They concluded that although GAG and type II collagen accumulation under low oxygen tension were enhanced in human articular chondrocytes, human nasal chondrocytes were much less responsive to culture under low oxygen tension.
Although the current study supports the conclusion that low oxygen tension does not promote redifferentiation of nasal septal chondrocytes, it is possible that further studies with extended culture under low oxygen tension may reveal differences in the structural organization of collagen fibers and degree and types of collagen crosslinks formed—this, in turn, may lead to enhanced biomechanical properties of engineered nasal septal cartilage.
Footnotes
The authors have no conflicts of interest to declare pertaining to this article
