Abstract
Minivelle® (Noven Therapeutics, LLC, FL, USA) is an estradiol transdermal delivery system that has recently been approved in the USA for prevention of postmenopausal osteoporosis. The decline in estrogen during menopause leads to bone resorption, increasing the risk of fractures. Transdermal estradiol has been shown to increase bone mineral density. Safety studies of transdermal estradiol have shown a decreased risk in cardiovascular disease as compared with oral estrogen therapy. Minivelle is currently the smallest available transdermal estradiol patch, providing the lowest effective dose of estrogen.
Osteoporosis, the most prevalent bone disorder [1], affects 30% of postmenopausal women in the USA [2]. The dramatic reduction in estrogen after menopause is associated with rapid bone loss, highlighting the influence of hormonal changes on bone density [3]. During menopause, serum 17β-estradiol levels decrease by 85–90% [4]. This leads to an imbalance between bone resorption and bone formation [4]. Estrogen deficiency also increases receptor activator of RANKL, which leads to an increase in osteoclast recruitment and activation and subsequent bone loss [4,5]. Postmenopausal osteoporosis is characterized by compromised bone strength, impaired bone architecture and an increased risk of fracture [1]. Osteoporotic fractures are associated with a high level of morbidity and mortality [3]. Hip fractures can cause serious disability, increasing the risk of mortality by 25% within 1 year [1]. Therefore, prevention of fracture is of extreme importance.
Prevention of osteoporotic fractures in postmenopausal women has been demonstrated with oral estrogen therapy, and the significant effects on bone mineral density (BMD) of both oral and transdermal therapies have been well established [6]. Minivelle® (Noven Therapeutics, LLC, FL, USA), a transdermal estradiol patch, previously indicated for the treatment of moderate-to-severe vasomotor symptoms associated with menopause, has recently been approved for the prevention of osteoporosis. This approval was based on a study conducted with Vivelle®, a transdermal estradiol patch, deemed bioequivalent to Minivelle in a Phase I study [7].
Overview of the market
Several therapies are available for osteoporosis prevention in postmenopausal women at high risk of fracture. Currently, hormonal therapy is not considered first line; rather, nonhormonal options, such as the bisphosphonates, are recommended [1]. Estrogen has known risks, which has limited its use as a preventative measure, and is usually therefore only considered for women with a high risk for osteoporosis who are not candidates for the other available therapies [3], or at-risk women less than 60 years of age or within 10 years of menopausal onset [8].
Characteristics of current US FDA-approved therapies for postmenopausal osteoporosis prevention.
