Abstract

“Overweight postmenopausal women have a worse prognosis, partly because nodal positivity is associated with obesity.”

Obesity is the blight of the western world being implicated in myriad diseases and premature deaths. Public Health England estimate that 58% of women have a BMI of >25 kg/m2 with 33% overweight (BMI: 25 to <30) and 25% obese (BMI: >35). Overweight postmenopausal women have an increased risk of developing breast cancer and carry a worse prognosis [1,2]. Although the rationale for weight loss may appear self-evident, the likely benefits in breast cancer patients require closer examination. Obesity may worsen prognosis as a result of increased synthesis of estrogens in peripheral fat and one possible approach to this problem is to use progestins to counteract the estrogenic drive.
Obesity & prognosis of breast cancer
Overweight postmenopausal women have a worse prognosis, partly because nodal positivity is associated with obesity. Daniell examined the axillary lymph node status of 465 postmenopausal women with breast cancer and reported that nodal positivity was more frequently associated with obesity (p < 0.001) [3]. As is shown in Table 1, there was a significant increase in breast cancer mortality with increasing BMI [3]. Based on the hypothesis that large breast size in obese women might delay discovery of breast cancers Cui et al. investigated the relation between body mass and stage at diagnosis [4]. In 966 breast cancer cases from the Baltimore Metropolitan area, high body mass was significantly associated with late stage at diagnosis and this relationship was stronger among women <50 years compared with those ≥50 years. In the American Cancer Society's Cancer Prevention Study II (CPS-II), 2852 breast cancer deaths occurred among 424,168 postmenopausal women after 14 years of follow-up [5]. There was a substantial and sustained increase in breast cancer mortality rate with increasing BMI. For those with a BMI >40.0 compared with BMI <18.5 there was a trebling of risk, as shown in Table 1.
Breast cancer prognosis in relation to weight.
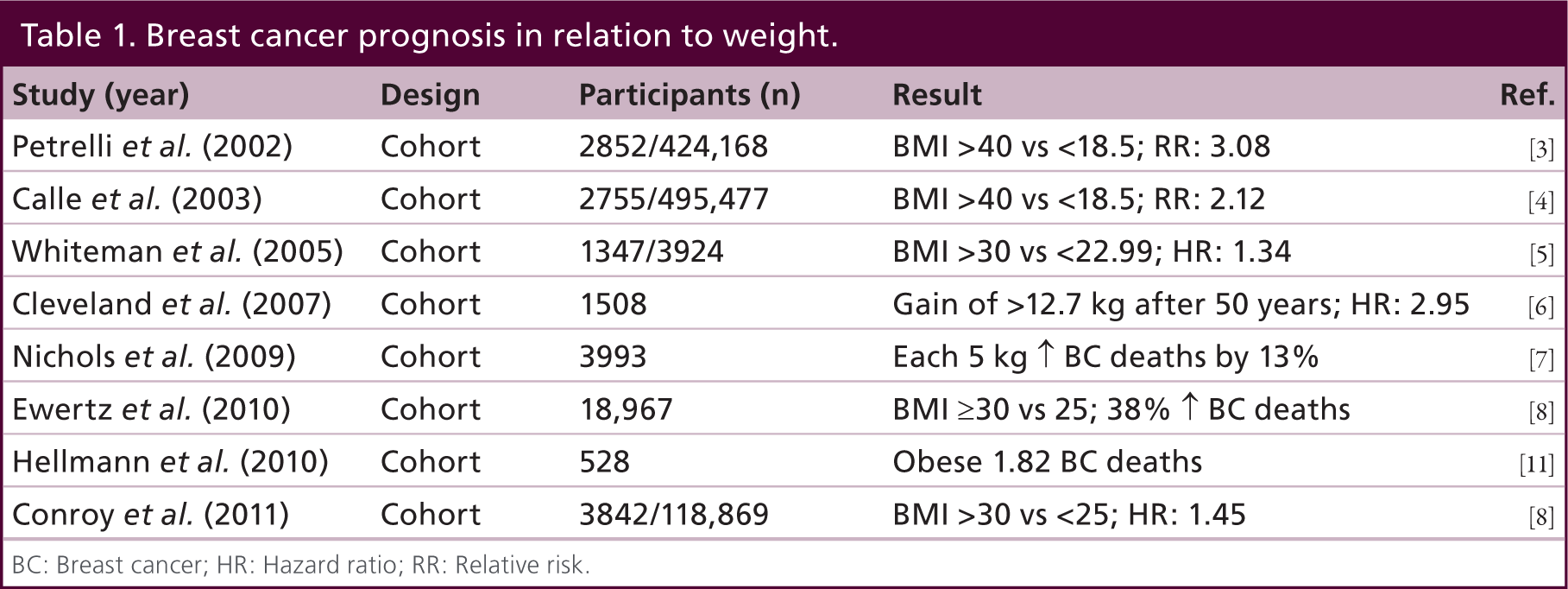
BC: Breast cancer; HR: Hazard ratio; RR: Relative risk.
