Abstract
Defecating disorders are a common and complex problem. There are a range of anatomical and functional bowel abnormalities that can lead to this condition. Treatment is difficult and needs a multidisciplinary approach. First line treatment for defecating disorders is conservative. For those that fail conservative treatment, some may respond to surgical therapy but with variable results. The aim of this review is to offer an overview of defecating disorders as well as provide an algorithm on how to diagnose and treat them with the help of a multidisciplinary and multimodal approach.
Keywords
Defecating disorders can be defined as the inability to evacuate contents from the rectum despite excessive straining and are frequently but not invariably associated with perineal descent [1]. Defecating disorders are not an uncommon problem, occurring in up to 7% of the population [2]. Defecating disorders can frequently be associated with constipation and in these cases it is unclear whether the primary etiology of defecating disorders are solely due to the underlying constipation [3].
Primary (or idiopathic) constipation as defined by the Rome III Criteria [4] can be subdivided into three pathophysiologic subtypes [5,6]: slow transit constipation (STC), irritable bowel syndrome with constipation (IBS-C) and defecating disorders; which may not be mutually exclusive. Chronic constipation is a common self-reported bowel symptom that affects 2–30% of people in the western world. It has a considerable impact on both health costs and quality of life. About 30–50% of constipated patients also have defecating disorders and these patients may frequently require a multidisciplinary approach in their management [5,7]. The prevalence of constipation is higher in women (median female-to-male ratio of 1.5:1) and women are also more likely to use laxatives and seek healthcare advice for these symptoms [8]. The true incidence and prevalence of constipation and defecating disorders are very difficult to calculate, due to the large variability of study parameters, heterogeneity of patients, historical diagnosis and definitions used.
STC is characterized by a delay of stool transit through the colon; this diagnosis is most commonly being made on the basis of imaging. Irritable bowel syndrome (IBS) is defined as recurrent abdominal pain or discomfort for at least 3 days per month in the last 3 months associated with two or more of the following: improvement with defecation; onset associated with a change in frequency of stool; onset associated with a change in form (appearance) of stool. IBS-C, a form of IBS, can be further characterized by predominantly symptoms of constipation [4]. The aim of this article is to provide an overview of the causes and symptoms of defecating disorders and to cover the various treatment options available for these complex conditions.
Causes of defecating disorders
The etiology of defecation disorders is unclear as although in many cases, an anatomical or functional abnormality can be detected, not all patients will improve despite those abnormalities being treated. Patients will frequently adapt their defecation technique to try to improve some of their symptoms [9,10], sometimes leading to worsening them.
The commonest anatomical abnormalities seen (which this article will describe later in greater depth) include perineal descent, rectocele formation, rectal intussusception, rectal prolapse, sigmoidocele and enterocele formation [3].
Dyssynergic defecation is a functional abnormality and is probably the most common cause of defecating disorders. Most specialists believe that dyssynergic defecation is mostly an acquired behavioral defecation disorder [11] and has been described in the literature by a plethora of alternative terms including anismus, pelvic floor dyssynergia, obstructive defecation, paradoxical puborectalis contraction, pelvic outlet obstruction and spastic pelvic floor syndrome [12]. This has occurred because there has been an extreme lack of understanding in this field of medicine, resulting in poor quality studies. However, the term dyssynergic defecation is now the recommended terminology [13].
Dyssynergic defecation may be conceptualized as maladaptive learning in the process of defecation which results in pelvic floor dysfunction; perhaps initiated by the avoidance of pain or trauma [14] or even due to neglecting the call to defecate [8].
Obstetric trauma during vaginal delivery and dyscoordinated pelvic relaxation has also both been cited as causative agents leading to the development of abnormalities on defecography studies [15].
Symptoms of defecating disorders
The classical symptoms of defecating disorders can include one or more of the following symptoms: pain on defecation, the need to strain in order to defecate, an extended time spent on the toilet, long intervals between bowel evacuations (e.g., 5–10 days), perineal pain/discomfort, a feeling of incomplete evacuation, fragmented defecation, the necessity to perform anal or vaginal digitation or the need to add perineal pressure in order to defecate [5].
Clinical assessment
History
A detailed medical, surgical, pregnancy, dietary and drug history can facilitate the recognition of constipation and identify the more specific diagnosis of defecating disorders. Patients should be encouraged to describe their bowel habit. Questions to clarify whether they experience a sense of incomplete evacuation, or if they need to strain or use digital maneuvers are useful [16].
It is also important to understand their usual stool consistency and stool form with the help of the Bristol Stool Scale [17]. In addition, it is useful to know if they ignore a call to stool, if there are any precipitating events which have led to this behavior, how their cultural beliefs and expectations affect their bowel patterns, and finally whether their problem started in childhood [18].
In patients with ‘red flag’ symptoms (such as a change in bowel habit, bleeding, weight loss), a test to rule out colorectal disease is performed either in the form of an endoscopy or radiological imaging.
Stool diaries and constipation questionnaires can help discern some of these symptoms, especially as many patients misinterpret or feel embarrassed to describe them [19,20]. A psychological history is equally important. Studies in constipated subjects have shown a history of sexual abuse in 22–48% of subjects (particularly women), and physical abuse in 31–74% of constipated subjects [20]. The impact of these symptoms on the patient's quality of life can be categorized to some degree with the help of questionnaires. All of these aides will help the surgeon to better assess the complexity of the patient's complaints [2]. The most common scoring system used is the Wexner constipation score. The Wexner score (minimum score 0; maximum score 30) correlates well with objective physiological findings and provides a baseline in the evaluation of the constipated patient [21].
More recently, the obstructed defecation syndrome score was published and has been prospectively validated [22]. It consists of eight items (the mean time spent on the toilet, number of attempts to defecate in 24 h, whether anal or vaginal digitation is required, use of laxatives, use of enemas, incomplete or fragmented evacuation, straining and stool consistency) scored from 0 to 4 (0 is good and 4 is poor) with a maximum score of 32.
Examination
In the outpatient clinic, a full physical examination should be performed including an abdominal, perineal and rectal examination, and a neurological examination if required.
Anal conditions such as hemorrhoids, anal fissures, warts, skin tags and scars due to previous obstetric trauma and anal surgery (such as previous fistulotomy or sphincterotomy) should be excluded. The anal canal needs to be examined to see whether it is ‘closed’ at rest before the patient is asked to squeeze and relax their anal sphincters. Patients are also asked to cough and push down, so that any perineal descent, mucosal or full thickness prolapse can be excluded.
