Abstract
Hypertensive disorders are the most common medical complication of pregnancy, with an incidence of 5-10%, and a common cause of maternal mortality in the USA. Incidence of pre-eclampsia has increased by 25% in the past two decades. In addition to being among the lethal triad, there are likely up to 100 other women who experience ‘near miss’ significant maternal morbidity that stops short of death for every pre-eclampsia-related mortality. The purpose of this review is to present the new task force statement and novel definitions, as well as management approaches to each of the hypertensive disorders in pregnancy. The increased understanding of the pathophysiology of hypertension in pregnancy, as well as advances in medical therapy to minimize risks of fetal toxicity and teratogenicity, will improve our ability to prevent and treat hypertension in pregnancy. Fetal programming and fetal origins of adult disease theories extrapolate the benefit of such therapy to future generations.
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint providership of Medscape, LLC and Future Medicine Ltd. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of 1.0
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test with a 75% minimum passing score and complete the evaluation at www.medscape.org/journal/whe; (4) view/print certificate.
LEARNING OBJECTIVES
Upon completion of this activity, participants will be able to:
Describe complications of hypertension in pregnancy, based on a review
Identify task force modifications, definitions, and classifications regarding hypertension in pregnancy
Assess diagnosis and management of complications of hypertension in pregnancy
Financial & competing interests disclosure
Background
Hypertension is the most frequent reason for office visits in the general population [1,2], as well as the most common medical condition, complicating up to 10% of pregnancies [2–5]. The incidence of hypertension has increased significantly over the last 10 years, with an estimated 40–50% rise [5,6], and therefore the above figures may be understated. This goes hand-in-hand with an increase in obesity rates in the USA, which may subsequently lead to higher incidence of diabetes mellitus [6,7]. Other possible reasons for the increase in hypertension in pregnancy include the increased rate of multiple gestation secondary to assisted reproductive technology and increase in the age of pregnant women due to delay in having children.
Hypertension is the second most common cause of maternal death in the USA [8] and African–American women have a fourfold increase in mortality rate [9]. The mortality rate is also increased in women over the age of 35 [9].
Box 1 summarizes the potential adverse effects and maternal and fetal complications of hypertension.
The American Congress of OBGYN Task Force on hypertension in pregnancy The Task Force on Hypertension in Pregnancy comprised 17 clinician–scientists from the fields of obstetrics, maternal–fetal medicine, hypertension, internal medicine, nephrology, anesthesiology, physiology and patient advocacy. These experts in the management of hypertension in pregnancy reviewed available data and provided evidence-based recommendations for clinical practice.
Its main contribution was in making evidence-based recommendations to modernize the definition and management of pre-eclampsia. Proteinuria was eliminated as a required criterion for diagnosis. In addition, preeclampsia is no longer classified as mild versus severe, but rather by having evidence of hypertensive pathology, and its severe form as defined by having severe features.
Management algorithms were provided accordingly for hypertensive disorders in pregnancy with and without severe features. Many of their management recommendations, particularly those for disease with severe features, provide much-needed clarity.
Definitions & classifications
Hypertension is defined as a systolic blood pressure (BP) ≥140 mmHg or a diastolic BP ≥90 mmHg. These measurements must be made on at least two occasions, no less than 4 h and no more than a week apart. If severe BP ranges are encountered, this time interval may be extended to less than that.
It is important to note that choosing the appropriate cuff size will help to eliminate inaccurate BP measurements. Abnormal proteinuria in pregnancy is defined as the excretion of ≥300 mg of protein in 24 h or a protein/creatinine ratio of ≥0.30. Twenty four-hour urine collection is the gold-standard tool for assessing total urinary protein excretion. Quantitative urine dipstick, however, may be used in some occasions especially when it is the only available measure to determine proteinuria.