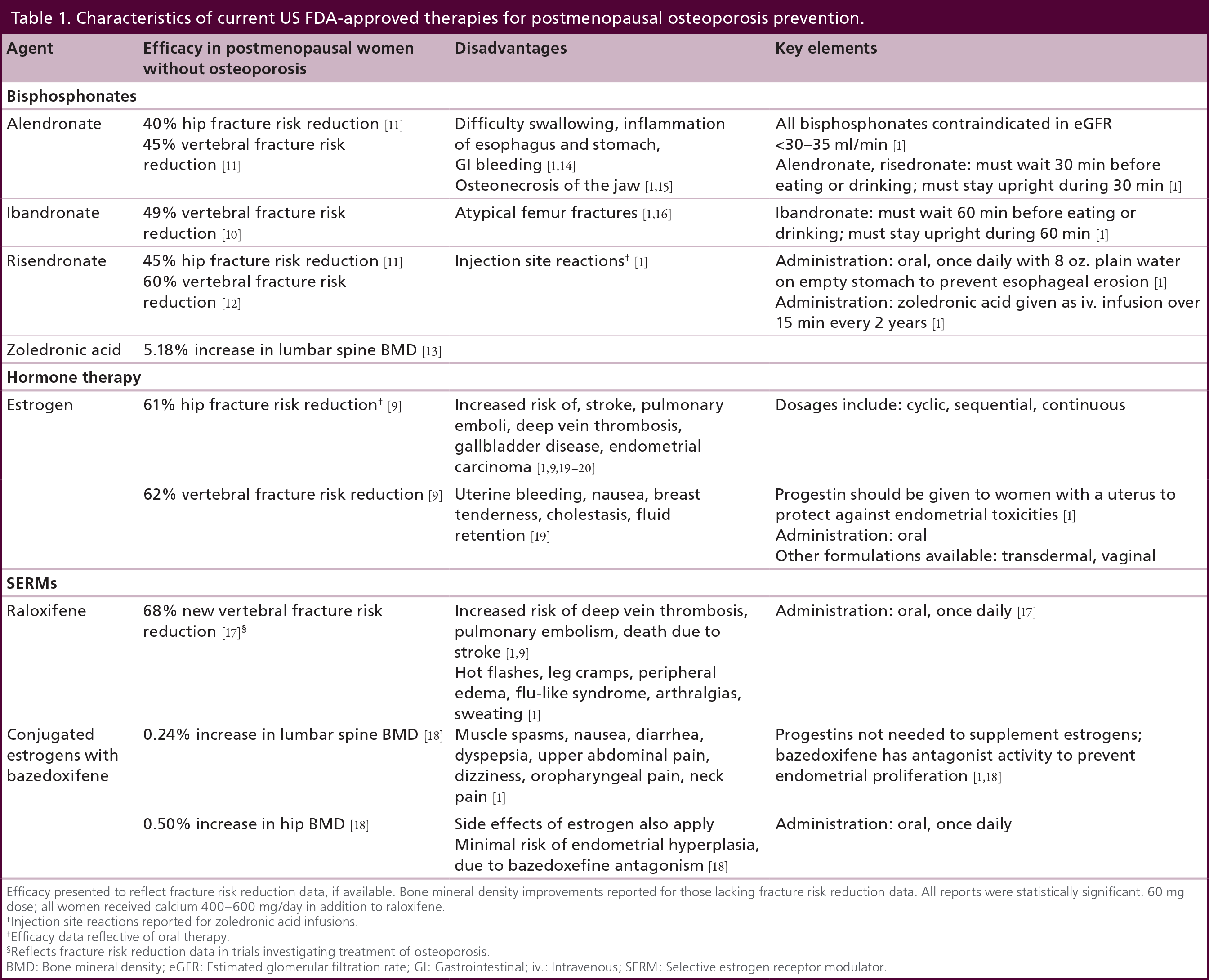
Efficacy presented to reflect fracture risk reduction data, if available. Bone mineral density improvements reported for those lacking fracture risk reduction data. All reports were statistically significant. 60 mg dose; all women received calcium 400–600 mg/day in addition to raloxifene.
Injection site reactions reported for zoledronic acid infusions.
Efficacy data reflective of oral therapy.
Reflects fracture risk reduction data in trials investigating treatment of osteoporosis.
BMD: Bone mineral density; eGFR: Estimated glomerular filtration rate; Gl: Gastrointestinal; iv.: Intravenous; SERM: Selective estrogen receptor modulator.
Estrogen therapy is an effective alternative for women such as those with contraindications to the nonhormonal options or for whom difficulties with long-term adherence, a common issue in nonhormonal options, exist [6]. Many of the nonhormonal therapy options have intolerable side effects or are available in undesirable dosage formulations. The bisphosphonates are associated with negative side effects such as osteonecrosis of the jaw, gastrointestinal discomfort or difficulty swallowing [1]. Raloxifene and oral estrogen therapies are associated with an increased risk of deep vein thrombosis, pulmonary emboli and stroke [1]. There appears to be a need for safer, more tolerable therapies.
Transdermal estrogen products have been approved for the prevention of postmenopausal osteoporosis and work by establishing physiological estradiol levels; they produce fewer nonreactive metabolites and exhibit decreased side effects compared with oral formulations [6]. The most commonly used transdermal estrogen delivery systems are patch and gel products. Estradiol patches are applied directly to the skin every 3.5 or 7 days and deliver a constant rate of estradiol via percutaneous absorption [6]. Use of the smallest dosage consistent with relief of symptoms is recommended [19].
Estrogen therapy alone is appropriate in women who have undergone a hysterectomy. However, it is recommended that a progestin be added to estrogen therapy in women with an intact uterus in order to reduce the risk of endometrial hyperplasia which is associated with an increased risk of endometrial carcinoma [21]. Oral medroxyprogesterone acetate 2.5 mg/day is commonly given in combination with transdermal estradiol therapy in nonhysterectomized women to decrease this risk [22]. Other available products, such as CombiPatch®, contain the progestin norethindrone acetate and estrogen within a transdermal system. Currently available estrogen-only transdermal products approved for osteoporosis prevention in postmenopausal women include: Alora®, Climara Pro®, Estraderm®, Menostar®, Minivelle and various generic products (see
A comparison of transdermal estradiol products indicated for postmenopausal osteoporosis prevention.

