Abstract
Shoulder dystocia is a complication of vaginal delivery and the primary factor associated with brachial plexus injury. In this review, we discuss the risk factors for shoulder dystocia and propose a framework for the prediction and prevention of the complication. A recommended approach to management when shoulder dystocia occurs is outlined, with review of the maneuvers used to relieve the obstruction with minimal risk of fetal and maternal injury.
Shoulder dystocia, a complication of vaginal delivery in which the fetal shoulders fail to deliver spontaneously after the head emerges, is uncommon but potentially treacherous. Its precise incidence is difficult to ascertain, owing to the different definitions used in the literature and uncertainty about how often its occurrence is documented in medical records. Estimates range between 0.15 and 2.0% [1–3]. Although most cases of shoulder dystocia can be relieved without permanent sequelae for the neonate, this is not always the case [4,5]. Complications include various degrees of brachial plexus injury (BPI) and, less commonly, asphyxial or traumatic central nervous system damage and long bone fractures [6]. Maternal adversity in the form of lacerations, hemorrhage and psychological stress occurs as well [7]. BPI occurs in about 1–20% of shoulder dystocia cases [1–2,6,8]. It is often a transient neuropraxis and recovers fully in hours to months; it is permanent in about 3–10% of cases [6,9], probably the result of avulsion of nerve tissue. The likelihood of intact survival after shoulder dystocia depends heavily on the skill with which it is managed; preemptive cesarean delivery of babies at high risk would be ideal, but the identification of such cases can be challenging.
Almost all BPIs associated with shoulder dystocia are Erb palsies, and result from overstretching of the C5–6 nerve roots during parturition, particularly in the presence of difficult shoulder delivery. There is enough evidence that BPI can occur in the absence of shoulder dystocia to conclude that not every injury is the consequence of excessive force applied by the obstetrician or midwife [10,11]. Moreover, it seems equally clear that BPI can occur in association with shoulder dystocia even when the complication has been managed optimally. The weight of the available information suggests, however, that inopportune medical intervention is probably a factor in most injuries.
Universal cesarean delivery would eliminate almost all cases of shoulder injury; but this approach is impractical, and would have an unfavorable risk/benefit balance. In this regard it should be noted that the incidence of shoulder dystocia seems to have increased in some places [12], even as the cesarean rate has risen substantially over recent decades. The incidence of Erb palsy, however, may have been be more stable [13,14], although a study from Sweden showed a dramatic increase between 1980 and 1994 [15]. Whether these differences in incidence and trend relate to ascertainment bias, variations in definitions and reporting or are the consequence of improved obstetric practice is unknown. Certainly, in order for the clinician to minimize the incidence of BPI or other adverse sequelae of shoulder dystocia she or he must be thoroughly familiar with existing risk factors, incorporate them into a decision matrix for each patient, offer cesarean delivery when risks are very high and be prepared to deal expertly with shoulder dystocia if it occurs.
Risk factors
To optimize outcomes we should aim to avoid injury from shoulder dystocia and, when possible, avert its occurrence. One step toward avoidance is to identify the patient at increased risk for shoulder dystocia and BPI.
There are a number of maternal and fetal characteristics associated with the development of shoulder dystocia and BPI, but many cases develop without recognized antecedent risk factors [16]. The ability to predict the occurrence in an individual delivery is limited, but not unachievable (see below), and the prevailing notion that shoulder dystocia is always an unexpected complication has done little to advance our understanding of how it might be prevented [17,18]. Several algorithms have been suggested to predict and thereby prevent shoulder dystocia based on identifiable predisposing factors [19–23]. Risk factors for shoulder dystocia and BPI can be usefully categorized into those identifiable in the patient's history, and those that arise or are identified during prenatal care or labor (Box 1).
Major risk factors for shoulder dystocia and Erb palsy.

