Abstract
A general characteristic of Parkinson's disease (PD) is the inability to adapt and change set quickly. The goal of this study was to assess whether PD affects performance in which changing set occurs over minutes, i.e., a slow and continuous form of sensorimotor set-change. Recovery from the postural lean aftereffect following prolonged stance on an inclined surface (Experiment 1) was tested to see if the addition of light-touch tactile feedback from the fingertips during inclined stance increased the aftereffect (Experiment 2). The percentage of responders in healthy Young, Older, and PD groups was similar, as were characteristics of the recovery towards vertical stance, namely the initial forward lean, range, time constant, and half-life. Tactile feedback increased the responder rate in all groups. A novel response was also observed in which the aftereffect did not dissipate; i.e., participants remained leaning forward throughout the post-incline stance period. PD does not appear to affect the ability to change sensorimotor set if the change is slow.
Recent studies have suggested that the basal ganglia form the critical neural substrates for changing set, a concept that refers to the internal (neural) constraints that compel cognitive or motor behaviors. The constraints reflect the preparatory state of the nervous system as influenced by contexts such as experience, expectation, instructions, or postural configuration (Cordo & Nashner, 1982; Prochazka, 1989). The context prompts the preferred motor or cognitive response even if the presenting stimuli may be similar across trials (Evarts & Tanji, 1974). For example, in the Wisconsin Card Sorting Test, a person is given a deck of cards to sort, either by color or by shape. Knowing which category to sort on depends on the tester's cues. The participant must sort using a different category when the tester prompts a change. Changing set thus refers to the ability to self-initiate a change of response at appropriate moments. When the context is different, set must change accordingly. Failure to do so in a timely manner reflects a breakdown in the set-changing mechanisms.
Individuals with Parkinson's disease (PD), the most widely studied human model of the basal ganglia system, have been found to exhibit set-changing difficulties in many types of cognitive and motor activities including delayed reaction time in executive control (Hayes, Davidson, Keele, & Rafal, 1998; Cools, Barker, Sahakian, & Robbins, 2001) independent of bradyphrenia (slowing of thought) (Rafal, Posner, Walker, & Friedrich, 1984), slower upper extremity perceptual visuomotor adaptation (Contreras-Vidal, Teulings, Stelmach, & Adler, 2002), and slower adaptation to changing task conditions during automatic stance postural control (Horak, Nutt, & Nashner, 1992; Chong, Horak, Frank, & Kaye, 1999; Chong, Jones, & Horak, 1999; Chong, Horak, & Woollacott, 2000), sitting (Horak, et al., 1992; Chong, Jones, et al., 1999), and rising onto the toes (Chong, Jones, et al., 1999; Frank, Horak, & Nutt, 2000). Set-changing difficulties are not prominent in people with Alzheimer's (Chong, Jones, et al., 1999) or cerebellar disorder (Horak & Diener, 1994; Ravizza & Ivry, 2001), suggesting that the basal ganglia are critically involved in changing set.
In PD, the ability to change set is facilitated by external stimuli (Rafal, et al., 1984, Horak, et al., 1996; Burleigh-Jacobs, et al., 1997). For example, focusing on horizontal lines or contrasting features on the floor may improve gait initiation and/or overcome freezing of walking (Stern, Lander, & Lees, 1980; Chong, Lee, Morgan, Mehta, Griffin, Merchant, et al., 2011). Thus, testing the ability to change set in PD is best studied under conditions in which there is ambiguity or an absence of a distinct external stimulus or rule for guiding action. Without a preceding stimulus that unambiguously specifies what the response should be, a change in set must consequently be self-initiated and willful. This is akin to declarative/explicit as opposed to procedural/implicit learning (Redding & Wallace, 1996; Teulings, Contreras-Vidal, Stelmach, & Adler, 2002; Fernandez-Ruiz, Diaz, Hall-Haro, Vergara, Mischner, Nunez, et al., 2003). Since set is a reflection of the current state of the brain, it is more difficult to alter the set if it needs to occur quickly. The reaction time or the number of trials to change set is used to infer the efficiency of the set-changing mechanism (Woollacott, Shumway-Cook, & Nashner, 1986; Hansen, Woollacott, & Debu, 1988; Horak, et al., 1992; Hayes, et al., 1998; Chong, Horak, & Woollacott, 1999; Chong, et al., 2000).
