Abstract
Cell counts in nasal secretions are not used in routine clinical practice to decide on anti-inflammatory or antimicrobial therapy. This study investigated the reproducibility, reliability (validity), and responsiveness of cell counts in blown nasal secretions with a view to implementing this in routine clinical practice. Nasal secretions were obtained from 19 subjects with allergic rhinitis on 3 days in random order (each separated by 1–2 days) by spontaneously blowing their noses (on 2 days) and by a nasal lavage by the modified Grunberg method on the 3rd day. Total and differential cell counts were performed after dispersing the solutions with dithiothreitol as described previously. At the end of the study, subjects had 1 week of open label treatment with nasal corticosteroids if they had nasal eosinophilia or an antibiotic if they had nasal neutrophilia. If the cell counts were normal, they were not treated. The proportion of eosinophil (%) was highly reproducible (intraclass correlation coefficient [ICC], 0.93), and the total cell count (X106/g) and the proportion of neutrophil (%) were modestly reproducible in blown nasal secretions (ICC, 0.46 and 0.55, respectively). The total cell count was consistently and significantly higher in the blown nasal secretions. The proportion of eosinophils (Rs = 0.4; p < 0.05) and neutrophils (Rs = 0.6; p < 0.05) showed modest correlation in the two types of samples. The responsiveness index for eosinophil count was 4.0 and for neutrophil count was 1.5. Total and differential cell counts can be reliably and reproducibly obtained from spontaneously blown nasal secretions. The cell counts are responsive to treatment and can help identify allergic and infective rhinosinusitis and guide therapy and are easy to implement in routine clinical practice.
Design and Methods
Nineteen atopic subjects (house-dust mites, n = 3; cats, n = 3; dogs, n = 3; ragweed, n = 2; trees, n = 4; grass, n = 4; 6 subjects were sensitive to more than one allergen) with rhinitis were recruited from the Chest and Allergy Clinics of Firestone Institute for Respiratory Health after obtaining informed consent and approval from the Research Ethics Board of St. Joseph's Health Care (Table 1). Skin-prick test and spirometry were performed and 12-hour reflective total nasal symptoms score (maximum score, 12) 3 and cold symptom score 4 were assessed. The severity of the cold was based on at least two or more symptoms of nasal discharge, sneezing, nasal congestion, sore throat, cough, headache, malaise, chills, and/or fever. The severity of each symptom was assessed by a severity scale: 0 = absent, 1 = mild, 2 = moderately severe, and 3 = severe (9 symptoms, maximum possible score, 27). Nasal secretions were obtained on 3 days in random order (each separated by 1–2 days) by spontaneously blowing their noses (on 2 days) and by a nasal lavage by the modified Grunberg method 5 on the 3rd day. Total and differential cell counts were performed after dispersing the solutions with dithiothreitol as described previously.5,6 At the end of the study, subjects had 1 week of open label treatment with nasal fluticasone, 50 μg, twice daily if they had eosinophilic rhinitis (>3% and a positive skin allergy test; n = 6) or azithromycin, 500 mg, on day 1, followed by 250 mg daily for 4 days if they had a neutrophilic rhinitis (>75% with a total cell count of >25 million cells; n = 5). If the cell counts were normal, they were not treated (n = 5).
Baseline subject characteristics of 19 subjects with seasonal allergic rhinitis
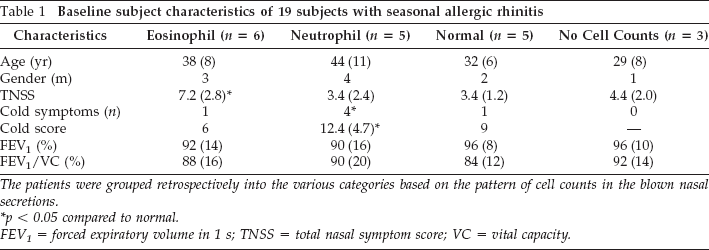
The patients were grouped retrospectively into the various categories based on the pattern of cell counts in the blown nasal secretions.
p < 0.05 compared to normal.
FEV1 = forced expiratory volume in 1 s; TNSS = total nasal symptom score; VC = vital capacity.
Analysis
Reproducibility was assessed by intraclass correlation coefficient (ICC). Validity of cell counts in blown nasal secretions was assessed by comparing with cell counts in lavage fluid (Spearman's correlation) and by comparing the cell counts to the baseline symptom scores. Although the treatment was not randomized, responsiveness was assessed as the ratio of the magnitude of change with treatment to the natural variance in the untreated patients. 7 Based on our previous experience of evaluating measurement properties of cell counts in sputum, 8 we assumed that a sample size of 15 subjects would be sufficient for this study.