A digital rectal examination is performed in order to exclude any palpable masses in the anus and lower rectum, to exclude the presence of a rectocele and to evaluate the resting pressure of the anal sphincters. Patients are then asked to squeeze and cough, and to bear down in order to evaluate their squeeze pressures and whether there is anal relaxation [23].
Straining proctoscopy will allow some degree of assessment of rectal mucosal descent.
Diagnostic procedures
The main diagnostic procedures for the evaluation of defecating disorders include anorectal manometry (ARM), balloon expulsion test, barium defecating proctogram and/or magnetic resonance (MR) defecography. Colonic transit studies can also be helpful in the diagnosis of defecating disorders. In addition, it is our local practice as a surgical clinic, to utilize an examination under anesthetic if the patients symptoms and proctography fail to correlate (e.g., if symptoms suggest a rectal prolapse but the imaging fail to demonstrate this, an examination under anesthetic is extremely helpful). If treatment therapy for STC fails, it is our recommendation that the condition of defecation disorders be excluded and vice versa as correct treatment of the associated disorder may improve the patient symptoms.
Anorectal manometry
ARM is not a single test but consists of a series of measurements. The parameters that should be determined by a standard ARM include: anal sphincter resting pressure, maximum anal sphincter voluntary squeeze pressure and squeeze duration, anal and rectal pressures during straining, anal and rectal pressures during coughing, the presence or absence of a recto-inhibitory reflex, and thresholds of rectal sensory perception (including threshold volumes for first sensation, desire to defecate, urgency to defecate and maximum tolerable volume) [24]. ARM can help with the detection of abnormalities including Hirschsprung's disease and dyssynergic defecation [25]. The absence of a rectoanal inhibitory reflex is considered pathognomonic for Hirschsprung's disease [25,26]. In a prospective study of 111 children, ARM had a sensitivity of 83% and specificity of 93% when compared with rectal suction biopsy (sensitivity 93% and specificity 100%) for detecting Hirschsprung's disease [26].
When normal subjects without symptoms attempt to defecate, there is usually arise in intrarectal pressure, which is synchronized with a fall in anal sphincter pressure due to relaxation of the puborectalis and external anal sphincter [20,27]. This maneuver is under voluntary control and is primarily a learned response [20]. The inability to perform this coordinated maneuver represents the chief pathophysiological abnormality in patients with dyssynergic defecation [20,27–28].
Manometry can be performed with either open-ripped or side-opening water-perfused catheters or direct online solid-state microtransducers, or air- or water-filled balloons [29].
The reproducibility of ARM is high, but its reliability depends on the operator's experience, and its use is limited by the absence of standardized protocols [30,31] and lack of data from large numbers of normal subjects [32]. Manometry results will also vary with age and sex and as normal values based on a large cohort of healthy individuals are still lacking, it causes a difficulty in interpreting manometry results [33].
It must be noted that up to 20% of asymptomatic healthy adults cannot produce a normal relaxation during attempted voluntary defecation [34]. The body position, whether sitting or lying down, the presence of stool-like sensation, and the consistency of stool may each influence the occurrence of dyssynergia and the inability to expel stools [35]. Hence, the finding of a dyssynergic pattern alone on a patients' manometry should not be considered as diagnostic of dyssynergic defecation [36]. For most patients, a complete history, examination with ARM can help to diagnose defecation disorders.
The maximum tolerable volume or pain threshold may be reduced in patients who have a noncompliant rectum (e.g., due to previous surgery such as an abdominoperineal pull-through or due to disease processes such as proctitis) [37]. The pain threshold may also be reduced in patients with IBS [38]. In contrast, a higher threshold for sensory perception suggests impaired rectal sensation or rectal hyposensitivity [38]. Subjects with dyssynergic defecation can demonstrate abnormal sensory perception and/or altered rectal wall contractility together with increased rectal tone [38]. Alterations in rectal compliance may result in a decreased or increased rectal capacity, an impaired ability to perceive rectal distention and an altered threshold for reflex internal anal sphincter inhibition. Conditions that decrease rectal compliance include ulcerative colitis, radiotherapy, previous surgery (e.g., previous anterior resection) and drugs [38].
Although not available in many institutions, 3D imaging from new anorectal catheters can give excellent information. 3D high-resolution ARM has been shown to correlate with conventional manometry but also have an advantage of providing pressure recording over the entire circumference and length of the anal canal [39].
Balloon expulsion test
A balloon expulsion test to diagnose dyssynergic defecation was first proposed by Barnes and Lennard-Jones. Patients with normal anorectal function should easily expel a balloon with 60 cc of air/water within 1 min [2]. However, it is now known that many patients with dyssynergic defecation can expel this balloon, hence this isolated test is insufficient to make a diagnosis of dyssynergic defecation [27]. Barium defecating proctogram is now more commonly used as a useful investigation for the diagnosis of dyssynergic defecation.
Defecating proctography
Although within our institution a defecating proctogram is the gold standard for the evaluation of defecating disorders, we acknowledge that in most institutions, a combination of history, focused anorectal physical exam, ARM, balloon expulsion and defecating proctography are important. Equally a defecating proctogram is able to assess the functional aspect of defecation, and provides specific measurements of the anorectal angle, the degree of perineal descent, and the puborectalis length [40]. The two most common defecating proctograms in use are described below.
Barium defecating proctogram
Barium paste is inserted into the rectum transanally, following which the patient is asked to sit on a portable toilet and asked to attempt to defecate. Simultaneously, video fluoroscopic images are recorded. This is a valuable test for the evaluation of the structural and functional disorders of the anorectal region [41]. Colpocysto-enterodefecography (i.e., the study of the anterior, middle and posterior pelvic floor compartments with fluoroscopy) can be performed when multiple compartment defects are suspected, including the presence of a cystocele, enterocele or descending perineum syndrome [42]. It must be noted that the presence of structural abnormalities within the pelvic floor may not cause defecating disorders. A recent study evaluated the variability of proctographic findings within a group of normal individuals [43]. In 28 asymptomatic women and 18 asymptomatic men, 26 women had a rectocele with a mean depth of 2.5 cm and there was recto-rectal intussusception in nine patients, but did not give overt symptoms.