The concept of set is extensive (Prochazka, 1989). It is unlikely that the basal ganglia are involved in all set-changing activities (Hayes, et al., 1998; Aron, Watkins, Sahakian, Monsell, Barker, & Robbins, 2003; Monsell, 2003). For example, one aspect of the sensorimotor set-changing hypothesis for postural orientation that has received little attention is whether individuals with PD are impaired when the change occurs over the course of several minutes rather than within seconds or within one trial. A task in which the time course to change sensorimotor set occurs over such an extended period may present an opportunity to further explore the concept and mechanisms of changing set. In the current study, the nature of changing set was examined in relation to postural orientation. It has been shown that when healthy young adults were subjected to prolonged stance on an inclined surface and subsequently returned to a level surface, an aftereffect was observed in some, whereby they stood leaning for several minutes. The lean occurred in the direction of the incline, e.g., standing on an upslope resulted in a forward lean. During this time, participants gradually drifted back toward baseline vertical posture (Kluzik, Horak, & Peterka, 2005).
The effect of light-touch fingertip tactile feedback during incline stance was also explored. Tactile feedback does not appear to improve reaction time or force production for balance control. However, it may improve or augment the central integration of proprioceptive inputs (Dickstein, Peterka, & Horak, 2003), i.e., thought to be defective in PD (Machado, Cunha, Velasques, Minc, Teixeira, Domingues, et al., 2010). Anecdotal and empirical findings suggest that individuals with PD move more slowly or require more trials to modify their responses in part to compensate for their inability to change set quickly, but can do so if speed is not critical.
Method
Participants
Healthy young and elderly persons, and individuals with Parkinson's disease participated in the study which was approved by the institutional review board. Experiment 1 comprised a convenience sample of 50 participants. Thirteen participants (10 men, three women) were recruited based on a diagnosis of idiopathic Parkinson's disease (PD, Table 1) (Hughes, Daniel, Kilford, & Lees, 1992). Four participants were rated at 1 or 2 on the Hoehn and Yahr (H&Y) disease severity scale (Goetz, Poewe, Rascol, Sampaio, Stebbins, Counsell, et al., 2004). The remaining nine participants were rated between 2.5 to 4. All PD participants participated in the study while taking their usual anti-PD drug regimen. Fourteen age-matched participants who reported no significant musculoskeletal or neurological impairments formed the Older control group (five men, nine women; M age = 65 yr., SD = 7). The remaining 23 participants served as the Young control group (11 men, 12 women; M age = 25 yr., SD = 2). Mean height and foot length were not different among the groups.
Clinical Profiles of Participants in Parkinson's Disease Group

Experiment 2 consisted of 34 participants. Six were individuals with PD (four men, two women; see Table 1). Three subjects were rated at 2 on the H&Y, one was rated at 2.5, and two were rated at 3. The Older group consisted of eight individuals (six men, two women; M age = 71 yr., SD = 6.3). The remaining 20 participants (10 men, 10 women; M age = 25.2 yr., SD = 1.7) served as the Young group.
Experiment 1
Participants stood quietly on a 5° inclined board (toesup) for 5 min. Vision was occluded with a blindfold. Tapes were placed around the participant's feet to ensure consistency. After the 5-min. period, the incline board was removed and participants continued standing on the level surface for the next 3 min. still wearing the blindfold. Blocking vision is critical for studying the adaptation of postural orientation, as vision provides a frame of reference that significantly interferes with the involuntary lean aftereffect (Earhart, Henckens, Carlson-Kuhta, & Horak, 2010). Vertical ground reaction forces (100 Hz) were collected 5 sec. after the participants were returned to level stance (Kluzik, et al., 2005) (Figure 1). Center of pressure was then calculated from the force data to quantify each participant's antero-posterior (A/P) body sway. Data underwent a 0.1 Hz low-pass filter to analyze the aftereffect (i.e., slow drift back to upright posture) and differentiate it from higher-frequency physiological tremors and corrections of postural sway (Kluzik, et al., 2005; Kouzaki & Masani, 2012).