Another large population investigation of US adults participating in Cancer Prevention Study II reported that those with a BMI of >40 kg/m2 had death rates from all cancers combined that were 62% than those of normal weight [6]. For breast cancer the relative risk in those with a BMI of >40kg/m2 was 2.12. Whiteman et al. conducted a cohort analysis of 3924 women aged 20–54 years with breast cancer enrolled in the CASH study [7]. Obese women with a BMI of >30 were significantly more likely than lean women to die of breast cancer (hazard ratio [HR]: 1.34).
Cleveland et al. interviewed 1508 breast cancer patients soon after diagnosis regarding height and weight at each decade of life from age 20 years until 1 year before diagnosis [8]. Obese women had increased mortality due to breast cancer compared with ideal weight women among those premenopausal at diagnosis (HR: 2.85) and those postmenopausal at diagnosis (HR: 1.91). Women with postmenopausal breast cancer who gained >12.7 kg after age of 50 years up to the year before diagnosis had a two- to three-fold increased risk of death due to all-causes (HR: 2.69). In a cohort of 3993 breast cancer patients completing a structured telephone interview 1−2 years after diagnosis and a mailed follow-up questionnaire, each 5-kg gain was associated with a 12% increase in all-cause mortality and a 13% increase in breast cancer-specific mortality [9], In a Danish study of 18,967 women with early breast cancer those with a BMI of ≥30 kg/m2 were older with more advanced disease at diagnosis compared with those having a BMI of <25 kg/m2 [10]. After adjustment for disease stage, risk of developing distant metastases after 10 years was significantly increased by 46%, and the risk of dying from breast cancer after 30 years was significantly increased by 38% for women with a BMI of ≥30 kg/m2. Additionally the heavier women were less likely to benefit from systemic adjuvant chemotherapy or endocrine therapy.
Expansion of adipocyte population
Postmenopausal breasts show irreversible lobular involution and at the same time, intralobular and interlobular stroma is replaced with dense connective tissue and fat, respectively [12]. Expansion of the numbers of the adipocyte population changes the endocrine milieu with elevated aromatase activity increasing estradiol conversion. In a study of 505 women with breast cancer, McTiernan et al. reported that a BMI >30 could lead to a tripling of free estradiol levels [13].
Invasion of macrophages secreting TNF-α, TGF-β, IL-1β and HGF may induce an insulin-resistant state with hyperinsulinemia stimulating mitosis in breast cancer cells [14]. Necrotic adipocytes surrounded by macrophages form crown-like structures (CLS) present in 47% of breast biopsies [15]. Severity of inflammation was significantly related to both BMI and adipocyte size with increased aromatase activity in the inflamed breast tissue of obese cases.
Higher fasting insulin levels in breast cancer patients increase risk of both recurrence and death [16,17]. Adipocyte-derived leptin may mediate obesity-associated mitogenesis and angiogenesis in vitro [18]. Insulin-related variables are prognostically significant only during the first 5 years whereas obesity-related variables (BMI, weight, leptin) have adverse associations constant over time [19]. High BMI was associated with increased mortality up to and beyond 10 years.