MR defecography
MRI of the pelvic floor using MR defecography is now frequently recommended as a valid alternative to contrast radiography, particularly in patients with suspected multicompartment disorder due to the panoramic view that it provides and the absence of ionizing radiation [32].
Defecographic techniques using both closed and open magnets, with and without evacuation, have been described, and this has further increased the attraction of dynamic MRI for rectal obstruction [44].
Closed-configuration MR systems permit imaging in the supine position only with or without evacuation. The problem with this technique is that in situations of incomplete evacuation, the diagnosis may be attributed to the patient's position rather than representing a true abnormality [40]. Supine MR defecography also reduces its ability to detect the descending perineum, the presence of an enterocele, rectal intussusception as well as peritoneal sac herniations [45]. Seated MR (open MRI) using open-configuration magnets are superior to the supine MRI for identifying the above abnormalities, although few centers have the equipment to perform this test [8].
MR defecography is a relatively new technique for the diagnosis of pelvic floor disorders. It is a more expensive technique and at the moment is not proven to be superior to a barium defecating proctogram and therefore we recommend that it should be used only in selected patients until further evidence in the literature is available
Until there is widespread availability of open MRI to allow patients to sit while evacuating and more prospective trials to validate the findings of MR defecography, traditional barium defecating proctogram remains the gold standard imaging technique for evaluation of pelvic floor disorders [40].
3D transperineal ultrasound
Lately, 3D transperineal ultrasound (3DTPUS) is being used for defecating disorders. 3DTPUS enables dynamic investigation of all three pelvic compartments at low cost. Dietz et al. [47,48] have reported that 3DTPUS can differentiate between different forms of posterior anatomic abnormalities such as rectocele and enterocele [49]. Unfortunately, 3DTPUS is not currently widely available, and more studies are needed to help evaluate it in its use in defecating disorders.
Pathology identified by defecating proctography
Anatomical changes & abnormalities seen on proctography
Rectoceles are identified as outward herniations of the rectal wall through an area of weakness, usually anterior through the rectovaginal septum into the vagina, a lower pressure region
The most common findings seen on a defecating proctogram in patients with defecating disorders include the prolonged retention or inability to expel barium paste, the presence of a mucosal intussusception and the presence of a rectocele [37,51].
Although defecography reveals abnormalities in 77% of all subjects, there is no direct relationship between the presence of symptoms with radiological abnormalities as many of these abnormalities were asymptomatic [37,43,51]. Because of this, it is important to treat the patient's symptoms rather than what is shown on the proctogram.
As mentioned previously, normal perineal descent can vary up to 3 cm. Further descent is defined as the descending perineum syndrome which is characterized by the ballooning of the perineum, several centimeters below the bony outlet of the pelvis during a straining effort. Approximately 75% of patients with descending perineum syndrome present with defecation disorder symptoms [52].
Hysterectomy, childbirth, menopause, age, previous perineal surgery, obesity, diabetes, straining at stool and pudendal neuropathy are associated with descending perineum syndrome [53].
Descending perineum syndrome is indicative of a pelvic floor weakness and is usually associated with other anorectal disorders such as rectal prolapse, rectocele or enterocele formation [54] and is diagnosed using a proctogram [55].
Like most of the pelvic floor conditions, the main treatment for descending perineum syndrome is conservative, using a high-fiber diet, laxatives, enemas and biofeedback [3]. Also patient can use an artificial device for reduction of extensive straining [55].

In the following paragraphs, we list the common conditions seen in defecating disorders and the possible treatment options available:
Dyssynergic defecation
Dyssynergic defecation is the most common cause of defecating disorders [56]. It is an acquired behavior and in most patients, dyssynergic defecation is a result of either poor toileting habits, the possible presence of painful defecation, a history of an obstetric or back injury, or the presence of a brain–gut dysfunction [12,38]. Some patients may have a history of sexual or physical abuse, or have evidence of an eating disorder [57]. Anxiety and psychological stress may also contribute to its development although there lacks substantial evidence to support this being the sole etiology of these conditions. [58].
Patients with dyssynergic defecation demonstrate an inability to coordinate their abdominal, rectoanal and pelvic floor muscles to facilitate defecation, which leads to paradoxical anal contraction (78%) or inadequate anal relaxation. In addition, 50–60% of patients also demonstrate a degree of impaired rectal sensation [28].
Dyssynergic defecation is diagnosed when a patient exhibits a dyssynergic pattern of defecation on ARM and demonstrates one other quantifiable measure of abnormal defecation such as an abnormal balloon expulsion test or a prolonged delay in colonic transit or incomplete evacuation during defecography [12,13]. In our own practice and experience, a barium defecating proctogram has been the gold standard for diagnosis. However, it is acknowledged that it can produce a false-positive diagnosis as defecation in a clinical setting can never reproduce the safety and familiarity of the home environment [59].