History of shoulder dystocia or baby with BPI
Maternal diabetes
Maternal obesity
Macrosomia (risk increases as fetal weight increases)
Gestational diabetes
Excessive weight gain
Clinical pelvimetry and estimated fetal weight concerning for CPD
Protracted active phase dilatation
Arrest of dilatation
Prolonged deceleration phase
Failed, protracted or arrested descent
Long second stage
Precipitate second stage
Instrumental delivery
BPI: Brachial plexus injury; CPD: Cephalopelvic disproportion.
Obstetric history
A woman with a prior pregnancy complicated by shoulder dystocia or BPI, neonatal macrosomia or diabetes mellitus is at increased risk for difficult shoulder delivery [24,25]. A previous shoulder dystocia increases the risk of recurrence several fold; up to 10–20% of patients have the complication with a subsequent birth [24–26], often with a higher incidence of associated BPI than the primary case [24]. These observations make it reasonable to offer cesarean delivery to patients with a history of shoulder dystocia in a previous pregnancy, particularly if there are other associated risk factors present.
Having had a macrosomic infant previously also increases the risk of shoulder dystocia [27]. It is uncertain whether a family history of shoulder dystocia in a sister or mother predisposes the patient to the complication or if differences in racial background or body type of the father of the pregnancy plays a role. In general, fetal growth is more dependent on maternal than paternal body composition and stature, although paternal stature contributes [28,29].
Other factors that some studies have associated with elevated rates of fetal macrosomia (and presumably shoulder dystocia) include high maternal birth weight, short stature, preexisting obesity, diabetes, excessive weight gain in pregnancy and advanced maternal age [30–33].
Prenatal care
A number of further risk factors are identifiable during the course of prenatal care. Some of these are potentially modifiable, but most are not. Prominent among these are maternal obesity [30,34], excessive weight gain [27,35], various degrees of glucose intolerance [36,37] and multiparity [30]. Ideally, maternal obesity should be addressed before pregnancy. Substantial weight loss is not prudent during gestation because it may predispose to small for gestational age infants, especially in women with relatively mild obesity [38]. Moderation of caloric intake and careful attention to weight gain during pregnancy does reduce the risk of macrosomia in women with class II and III obesity [38].
Maternal diabetes mellitus has long been recognized as a strong risk factor for shoulder dystocia [39–41]. In addition to predisposing to macrosomia [42], diabetes, especially if glycemic control is poor, confers differences in body proportions that probably explain why, at any given birth weight, the fetus of a diabetic mother is more likely to experience shoulder delivery obstruction than one of a nondiabetic [41,43–44]. The fetus of a diabetic is prone to central growth and adiposity, with the trunk relatively large compared with the head [37,45]. In fact, this disproportionate growth affects large babies of nondiabetics, and helps explain the high incidence of shoulder dystocia among them [45,46].
Glucose tolerance exists on a continuum, and it is therefore not surprising that the incidence of neonatal macrosomia, shoulder dystocia and BPI is increased among women who have a positive 50 g glucose challenge test followed by a negative glucose tolerance test, particularly if the latter has one abnormal value [36,37].
Intrapartum observations
Careful evaluation of a parturient can reveal risk factors not previously recognized. Clinical or ultrasonographic estimation of fetal weight is valuable. Indeed, fetal macrosomia (variously defined) is the strongest risk factor for shoulder dystocia and BPI in both diabetic and nondiabetic pregnancies (Box 1) [47,48].
Estimation of fetal weight can, however, be challenging, especially in the large fetus, for which measurement errors can be substantial. Even ultrasonography does not always provide highly accurate fetal weight estimates [49,50]. In part for that reason, recommendation for cesarean delivery based solely on a high estimated fetal weight is not likely to be cost effective and would result in an excessive number of unnecessary cesareans [51–53]. Moreover, approximately half of shoulder dystocia events occur in fetuses weighing <4000 g [3,52], so simply using a weight cutoff to preclude a trial of labor will not prevent many cases [50,52]. The American Congress of Obstetricians and Gynecologists has suggested consideration of cesarean section to prevent a shoulder dystocia at an estimated fetal weight of 4500 g in a diabetic and 5000 g in a nondiabetic patient, although several hundred cesareans would probably required for prevention of each BPI using such weight criteria [51].