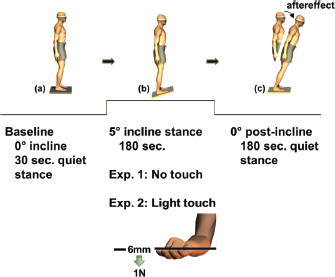
Illustration of the slow set-change protocol: In Experiment 1, (a) after quiet baseline stance (b) participants stood on a 5° incline board for 3 min.; (c) the board was then removed and participants stood on a level surface for another 3 min. The forward leaning posture in the figure illustrates the involuntary lean aftereffect as a result of the preceding stance on the inclined board. Participants wore a blindfold with eyes closed throughout the protocol to prohibit them from using vision to correct their lean aftereffect. In Experiment 2, participants lightly touched a rigid board (1N, 6mm thickness) during the inclined stance. They also underwent the protocol described in the first experiment as the control condition.
Participants also underwent the standard Sensory Organization Test (SOT) protocol to determine whether responders (i.e., those who displayed the aftereffect) were different from non-responders (see Analyses section below). The SOT protocol comprises six conditions with permutations of eyes open, eyes closed, fixed/sway-referenced support surface, and fixed/sway-referenced visual surround (NeuroCom International Inc., Clackamas, Oregon) (Nashner, 1997; Chong, Mills, Dailey, Lane, Smith, & Lee, 2010; Figure 2). In the sway-referenced conditions, the floor and/or surround rotated in proportion to A/P body sway. For example, if the participant swayed forward by 1° (at the ankles), the floor and/or the surround will rotate downward by a corresponding 1°. The proportionate rotation of the floor results in less than optimal input to the somatosensory system and the participant incorrectly perceives that there is little or no postural sway. Similarly, by sway-referencing the surround, inputs from the visual system no longer accurately specifies how the body is swaying. Each condition was tested three times in a block in ascending order of difficulty, from baseline quiet stance to moving floor and surrounding walls (in proportion to body sway) (Chong, 2013). The upper (anterior) and lower (posterior) limit of A/P sway over each 20-sec. trial was used to infer the postural sway range for that trial. Each sway value was converted to a standardized index by expressing it as a function of the theoretical maximum sway range of 12°. A higher value indicates better postural control performance, while a low score (i.e., high sway range) indicates difficulty with postural control. Participants received a score of zero in a trial if they moved their foot/feet out of position, opened their eyes in conditions 2 or 5 (eyes-closed conditions) or reached out and touched the surround to catch their balance.
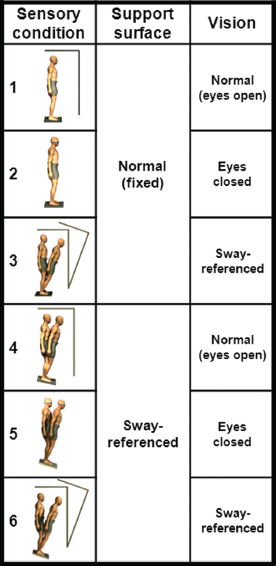
The six conditions of the Sensory Organization Test protocol
The SOT was also administered to the Young and Older groups to assess whether there may be a preference to rely on the somatosensory system in some participants or the vestibular system in others for whole-body postural orientation during the period of incline stance (Kluzik, et al., 2005). A “proprioceptive” deficit in the case of PD and reliance on the somatosensory system in the healthy groups should produce less favorable performances in SOT conditions 2 and 3, while reliance on the vestibular system should result in poorer performances in conditions 5 and 6 (Nashner, 1997). The order of the SOT and incline stance tests was counterbalanced among the participants.
Experiment 2
It has been suggested that there may be a deficiency in mapping proprioceptive inputs for motor control in individuals with PD (Konczak, Corcos, Horak, Poizner, Shapiro, Tuite, et al., 2009). To assess the possibility of an association between adaptation to prolonged incline surface stance and proprioceptive deficits, participants underwent a one-session counter-balanced cross-over protocol in which they lightly touched the under-surface of a 6-mm-thick 1 N rigid cardboard with their fingertips of one hand while standing on the incline (Figure 1). The participants were continuously monitored to ensure that they did not exceed the 1 N vertical force as evidenced by a slack in the twines that suspended the board. Although small movements of the board were inevitable, the board provided a significant source of somatosensory augmentation to stabilize body sway (Oshita & Yano, 2013). Participants also underwent the protocol described in Experiment 1, which served as the control condition. The period of the incline stance was shortened to 3 min. (from 5 min. in the first experiment) to avoid fatiguing the PD participants.
