Abstract
Prefrontal brain functions are reported to be involved in postural control. This study examined the correlation between a postural control task and a Stroop/reverse-Stroop test that also taps the prefrontal brain functions. Twenty healthy elderly people (3 men, 17 women) ages 61–81 years participated. Stroop interference and reverse-Stroop interference were measured. The postural control task required participants to stand as still as possible for 30 sec. with eyes open and then with eyes closed. Reverse-Stroop interference, which is reported to activate more prefrontal function, correlated positively with postural sway when standing with eyes closed. Prefrontal function may be an important component of postural control when sensory information is insufficient.
Postural control is involved in two important behavioral goals, postural orientation and postural equilibrium, whether in static or dynamic conditions. It is a complex process involving the sensation of position and motion from visual, somatosensory, and vestibular systems, the processing of the sensory information to assess orientation and movement, and the selection of motor responses that maintain or bring the body into equilibrium (Schmidt, 1975; Horak, 2006). Some features of postural control have been investigated in terms of the amount of motion of the center of pressure away from the equilibrium point: the greater it becomes, the less stable the posture is (Winter, 1995; Balasubramaniam & Wing, 2002; Horak, 2006).
Recent studies have suggested that significant attentional requirements exist for postural control, although such control has been traditionally regarded as an automatic or reflex-controlled task, suggesting that postural control systems demand minimal attentional resources (Woollacott & Shumway-Cook, 2002). The interaction between postural control and attention is investigated typically in dual-task paradigms in which the two tasks can mutually compete. Specifically, many studies have examined how postural control or gait affects the performance of concurrent cognitive tasks and demonstrated the decline in performance of either postural control or cognitive task relative to either task performed by itself, which is taken as evidence that the concurrent tasks compete for particular attentional resources (Dault, Geurts, Mulder, & Duysens, 2001; Barra, Bray, Sahni, Golding, & Gresty, 2006; Slobounov, Wu, & Hallett, 2006; Springer, Giladi, Peretz, Yogev, Simon, & Hausdorff, 2006; Hollman, Kovash, Kubik, & Linbo, 2007; Hirota, Watanabe, Sun, Tanimoto, Kono, Takasaski, et al., 2010; Olivier, Cuisinier, Vaugoyeau, Nougier, & Assaiante, 2010; Boonyong, Siu, van Donkelaar, Chou, & Woollacott, 2012; Ikeda, Okuzumi, & Kokubun, in press).
Particularly, earlier studies have implicated an important role of executive function or inhibitory control in postural control (Redfern, Jennings, Martin, & Furman, 2001; Reilly, van Donkelaar, Saavedra, & Woollacott, 2008; Yogev-Seligmann, Hausdorff, & Giladi, 2008; Redfern, Jennings, Mendelson, & Nebes, 2009). Executive function, including inhibitory control, refers to the goal-oriented regulation of one's own thoughts and actions based on brain activities primarily of the prefrontal cortex (Luria, 1966; Shallice, 1982; Lezak, 1983; Zelazo & Müller, 2002, 2011). Specifically, recent studies have suggested the potential involvement of a network of several brain areas related to executive function in postural control, including the parietal cortex, cerebellum, and two prefrontal cortices: the anterior cingulate cortex (ACC) and the dorsolateral prefrontal cortex (DLPFC). Although the role of the parietal cortex and cerebellum in postural control is still debated (e.g., Horak & Macpherson, 1996; Pellijeff, Bonilha, Morgan, McKenzie, & Jackson, 2006; Slobounov, et al., 2006; Mihara, Miyai, Hatakenaka, Kubota, & Sakoda, 2008), the role of the ACC and the DLPFC have been suggested. The ACC is regarded as playing an important role in detecting postural instability (Slobounov, et al., 2006) or signaling an error between expected and actual sensory information related to postural status (Jacobs & Horak, 2007). The DLPFC is regarded as playing an important role in adequate allocation of visuospatial attention (Mihara, et al., 2008).