Menostar® brand product no longer available In USA; currently available as generic.
BMD: Bone mineral density.
New treatment modalities for the prevention of osteoporosis are currently in clinical trials and late development. A new antiresorptive agent, odanacatib, is a capthepsin-K inhibitor. Romosozumab and blosozumab, antisclerostin monoclonal antibodies and PTHrp 1–34 (abaloparatide) have been shown to work as anabolic agents [26]. These new agents are being studied and developed in an effort to result in greater fracture risk reduction, improved patient tolerance and intermittent administration; further, they may become additional treatment options for patients who are poor responders, or who have contraindications to currently existing drugs [26].
Introduction to the compound
Minivelle is a transdermal estradiol patch indicated for the prevention of osteoporosis in postmenopausal women, in addition to the treatment of moderate-to-severe vasomotor symptoms caused by menopause [22]. Minivelle is the smallest available estrogen patch (1.65cm2), about the size of a dime, allowing for discreet application and wear [22]. It is comprised three layers: a polyester film laminate (the layer most distant from the skin); an adhesive formulation containing estradiol, acrylic adhesive, silicone adhesive and oleyl alcohol; and a polyester release liner which is attached to the adhesive surface and must be removed before applying the patch for use [22]. Estradiol, the active drug in the Minivelle patch and an estrogen receptor agonist, is more potent than estrone and estriol, which are the primary hormones produced by the body after menopause [19]. Estradiol is widely distributed in the sex hormone target organs and is highly bound to sex-hormone binding globulin and albumin and is excreted primarily in the urine [19].
In postmenopausal women, bone loss can be rapid due to the decreased ovarian function and subsequent decreased estrogen production [19]. Estradiol replacement helps to reduce this bone resorption and minimize bone loss in postmenopausal women [21]. The matrix adhesive platform found in Minivelle is designed to release estradiol continuously. The estradiol is easily absorbed through the skin and results in the increase of a woman's baseline estradiol levels within 4 h of application [19,22]. Minivelle therapy for the prevention of osteoporosis should be started with 0.025 mg per day, applied to the skin twice weekly; the dose can be adjusted as necessary [22]. Other available dosage strengths of Minivelle, indicated for the treatment of vasomotor symptoms associated with menopause and for the prevention of osteoporosis, include: 0.0375, 0.05, 0.075 and 0.1 mg/day [22]. The transdermal system is applied topically to a clean, dry and hairless area of intact skin on the trunk of the body [21].
Chemistry & pharmacodynamics
Estradiol is a naturally occurring steroidal estrogen that can also be prepared synthetically [27]. Estradiol is the most active endogenous estrogen and the major secretory product of the ovary [19,21]. Figure 1 illustrates the chemical structure of estradiol [28]. Estrogen receptors are found primarily in the nucleus of estrogen-responsive tissues and undergo a conformational change when estradiol binds, which leads to gene transcription. There are two estrogen receptors that have been identified, estrogen receptor (ER)-α and ER-β. Both receptors have a similar affinity for estradiol, but are expressed in different tissue. In estrogen sensitive tissues ER-α is the receptor expressed most frequently and that is most affected. It is thought that ER-β plays a regulatory role and reduce ER-α regulated gene transcription [21]. Exogenous estrogens produce many of the pharmacologic responses observed with endogenous estrogens. Estrogen therapy is administered continuously or cyclically [21], and the route of administration can have a varying consequence on biomarkers [6]. For example, inflammatory markers, and coagulation proteins are altered with oral estrogen, which differs from transdermal estrogen. In an observational study looking at oral estrogen and its effect on the coagulation pathway, it was found that there was an increased risk of venous thromboembolism (VTE) when taking oral estrogen. However, no such risk has been associated with transdermal estrogen use [29]. Oral and transdermal estrogen therapy have been shown to increase high-density lipoprotein and reduce low-density lipoprotein, although transdermal therapy appears to have a lesser impact on lipoprotein profiles compared with oral therapy [30]. Transdermal estrogen has also been shown to have a lowering effect on triglycerides as compared with oral therapy [31].