Analyses
PD participants in the earlier (H&Y 1–2) and later stages (H&Y 2.5–4) of the disease condition did not differ in their postural incline performance. Their data were therefore combined for analysis as the PD group. Initial analyses of postural adaptation data in all the three groups showed that some participants showed a lean aftereffect (responders) whereas others did not (non-responders). This observation was similar to previous findings in which non-responders were participants whose average baseline sway was not different from their post-incline sway, indicating that they did not show a lean aftereffect (Kluzik, et al., 2005).
Among the responders, a first-order exponential or polynomial least-squares ordinary fit for each participant followed by independent one-way analyses of variance (ANOVA) for main effect of Group were carried out to characterize the aftereffect, namely the initial forward lean amplitude following incline stance, the range of the antero-posterior (AP) sway amplitude, time constant (τ), and half-life (t½). Time constant and half-life were not applicable in eight of the 30 responders whose aftereffect approximated a polynomial straight line trajectory instead (Kluzik, et al., 2005). Chi-squared statistics were used to compare the frequency count of responders among the groups. Mann-Whitney U tests were used to compare the average sway performance in each of the six SOT conditions between the responders and non-responders within each group of participants. The alpha level for a significant outcome was set at p < .05 for all analyses. Data are expressed as mean ± SE.
Results
Experiment 1
Thirty of the total 50 participants (60%) which comprised the three groups showed the lean aftereffect. The responder rates were 56.5% in the Young group, 57% in the Elderly group, and 69% in the PD group. The chi-squared test of independence was not significant [χ2 (2, N = 50) = 0.25, p = .88], indicating that the number of responders in each group did not differ from the expected responder rate of 60%.
Figure 3 shows the post-incline adaptation of the responders among the Young, Elderly, and PD groups. Following a similar initial forward lean at the beginning of the post-incline stance, all groups also showed a similar trend towards returning to upright stance over the next 3 min. The range of the A/P forward lean magnitude was 2 cm (SE = 0.2). The time constant (τ) was 39.6 sec. (SE = 8.3) and half-life (t½) was 27.5 sec. (SE = 5.7). The coefficient of determination, R2, which represents the proportion of the variance in the aftereffect, i.e., explained by the predictor variable (time) was 0.44 (95% CI = 0.36–0.52). The PD group presented an average of 0.5 cm anterior shift of the center of pressure compared to the Young and Elderly groups, as seen in Fig. 3. This may be due to their tendency to spontaneously adopt a more anterior posture (Blaszczyk, Orawiec, Duda-Klodowska, & Opala, 2007).

Post-incline adaptation of the responders among the Young, Elderly and PD groups. Each trace represents the mean anteroposterior (A/P) center of pressure (CoP) sway of the group. All groups showed a similar trend towards returning toward baseline stance following the initial forward lean (aftereffect). The grey bar represents the 95% CI of the half-life range of the first order exponential decay averaged across the three groups (15.8–39.2sec.).
There was no difference between the responders and non-responders in the SOT within each group.
Experiment 2: Effect of Light-touch Tactile Feedback
Light-touch tactile feedback from the fingertips increased the responder rate from 18 to 26 out of 34 participants in the three groups (52.9 to 76.5%) [χ2 (2, N = 34) = 6.53, p = .04, φc = 0.44]. Standardized residual analyses indicated that the Elderly group contributed the most to the significance of the χ2 test (2.1), followed by the PD (1.2) and the Young (0.83) groups.
Participants showing a novel response during the tactile feedback condition were referred to as touch-responders. They displayed the forward lean aftereffect similar to the responders in Experiment 1, but they did not drift back to vertical posture during the 3 min. of post-incline stance on the level surface (Fig. 4). In the Young group (n = 20), there was an increase from 13 responders in the No-touch condition to 16 responders in the Touch condition (from 65% to 80%), of which 4 were touch-responders. In the Elderly group (n = 8), the frequency of responders increased from two to five (from 25% to 62.5%) including two touch-responders. In the PD group (n = 6), the number of responders increased from three to five participants (from 50% to 83.3%).
