Abstract
Aim:
To investigate whether anti-Müllerian hormone (AMH) is associated with IVF cycle outcomes in young patients with diminished ovarian reserve.
Materials & methods:
Retrospective study of patients <35 years of age undergoing fresh IVF who had at least two 8-cell, day-3 embryos transferred with grades 1, 1.5 or 2. Patients were subgrouped,
Results:
In total, 1005 patients were included. Patients in the >1 ng/ml group required lesser gonadotropins compared with the <1 ng/ml and the <0.5 ng/ml group. More oocytes were retrieved from the same group compared with the latter two (p < 0.001). Despite these differences, the overall rates of clinical pregnancy, spontaneous abortion and live birth were comparable between the two groups.
Conclusion:
In patients with diminished ovarian reserve who have good quality embryos, AMH is not associated with clinical pregnancy, spontaneous miscarriage or live birth rates.
Introduction
Diminished ovarian reserve (DOR) refers to a decreased reproductive potential, primarily based on the quantity of remaining oocytes [1]. While a decline in reproductive potential is considered part of the natural aging process [2], it may also be encountered in young women [3]. Several clinical markers for DOR are currently used, including ultrasonographic measurements of antral follicle count, early follicular levels of follicle-stimulating hormone (FSH) and anti-Müllerian hormone (AMH) [4]. Although a serum FSH level >10 mIU/ml is a commonly accepted cut-off for DOR [5,6], cycle-to-cycle variations in FSH levels are frequently encountered in the early follicular phase [4,7]. Furthermore, recent data suggests that AMH levels better correlate with the number of early antral follicles compared with other hormonal markers [8,9]. Given these advantages of AMH over FSH, serum AMH has gained popularity in the recent years.
The utility of serum AMH in predicting response to ovarian stimulation has been well established [10]; however, the use of AMH as a marker of pregnancy outcomes remains contentious. Previous studies have suggested that patients with serum markers of DOR have lower rates of clinical pregnancy (CP) and live birth (LB) after IVF–embryo transfer (ET) cycles [11–13]. However, some recent studies, including work from our group have questioned the utility of AMH as a predictor of pregnancy outcomes after IVF–ET [14–16]. To test these hypotheses, we investigate whether AMH levels are associated with CP, spontaneous abortion (SAB) and LB rates in patients <35 years of age with DOR and good quality embryos.
Materials & methods
Cycle inclusion criteria
The institutional review board at Weill Cornell Medical College approved our retrospective cohort study. All fresh IVF cycles initiated at the Ronald O Perelman and Claudia Cohen Center for Reproductive Medicine between January 2010 and July 2013 resulting in ET were analyzed for potential inclusion. Only patients who had two 8-cell, day-3 embryos transferred with grades 1, 1.5 or 2 were included in the analysis. Patients with unilateral oophorectomy, known polycystic ovarian syndrome or adrenal disease were excluded. Cycles canceled prior to oocyte retrieval, those utilizing donor oocytes, or those with incomplete records were also excluded.
Clinical & laboratory protocols
All AMH measurements were performed at our center's laboratory. Serum samples were analyzed using the GenII Beckman ELISA assay (Beckman Coulter Inc., CA, USA). Serum AMH levels were measured prior to initiating ovarian stimulation in each patient. The intra- and inter-assay variability for AMH were 5.8 and 13.4%, respectively [17].
Controlled ovarian stimulation (COS), human chorionic gonadotropin (hCG) trigger and oocyte retrieval were performed per our center's standard protocols [17]. Initial gonadotropin dosages were based on patient age, weight, antral follicle count and previous response to stimulation, if any. COS was achieved with gonadotropins (Follistim, Merck, NJ, USA; Gonal-F, EMD-Serono, Geneva, Switzerland; and/or Menopur, Ferring Pharmaceuticals Inc, NJ, USA). Ovulation was suppressed using 0.25 mg Ganirelix Acetate (Merck) or Cetrotide (EMD-Serono).
hCG was used as the ovulation trigger. The hCG trigger was generally given when the two lead follicles attained a mean diameter >17 mm and was administered according to a sliding scale (10,000 IU for estradiol [E2] <1500 pg/ml; 5000 IU for E2 1501–2500 pg/ml; 4000 IU for E2 2501–3000 pg/ml and 3300 IU for E2 >3001 pg/ml) [17,18]. Oocyte retrieval was performed under conscious sedation and transvaginal ultrasound (TVUS) guidance with a 30 cm 16 G oocyte aspiration needle (Cook® Medical, IN, USA) 35–36 h after the ovulatory trigger. Luteal support with 50 mg of intramuscular progesterone daily was begun the day after retrieval. Fertilization of oocytes was performed with conventional insemination or intracytoplasmic sperm injection (ICSI) based on the couple's history and the male partner's semen analysis. All embryos were cultured using in-house culture media. Cleavage embryos were assessed on day 2 (44–46 h after insemination or sperm injection) and on day 3 (66–72 h insemination or sperm injection) [18]. Embryos were graded based on criteria described by Veeck [19]. ETs were performed with Wallace catheters (Smiths Medical, OH, USA) at approximately 1 cm less than the uterine depth identified at prior trial transfer.