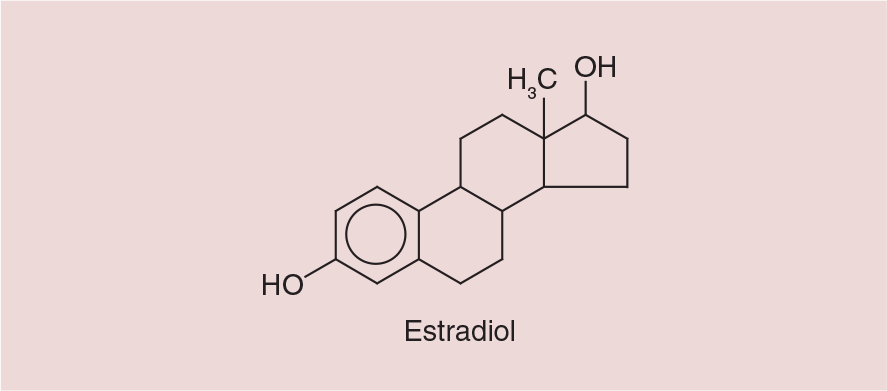
Chemical structure of estradiol.
Pharmacokinetics & metabolism
The pharmacokinetic profile for Minivelle 0.1 mg/day has been deemed bioequivalent to Vivelle 0.1 mg/day in a Phase I study [7]. This study was a randomized, single-dose, two-way crossover study completed in 100 healthy, nonsmoking postmenopausal women whose ages ranged from 40 to 65 years [7]. Each patient received a single dose of Vivelle and Minivelle, that was worn for 84 h. There was a washout period of at least 14 days between the two treatments [7,32]. Both products are transdermal patches that are to be worn for 3–4 days. Using the estradiol peak serum levels, Minivelle was deemed to be bioequivalent to Vivelle [7,22].
In a study of 36 healthy, nonsmoking postmenopausal women, the mean serum pharmacokinetic parameters were reported after a single dose of Minivelle 0.1, 0.05 or 0.025 mg/day. The estradiol peak concentration (Cmax) for Minivelle 0.1, 0.05 and 0.025 mg/day was 117, 56.6 and 30.3 pg/ml, respectively. The estradiol time to peak concentration (Tmax) for Minivelle 0.1, 0.05 and 0.025 mg/day was 24, 24 and 36 h, respectively. The estradiol exposure (AUC84) for Minivelle 0.1, 0.05 and 0.025 mg/day was 5875, 3057 and 1763 pg/h/ml, respectively [22].
At this time, there have been no studies evaluating the extent of tissue distribution of estradiol absorbed from Minivelle in humans. Endogenous estrogens are concentrated in fat deposits throughout the body and are 50–80% bound to plasma proteins [21]. However, estrogens are widely distributed in the body and are generally found in higher concentrations in the sex hormone target organs [22].
Free estradiol is converted by the liver to estrone and estriol and their 2-hydroxylated derivatives and conjugated metabolites, which are then excreted into bile, reabsorbed from the GI tract and recirculated through the liver [21]. Conjugates may be hydrolyzed in the intestine and reabsorbed [19]. Estradiol, estrone and estriol are excreted in the urine along with glucuronide and sulfate conjugates [7]. In comparison to estradiol, estrone and estriol have lower affinity for the estrogen receptor. Estrogens are metabolized partially by CYPP450 3A4 enzymes, therefore CYP3A4 inhibitors and inducers may reduce estradiol drug metabolism [22], potentially leading to increased estrogen effects.
Transdermal estradiol avoids first-pass metabolism, allowing for more consistent and physiological estrogen blood levels [6]. Transdermal estradiol in postmenopausal women re-establishes serum estrogen levels to that similar to a premenopausal women and produces a estradiol to estrone ratio of approximately one [6]. From the bioequivalence study, the calculated half-life ranged from 6.2 to 7.9 h. Once the patch is removed, serum concentrations of estradiol return to baseline within 24 h [7].
Clinical efficacy
The efficacy of oral estrogens in osteoporosis was demonstrated and supported by the large-scale, 2004 WHI trial [20]. Estrogen reduced the risk of fractures in nonosteoporotic women, compared with those with established osteoporosis or osteopenia. There was a 62% risk reduction in vertebral fractures and a 61% risk reduction in hip fractures. This study, however, only assessed oral estrogens. Multiple studies have shown positive impact on BMD [33]. Although BMD increases are a less robust end point than fracture risk reduction, they may serve as a surrogate marker for fracture prevention efficacy.