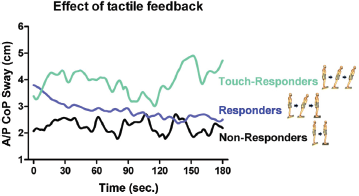
Traces are averaged data from the Young, Elderly, and PD groups. Non-responders were participants who did not display the post-incline aftereffect. Responders were participants who displayed the aftereffect and gradually drifted back to upright posture. Touch-responders were those who displayed the aftereffect during the entire post-incline period and did not return to upright posture.
Discussion
The results of the current study suggest that the PD group was not affected by a slow sensorimotor set change in postural orientation. It has been posited that responders (i.e., participants who displayed the lean aftereffect) tend to align their postural orientation more with the somatosensory system, whereas non-responders aligned their orientation more with the vestibular system (Kluzik, et al., 2005). Although the SOT failed to detect such sensory patterns, the aftereffect appeared to originate in part from modifications to central integrative areas of the brain. Healthy participants exhibited the aftereffect by bending at their hips when their lower extremities were kept vertical by a rigid bar that was placed across the greater trochanter of the hip (Kluzik, Peterka, & Horak, 2007). Future research should include testing participants with other types of dysfunction such as vestibular and somatosensory disorders to examine the responder vs non-responder postural adaptation to prolonged incline stance.
Effect of Prolonged Inclined Stance
In conjunction with this, it has been suggested that postural control can be affected by prolonged stretching (Chong & Do, 2002; Vaugoyeau, Hakam, & Azulay, 2011) and that “proprioception,” or its integration with other sensory systems, is decreased in PD (Dietz, 2002; Abbruzzese & Berardelli, 2003; Seiss, Praamstra, Hesse, & Rickards, 2003; Jacobs & Horak, 2006; Konczak, et al., 2009; Tagliabue, Ferrigno, & Horak, 2009) which may be worsened by anti-PD mediations (O'Suilleabhain, Bullard, & Dewey, 2001). The PD group in the current study was tested while taking their usual medications. This should have resulted in a significantly higher occurrence of non-responders in the PD group as they would have been more reliant on the vestibular system for postural orientation. Based on the characteristics of the aftereffect, no evidence was found in support of the so-called proprioception deficit hypothesis in neither the current nor previous studies (Bronstein, Hood, Gresty, & Panagi, 1990; Chong, Jones, et al., 1999; Bronte-Stewart, Minn, Rodrigues, Buckley, & Nashner, 2002).
Other studies have, however, reported opposite results. In a study by Jenkinson and colleagues, participants moved their hands inside a mirror box in which the reflected and moving hands moved in opposite directions while the obscured and visible hands moved in the same direction. PD participants with no anosognosia of dyskinesia described the incongruent movements as feeling stranger than the congruent movements, whereas those with anosognosia reported that all movements felt equally strange (Jenkinson, Edelstyn, Stephens, & Ellis, 2009). In another study, PD participants were unable to distinguish between one vs two flexions of the index finger or wrist as accurately as age-matched controls when the time interval between pairs of muscle stimulations was very short (Fiorio, Stanzani, Rothwell, Bhatia, Moretto, Fiaschi, et al., 2007). PD patients also showed a smaller magnitude of under-shooting error when the wrist flexor was vibrated while performing an extension at the wrist (Rickards & Cody, 1997). The under-shooting was not due to reciprocal inhibition of the wrist extensor muscles. A similar finding of vibration-induced under-shooting was observed during ankle dorsiflexor movements (Khudados, Cody, & O'Boyle, 1999). The above studies suggest that PD does affect the processing or integration of proprioceptive inputs in some instances.