One cognitive test that taps a component of executive function, i.e., inhibitory control, is the Stroop/reverse-Stroop test (Stroop, 1935; MacLeod, 1991; Ikeda, Okuzumi, Kokubun, & Haishi, 2011). In this test, individuals are presented with incongruent-color word stimuli, for instance the word red printed in blue ink. They are then requested to name the ink color while inhibiting the prepotent tendency of word-reading. The response time (RT) cost in color-naming of the incongruent stimuli versus neutral stimuli, such as a blue rectangle, indicates Stroop interference, also known as the Stroop effect. Reading the incongruent stimuli while inhibiting response to ink color elicits reverse-Stroop interference when compared with reading color words written in black, especially in a manual response, e.g., a paper-and-pencil version of the test (Flowers, 1975; Ikeda, Hirata, Okuzumi, & Kokubun, 2010; Song & Hakoda, 2011; Ikeda, Okuzumi, & Kokubun, 2013; Yasumura, Kokubo, Yamamoto, Yasumura, Nakagawa, Kaga, et al., in press). Higher Stroop/reverse-Stroop interference has been regarded as an index of lower inhibitory control. To resolve the Stroop/reverse-Stroop interference, a network of several brain areas including parietal cortex, cerebellum, and the two prefrontal cortices are inferred as involved, with the ACC detecting conflict between possible responses and the DLPFC allocating adequate attention (MacDonald, Cohen, Stenger, & Carter, 2000; Ruff, Woodward, Laurens, & Liddle, 2001; Botvinick, Cohen, & Carter, 2004; Kerns, Cohen, MacDonald, Cho, Stenger, & Carter, 2004).
In elderly adults, prefrontal brain activities have been suggested to be altered compared to young adults (Milham, Erickson, Banich, Kramer, Webb, Wszalek, et al., 2002). An earlier study has showed an increase in the ACC's sensitivity to the occurrence of competition between two sources of color information in the Stroop test (ink color and word identity). Elderly adults showed increased activity in the ACC even when they were faced with congruent-color word stimuli; for instance, the word red printed in red ink, which has no conflict but competition. Therefore, more activation in a network of brain areas, especially in the ACC, is expected for elderly adults in the Stroop test as well as in the reverse-Stroop test.
The purpose of this current study was to determine if inhibitory control as assessed by the Stroop/reverse-Stroop test is associated with postural control in healthy elderly adults. Considering a similarity of the prefrontal brain activities postulated between postural control and inhibitory control, individual differences in inhibitory control should relate to the efficiency of maintaining balance. Postural control was assessed by measuring sway with eyes open and eyes closed.
Hypothesis 1. Inhibitory control will be more strongly correlated with postural sway with eyes closed, that is, more unstable postural sway that could be derived from a greater error between expected and actual sensory information related to postural status.
Hypothesis 2. If the ACC is more activated in the reverse-Stroop interference task than in the Stroop interference task with manual response (Ruff, et al., 2001), the reverse-Stroop interference should have a stronger correlation with postural sway.
Hypothesis 3. Stroop/reverse-Stroop test and the postural control task will replicate prior well-known patterns (showing the data are valid).
Method
Participants
Twenty healthy elderly people (3 men, 17 women; M age = 70.8 yr., SD = 6.0, age range = 61–81) volunteered in this study. No participants had a prominent deficit in sensorimotor or cognitive skills based on their interview with a medical doctor.
Measures
Inhibitory control task. —The Stroop/reverse-Stroop Test (Hakoda & Watanabe, 2005; Song & Hakoda, 2011) was composed of four conditions in which color words and colored rectangles were printed on four separate sheets of paper: the Stroop interference condition, the reverse-Stroop interference condition, and the control conditions for each. In the reverse-Stroop control condition (Test 1), five words (“red,” “blue,” “yellow,” “green,” and “black” in Japanese) printed in black were presented one by one, requiring participants to make a choice from the five matching colored rectangles printed on the right side of the test sheet corresponding to the color the stimulus represented. In the reverse-Stroop interference condition (Test 2), the five words printed in a non-matching color ink (from among the same five colors) were presented one by one, requiring participants to make a choice from the five matching colored rectangles printed on the right side of the test sheet corresponding to the color the stimulus represented. In the Stroop control condition (Test 3), five colored rectangles (red, blue, yellow, green, and black) were presented one by one, requiring participants to choose from the five matching color words printed in black on the right side of the test sheet corresponding to the color of the stimulus. In the Stroop interference condition (Test 4), the five words printed in a non-matching color ink (from the same five colors) were presented one by one, requiring participants to make a choice from the five matching color words printed in black on the right side of the test sheet corresponding to the ink color of the stimulus.
Postural control task.—Postural sway was measured as an index of postural control. On a strain-gaugeforce platform (EB1101; NEC Corp., Tokyo, Japan), participants were asked to stand as still as possible with their feet together (Romberg stance) for 30 sec., with eyes open and eyes closed, respectively. When their eyes were open, the participants stood 1 m from a wall and fixated on a small object on the wall. When their eyes were closed, both eyes were covered with a blindfold.
Design
Participants were tested individually in a well-lit, quiet room at a university. Before the assessment session, participants were confirmed not to have color vision deficiency by a preliminary test which required them to match color words with colored rectangles in five colors: “red,” “blue,” “yellow,” “green,” and “black” (otherwise, a participant was not administered the tasks any further and excluded from participating in this study). This preliminary test was conducted for a single trial and found one participant who had color vision deficiency. Then, each participant was administered the Stroop color-word test and the postural control task sequentially. The order of the two tasks was counterbalanced among participants.