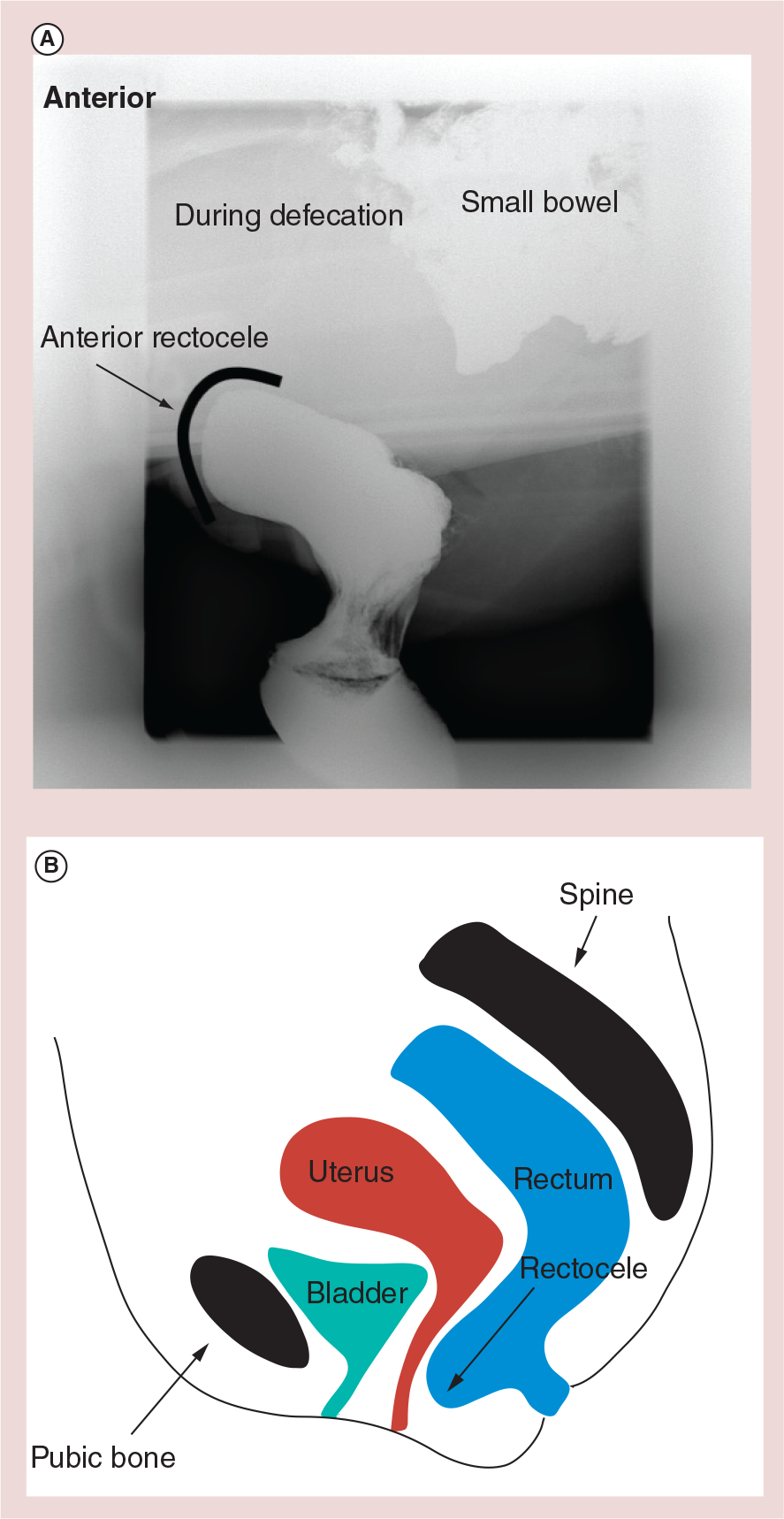
The initial management of dyssynergic defecation is conservative and will include advice on dietary, activity levels and eating habits in order to achieve a normal stool type and a regular bowel pattern. Patients are advised to increase dietary fiber and fluids and to adopt a regular pattern of eating and increase activity levels. In addition, pelvic floor and abdominal muscle re-education is used as first-line therapy [37,60–61]. If this fails, patients are treated with biofeedback therapy.
Biofeedback is defined as the technique of making unconscious or involuntary bodily processes perceptible to the senses (in this case by use of electromyography or balloon manometry) in order to manipulate them by conscious mental control. The goal of muscle re-education and biofeedback therapy is to retrain the normal defecation process. This includes pelvic floor muscle re-education to improve cortical awareness of pelvic floor muscle contraction and relaxation and abdominal muscle activity during abnormal and normal straining (increase intra-abdominal pressure). These key elements combine with optimal toilet positioning to achieve defecation. By following this ‘relearning to defecate process’, the nonrelaxing pelvic floor is gradually suppressed allowing normal coordination to be restored [23]. Randomized controlled trials have shown a success rate of approximately 70% and a long-term success rate of approximately 50% with biofeedback [62]. A Cochrane metanalysis failed to provide firm conclusions in the efficacy and safety of biofeedback when used in the management of people with chronic constipation and dyssynergic defecation, but the reason for this conclusion may be due to the lack of high quality studies in the literature [63].
Patients who fail a course of such conservative management, can be considered for botulinum toxin (BT) injection into the puborectalis muscle and external anal sphincter. Based on small, uncontrolled studies, injection of BT into the puborectalis muscle, has a variable effect [64,65]. In total, 75% of patients with BT injections improve, although the benefit is usually short term, ranging from 1 to 3 months [66,67]. The most significant side effect is transient fecal incontinence, which occurs in 25% of patients, but resolves in 1–3 months. Many also seem to have some degree of pain as well [68]. A recent study from the Oxford group showed a 95% response rate in patients without rectal intussusception, and 95% of these patients sustain a response following a median follow-up of 19.2 months [69].
Rectal intussusception
Rectal intussusception can be described as the invagination of rectal wall during defecation. The intussusception may be entirely contained within the rectum, can reach the dentate line, can protrude into the anal canal (recto-anal intussuception) or even progress out through the anus (therefore becoming an external rectal prolapse) [70]. Rectal intussusception can progress to an overt rectal prolapse in approximately 2% of patients [71–73]. In total, 50% of patients with rectal intussusception, in addition to the usual symptoms associated with obstructed defecation will also complain of fecal incontinence [74,75]. Possible mechanisms for this include an occult defect of the external anal sphincter or a traction-induced pudendal neuropathy resulting from a longstanding, excessive straining to defecate [70]. Patients have also been shown to have other structural abnormalities including the presence of a rectocele or a solitary rectal ulcer [76].
The initial treatment of rectal intussusception is conservative therapy, including the use of dietary changes, laxatives and biofeedback [68]. The reported success rate of biofeedback therapy ranges from 37 to 100% for patients with rectal intussusception presenting with obstructive defecation and 50–90% for those presenting with fecal incontinence [77].
Surgery is an option if conservative management fails. These include performing an internal Delorme's procedure (redundant rectal mucosal is dissected and excised cephalad through the anus). An abdominal rectopexy, laparoscopic ventral rectopexy (LVR), or the use of the stapled transanal resection of rectum (STARR: is a full thickness circumferential resection of lower rectum using stapling devices [78]) procedure have also been used to treat this condition with variable results [79,80].
In 2012, the results of 167 patients who underwent an internal Delorme's procedure for rectal intussusception were published; the authors reported an overall success rate of 73.7% and a recurrence rate of 5.4% [81].
Abdominal posterior rectopexy for rectal intussusception has been reported as having poor results, especially being linked with the development of constipation following surgery [82].