A multicenter, randomized, double-blind, active-controlled, noninferiority trial of 500 osteopenic postmenopausal women compared transdermal estradiol (0.014 mg/day) with oral raloxifene (60 mg/day). Following 2 years of administration, 77.3% of the estradiol recipients and 80.5% of the raloxifene recipients had no bone loss in the lumbar spine. Both treatments were well tolerated and there were no clinically significant effects observed relative to either endometrial or breast density [34].
The efficacy and safety of Vivelle, the pharmacologic bioequivalent of Minivelle, was studied in a 2-year double-blind, randomized, placebo-controlled, parallel group study of 261 participants [3]. At study inception, no subjects exhibited any evidence of osteoporosis, which was defined as a lumbar spine BMD within two SDs of average peak bone mass. One hundred ninety-four patients were randomized to receive one of the four doses of Vivelle (0.0375, 0.05, 0.075 or 0.1 mg/day), and 67 patients were randomized to receive placebo. There was an increase in BMD of the lumbar spine in all four Vivelle dose groups. The placebo group was observed to experience a decrease in lumbar spine BMD. While the Vivelle 0.05 mg/day dose was not noted to exhibit results significantly greater than placebo at 6 months, all other Vivelle doses did result in significant increases in BMD compared with placebo. The highest dose of Vivelle-Dot® (0.1 mg/day) was superior to all other doses, exhibiting the greatest percentage increase in BMD [3]. Table 2 provides a comparison of other available transdermal estradiol estrogen products indicated for the prevention of postmenopausal osteoporosis.
Safety & tolerability
The WHI study of 2002 was the first to demonstrate the significant efficacy of estrogen alone in the prevention of osteoporotic fractures in postmenopausal women [20]. This 15-year-long trial also established the association between oral estrogen hormonal therapy and an increased risk of complications.
The WHI studies demonstrated the increased risk of breast cancer, coronary heart disease and stroke with the combination of estrogen and progestin. Estrogen alone was also found to increase the incidence of VTE and stroke, but showed decreased risk of breast cancer [20,35]. These findings impacted the prescribing of hormonal therapy, leading to an 80% decrease in use of oral estrogen [36]. However, more recent evidence shows that the absolute risk of VTE and stroke with unopposed estrogen use is decreased in women before the age of 60 years or those within 10 years of menopause [8].
The risks demonstrated with oral therapy may not be comparable to those of transdermal products. The transdermal route bypasses first pass metabolism, leading to estrogen's direct entry into the circulation [32]. By doing so, no gastrointestinal conversion of estradiol to estrone takes place, reducing the higher concentration of estrogen in the liver [37], and the hepatic induction of prothrombotic substances and inflammatory mediators believed to mechanistically contribute to oral estrogen's fourfold increase in the risk of VTE compared with nonusers [37–40].
In comparison, transdermal estrogen may have a neutral or suppressive effect on inflammatory mediators and clotting protein synthesis [35,39–40], and decrease triglyceride levels [40]. Transdermal administration results in lower doses necessary to achieve physiological effects and maintains constant serum levels, eliminating the peaks and troughs of oral therapy. As such, the transdermal route may also confer less initial risk of VTE [35].
Characteristics of studies comparing transdermal estradiol and oral estrogen therapy in the risk of venous thromboembolism.