Outcome variables
Demographic characteristics included age, parity, BMI (kg/m2), infertility diagnosis and previous IVF attempts. Baseline IVF cycle characteristics included basal FSH level and type of COS protocol. COS parameters recorded were total days of COS, total gonadotropins administered (IU), peak E2 level (pg/ml), peak endometrial stripe (mm), total number of oocytes retrieved, total number of mature oocytes and fertilization rate (%). Pregnancy outcomes following ET were also noted. Implantation rate was defined as number of gestational sacs seen via transvaginal ultrasound out of the total number of embryos transferred. Clinical pregnancy (CP) rate was defined as the number of intrauterine gestations with fetal cardiac activity per IVF–ET cycle. Pregnancy loss after visualization of an intrauterine gestation was considered a SAB. Any birth after 24 weeks of gestation was considered a LB.
Statistical analyses
For the purpose of this study, patients were subgrouped, a priori, based on serum AMH levels: <1 or >1 ng/ml and <0.5 or >0.5 ng/ml. At our center, serum AMH levels ≤1 ng/ml and ≤0.5 ng/ml are considered the cut-offs for diminished and very low ovarian reserve, respectively [17]. For the first part of our analysis, we compared the demographics, baseline IVF characteristics, ovarian stimulation parameters and pregnancy outcomes after ET in patients with AMH ≤1 ng/ml and >1 ng/ml. Thus, this analysis compared outcomes of patients with DOR (AMH ≤1 ng/ml) and normal ovarian reserve (AMH >1 ng/ml) having good quality embryos. The aforementioned analysis was replicated for patients with very low ovarian reserve (AMH ≤0.5 ng/ml) and controls (AMH >0.5 ng/ml) with good quality embryos.
Continuous variables were expressed as mean ± standard deviation (SD). Categorical variables were expressed as number of cases (n) and percentage of occurrence (%). Student's
Results
In total, 1005 patients met inclusion criteria. Table 1 compares the demographic and baseline IVF characteristics of patients with AMH ≤1 and >1 ng/ml. There was no statistical difference in the mean age, parity, BMI, distribution of infertility diagnosis, previous IVF attempts or basal FSH level. The distribution of COS protocols was also similar. Table 2 compares COS response between the two groups. Patients in the ≤1 ng/ml group required more gonadotropins compared with the >1 ng/ml group (2517.1 [±1056.8] IU vs 1952.1 [±968.4] IU; p < 0.001). The peak E2 level was 1890.5 (±678.3) pg/ml in the >1 ng/ml group, which was higher than the peak E2 level of 1687.4 (±637.8) pg/ml in the ≤1 ng/ml group (p < 0.001). More oocytes were retrieved from the former group compared with the latter [13.8 (±5.51) vs 9.40 (±4.62); p < 0.001]. Table 3 compares the pregnancy outcomes of patients with AMH ≤1 and >1 ng/ml after fresh IVF–ET cycles. The CP, SAB and LB rates in the ≤1 ng/ml group were 49.2, 5.97 and 43.2%, respectively. The corresponding rates in the >1 ng/ml group were 50.2, 6.86 and 43.3%, respectively. The adjusted OR after multivariate logistic regression analysis accounting for total gonadotropins administered, peak E2 level and number of oocytes retrieved was 0.95 (95% CI: 0.54–1.65) for CP, 0.86 (95% CI: 0.28–2.68) for SAB and 0.97 (95% CI: 0.56–1.73) for LB. Overall, Tables 1–3 compare outcomes of patients with normal ovarian reserve (AMH >1 ng/ml) and DOR (AMH ≤1 ng/ml) having good quality embryos.
Demographic and baseline IVF characteristics of patients with anti-Müllerian hormone ≤1 and >1 ng/ml (n = 1005).

Data are presented as mean (±standard deviation) and n (%).
AMH: Anti-Müllerian hormone; COS: Controlled ovarian stimulation; FSH: Follicle-stimulating hormone; GnRH-a: Gonadotropin-releasing hormone agonist; GnRH-ant: Gonadotropin-releasing hormone antagonist.
Ovarian stimulation outcomes of patients with anti-Müllerian hormone ≤1 and >1 ng/ml (n = 1005).

Data are presented as mean (±standard deviation).
AMH: Anti-Müllerian hormone; E2: Estradiol; ICSI: Intracytoplasmic sperm injection.
IVF cycle outcomes of patients with anti-Müllerian hormone ≤1 and >1 ng/ml (n = 1005).

Data are presented as mean (±standard deviation) and n (%).
AMH: Anti-Müllerian hormone.
Table 4 compares the demographic and baseline IVF characteristics of patients with AMH ≤0.5 and >0.5 ng/ml. Overall, there was no difference between the two groups. Table 5 compares COS response between the two AMH groups. Similar to patients in the AMH ≤1 and >1 ng/ml groups, patients in the ≤0.5 ng/ml group required more gonadotropins (3328.2 [±1765.9]) IU compared with the >0.5 ng/ml group (2815.1 [±1405.6]) IU (p < 0.001). The peak E2 level was higher in the >0.5 ng/ml group compared with the ≤0.5 ng/ml group (1595.9 [±640.9] pg/ml vs 1190.1 [±672.2] pg/ml; p < 0.001). The total and mature number of oocytes in the >0.5 ng/ml group was higher than the ≤0.5 ng/ml group. Table 6 compares