Results
Reproducibility
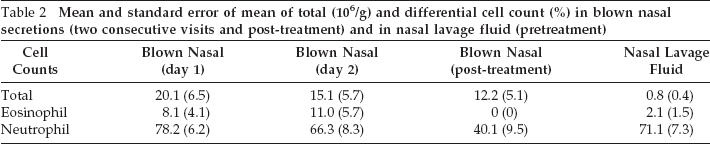
Sufficient samples were obtained from 16 subjects (9 men; mean age, 40 years) on all 3 days. The mean and standard error of the mean of cell counts are shown in Table 2. The proportion of eosinophil (%) was highly reproducible (ICC, 0.93), while the total cell count (X10 6 /g) and the proportion of neutrophil (%) were modestly reproducible in blown nasal secretions (ICC, 0.46 and 0.55, respectively). The total cell count was consistently and significantly higher in the blown nasal secretions than in the lavage fluid, indicating that this is a more efficient method of collecting nasal secretions.
Mean and standard error of mean of total (106/g) and differential cell count (%) in blown nasal secretions (two consecutive visits and post-treatment) and in nasal lavage fluid (pretreatment)
Validity
Subjects who had greater symptoms suggestive of allergic rhinitis had higher eosinophil count in the blown nasal secretions than the other groups, whereas patients with higher cold symptoms had higher neutrophil count, providing evidence to the validity of the cell counts (Table 2). The proportion of eosinophils (Rs = 0.4; p < 0.05) and neutrophils (Rs = 0.6; p < 0.05) showed modest correlation in the two types of samples, providing further support to external validity of cell counts in blown secretions.
Responsiveness
All of the subjects reported clinical improvement with their respective treatment. One subject required an additional week of antibiotic treatment. The changes in eosinophil and neutrophil percent in blown nasal secretion in those patients who were treated with nasal corticosteroids or azithromycin are shown in Fig. 1. The responsiveness index for eosinophil count was 4.0 and for neutrophil count was 1.5.

Figure showing the eosinophil percent and neutrophil percent pre- and post-treatment (5 days) with nasal fluticasone and oral zithromycin in patients with baseline eosinophils of >3% (n = 6), baseline neutrophils of >75%, and total cell count of >25 million cells/g (n = 5).
Discussion
This study shows the measurement properties of cell counts in blown nasal secretions. They are easy to obtain, reproducible, reliable, and appear to be responsive to treatment. It ought to be possible to implement them in routine clinical practice to guide therapy.
Objective measurements are necessary to guide therapy. When the type of inflammation is guessed, it is usually wrong. Although this has not been directly investigated for rhinitis, it is not likely to be different from that in asthma.9,10 Rhinitis associated with nasal eosinophilia almost always responds to treatment with nasal corticosteroids or a leukotriene receptor antagonist. 11 Some patients with rhinitis that is not associated with eosinophilia may also respond to treatment with nasal corticosteroids. 12 Rhinitis associated with infective sinusitis and neutrophilia responds to antibiotics. Therefore, it is reasonable that the type of inflammation is measured to recommend appropriate therapy. Although there are a number of excellent ways of assessing nasal inflammation in research studies such as nasal lavage, brushing, scraping, biopsy, and nasal nitric oxide, 13 they are not usually used in routine clinical practice. Our observations of the excellent measurement properties of cell counts in dithiothreitol-dispersed nasal secretions suggest that spontaneously blown nasal secretions have a role in clinical practice. Although the intervention was not placebo controlled, all patients with raised eosinophil count improved with nasal fluticasone and all patients with raised neutrophil count, who also had a high “cold symptom score,” improved with zithromax. These need additional evaluation in a placebo-controlled clinical trial. The high responsiveness index suggests that there is a high signal-to-noise ratio in our measurements. Assuming that a 25% change in the percent eosinophil and neutrophil count is the minimal clinically significant difference, a clinical trial with −10 paired observations or 20 nonpaired observations would have 80% power to show the required difference with an α = 0.05. 14
This method may have a few disadvantages as well. This was a small study and the success rate was only 85%. The cells represent luminal cells and may not be a true representation of tissue inflammation. The yield of metachromatic cells is very low in blown nasal secretions. The reproducibility of neutrophil count was low. Nevertheless, it is useful to distinguish allergic from infective rhinitis and guide response to therapy. Because they are noninvasive and easy to perform without any technical assistance, they are easy to implement in routine clinical practice. Because we did not have a placebo-controlled arm to this study, the responsiveness index may lack precision. A prospective larger controlled clinical trial is necessary to validate these observations and to investigate whether routine measurement of nasal inflammation improves diagnosis of nasal disease and improves therapy and outcomes.