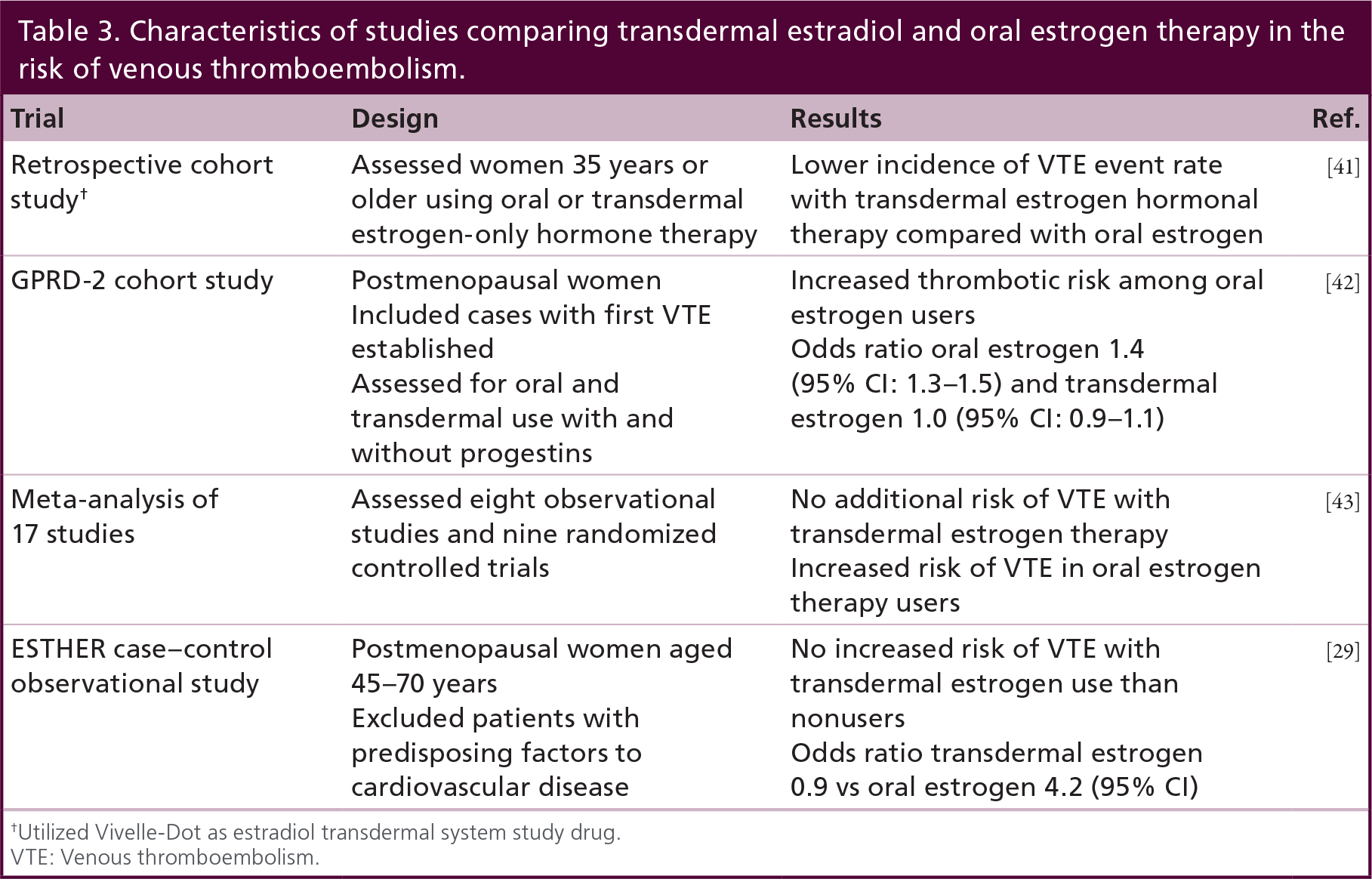
Utilized Vivelle-Dot as estradiol transdermal system study drug.
VTE: Venous thromboembolism.
Based on the growing safety evidence for transdermal estradiol delivery systems, current guidelines from major organizations do support its use. For example, the North American Menopause Society 2014 recommendations identify transdermal estrogen therapy to have a lower risk of VTE and gallbladder disease than oral therapy [43]. The transdermal route has also been suggested for women with chronic conditions of hypertension or hypertriglyceridemia [40] and may be considered for those with history of VTE or contraindications to oral therapy [38].
No clinical trials have been conducted specifically for the safety of Minivelle, as it was approved based on its bioequivalence to Vivelle. Reports of adverse events are thus reflective of Vivelle Phase III clinical trials and pharmacokinetic bioequivalence studies. Trials of Vivelle, using the Minivelle-indicated dose of 0.025 mg/day, reported the most frequent treatment-related adverse events among 47 treated patients, including breast tenderness (n = 8), headache (n = 7), dyspepsia (n = 4) and back pain (n = 4) [44]. A summary of the most common adverse events reported for subjects treated with a dose of 0.025 mg/day are provided in Table 4 [44].
Adverse events reported by greater than 3% of subjects treated with Vivelle® 0.025 mg/day by system organ.

NOS: Not otherwise specified.
Pharmacokinetic studies used to determine bioequivalency of Minivelle to Vivelle reported total rates of adverse events possibly related to study drug to be 22% of subjects receiving Minivelle and 30% in those receiving Vivelle [7].