Parkinson's disease does not appear to affect the time course or nature of the aftereffect observed in the current study. One possible reason relevant to the set-changing hypothesis is that the proprioceptive deficits observed in previous studies occurred where either the participant's performance was measured within a short and discrete trial or the experimental stimulus was brief. This is quite different from the task investigated in the current study, which involved a long-lasting stimulus of standing on an incline, and an equally long-lasting adaptive response, i.e., expression and dissipation of the aftereffect. It may be that this type of postural adaptation does not significantly involve the basal ganglia (Forgacs & Bodis-Wollner, 2004). Instead, other neural substrates such as the cerebellum (Martin, Keating, Goodkin, Bastian, & Thach, 1996) may encompass this sort of postural adaptation.
Effect of Light-touch Tactile Feedback
The results of the second experiment also lend some support to this conjecture. Like the Young and Elderly groups, sensory augmentation increased the lean aftereffect in the PD group. However, it is not clear why there were no touch-responders among the PD participants. This needs further investigation, as the experiments were not designed to answer this question. It may be that this type of postural adaptation reflects the ability to maintain the aftereffect, i.e., maintaining a new sensorimotor set. If so, then the results are somewhat consistent with the suggestion that some PD individuals have trouble maintaining a new set (Flowers & Robertson, 1985; Robertson & Flowers, 1990). When required to maintain a set by applying the same mental strategy or motor sequence, PD individuals showed erratic behavior by switching to the previous organization of responses at inappropriate times. This was not due to problems with memory, distraction, or judgment, as they were able to reproduce the required responses when external cues were provided or when a non-switching stable response was requested.
Although the tactile feedback provided in the current study appeared to have been incorporated into the central sensory integrative mechanisms, the relatively weak sensorimotor set meant that PD participants would more easily drift back to upright stance (Chong, et al., 2000). There was a trend that this may have been the case in the current study with some PD participants. In the first experiment, the aftereffect in the PD group showed a rapid initial decline in the anterior displacement of the body center of pressure (Fig. 1).
General discussion
The ability to stand upright requires effective integration of visual, somatosensory, and vestibular inputs to create a reference frame when adapting to postural changes (Horak & Macpherson, 1996). Many patients with PD who are referred to physical therapy present with rounded shoulders, hunched posture, and positioning their center of gravity more anteriorly than normal (Blaszczyk, et al., 2007). The anterior displacement of body mass toward the limit of stability increases the risk of falling. The finding that adaptation to stance on an inclined surface is not detrimental in PD suggests that standing on an angled surface may encourage patients to learn to re-adjust their postural alignment following return to a level surface. For example, placing patients on a toes-down slope may help to reverse their forward lean and encourage a more vertical posture. Since their center of gravity is displaced anteriorly, the adaptation should not cause an abnormally extended (backward) posture. Instead, it should encourage standing up straight and centering their center of mass safely within their base of support. This change in posture will hopefully lead to a decreased risk of falls and improved postural control.
The ability to adapt to slow set changes in PD patients may also be translated for use in the clinic when treating this population. Education on taking their time while changing directions when walking or when participating in faster forms of set-changing activities such as transitioning from sit-to-stand or walk-and-turn may help to reduce episodes of freezing and instability (Chong, Barbas, Garrison, Herolz, Teheng, & Sethi, 2001). This principle of slowing down movements may be applied to improving other activities, e.g., those that are specifically evaluated within the Berg Balance or Tinetti instruments, in which the time taken to complete a movement is not used to grade the patient's performance, thereby improving outcome scores while also improving balance control and functional independence in daily living. Freezing of gait which can be exacerbated as a result of frustration and may be alleviated by reducing the patient's stress and apprehension through the use of slower movement strategies rather than attempting to restore normality. By affording adequate time to complete a task, they may assist with compensatory strategies by switching to another substrate, possibly the cerebellar system (Ouchi, Okada, Yoshikawa, Nobezawa, & Futatsubashi, 1999). Over time, the internal representation of the postural orientation may undergo a postural sensorimotor transformation which may be shaped by parietal and cerebellar circuitries including the cerebrocerebellar, spinocerebellar and vestibulocerebellar tracts (Ghez & Thach, 2000).
It remains to be determined whether long-term training in sensory augmentation may be an effective technique for postural adaptation in individuals with PD. Overall, the results of the current study should prompt a confluence of descriptive and hypothesis-based studies to better understand how the proprioceptive system affects rapid vs extended postural adaptations in Parkinson's disease.