STARR has been used for the surgical treatment of rectal intussusception. A recent prospective study reports an improvement of both the obstructive defecation syndrome and fecal incontinence scores. Recurrence of obstructed defecation symptoms was 4.9%, but this was a relatively short follow-up of 98 weeks. It was noted that there was an early complication rate of 16.2% with problems of fecal urgency after this procedure, although these urgency symptoms did seem to improve with time [83].
Enterocele/Sigmoidocele
Both an enterocele and sigmoidocele are defined as a herniation of the peritoneal sac between the vagina and the rectum. This hernia may contain either the sigmoid colon (sigmoidocele) or more commonly the small bowel (enterocele) [84]. The epidemiology of enterocele or sigmoidocele formation is currently unknown [85]. However, there is a high correlation between women who have had a previous hysterectomy and enterocele formation [78]. Enteroceles are most frequently found in elderly, multiparous females [84]. Usually, patients with enteroceles have concomitant anorectal morphological abnormalities including the presence of a rectocele, excessive perineal descent or rectal intussusception [78,85]. Patients usually present with either pelvic pain or heaviness (on standing), with symptoms of difficult defecation and the feeling of incomplete evacuation. Both enteroceles and sigmoidoceles are diagnosed with a barium defecating proctogram, with oral contrast, or with MR defecography [3].

For both conditions, conservative management is the initial treatment. If symptoms persist, surgical treatment may be beneficial [76]. For enterocele repair, a laparoscopic ventral rectopexy is proposed as the new treatment modality if other anatomical anomalies are also present [86]. For sigmoidoceles, sigmoid resection and rectopexy with obliteration of the Douglas pouch have been performed.
Rectocele
A rectocele is the protrusion of the rectal wall, usually anteriorly and toward the vagina [56]. The true incidence of this anomaly is unknown. A prospective study, on healthy asymptomatic subjects, showed that up to 93% of female subjects, regardless of parity had an anterior rectocele [43]. It is believed that rectoceles are caused by weakness of the pelvic floor and the rectovaginal septum. Childbirth and/or excessive and inappropriate straining weaken the vaginal septum, pelvic floor muscles and damage the pudendal nerve [76]. However, rectoceles are also seen in 40% of nulliparous women [87]. As you can see, there is a large variation of rectocoele incidence within the literature as the demographics of these volunteers and patients within these studies can be quite different. What is apparent though is that patients can have a rectocoele but can be fairly asymptomatic with it.
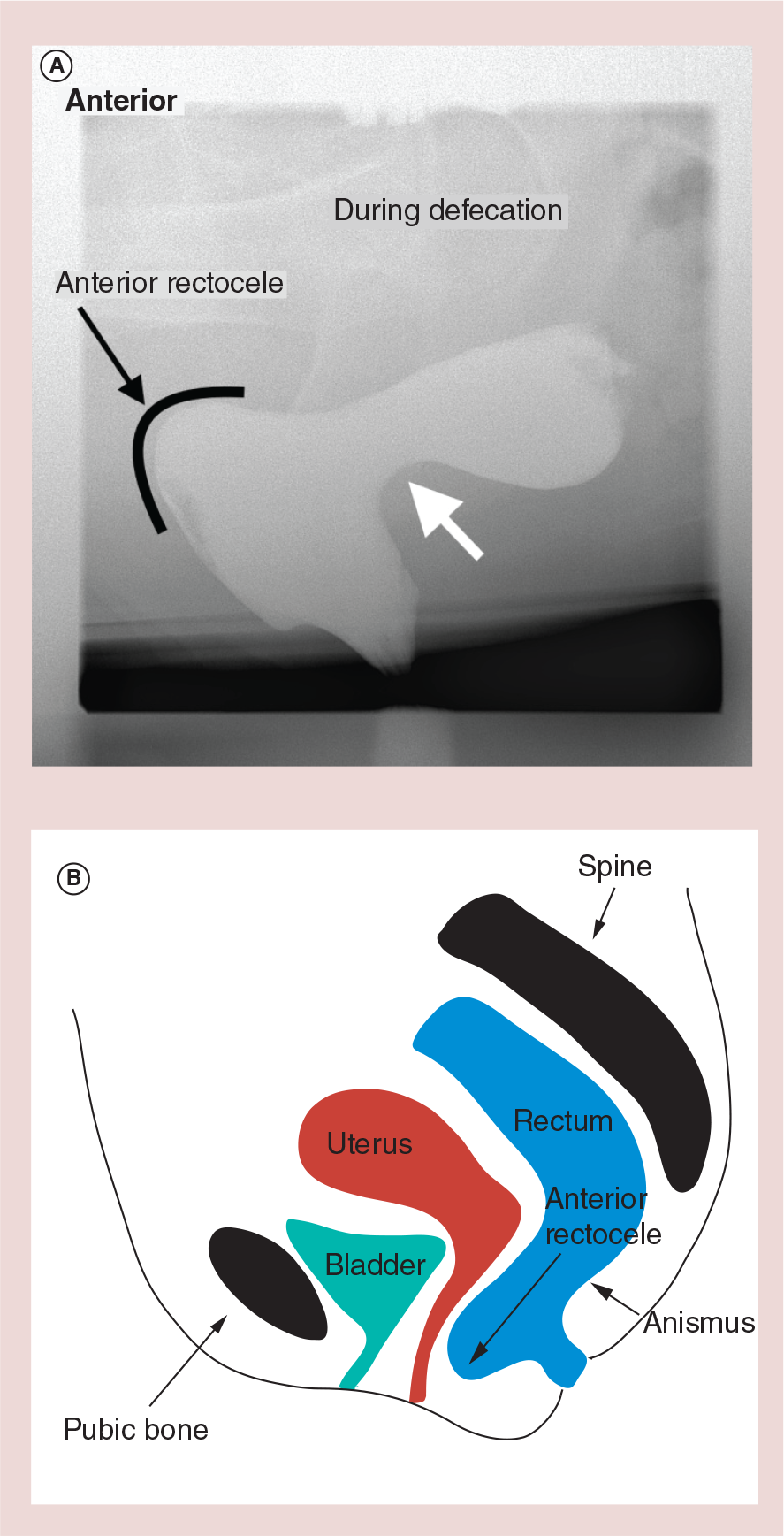
Dyssynergic defecation and intussusception are often associated with rectoceles, [76] but it is unclear if rectoceles are the cause or the result of dyssynergic defecation [58]. A previous study had reported that a dyssynergic pattern of defecation was seen in 60% of patients with a rectocele as compared with 24% without [88].