The tolerability to Minivelle was determined via pharmacology studies, which reported less skin irritation than larger transdermal products [7]. Less than 35% of subjects reported ‘barely perceptible erythema’ after 84 h of wear [22], its approved duration of use based on twice weekly application. There were no reports of removal of the patch due to irritation [22]. It was found that the Minivelle transdermal estradiol patch had good adhesion rates with 99% of subjects reporting the adhesion to be greater than 90% at 84 h [7]. One subject experienced the patch becoming detached during the 24 h time period with Minivelle. After Minivelle was removed, 54% of the 208 subjects had light adhesive residue and 46% had no adhesive residue [22].
The 0.025 mg/day Minivelle dose approved for osteoporosis prevention, currently the lowest dose available, aligns with current guideline recommendations for the lowest effective dose at the shortest possible duration [45].
Regulatory affairs
This estradiol transdermal system has been approved in the USA under the name Minivelle for the prevention of postmenopausal osteoporosis as well as the treatment of moderate-to-severe vasomotor symptoms associated with menopause [22]. It is available in five dosage strengths; the lowest dose, 0.025 mg/day is indicated in osteoporosis prevention to be applied to the skin twice weekly, while the higher doses have both the indication for the prevention of osteoporosis and the treatment of menopausal vasomotor symptoms. The US FDA has deemed Minivelle a bioequivalent product of Vivelle, a transdermal estradiol product indicated for treatment of severe vasomotor symptoms and severe symptoms of vulvar and vaginal atrophy due to menopause [3]. No clinical trials were conducted for Minivelle. Approval was based on pharmacokinetic and pharmacodynamic studies that deemed it equivalent to Vivelle. Minivelle is classified as an estrogen-alone therapy, and as such requires a black box warning for: endometrial cancer, cardiovascular disease, breast cancer and probable dementia risks. Hormone therapy using estrogen is recommended at the lowest effective dose for the shortest duration of time to minimize risk.
Conclusion
Although hormonal therapy is not a first-line therapeutic option for osteoporosis prevention, transdermal estradiol systems provide a safe and effective alternative to nonhormonal therapies and have potential advantages in their delivery. Minivelle has been approved, in a new lower dosage form, for the prevention of osteoporosis in postmenopausal women. The 0.025 mg/day patch is smaller than the size of a dime. This new size allows for more discrete wear for patients [22]. Minivelle eliminates the necessity for strict, daily oral dosing requirements or the need for physician office visits for administration of injectable products. Transdermal delivery also infers a reduced risk of serious side effects, such as VTE and cardiovascular disease when compared with oral estrogen therapy. Transdermal estradiol bypasses first pass metabolism in the liver [32], leading to decreased levels of estrogen in the liver and a perceivably decreased risk of VTE [37–40]. Transdermal Minivelle thus provides an effective alternative for women who cannot receive nonhormonal therapy for the prevention of osteoporosis.
Executive summary
Minivelle® (Noven Therapeutics, LLC, FL, USA) is a transdermal estradiol patch (0.025, 0.0375, 0.05, 0.075 and 0.1 mg/day) used in the treatment of postmenopausal vasomotor symptoms and for the prevention of osteoporosis.
After absorption, estradiol replaces the estrogen lost during/following menopause.
Estradiol peak concentration (Cmax) for Minivelle 0.025 mg/day was 117 pg/ml.
Transdermal estradiol bypasses first past metabolism in the liver.
Estradiol is excreted in the urine along with glucuronide and sulfate conjugates.
Half-life ranges from 6.2 to 7.9 h.
Transdermal estradiol has been shown to be effective in the prevention of postmenopausal osteoporosis.
It has been shown to have a positive impact on the bone mineral density of the lumbar spine.
Transdermal estradiol carries a lower risk of venous thromboembolism and gallbladder disease than oral estrogen.
Lower risk of hypertension and dyslipidemia with transdermal versus oral estrogen.
Adverse effects include: breast tenderness, headache, dyspepsia and back ache.
No significant drug interactions noted.
The Minivelle transdermal estradiol 0.025 mg/day patch is applied to the skin twice weekly.
Footnotes
In addition to the peer-review process, with the author(s) consent, the manufacturer of the product(s) discussed in this article was given the opportunity to review the manuscript for factual accuracy. Changes were made at the discretion of the author(s) and based on scientific or editorial merit only.
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