Data Analysis
Stroop color-word test.—Each test sheet consisted of 10 practice trials and 100 test trials. The participants were asked to perform the practice and test trials as fast and as accurately as possible for 10 and 60 sec., respectively. For each test, the quantities of items completed and the correct responses out of 100 test trials were recorded. As indices of inhibitory control, the interference scores were calculated using the formula shown below:
Postural control task.—The stationary postural sway was analyzed for 20.48 sec. of the total 30 sec. (Baumberger, Isableu, & Flückiger, 2004), clipping off both the beginning (9 sec.) and the end (0.52 sec.). Both lateral and foreaft sways were conditioned before conversion to digital signals through an analog to digital (A/D) converter (NR-2000; Keyence Co.) at a sampling frequency of 100 Hz. As indices of postural sway, total two-dimensional (planar) sway length (cm) and sway area surrounding maximal amplitude rectangle (cm2) were calculated (Shumway-Cook, Woollacott, Kerns, & Baldwin, 1997; Okuzumi, Furuna, Nishizawa, & Sugiura, 2000).
Statistical Analysis
The data of the Stroop/reverse-Stroop test and the postural control task were analyzed using an analysis of variance (ANOVA). Specifically, in the Stroop/reverse-Stroop test, two-way ANOVAs with within-participant factors of task (Stroop and reverse-Stroop) and condition (interference and control) were conducted for number of items completed and number of correct responses, and one-way ANOVA with within-participant factor of task (Stroop and reverse-Stroop) for the interference score. One-way ANOVAs with within-participant factor of condition (with eyes open and closed) were conducted for the sway area and sway length. Moreover, Pearson product moment correlations between the performances and age were examined to see if the data replicated prior findings. Then, correlational analysis between the Stroop/reverse-Stroop test and the postural control task was conducted to assess whether individual differences in inhibitory control correlated to the efficiency of maintaining balance. For statistical analyses, SPSS 19.0 for Windows (SPSS Japan Inc., Tokyo, Japan) was used. The .05 level of significance was adopted throughout all statistical analyses.
Ethical Approval
The experimental protocol was administered in accordance with the guidelines of the Declaration of Helsinki and approved by the institutional review board. Informed consent was obtained from all participants before the assessment session. There was no participant who showed any hesitancy in performing the task.
Results
In the Stroop/reverse-Stroop test, the participants showed very accurate performances. Because errors were few and not normally distributed, the analysis of errors was not conducted further. Table 1 presents means and standard deviations of performances for the Stroop/reverse-Stroop test and the postural control task. Here, 2 (task; Stroop and reverse-Stroop) × 2 (condition; interference and control) ANOVAs were conducted for the number of items completed and the number of correct responses, respectively. For the number of items completed, analysis showed a statistically significant main effect for task (F1,19 =212.03, p < .001; partial η2 = 0.92) and for condition (F1,19 =37.05, p < .001; partial η2 = 0.66), but not for interaction between the task and condition (F1,19 =2.45, ns). For the number of correct responses, the analysis showed a statistically significant main effect for the task (F1,19 = 179.14, p < .001; partial η2 = 0.90) and for the condition (F1,19 = 39.20, p < .001; partial η2 = 0.67), but not for interaction between the task and condition (F1,19 =2.73, ns). These results showed that participants responded faster to the reverse-Stroop interference task than the Stroop interference task. Results also show interference in both the Stroop and reverse-Stroop tasks. The results of one-way ANOVAs conducted for the interference scores support the result presented above. There was no statistically significant difference between the Stroop and the reverse-Stroop interference scores (F1,19 = 0.33, ns). In the postural control task, because of concern about violations of normality assumptions for ANOVA, compliance of residuals to a normal distribution was evaluated by Kolmogorov-Smirnov goodness-of-fit tests. The sway area and sway length did not differ significantly from a normal distribution. Then, the sway area and sway length were compared between conditions (with eyes open and closed) using one-way ANOVA. Results showed a statistically significant effect for condition (sway area: F1,19 = 26.77, p < .001, partial η2 = 0.59; sway length: F1,19=29.46, p < .001, partial η2 = 0.61). Postural sway was greater when standing with eyes closed than with eyes open.
Means and Standard Deviations of Task Performances and Their Correlation With Age

Note Postural stability variables (sway area and total sway length) were taken from center of pressure. *p < .05.
Table 1 presents Pearson product-moment correlation coefficients between the performances and age. For the Stroop/reverse-Stroop test, age correlated with number of items completed and with number of correct responses across conditions. For the postural control tasks, age was not correlated with any performance.