Widespread alterations in gene expression occur in obese patients with the obesity gene signature correlated with IGF signaling and reduced estrogen receptor (ER) levels expression [20]. Estrogens induce the obesity transcription program in vitro, while agents targeting neurotransmitter receptor pathways cause repression.
Adiponectin acts in a protective capacity against obesity-related disorders including Type 2 diabetes and cardiovascular disease [21]. Additionally it inhibits immune cells and downregulates vascular adhesion molecules in endothelium [22]. Adiponectin can inhibit leptin-stimulated invasion of breast cancer cells [23] and two case–control studies have found an inverse relationship between serum adiponectin levels and breast cancer risk, independent of known risk factors [24,25]. In vitro exposure of human adipocytes to hypoxia produces a rapid threefold reduction in adiponectin levels [26]. In contrast, hypoxia stimulates leptin and VEGF gene expression so that obesity can give rise to a potentially lethal combination of reduced protection and increased malignant proliferation and metastasis.
Aromatase
The majority of postmenopausal women with ER positive tumors receive aromatase inhibitors which achieve castrate levels of estrogen, irrespective of BMI [27]. In the ATAC study women with BMI of >35 kg/m2 were more likely to relapse than those with a BMI of <23 kg/m2 and had significantly more distant recurrences [28]. Similar results were recorded in the German BRENDA-cohort [29]. In the Japanese neoadjuvant study JFMC 34-0601, response rates to neoadjuvant exemestane were 22, 56 and 61% in tertiles of BMI, with low BMI emerging as an independent negative predictor of clinical response [30]. In a prospective Japanese study of 501 patients with ER-positive cancers, 102 were of normal weight (BMI <25 kg/m2) and 399 were overweight or obese and they all received an adjuvant aromatase inhibitor [31]. There were no significant clinicopathological differences according to BMI. At 3 years disease-free survival was 78% in the nonobese compared with 86% in the obese.
Lymphovascular invasion & obesity
Abe et al. reported that poorer prognosis in obese women was associated with increased lymphovascular invasion (LVI) present in 73% [32]. A study of 393 postmenopausal patients at Guy's Hospital (London, UK) showed a direct relationship between LVI and BMI [33]. In the specimens from those <50 kg, LVI was observed in 11% compared with 45% from women >80 kg (p = 0.02). 5-year survival of women with LVI was 74% compared with 91% when absent (p < 0.0001). This was confirmed in another study of 1312 breast cancer cases where on multivariate analysis, severe obesity was independently associated with the presence of LVI (odds ratio: 1.80; p = 0.03) [34].
Most studies have not distinguished (LVI) and blood vessel invasion (BVI). Using an endothelial subtype specific immunohistochemical stains, Lee et al. reported a significant correlation between D2–40 LVI positivity and lymph node metastasis and recurrence [35]. There was no correlation between BVI and recurrence. A large study from Nottingham showed clearly that LVI was strongly inversely associated with distant metastasis-free survival, similar to the effect of having one to two lymph nodes involved [36].
Weight loss & prognosis
De Waard et al. set up a feasibility study with weight reduction achieved by calorie restriction and this was maintained for another 2 years but no end results were published [37]. In a prospective observational study Holmes et al. examined regular exercise and risk of relapse [38]. Compared with women engaging in <3 metabolic equivalent task (MET) hours per week, RR of breast cancer death was 0.80 for 3–8.9 MET, as shown in Table 2.
Studies examining weight loss and outcome after breast cancer treatment.

MET: Metabolic equivalent task hours; RCT: Randomized controlled trial; RR: Relative risk.
In a randomized multicenter clinical trial of 2437 women, after 5 years median follow-up (when funding ceased) fat intake was significantly lower in the intervention group with a hazard ratio for relapse events in the intervention group versus controls of 0.76 (95% CI: 0.60–0.98; p = 0.034) [39]. There was a significant reduction in mean bodyweight within the intervention group (6 pounds; 2.7 kg).
Against this, Caan et al. reported that weight gain up to 4 years after diagnosis did not increase risk of recurrence or death from any cause [40]. Women with weight losses (≥10%) had increased risk of death from to any cause versus those with unchanged weight. This risk was more pronounced among those obese before diagnosis.
The RENEW trial tested a mailed diet-exercise intervention and telephone counseling in cancer survivors either immediately or after a 1-year delay [41]. The immediate intervention group experienced significant improvements in diet, activity and BMI but no impact on survival was reported. Only 6% of invited patients actually participated in the study and the attrition rate at 2 years was 25% [42]. In a meta-analysis of body compositional changes after breast cancer diagnosis bodyweight did not consistently increase nor did it accurately reflect lean or adipose tissue changes [43]. Behavioral interventions produced inconsistent or insignificant effects on body composition.
Some patients are more likely to accept a drug rather than a life style change to enable weight loss. Veerman et al. constructed a multistate life-table based Markov model using Australian data on effectiveness, mortality and disease costs, evaluating 1-year interventions with the drugs sibutramine and orlistat [44]. With reductions in bodyweight related disease burdens of 0.2 and 0.1%, respectively, the disability-adjusted life year avoidance cost was almost three times that deemed good value for money.
A meta-analysis of 43 studies examining weight and prognosis, confirmed worse overall and breast cancer-specific survival in the obese [45]. Survival rates in treatment and control cohorts were not statistically different. The authors concluded there was no current evidence of weight loss improving survival.
Changing the perioperative endocrine milieu
The hyperestrogenisation associated with obesity stimulates VEGF and is associated with a higher risk of LVI [35,36]. Before and during surgical excision there may be an increased risk of tumor dissemination. Studies in premenopausal women with breast cancer had shown that surgery in the luteal phase of the cycle was associated with better outcome [46–48]. Furthermore, higher blood levels of progesterone were also predictive of improved prognosis [49]. This being so, it was reasoned that progesterone treatment could be used in the postmenopausal to try and simulate the endocrine status in the luteal phase of the cycle of the premenopausal.
To investigate this prospectively Badwe et al. conducted a randomized trial of 1000 women with operable breast cancer treated by surgery alone or with a single intramuscular hydroxyprogesterone 500 mg injection between 5 and 14 days before surgery [50]. After a median follow-up of 65 months, there had been 273 recurrences and 202 deaths. 5-year overall survival rates for the intervention and control groups were 80 versus 78%, respectively. Among the 471 node-positive patients, the 5-year overall survival rates were 76 versus 67% (p = 0.04). There were no reported adverse events nor were there any postoperative deaths. This result was described by the authors as ‘hypothesis generating’ and warrants further investigation since it could represent a simple and effective adjuvant therapy with progesterone inhibiting shedding of viable tumor during surgery. This is a pragmatic approach to examine the impact on survival but biological effects of progesterone on proliferation and apoptosis could be determined as bolt-on projects.
Conclusion
In postmenopausal women with breast cancer, although weight loss may improve health and self-esteem there seems little evidence to support its use as a means of reducing risk of recurrence compared with that achieved by aromatase inhibitors. Additionally although this may be achieved by some motivated individuals it is unlikely to work on a population basis. The harm may already have been done in terms of tumor angiogenesis. An agent that can be simply administered and well tolerated is likely to be more acceptable to patients at a time of great stress. Perioperative progesterone does provide a nontoxic potentially effective intervention and it is important that this approach is investigated by further randomized controlled trials.
Future perspective
Until now, neoadjuvant treatments for breast cancer have been able to sometimes downsize the tumor but have not led to any improvement in survival compared with postsurgical adjuvant therapy. We need therapies which will stabilize the cancer possibly by reducing mitosis or increasing apoptosis and thereby prevent local or distant spread as a result of surgical interventions. Perioperative progesterone may offer such an approach which requires confirmation by further randomized trials since this could have an important impact on survival for postmenopausal women with operable breast cancer.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