Although the American Congress of OBGYN Task Force on Hypertension in pregnancy has made some modifications to the components of the classification of hypertension, it chose to continue using the basic classification first introduced by the college in 1972. This classification considers hypertension during pregnancy in four categories: gestational hypertension; pre-eclampsia–eclampsia; chronic hypertension; and chronic hypertension with superimposed pre-eclampsia [10]. Box 2 lists the classification of hypertension during pregnancy.
NICE categorizes hypertensive disorders in pregnancy into the following: gestational hypertension, pre-eclampsia, severe pre-eclampsia, eclampsia, HELLP syndrome and chronic hypertension. Chronic hypertension itself is classified itself as mild, moderate and severe hypertension. Mild hypertension is defined as diastolic BP of 90–99 mmHg and systolic BP 140–149 mmHg. Moderate hypertension is defined as diastolic BP of 100–109 mmHg and systolic BP of 150–159 mmHg. Severe hypertension includes diastolic BP of 110 mmHg or greater and systolic BP 160 mmHg or greater [11].
International Society For The Study of Hypertension In Pregnancy's (ISSHP) classification of hypertensive disease in pregnancy include pre-eclampsia, gestational hypertension, chronic hypertension (including essential or secondary) and pre-eclampsia superimposed on chronic hypertension [12].
Gestational hypertension
Gestational hypertension is characterized by new onset of elevated BP during the second half of pregnancy (after 20 weeks of gestation) or in the first 24 h postpartum, without accompanying proteinuria or abnormal blood tests (elevated liver enzymes, low platelets or elevated serum creatinine), and in the absence of symptoms. Normalization of BP occurs in the postpartum period, usually within 10 days. The failure of BP to normalize during the postpartum period requires changing the diagnosis to chronic hypertension. Treatment is generally not warranted in this condition, since most patients will have mild hypertension. Gestational hypertension has little effect on maternal or perinatal morbidity or mortality when it develops at or beyond 37 weeks gestation. However, approximately 40% of patients diagnosed with preterm gestational hypertension will subsequently develop pre-eclampsia, or severe features. In addition, these pregnancies may result in fetal growth restriction and placental abruption.
Patients with severe features in the setting of gestational hypertension are at risk for developing adverse maternal and perinatal outcomes. Management of these patients should be similar to patients with pre-eclampsia with severe features. Use of antihypertensive therapy thus should not be part of outpatient management of patients with severe disease.
Although transient in nature, gestational hypertension can be a sign of future remote chronic hypertension. Therefore, even in the benign cases, it is an important marker for follow-up and prevention of development of chronic hypertension [13].
Pre-eclampsia–eclampsia
The classic definition of pre-eclampsia with hypertension and proteinuria has been challenged and modified per the Task Force. Currently, the syndrome of pre-eclampsia requires meeting two criteria; the development of hypertension after 20 weeks of gestation in a woman with previously normal BP [10,14], in addition to presence of proteinuria or new onset of symptoms. Certain laboratory abnormalities are consistent with severe disease, and are used interchangeably or in addition to symptoms.
Adverse outcomes in severe hypertensive disorders of pregnancy.
Abruptio placentae
Disseminated intravascular coagulopathy
Eclampsia
Acute renal failure
Liver hemorrhage or failure
Intracerebral hemorrhage
Hypertensive encephalopathy
Pulmonary edema
Death
Atherosclerosis
Cardiovascular disease
End-stage renal disease
Stroke
Retinopathy
Severe intrauterine growth retardation
Oligohydramnios
Preterm delivery
Hypoxia-acidoslis
Neurologic injuliy
Death
Cerebral palsy
Fetal programming
Cardiovascular disease
Hypertension
Symptoms of pre-eclampsia include cerebral/visual symptoms, severe persistent right upper quadrant/epigastric pain unresponsive to treatment and pulmonary edema. Laboratory abnormalities include thrombocytopenia with a platelet count <100,000, serum creatinine level >1.1 mg/dl, and elevated liver enzymes (>2× normal).