Patients with rectoceles can present with a myriad of gynecological symptoms in addition to defecation disorders and may present to a gastroenterologist, gynecologist, urologist or colorectal surgeon depending on what symptom bothers them the most [89]. Symptoms include pelvic pain, feeling of a prolapse, the feeling of a constant pressure, backache and having a vaginal or perineal bulge. Others may describe a sensation of blockage or an inability to evacuate, the presence of post-defecation soiling, urological complaints and/or the presence of dyspareunia [76]. A proportion of patients describe vaginal splinting using digital support which can sometimes help with either initiating or completing defecation in the presence of a rectocele [56].
Rectal examination may reveal an anterior out-pouching of the rectal wall, particularly during straining. Bimanual rectal and vaginal examination can further help to confirm or facilitate a diagnosis. A barium defecating proctogram is considered the gold standard for the diagnosis of an anterior or posterior rectocele, to measure its size and quantify whether there is any stool retention. It also provides information regarding coexisting conditions such as rectal mucosal intussusception, excessive perineal descent or dyssynergic defecation [90].
The initial management of all symptomatic rectoceles is dietary with adequate fluid and fiber intake. Biofeedback can be considered for bowel retraining and treatment of the associated dyssynergic defecation that will be present in 60% of patients [91,92]. If conservative management fails, many surgeons recommend operative repair [68].
The surgical indication to rectocele repair is controversial as patients who go onto a repair may not have any symptomatic relief. However, most surgeons advocate it when a rectocele is symptomatic and is of a large dimension (>4 cm), or if the rectum fails to empty sufficiently on defecography [93]. Although many surgeons base surgical treatment on whether there is a preoperative need to digitate to help evacuate, this is not predictive of a good result after surgical intervention [92]. A poor prognostic factor is the presence of a descending perineum [94].
A range of operations have been proposed for rectoceles including transvaginal, transanal or transperineal repair, LVR and the STARR, but with varying success rates [92,95–99].
Most commonly, rectocoeles are repaired trans-vaginally. Transvaginal repairs are reported to improve evacuation difficulty between 28 and 63%, but postoperative dyspareunia will be present in 25% of patients [100,101]. The recurrence rate for this technique ranges from 5.7 to 7% [102].
In transrectal repairs, the defect in the rectovaginal septum is closed transanally. The incidence of postoperative dyspareunia is significantly less, compared with the transvaginal repairs and the results reported are variable, with an improvement of defecation between 47 and 84% [100,101].
Transperineal repair with or without levatorplasty is also used in the repair of rectoceles. Short-term results showed an improvement in 75% of patients [103]. A more recent trial of perineal repair with levatorplasty with a 2-year follow-up showed excellent results in 50% of the patients [104].
Unfortunately, few prospective randomized controlled trials have been done to evaluate these different forms of rectocele repairs. Kahn et al., compared posterior colporrhaphy (transvaginaly plication of rectovaginal septum [105]; n = 24) with transanal repair (n = 33), finding that the success rate was higher in the vaginal repair group 87.5% compared with 69.7% in anal group [105]
LVR has also been shown to improve defecating disorder symptoms in patients with a symptomatic complex rectocele, with no worsening of preoperative symptoms or new complaints of constipation, dyspareunia or fecal incontinence. Overall, 88% of patients (n = 84) reported an improvement in overall well being. The complication rate was 4.8% and the mean follow-up in this group was 29 months [106].
Recent publications showed improvements in quality of life and sexual function after ventral rectopexy (n = 154) [107–109]. A consensus panel has agreed that ventral rectopexy could be performed for complex rectoceles of more than 3–4 cm, accompanied by a high-grade internal rectal prolapse and an enterocele [86].
Finally the STARR procedure has also been used for the treatment of defecation disorders, but mainly for the indications of a rectocele with or without the evidence of rectal intussusception.
The European STARR Registry (2006–2008) recruited patients who had the STARR procedure from three countries. Out of 2838 enrolled patients, 2224 received a minimum follow-up of 12 months. Complete dataset of function-related results after 12 months exists for only 41% of the recruited patients. There was also a very high complication rate (36% in total number of patients) where there was a relatively high proportion of patients developing both urge incontinence (20%) and persistent pain (7%) postoperatively. Rectocele recurrence occurred in up to 33% at the end of the first year [110].
Harris et al. [111] compared the STARR procedure with transvaginal rectocele reduction. The authors observed similar success rates for both procedures (82.3 vs 78.4%), but also reported a higher rate of rectocele recurrence after the transvaginal procedure (2.7 vs 10.8%); the clinical impact of this finding on patient postoperative function, however, was not defined. The complication rate after the STARR procedure was significantly higher (61 vs 18%). A noteworthy finding was the high proportion of postoperative rectal bleeding and of patients with sensations of rectal pressure; their clinical relevance, however, was not differentiated. Lastly, a notable feature is the equally high incidence in both groups of dyspareunia (10.5 and 12%, respectively).
A more recent technique using a contour Transtar device for STARR procedure has had its first year follow-up outcome published from the European Transtar registry. A total of 100 patients were recruited of which 11% had complications (bleeding 5%, staple line complications 3%, persistent pain 1%) [112]. It is difficult to compare all these studies as patient factors and outcome measures were highly variable.
It is clear from the large number of procedures which exist for rectocele repair, that there is no solid evidence which supports the use of one procedure over another. What is clear is that it is sensible for all patients to be initially commenced on conservative management before any surgery is contemplated since a large proportion (71.1%) of patients responded to conservative treatment, including dietary change and pelvic floor muscle retraining [113].
Overview of the managment of patients with defecating disorders
Patients with defecating disorders should be ideally managed by a multiprofessional team of specialists. Within our unit all patients are assessed in a multidisciplinary clinic and have a comprehensive history and examination performed.
Patients with a history suggestive of defecating disorders are sent for barium defecating proctogram and ARM. Patients with multicompartment symptoms will have MR defecography.
Patients are then discussed at a multidisciplinary pelvic floor team meeting where their treatment plan is discussed, consisting of specialist radiologists, specialist therapists, urogynecologists, and colorectal surgeons with support from gastroenterologists, nutritionists, psychology and psychiatry.