the pregnancy outcomes of patients with AMH ≤0.5 and >0.5 ng/ml after fresh IVF–ET cycles. The CP, SAB and LB rates in the ≤0.5 ng/ml group were 50.5, 6.93 and 43.6%, respectively. The corresponding rates of CP, SAB and LB in the >0.5 ng/ml group were similar: 52.2, 7.63 and 44.6%, respectively. Following multivariate logistic regression analysis, the adjusted OR was 0.94 (95% CI: 0.54–1.63) for CP, 0.90 (95% CI: 0.31–2.62) for SAB and 0.96 (95% CI: 0.55–1.68) for LB.
Baseline characteristics of patients with anti-Müllerian hormone ≤0.5 and >0.5 ng/ml (n = 1005).
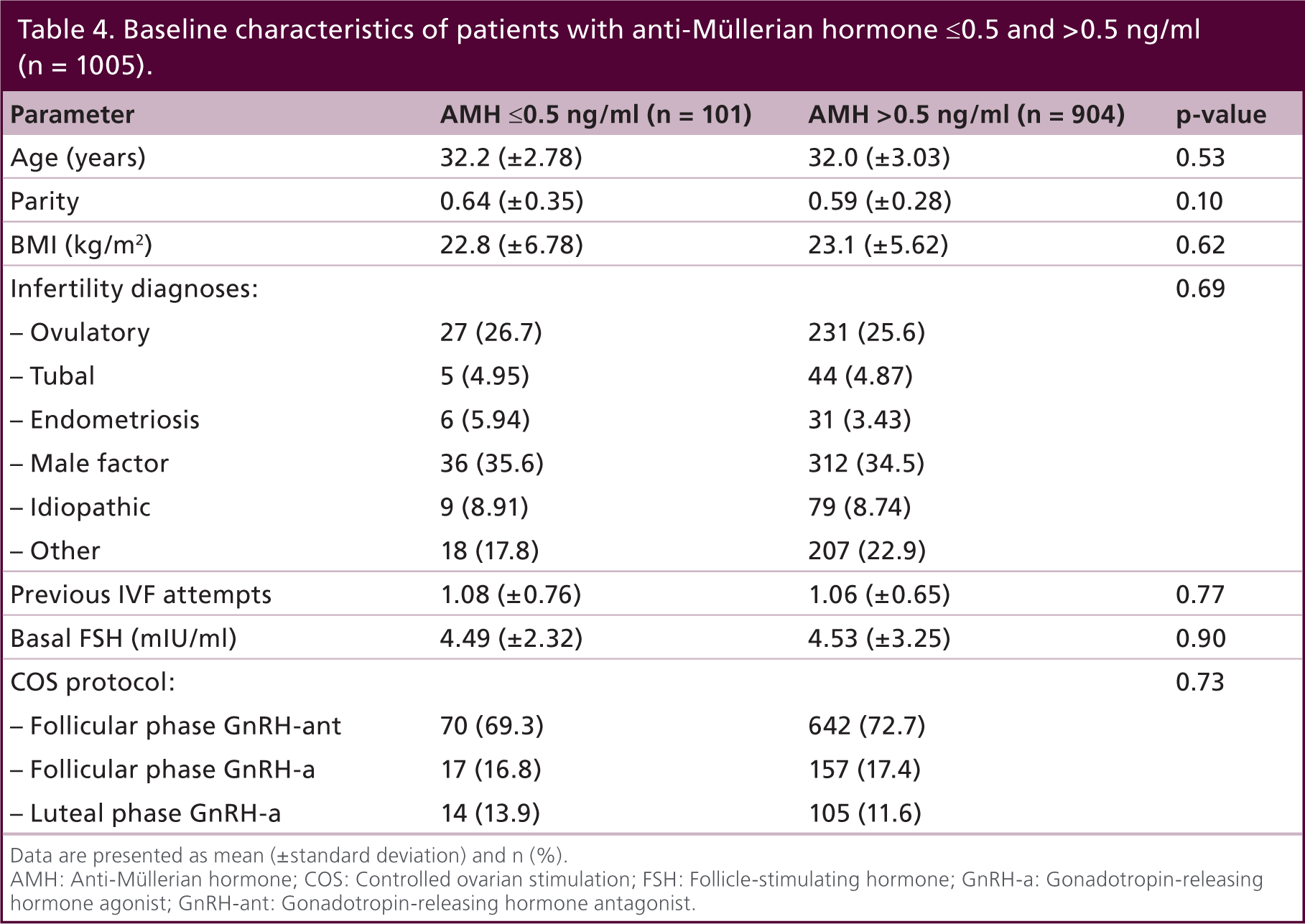
Data are presented as mean (±standard deviation) and n (%).
AMH: Anti-Müllerian hormone; COS: Controlled ovarian stimulation; FSH: Follicle-stimulating hormone; GnRH-a: Gonadotropin-releasing hormone agonist; GnRH-ant: Gonadotropin-releasing hormone antagonist.
Ovarian stimulation outcomes of patients with anti-Müllerian hormone ≤0.5 and >0.5 ng/ml (n = 1005).

Data are presented as mean (±standard deviation).
AMH: Anti-Müllerian hormone; E2: Estradiol; ICSI: Intracytoplasmic sperm injection.
IVF cycle outcomes of patients with anti-Müllerian hormone ≤0.5 and >0.5 ng/ml (n = 1005).
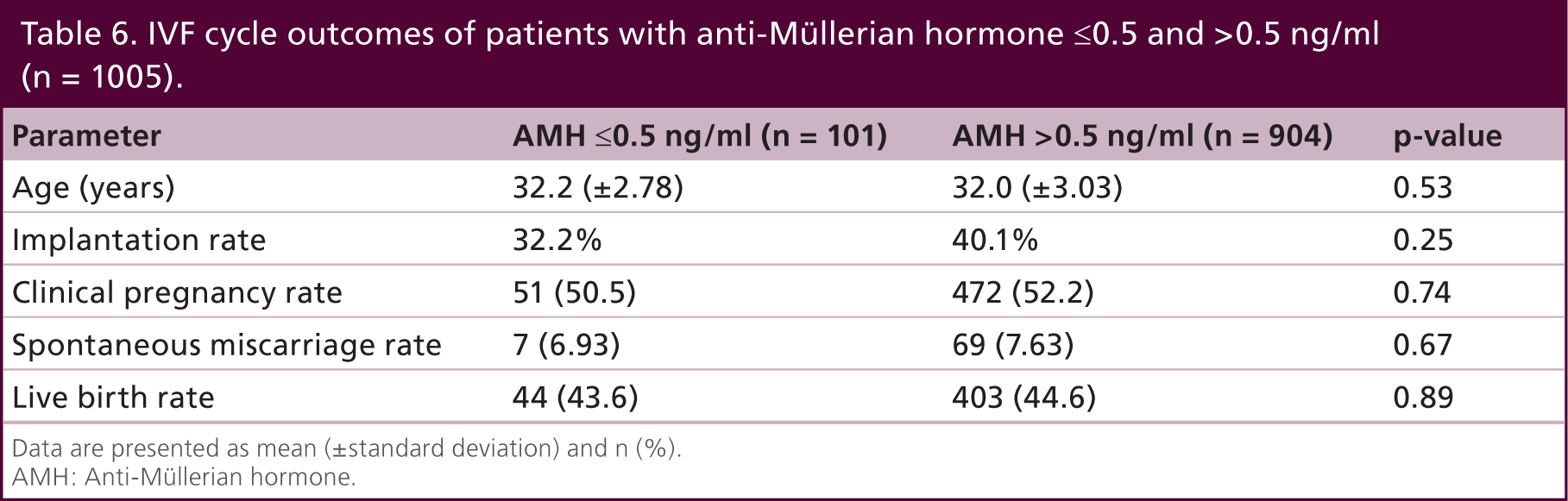
Data are presented as mean (±standard deviation) and n (%).