Induction of labor in cases of suspected fetal macrosomia does not decrease the incidence of shoulder dystocia, except perhaps in some diabetics [54–56]. Likewise, although a male fetus is more likely to have shoulder dystocia than a female [57], male fetal sex is not a sufficiently compelling reason to recommend induction of labor or cesarean to prevent shoulder dystocia.
Thorough clinical cephalopelvimetry can be especially helpful to the clinician in assessing risk, because certain pelvic features predispose to difficult shoulder delivery. The presence of a narrow anteroposterior outlet diameter (common in a pelvis with android or platypelloid features), or a long and steeply inclined pubic symphysis (seen primarily in an anthropoid pelvis), should alert the clinician to an increased risk, especially if other predisposing factors exist. Shoulder dystocia can even occur in a gynecoid pelvis if it is unusually small or descent is precipitate.
As labor progresses, certain abnormalities of dilatation and descent signal further risk, probably because of their association with fetal macrosomia or fetopelvic disproportion. In at least half of BPI cases a preceding dysfunctional labor pattern can be identified [37]. Arrest and protraction disorders of the active phase and second stage probably predispose, as does (seemingly paradoxically) precipitate descent [58]. But one abnormality of the first stage, a prolonged deceleration phase, has a particularly strong association with shoulder dystocia and neonatal BPI [27,30,59]. This abnormality, which occurs when final retraction of the cervix around the fetal head is delayed because fetal descent cannot be timely initiated, is an important bellwether for second stage abnormalities and shoulder dystocia. When a prolonged deceleration phase is combined with a second stage longer than 2 h the odds of BPI increase 20-fold [20].
Decisions about assisted vaginal delivery in the setting of a prolonged second stage in a patient with a suspected macrosomic fetus should be informed by the fact that the use of forceps or vacuum extractor substantially increases the risk of shoulder dystocia [60]. This is especially true if the delivery is done from the midpelvis, but applies as well to all instrumental deliveries [2,61–62]. Moreover, the shoulder dystocia associated with these deliveries is more likely to require complex maneuvers and result in fetal injury [61].
Prediction
It is often assumed that shoulder dystocia is an unpredictable and, therefore, unpreventable complication [17,63] but this fatalism is unwarranted. Although predicting with certainty that shoulder dystocia or BPI will occur in a particular case is rarely possible, our ability to identify cases in which shoulder problems are likely is improving and can help guide clinical decisions.
Three kinds of systematic attempts to identify cases at high risk have been promulgated. One uses late pregnancy or intrapartum sonographic measurements of body weight and dimensions, an attempt to identify the fetus with macrosomia or a disproportionately large trunk or bisacromial diameter. Empiric risk scores have also been used, based on the assumption that there might be a direct relationship between the number and type of risk factors and likelihood of shoulder complications. Finally, multivariate statistical techniques can assess risk factors based on their strength and interaction in a population so that risk can be assessed for an individual patient [20–21,64]. These approaches have yielded mixed results.
Ultrasonography has not proved very helpful in identifying candidates for preemptive cesarean delivery. Shoulder dystocia and BPI are strongly associated with large fetal weight [65] but, although using a weight threshold for preventive cesarean will forestall many cases, the trade-off in higher economic cost and in maternal and neonatal morbidity is substantial [65–68]. A more focused approach that relies on identifying the fetus with disproportionate trunk or shoulder girdle growth seems more promising, but the predictive values and false positive rates are not encouraging [69]. Gerber et al., for example, found that for an abdominal/head circumference ratio above 1.05 the sensitivity and specificity for the prediction of shoulder dystocia were 46 and 75%, respectively, with a positive predictive value of only 5.7% [70]. Similar results were obtained by other investigators [71].