All patients are offered conservative treatment in the form of dietary and activity advice and those who fail with this initial management, will be offered biofeedback therapy [62].
A significant number of patients with defecating disorders (80%) will respond to conservative treatment alone or with the addition of biofeedback therapy [114].
For those who fail, we have listed the general management principles for the following patient conditions:
Patients who have dyssynergic defecation in our unit who have failed initial biofeedback therapy will be offered BT injections into the pelvic floor followed by further biofeedback therapy. If they have an associated history of constipation, the following points are explored as part of their therapy;
Patients with coexisting constipation are treated medically for their constipation.
There is good evidence [62] that both laxatives and fiber supplements help. In addition, in some patients either suppositories or enemas may be of benefit. If these simple treatments fail, then either prosecretory or serotoninergic enterokinetic agents can be offered. It must be noted that patients with a predominant defecating disorder may not completely respond to this type of medical therapy and they may eventually require additional biofeedback therapy with the use of suppositories or transanal irrigation to help with the constipation treatment [115]. Transanal irrigation is a useful technique of using a balloon rectal catheter with the instillation of water which stimulates the flushing of bowel contents with the aim of emptying the rectum for the treatment of constipation [116]. In a study of patients with symptoms of defecating disorders, chronic constipation and slow transit constipation, symptoms of constipation improved with the use of irrigation, with very little side effects (mainly rectal bleeding).
Novel drug treatments for constipation
Traditionally, clinicians have relied on a limited choice of laxatives. Over the last few years, there have been novel drug therapies available to help with constipation treatment. These can be classified as either prosecretory or serotoninergic enterokinetic agents.
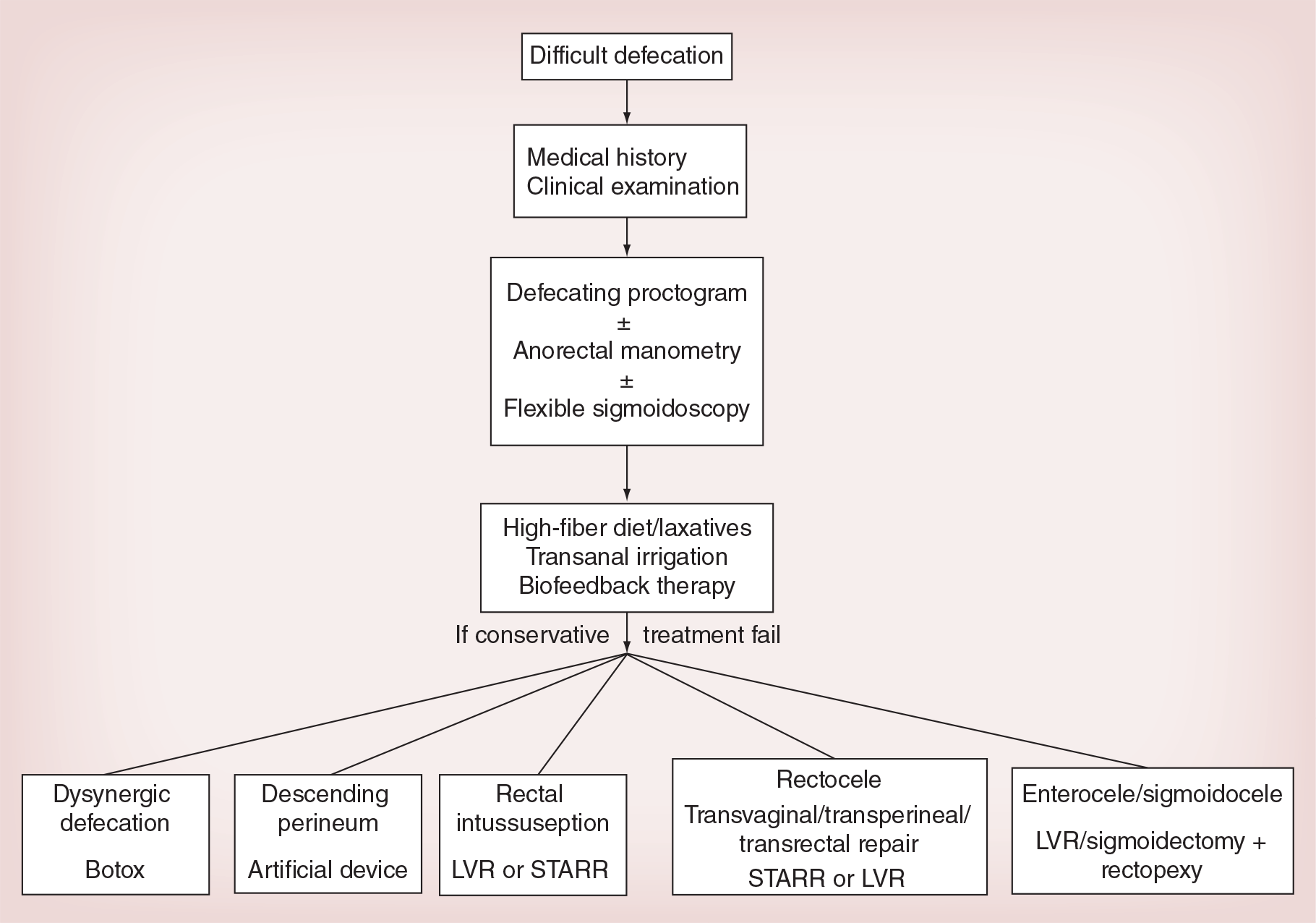
Algorithm in the diagnosis and management of defecating disorders.
Prosecretory drugs: linaclotide & lubiprostone
These two drugs both stimulate bowel movement through increasing electrolyte excretion into the gut lumen.
Linaclotide has been shown to be useful in treatment of symptoms in patients with IBS-C including an improvement of abdominal pain and bowel movements. Linaclotode acts on the guanylate cyclase-C that leads to a rise in cyclic guanosine monophosphate and therefore leads to an increase in chloride and bicarbonate secretion in the gut lumen [117], accelerating colonic transit [8]. Diarrhea is the most common side effect leading to its discontinuation in 5% of patients [117].
Lubiprostone is a type 2 chloride channel activator which facilitates spontaneous bowel movements in patients with chronic idiopathic constipation [118] through increasing intestinal fluid secretion, accelerating small bowel and colonic transit [8].