AMH: Anti-Müllerian hormone.
Discussion
In the current study, we investigate the association between AMH and pregnancy outcomes after fresh IVF–ET cycles in patients <35 years with DOR. Our data suggests that patients with AMH >1 ng/ml compared with ≤1 ng/ml and AMH >0.5 ng/ml compared with ≤0.5 ng/ml require less gonadotropins, have higher peak E2 levels and have more oocytes retrieved. Yet, the overall CP, SAB and LB rates are comparable across all groups, suggesting that there is no association between serum AMH levels and pregnancy outcomes in young patients with DOR who have good quality embryos.
The role of serum AMH in predicting the likelihood of IVF–ET success continues to be a subject of debate. Several meta-analyses [11–13] and individual studies have suggested that low AMH correlates with lower rates of clinical pregnancies [20–24]. In their retrospective study of 892 women undergoing 1230 IVF–ICSI cycles, Brodin
Contrary to the aforementioned findings, several recent studies have questioned the utility of AMH as a predictor of pregnancy outcomes after IVF–ET [14–16]. In one study of 2760 patients undergoing 4072 cycles, Reichman
Our findings further substantiate that AMH is not associated with pregnancy outcomes in young patients with DOR – that is, poor ovarian reserve or response in young patients does not equate to poor IVF outcomes. The strengths of this study include its large sample size and the specific patient cohort of young patients with DOR. By including only young patients with good quality embryos, we are able to more precisely isolate the role of serum AMH levels, reflecting quantitative ovarian response, as a correlate of various pregnancy outcomes. By excluding canceled cycles, our results demonstrate per-cycle pregnancy outcomes rather than cumulative pregnancy outcomes, which would be negatively affected by the low oocyte yield expected in a low AMH subgroup [33]. A limitation to the generalizability of this retrospective study is that we did not account for the burden of canceled cycles, which in practice is a significant barrier to LB rates per cycle start. We also did not evaluate the progression of day-3 embryos to the blastocyst stage in the current study cohort. Finally, as with any retrospective study, it is important to analyze prospective data to validate our findings.
In conclusion, the findings of this study are consistent with previous studies highlighting that AMH levels correlate well with the number of oocytes retrieved per IVF cycle [14]. Despite a lower oocyte yield, in young patients with DOR who have good quality embryos, AMH is not associated with CP, SAB or LB rates after fresh IVF–ET cycles. As evident from the study cohort, young patients with DOR can achieve reasonable IVF–ET outcomes when good quality embryos are available. While these patients should not be discouraged from pursuing IVF treatment based on low or undetectable AMH levels, they should be aware of the risk of IVF cycle cancellations [14,30].
Executive summary

Diminished ovarian reserve (DOR) refers to a decreased reproductive potential, primarily based on the quantity of remaining oocytes.
The utility of serum anti-Müllerian hormone (AMH) in predicting response to ovarian stimulation has been well established; however, the use of AMH as a marker of pregnancy outcomes remains contentious.
Previous studies have suggested that patients with serum markers of DOR have lower rates of clinical pregnancy (CP) and live birth (LB) after IVF – embryo transfer (ET) cycles.
Our data suggests that patients with AMH >1 ng/ml compared with <1 ng/ml and AMH >0.5 ng/ml compared with <0.5 ng/ml require less gonadotropins, have higher peak estradiol levels and have more oocytes retrieved.
Yet, the overall CP, spontaneous miscarriage and LB rates are comparable across all groups, suggesting that there is no association between serum AMH levels and pregnancy outcomes in young patients with DOR who have good quality embryos.
This study confirms that AMH levels correlate well with the number of oocytes retrieved per IVF cycle.
Despite a lower oocyte yield, in young patients with DOR who have good quality embryos, AMH is not associated with CP, spontaneous miscarriage and LB rates after fresh IVF–ET cycles.
As evident from the study cohort, young patients with DOR can achieve reasonable IVF–ET outcomes when good quality embryos are available.
While these patients should not be discouraged from pursuing IVF treatment based on low or undetectable AMH levels, they should be aware of the risk of IVF cycle cancellations.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