Risk factors, as discussed above, are characteristics or events shown to occur with a significantly higher frequency in association with the outcome under study. But a distinction must be made between risk factors and predictive factors. Many strong risk factors for an outcome are poor predictors of it, particularly if they are prevalent in the population. For example, maternal obesity has a strong association with shoulder dystocia and BPI; but the presence of obesity in an individual is a poor predictor of BPI because obesity is common, and only a small proportion of obese parturients have shoulder dystocia or BPI. Further complicating this issue is that many of the strong risk factors for shoulder dystocia are not mutually exclusive, and their interactions are not well understood. For example, gestational diabetes mellitus, obesity, fetal macrosomia, excessive weight gain and dysfunctional labor frequently coexist, and whether their individual influences on risk overlap, are additive, or multiplicative may be difficult to discern statistically. Moreover, the independent effects of variables may vary among patients according to the complex clinical situation in which they occur. That may be why empiric risk scoring systems have not been fruitful [22,23].
Multivariate analytic techniques, which can consider the interaction of risk variables associated with shoulder dystocia or injury, hold the most promise for prediction [20–21,64]. One study created an Erb palsy risk score from a series of 45 cases. Applied to a theoretical population, the risk scoring system would prevent 36% of BPI cases and result in 14 (in retrospect, unnecessary) cesarean deliveries for each BPI averted [20].
To optimize the value of these multivariate predictive systems they must be unique to the population in which they are used because the influence of demographic variables will depend on their prevalence in the population. Even the baseline prevalence of shoulder dystocia and BPI will affect the value of these systems. In addition, only variables the practitioner can discern and act upon before delivery should be used. Thus, in a decision matrix aimed to reduce the incidence of BPI, variables like birth weight (which cannot be known accurately before delivery) or the presence of shoulder dystocia (which, once present, precludes preemptive cesarean) should not be incorporated as variables. Unfortunately, variables that could be valuable in a risk scoring system, such as pelvic architectural features and details of obstetric history, are often not recorded, or are not assessed with sufficient clarity to be of value.
Given the current state of our knowledge, and the fact that multivariate risk scoring systems are not yet suitable for general use, what approach should the practitioner take to minimize the incidence of BPI without doing an excessive number of cesarean deliveries? A practical approach is to consider the presence of risk factors in the three categories described above (historical, prenatal and intrapartum). The presence of strong risk factors in two or three categories should prompt strong consideration of cesarean delivery. It is necessary to consider that the adverse influence of a risk factor is affected by its severity. Poorly controlled diabetes presents greater hazard than a case with euglycemia; an estimated fetal weight of 5 kg is more concerning than one of 4 kg; a prior shoulder dystocia that resulted in permanent BPI is of greater importance than one that resolved without injury. Moreover, risk assessment is a serial process, and as new problems are identified during the course of care, especially during labor, plans can be altered accordingly. For example, the development of a prolonged deceleration phase in a woman with other risk factors may well be sufficient to tip the scales in favor of cesarean section.
Clinical management
When shoulder dystocia occurs there is an understandable urgency perceived by everyone in the room, not least the patient, who is generally quick to react with anxiety to fears expressed by the staff or to chaotic behavior. Although the situation needs to be addressed promptly and efficiently, hasty management can do serious damage. Deliberate and logical steps should be taken, and, assuming that fetal oxygenation was normal at the onset of the dystocia, taking several minutes to deliver the fetus rarely causes significant morbidity. To ensure optimal outcome the primary provider and the rest of the team must institute a coordinated prescriptive plan of action. There is some evidence that staff training approaches including drills, checklists and debriefings after shoulder dystocia cases can be helpful. They improve documentation and result in superior outcomes [72,73]. It is important that all staff are included in these training exercises so that everyone understands her or his role when this emergency occurs [34,73–75]. Our suggested management paradigm is outlined in Figure 1 and described below [75].