It has been shown to be useful and safe in patients with functional bowel disease [115] with the main side effects being nausea.
Serotoninergic enterokinetic agent: prucalopride
Prucalopride is a highly selective 5-HT4 agonist which has been shown in clinical trials to be effective in patients with chronic constipation with little side effects. Currently prucalopride is only licensed for use in women and has the main side effect of headaches [119].
In our unit, if one type of constipation drug therapy fails to work, then an alternate drug will be tried:
Patients failing conservative treatment and have a rectocele may be offered surgical repair depending on their presenting symptoms and provided dyssynergic defecation is excluded, and constipation symptoms are treated;
If surgical treatment is being considered, we would advocate a multidisciplinary discussion on the proposed management before this is offered to the patient.
Conclusion
Defecating disorders are a condition which is predominantly seen in women, and these lead to a difficulty with defecation. A wide range of conditions can cause this syndrome and therefore leads to patients presenting in different ways.
The gold standard in diagnosing the cause of this condition is a targeted clinical history and examination, coupled with ARM and a barium defecating proctogram.
All patients should initially be offered conservative therapy, which includes dietary and lifestyle advice coupled with muscle re-education and biofeedback therapy, as most (70%) patients can be treated successfully this way. Relatively fewer patients who fail this treatment will go on to have surgery.
There are a variety of surgical treatments available. However, the literature fails to make firm evidence-based recommendations as to which procedure is the most appropriate. An algorithm in the diagnostic steps and management of defecating disorders is proposed in
Future perspective
The treatment of pelvic floor disorders is evolving. The traditional focus on individual compartments by separate specialties has changed and has regionalized to specialized multidisciplinary teams involving the gastroenterologists, colorectal and urogynecological surgeons as well as specialist radiologists and specialist practitioners. In the future, there will be more regional and national collaboration, education and mentorship.
Research on pelvic floor disorders is still in its infancy. In particular, there needs to be further research into how radiological findings correlate with clinical features and how imaging findings change following surgery in patients with defecating disorders.
In addition, the use of MR defecography will be more prevalent as clinicians get more experience in this technique.
Finally, pelvic floor disorders will present more often as patients and community practitioners become more aware of its existence. Because of this, there will need to be an expansion of pelvic floor specialists to deal with this new demand.
However, despite all this, the field of pelvic floor disorders will continue to be fulfilling as there has been so much treatment evolution over the last few years which we can use to help patients with such a potentially disabling condition.
No writing assistance was utilized in the production of this manuscript.
Executive summary
Defecating disorder is defined as the inability to evacuate contents from the rectum despite excessive straining.
Causes of defecating disorders are multifactoral. Patients may have anatomical or functional abnormalities of their anorectum and pelvic floor or both.
The commonest anatomical abnormalities seen are descending perineum syndrome, rectocele, rectal intussusception, rectal prolapse, sigmoidocele and enterocele formation.
Functional abnormalities are mainly in the form of dyssynergic defecation.
The main symptoms of defecating disorders can include extreme straining during defecation, extended time spent on the toilet, long intervals between bowel evacuation, feeling of incomplete evacuation, fragmented defecation and the need to perform anal, vaginal or perineal pressure in order to evacuate.
The main diagnostic procedures for the evaluation of defecating disorders include anorectal manometry, balloon expulsion test and defecating proctography.
Dyssynergic defecation is the most common functional cause of defecating disorders. Patients with dyssynergic defecation demonstrate an inability to coordinate their abdominal, rectoanal and pelvic floor muscles to facilitate defecation. Initial management of dyssynergic defecation is conservative and will include advice and change of eating habits in order to archive normal stool type and regular bowel pattern. In addition pelvic floor and abdominal muscle re-education with or without biofeedback is used as first-line therapy. Patients who fail conservative treatment can be considered for Botox injection into the puborectalis muscle and external anal sphincter.
Rectal intussusception can be described as the invagination of the rectal wall during defecation. Fifty percent of the patients with rectal intussusception, in addition to the usual symptoms associated with defecating disorders, may complain of fecal incontinence. Patients may also have other structural abnormalities such as a rectocele. The initial treatment of rectal intussusception is conservative therapy, including the use of dietary changes, laxatives and biofeedback. The reported success rate of biofeedback therapy ranges from 37 to 100% for patients with rectal intussusception presenting with a defecating disorder and 50–90% for those presenting with fecal incontinence. Surgery is offered if conservative management fails but with variable results.
Both an enterocele and sigmoidocele are defined as a herniation of the peritoneal sac with either small or large bowel herniating between the vagina and the rectum. Patients usually present with either pelvic pain or heaviness, with symptoms of difficult defecation and the feeling of incomplete evacuation. For both conditions, conservative management is the initial treatment. If symptoms persist, surgical treatment may be beneficial. For enterocele repair, a laparoscopic ventral rectopexy is proposed as the new treatment modality if other anatomical anomalies are also present. For sigmoidoceles, sigmoid resection and rectopexy with obliteration of the Douglas pouch can be performed.
Descending perineum syndrome is characterized by the ballooning of the perineum, several centimeters below the bony outlet of the pelvis during a straining effort. Approximately 75% of patients with descending perineum syndrome present with defecating disorder symptoms. The main treatment for descending perineum syndrome is conservative, using a high-fiber diet, laxatives, enemas and biofeedback.
A rectocele is the protrusion of the rectal wall, usually anteriorly toward the vagina. Patients can present with pelvic pain, feeling of a prolapse, feeling of a constant pressure, having a vaginal or perineal bulge, a sensation of blockage or an inability to evacuate, the presence of post-defecation soiling, urological complaints and/or the presence of dyspareunia. Some patients describe vaginal splinting using digital support, can sometimes help with either initiating or completing defecation in the presence of a rectocele. The initial management of all symptomatic rectoceles are dietary with adequate fluid and fiber intake. Biofeedback can be considered for bowel retraining and treatment of the associated pelvic dyssynergy that will be present in 60% of patients. If conservative management fails, surgical repair may be offered. A range of operations have been proposed for rectoceles including transvaginal, transanal or transperineal repair, laparoscopic ventral rectopexy and stapled transanal resection of rectum, but with varying success rates.
Footnotes
Leicester Royal Infirmary received a grant from the Prucalopride manufacturers, SHIRE, for the funding of specialist practitioner salary. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed