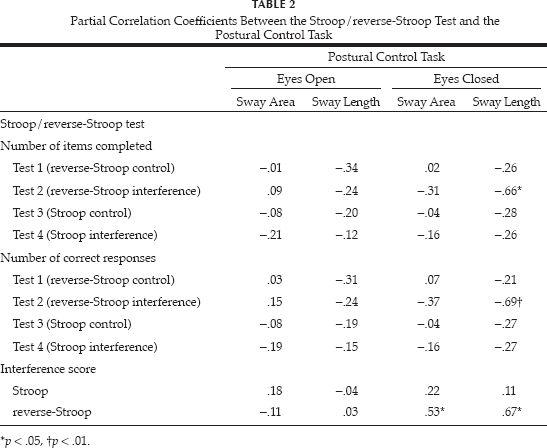
Table 2 presents the partial correlations, controlled for age, between performances of the Stroop/reverse-Stroop test and the postural control task. The sway length of the eyes-closed condition correlated negatively with the number of items completed and correct responses of the reverse-Stroop interference condition, and positively with the reverse-Stroop interference score. The reverse-Stroop interference score also correlated with the sway area of the eyes-closed condition. No other comparison was statistically significant.
Partial Correlation Coefficients Between the Stroop/reverse-Stroop Test and the Postural Control Task
p < .05,
p < .01.
Discussion
This study investigated if inhibitory control is associated with postural control in healthy elderly adults, considering a similarity of the prefrontal brain activities postulated between these controls. This study examined whether the data replicated prior findings. Then, this study examined whether postural sway when standing with eyes open and closed correlated with performances of the Stroop/reverse-Stroop test, which taps inhibitory control.
Performances on the Stroop/reverse-Stroop test were mostly in line with those of previous studies. This study showed better performance on the reverse-Stroop task than the Stroop task and robust Stroop and reverse-Stroop interference, as in a study conducted by Ikeda, et al. (2010). The negative correlation between age and the number of items completed and the number of correct responses accords with results of a previous study showing that responses got slower with age in older adults aged between 60 and 80 (Houx, Jolles, & Vreeling, 1993). In contrast, age did not correlate with the interference scores, i.e., inhibitory control, which is inconsistent with the result of Houx, et al. (1993) that demonstrated the Stroop interference score increased with age. This result might be a characteristic of healthy elderly adults, given that the age-related trend was weakened by controlling health-related factors that are purported to play an important role in age-associated decline in performance (Houx, et al., 1993).
The performances of the postural control task were also mostly in line with previous studies, which have shown that postural sway is greater when standing with eyes closed than with eyes open (Dornan, Fernie, & Holliday, 1978; Hageman, Leibowitz, & Blanke, 1995). Inconsistent with an earlier study that documented increased postural sway with age (Okuzumi, et al., 2000), age did not correlate with any index of postural sway. This result might be attributable to the small sample, given that positive but small correlation was found for all indices of postural sway.
Consistent with the hypotheses, there was a significant relationship between postural control with eyes closed and inhibitory control of the reverse-Stroop interference. It should be emphasized that the basis of this relationship is not unknown. However, a possible explanation relates to the role of the ACC. In the Stroop paradigm, the ACC is involved in the detection of conflict in potential responses (color-naming versus word-reading), triggering compensatory adjustments to resolve the Stroop and reverse-Stroop interferences based on activities in the DLPFC (MacDonald, et al., 2000; Botvinick, et al, 2004; Kerns, et al., 2004). In an fMRI study using the Stroop and reverse-Stroop tests with manual response (a button press), Ruff, et al. (2001) reported that the ACC was more activated in the reverse-Stroop interference task than in the Stroop interference task. On the other hand, the ACC is believed to be involved in postural control. The ACC is regarded as playing an important role in detecting postural instability (Slobounov, et al., 2006) or signaling an error between expected and actual sensory information related to postural status (Jacobs & Horak, 2007). In fact, recent research points to the overall role of the ACC in conflict monitoring across cognition and action (Botvinick, et al., 2004).
The fact that inhibitory control related with postural sway when eyes were closed, but not postural sway when eyes were open may facilitate understanding of the role of the ACC in postural control. Postural control via the ACC activities may be necessary when visual information is not available for the sensation of position and motion and the processing of the sensory information to assess orientation and movement. In other words, attentional control may not be so necessary for postural control when visual information is at hand because our balance strongly depends on visual information (Soechting & Berthoz, 1979; Remaud, Boyas, Caron, & Bilodeau, 2012; Mohapatra & Aruin, 2013).
Limitations of this study should be noted. This study used a relatively small sample in which most of the participants were female. Future research is expected to use more diverse samples with balanced gender ratio. Moreover, further research must be undertaken to identify the underlying mechanisms that explain the relationship between postural control and inhibitory control, e.g., in a dual-task paradigm in which an auditory inhibitory control task competes against postural control task with little visual information.