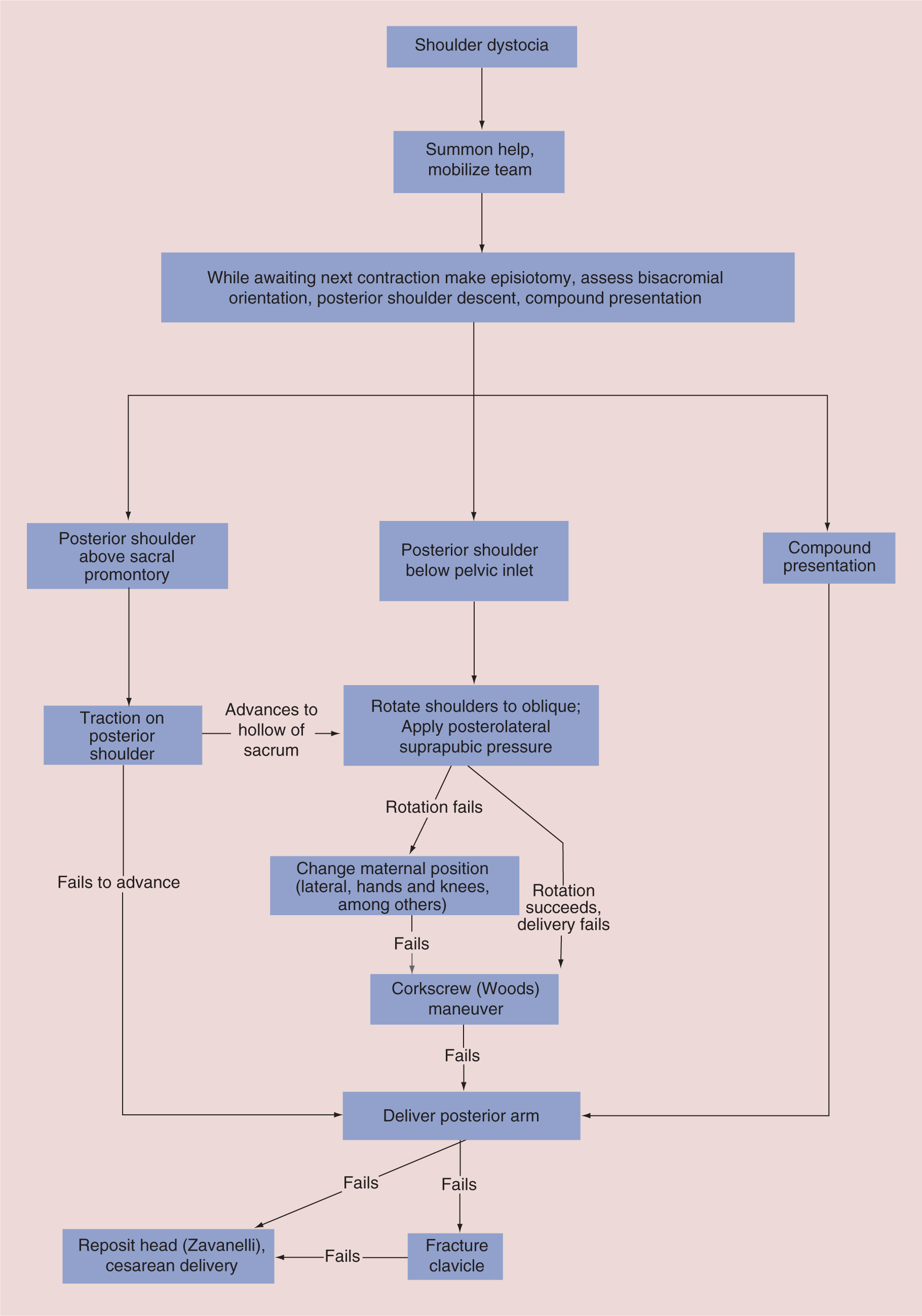
While its details may be modified to address individual or institutional preferences, availability of clinical resources and extant circumstances, the need to proceed with a logical sequence of assessments and maneuvers is paramount. A detailed description of the technical aspects of maneuvers to resolve shoulder impaction is beyond the scope of this review. For such information the reader is referred to references 75–78. There is not strong evidence to support any sequence of manipulations as being superior to others. Most recommendations are based on clinical experience. Whatever maneuvers are used, it is important not to rush, to remain calm and to give clear instructions to the patient and assistants. Intense fundal pressure, downward traction on the fetal head or repeated forceful suprapubic pressure should be avoided, as these actions are likely to result in injury. An episiotomy may be appropriate. It will not relieve the obstruction, but will facilitate intravaginal or intrauterine manipulations.
When shoulder dystocia is suspected, often – but not always – by finding the baby's head retracted tightly against the mother's perineum, the management plan should be instituted. The best initial approach is not to touch the baby's head, no matter how tempting to do so. The brachial plexus may already be on tension, and it can take surprisingly little force to injure it. Rather, it is advisable to await the next contraction before instituting any maneuvers [76]. Use the intervening time to assess the situation by thorough examination, to summon necessary help and to explain to the patient and the team what is to be done. The mother should be encouraged not to push until a shoulder is emerging, and fundal pressure should generally be avoided. The most experienced obstetrician available should take charge of management.
The presence of a protocol, unique to each institution, is vital. Once triggered by personnel at the delivery a series of events should be set in motion so that help is summoned. An experienced obstetrician and obstetrical nurse are important, as may be a neonatologist and anesthesiologist. If the patient was deemed to be at very high risk extra help should be immediately available in anticipation of difficult shoulder delivery.
A careful examination of the patient should be done, with a focus on several things. Determine the orientation of the shoulders in the pelvis, assess whether the posterior shoulder has negotiated the sacral promontory and entered the midpelvis and rule out a compound presentation. Reassess the subpubic angle and the inclination of the lower sacrum and coccyx. If the posterior shoulder is unengaged or there is a limb presenting alongside the head, problems that complicate a few percent of shoulder dystocias, these should be dealt with promptly. If not, and the anterior shoulder does not stem beneath the symphysis pubis with the subsequent contraction, attempt rotation of the shoulders into an oblique diameter of the inlet (Rubin maneuver) [77]. If rotation is not possible, consider delivery of the posterior arm. If rotation occurs but delivery does not, continue rotation a full 180°, the Woods maneuver [78].
When the lead provider is attempting to rotate the shoulders it is advantageous for an assistant to facilitate rotation by applying pressure to the anterior shoulder suprapubically. This pressure should be directed posterolaterally in the same direction as vaginal attempts by the lead provider so as to encourage rotation of the trunk. Suprapubic pressure directed posteriorly will not accomplish that goal.
Delivery of the posterior arm
The posterior arm will usually be extended, and require flexion so that the hand can be grasped and the arm pulled across the fetal chest. Once the posterior arm is delivered, the anterior shoulder will usually emerge easily. Computer simulation suggests that this approach might involve less stretch applied to the brachial plexus when compared with suprapubic pressure or rotation of the fetal shoulders [79] and, when it can be accomplished, is highly effective in relieving the obstruction [80]. Fracture of the humerus is not an uncommon complication of posterior arm delivery, especially when flexion of the elbow is difficult or impossible. Most such injuries heal well.
Extreme maneuvers
Clavicle fracture
When more conservative approaches fail, intentional fracture of the clavicle may relieve a shoulder dystocia. In fact, spontaneous fractures are not often associated with BPI, probably because the collapse of the shoulder girdle precludes the problem. The posterior clavicle is generally most accessible. Careful technique is required to avoid injuring the subclavian vessels or the apex of the lung.
Zavanelli maneuver
When all attempts to relieve shoulder obstruction have failed, the Zavanelli maneuver is an option. The fetal head is flexed and pushed back into the uterus, reversing the movements of the preceding mechanism of labor. Expeditious cesarean is then done. There are case reports and case series detailing the use of this maneuver, with mixed results [81–83]. During the time before delivery, death or permanent peripheral or central nervous system injury may have already occurred [81]. However, the technique can be successful, with excellent maternal or neonatal outcome [82]. Because it is done so infrequently, and not all cases are reported, it is impossible to evaluate the relative risks and benefits of the Zavanelli maneuver.
Symphysiotomy
This procedure, which involves surgical division of the symphysis pubis via an incision in the mons veneris, is performed rarely in the developed world, although it does have advocates [84,85]. In cases of intractable shoulder dystocia, often after a failed Zavanelli maneuver, it has been used as a last resort [86]. Injury to the urethra, an unstable pelvis and chronic osteitis pubis can complicate the procedure and recovery.
Atypical presentations
Maneuvering the posterior shoulder into the sacral hollow
When initial examination after diagnosis of suspected shoulder dystocia finds the anterior shoulder impacted behind the symphysis and the posterior shoulder above the sacral promontory, most maneuvers will be of no avail until the posterior shoulder is engaged. To accomplish this, place traction on the fetal scapula or, if necessary, the axilla, pulling the shoulder into the sacral hollow. The movement of the shoulder into the pelvis from above the inlet will allow manipulation of the shoulders or posterior arm to facilitate delivery. The use of an axillary sling to manipulate the posterior shoulder has been advocated [87].
Compound presentations
The presence of a prolapsed arm or a leg adjacent to the head can cause shoulder dystocia, and identifying a compound presentation is vital before any of the usual maneuvers is initiated. The limb may remain in the vagina once the head delivers, or may be completely prolapsed. If it is a leg and cannot be easily repositioned, cesarean will be necessary. If it is the posterior arm, attempt to deliver it directly, and if the anterior arm presents, do a Woods maneuver to move it posteriorly to facilitate its delivery.
Rare sources of dystocia
Rarely, what appears to be shoulder dystocia is caused by other obstructive phenomena. A large sacrococcygeal teratoma, conjoined twins or fetal ascites can prevent delivery of the fetal body after the head emerges. In these situations, some clues from the physical examination can be helpful, and ultrasound imaging in the delivery room may make the diagnosis and guide therapy. Cesarean delivery is usually necessary, although in the case of ascites needle drainage can sometimes be done to diminish the abdominal circumference.
McRoberts maneuver
The McRoberts maneuver, which came into popular use in the early 1980s, is often recommended as the first maneuver to use in a case of shoulder dystocia. It has the advantages of simplicity, ease of application and no requirement for skillful manipulation. Full flexion of the mother's knees and hips against her abdomen may alter pelvic dimensions to favor delivery [88]. It is successful in a substantial number of cases [89]. However, in severe cases when the McRoberts maneuver does not work it may in fact contribute to the occurrence of BPI, and for this reason we do not recommend its use. The rotation of the pubic symphysis may draw the impacted anterior shoulder away from the head fixed at the introitus, thus introducing further tension on the already stretched brachial plexus roots. Moreover, when this procedure is used, it is often accompanied by unnecessary and potentially harmful downward traction on the head and suprapubic pressure. It does not work prophylactically, and potentially places more tension on the fetal neck than maneuvers that involve manipulation of the shoulders [2,90–92]. Adoption of McRoberts maneuver has been associated with an increased incidence of BPI over time [12], despite the increasing rate of cesarean delivery among macrosomic babies [93,94].
Maternal position
The importance of maternal posture requires emphasis. Most deliveries occur with the mother in a modified lithotomy position. All of the maneuvers described for manipulation of the fetus tend to be described from this perspective, which is most convenient for the attendant, but not always the most comfortable for the parturient. In fact, reports, admittedly anecdotal, from situations in which alternative positions for delivery are the norm suggest very low rates of shoulder dystocia. The use of the lateral position, or even squatting or kneeling on hands and knees should be considered for deliveries in which there is a high risk of shoulder dystocia. Moreover, these positions may be valuable in the management of obstructed shoulders and should be strongly considered if initial attempts at rotation of the shoulders are unavailing. These options can be limited by epidural anesthesia or by patients who find it difficult to change position. The Gaskin maneuver (moving the patient onto her hands and knees) and the lateral position often result in spontaneous or easily expedited delivery of the posterior shoulder [3]. If necessary, delivery of the posterior arm is often facilitated by these positions.
Conclusion
Shoulder dystocia is infrequent but has potentially serious consequences. It can be prevented by performing preemptive cesarean delivery on cases at very high risk, but our ability to identify such cases is still limited. Prompt diagnosis and optimal management of shoulder dystocia when it occurs is the key to preventing permanent neurologic sequelae. Management requires the coordinated efforts of a team with the requisite skills. The team leader should direct the management and institute a series of maneuvers to extricate the fetus with minimal risk to it and the mother. A thorough understanding of relevant pelvic and fetal anatomy is necessary as well as of the mechanisms through which the dystocia can be resolved. Maneuvers that involve manipulation of the fetal shoulder girdle confer less tractile force on the fetal brachial plexus than manipulations of the mother or of the fetal head or neck.
Future perspective
To reduce the frequency of BPI, especially that associated with difficult shoulder delivery, will require progress on two seemingly paradoxical fronts. First, we must harness the ability of decision support software in electronic obstetric records to present the clinician with updated risk assessments during the course of pregnancy and labor. The supporting algorithms must be population-specific and be modeled so as to consider all relevant variables that might impact risk while there is still time to avoid hazardous vaginal delivery. Each hospital or governing organization will need to set thresholds for the number of cesareans that is an acceptable trade-off for preventing a BPI.
In addition to this futuristic approach there is a need to revisit education in some basic obstetric principles. The importance of clinical cephalopelvimetry and accurate identification of dysfunctional labor patterns – both arguably diminishing skills – is necessary for the best possible information to be fed to the computer algorithms.
Study of the relative value of various maneuvers or sequences of maneuvers for management of shoulder dystocia could, in theory, be accomplished in a series of randomized clinical trials. Such studies would be difficult to design because of the many potentially confounding variables to take account of, would require very large multi-institution samples, and would be very expensive and complex.
Executive summary

Shoulder dystocia occurs in 0.15–2% of all deliveries.
Brachial plexus Injury is diagnosed in up to 20% of newborns after shoulder dystocia. Injury is transient in most, but can lead to serious permanent disability.
Risk factors can be identified in the patient's history and during prenatal care and labor. Most prominent are a history of prior shoulder dystocia or brachial plexus injury, current fetal macrosomia, maternal obesity, diabetes mellitus, excessive weight gain and dysfunctional labor patterns, especially a long deceleration phase followed by a long second stage.
Prediction of this complication is imperfect, but many cases can be avoided by taking multiple risk factors into account and delivering the highest risk cases by preemptive cesarean. Risk assessment is best accomplished by multivariate analysis, but current systems can predict only about a third of cases.
Skillful use of a logical series of maneuvers can prevent injury in many cases. Staff training and simulation exercises are helpful to prepare for management of this emergency.
When shoulder dystocia is suspected it is useful to await the next contraction before initiating any maneuvers. Traction on the fetal head and neck should be scrupulously avoided.
Initial maneuvers should focus on rotation of the fetal shoulders. If unsuccessful, delivery of the posterior arm is usually helpful, as is moving the mother into a lateral or hands-and-knees position.
Limited data exist on the effectiveness and safety of intentional clavicle fracture, the Zavanelli maneuver and the use of symphysiotomy. They could be considered, however, in desperate cases.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