Pre-eclampsia syndrome can be subdivided into pre-eclampsia and pre-eclampsia with severe features. The distinction between the two is based on the severity of hypertension as well as the involvement of other organ systems (Box 2). Close surveillance of patients with pre-eclampsia is warranted, as either type may progress to fulminant disease.
Some maternal symptoms, even in the absence of a confirmed diagnosis of pre-eclampsia, should be considered as prediagnostic findings warranting increased surveillance and should prompt the healthcare provider to closely monitor maternal status for development of pre-eclampsia [15,16]. Women who demonstrate elevations in BP during pregnancy that exceed 15 mmHg diastolic or 30 mmHg systolic ‘warrant close observation’, as suggested by the National High Blood Pressure Education Program Working Group [10].
Classification of hypertension.
Systolic <160 mmHg, or
Diastolic <110 mmHg
No proteinuria and no symptoms
Proteinuria definitioli:
– >300 mg/24 h, or
– Protein/creatinine ratio >0.30, or
– ≥1 + on dipstick
Severe hypertensilin
– Systolic ≥160 mmHg or
– Diastolic ≥110 mmHg
– Persistently severe cerebral symptoms
– Thrombocytopenia: 100,000/mm3
– Elevated liver enzymes >2× upper limit normal
– Pulmonary edema
– Serum creatinine: 1.1 mg/dl
Hypertension before pregnancy Hypertension before 20 weeks gestation
Exacerbation† of hypertension, and/or New-onset proteinuria, and/or Sudden increase in proteinuria‡ Changes have to be substantial and sustained
†Exacerbation is when it gets to persistent and progressive changes in the severe range
‡There is a lack of evidence into what is the definition to follow for an increase in proteinuria; however, a doubling and a progressive persistent increase is key. Proteinuria by itself will not guide management as superimposed pre-eclampsia will need to have the severe features of which proteinuria is not.
Diabetes, obesity, nulliparity, extremes of age, renal insufficiency, pre-existing hypertension, personal history of pre-eclampsia, family history of pre-eclampsia, molar pregnancy, multifetal gestation, fetal hydrops and thrombophilia are among risk factors for the development of pre-eclampsia.
Eclampsia
Eclampsia is defined as pre-eclampsia accompanied by development of new-onset grand mal seizures or coma during pregnancy or the postpartum period, not attributable to other causes. Eclampsia can occur before, during, or after labor. Other causes of seizure during pregnancy can include a bleeding arteriovenous malformation, idiopathic seizure disorder or ruptured aneurysm [17].
Studies on magnesium sulfate for the management and prevention of eclampsia have shown its superiority to other anticonvulsants such as phenytoin and diazepam [18]. Patients being treated for eclamptic seizures should receive an intravenous loading dose of 4–6 g of magnesium sulfate followed by a maintenance dose of 1–2 g/h for at least 24 h. It is recommended that women with eclampsia should undergo delivery after initial stabilization. If undergoing cesarean delivery, the Task Force recommends intraoperative administration of parenteral magnesium sulfate.
HELLP syndrome
A particularly severe form of pre-eclampsia is hemolysis elevated liver enzymes and low platelets (HELLP) syndrome. HELLP syndrome is the acronym for hemolysis (H), elevated liver enzymes (EL), and low platelet count (LP). The diagnosis of HELLP can be elusive, as BP values may only be slightly elevated. Box 3 describes the laboratory criteria for the diagnosis of HELLP syndrome. Once a patient is diagnosed with HELLP, she will automatically be considered to have severe features.
The development of HELLP syndrome may occur antepartum or postpartum [19]. Delivery is indicated if this syndrome develops beyond 34 weeks of gestation or earlier with evidence of disseminated intravascular coagulation, liver infarction or hemorrhage, renal failure, pulmonary edema, abruptio placentae or nonreassuring fetal testing [19]. In women from the gestational age of fetal viability to 33 6/7 weeks of gestation, it is suggested by the Task Force to delay the delivery for 24–48 h if maternal and fetal conditions remain stable, so that the corticosteroid course can be completed.
Chronic hypertension
Hypertension in pregnancy is defined as chronic if the patient was diagnosed with hypertension prior to pregnancy, if hypertension is noted prior to 20 weeks gestation, or if it persists beyond 6 months postpartum. The incidence of chronic hypertension in pregnancy varies from 1 to 5% [20,21]. Essential or primary hypertension is the most common type of chronic hypertension contributing to 90% of chronic hypertension cases, while secondary hypertension accounts for only 10% of the cases. Chronic renal disease (glomerulonephritis, polycystic kidney disease or renal artery stenosis) is the most common cause of secondary hypertension. Other secondary causes of hypertension include polyarteritis nodosa, lupus erythematosus, endocrine disorders (primary hyperaldosteronism, Cushing disease, phaeochromocytoma or diabetes mellitus especially with renovascular involvement) and coarctation of the renovascular hypertension and pheochromocytoma, may be associated with poor pregnancy prognosis and fetal outcomes. Clinical features that may warrant additional work up for secondary hypertension include hypertension resistant to medical management, lack of family history of hypertension, extremes of age, electrolyte abnormalities (e.g., hypokalemia and hypernatremia) and elevated serum creatinine levels suggestive of chronic renal failure. Per Task Force recommendations, clinical suspicion for secondary hypertension warrants referral to a hypertension specialist for further work-up [17].
Women with chronic hypertension are at risk of developing superimposed pre-eclampsia. Women with chronic hypertension who develop superimposed pre-eclampsia have higher rates of adverse maternal–fetal outcomes. Chronic hypertension is also associated with a greater risk of cesarean delivery and development of postpartum hemorrhage [23]. Other adverse maternal outcomes of chronic hypertension include end organ damage, increased risk of development of gestational diabetes [23–25] and increased risk of abruptio placentae (threefold) [26–28]. Women with chronic hypertension are also more likely to be hospitalized for hypertension [25].
With regards to fetal outcomes, perinatal mortality and perinatal death are higher in pregnancies complicated by chronic hypertension [25,26,28]. Fetal growth restriction is more common with chronic hypertension and is usually associated with superimposed pre-eclampsia [29].
Chronic hypertension with superimposed pre-eclampsia
The Task Force suggests that superimposed pre-eclampsia can be divided into two groups: superimposed pre-eclampsia; and superimposed pre-eclampsia with severe features.
Laboratory criteria for the diagnosis of HELLP syndrome.
Abnormal peripheral blood smear (burr cells, schistocytes) Elevated bilirubin ≥1.2 mg/dl Low serum haptoglobulin Significant drop in hemoglobin levels unrelated to blood loss
Elevated aspartate transaminase or alanine transaminase ≥2× the upper limit of normal for the laboratory
Increased lactate dehydrogenase >2× the upper limit of normal for the laboratory
Superimposed pre-eclampsia is defined as an exacerbation of hypertension that was previously well-controlled requiring escalation of BP medications and/or new onset of proteinuria or sudden increase in pre-existing proteinuria that has to be substantial and/or sustained. Superimposed pre-eclampsia develops in 13–40% of women with chronic hypertension, depending on diagnostic criteria, etiology of chronic hypertension and its severity [30,31].
Superimposed pre-eclampsia with severe features is defined by presence of severe hypertension despite treatment, symptoms including cerebral/visual symptoms, persistent right upper quadrant/epigastric pain unresponsive to treatment, or pulmonary edema. Laboratory abnormalities meeting criteria for severe features include low platelets < 100,000 elevated liver enzymes (>two-times upper normal) and new elevation of serum creatinine to >1.1 mg (Box 4).
Criteria for the diagnosis of suspected superimposed pre-eclampsia, or superimposed pre-eclampsia with severe features.
Exacerbation of hypertension that was previously well controlled requiring escalation of blood pressure medications New onset of proteinuria Sudden increase in pre-existing proteinurlia – Substantial and/or sustained
Severe hypertension despite treatment
Cerebral or visual disturbances
Epigastric pain/right upper quadrant pain
Pulmonary edema
Abnormal liver function tests: aspartate transaminase or alanine transaminase >2× the upper limit for the laboratory
Thrombocytopenia (platelet count <100,000/mm3)
Serum creatinine >1.1 mg (new onset)
Women with superimposed pre-eclampsia can develop end-organ damage and adverse outcomes. Therefore, increased surveillance of these patients is warranted even in the cases when the diagnosis of superimposed pre-eclampsia is suspected and not definitive.
Management considerations
The Task Force statement has provided long awaited clarification. Hypertensive disorders in pregnancy without evidence of severe features are considered consistent with mild disease. Management guidelines are thus applicable to gestational hypertension, pre-eclampsia and superimposed pre-eclampsia as long as they are not associated with any symptoms or laboratory abnormalities consistent with severe features. Chronic hypertension falls under the same category; however, recommended gestational age for delivery is 38 weeks, in contrast to 37 weeks (Figure 1).
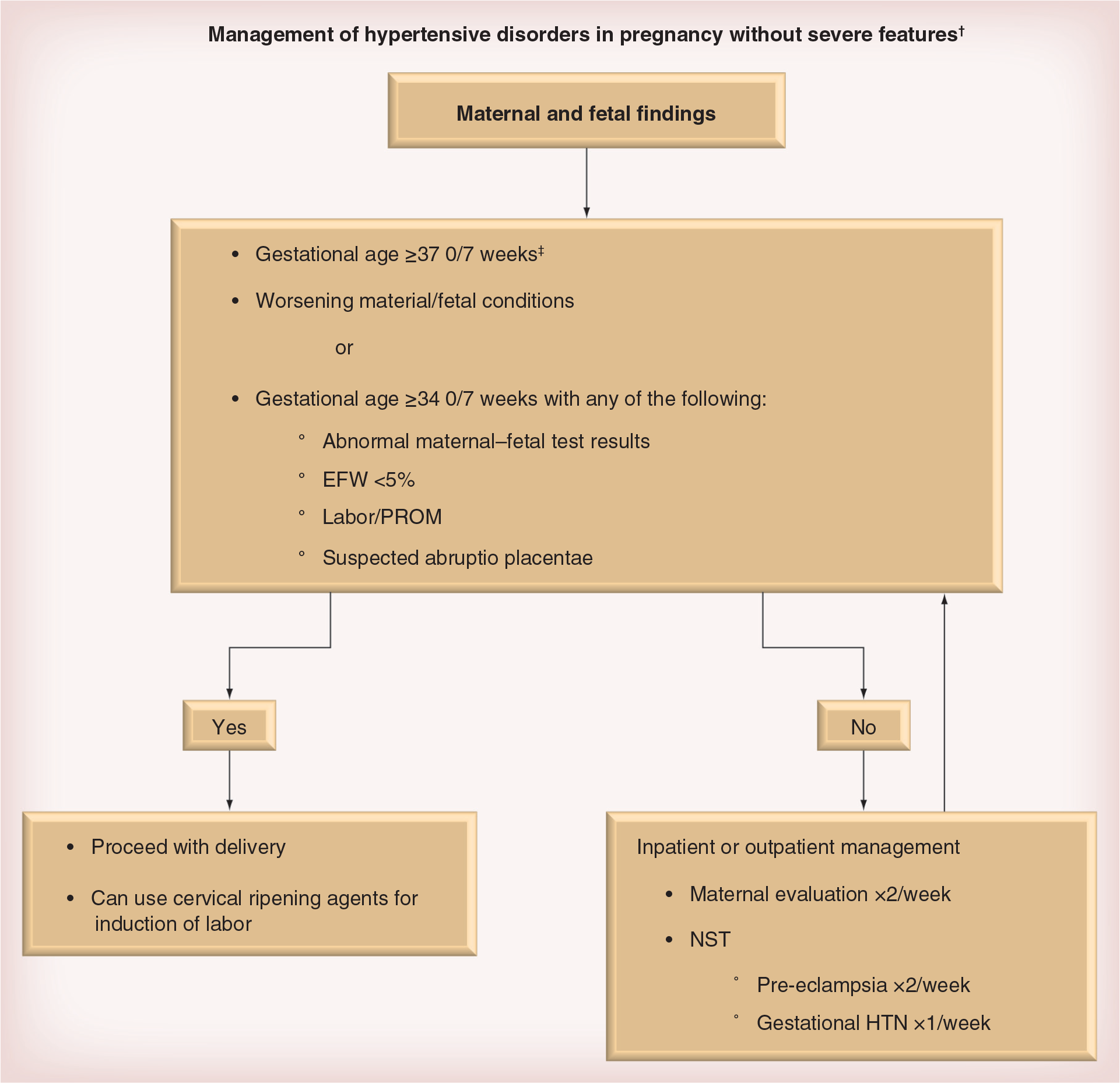
Management of hypertensive disorders in pregnancy without severe features (algorithm).
Hypertensive disorders in pregnancy without severe features
Antepartum management
All women diagnosed with hypertension in pregnancy should have a complete blood count, serum creatinine, liver enzymes, 24-h urine collection or urine protein to creatinine ratio to assess for urine protein. Fetal assessment should be done through ultrasonographic evaluation of estimated fetal weight and amniotic fluid index, nonstress test (NST), and biophysical profile (BPP).
Indications for hospitalization and delivery per the Task Force for mild gestational hypertension and pre-eclampsia without severe features are as follows (Figure 1):
37 weeks of gestation or more;
Suspected abruptio placenta;
34 weeks of gestation plus any of the following:
• Progressive labor or rupture of membranes;
• Estimated fetal weight less than fifth percentile on ultrasound;
• Oligohydramnios (amniotic fluid index less than 5 cm);
• Persistent BPP of 6/10 or less.
Monitoring includes fetal testing, as well as maternal evaluation. That includes daily kick counts, ultrasound for fetal growth evaluation every 3 weeks, as well as NST once-weekly for patients with gestational hypertension and twice-weekly for patients with pre-eclampsia without severe features. Maternal evaluation comprises laboratory evaluation with complete blood count, liver enzymes, and serum creatinine level at least once a week. Symptoms and serial BP checks should be monitored twice weekly with adequate patient education to access care with any severe feature development. The Task Force recommends assessment of proteinuria at least once a week in the office and weekly measurement of BPs at home or in the office for women with gestational hypertension.
In absence of severe features, antihypertensive therapy is reserved for chronic hypertension.
Management of chronic hypertension in pregnancy
Hypertensive women should be evaluated at the initial prenatal visit to identify end-organ damage since such findings can change their prognosis and consequently their management. Initial evaluation includes 24-h urine for total protein and creatinine clearance, renal panel or serum electrolytes, serum creatinine, liver enzymes, uric acid, platelet count, ophthalmoscopy or ophthalmology consult, electrocardiogram and cardiology consult if there is an abnormal electrocardiogram.
Ideally, preconception counseling and management should include establishing the etiology of hypertension and identification of any end-organ damage and adequate control of BP prior to conception. Assessment of renal function is very important in patients with hypertension especially if there is co-existing diabetes mellitus. Laboratory investigations include urinalysis, urine-specific gravity, urine culture and sensitivity studies, baseline concentrations of blood urea and plasma protein, 24-h urine collection for total protein loss and creatinine clearance. In the very rare event of a suspected pheochromocytoma or paraganglioma, the detection methods of choice include urinary metanephrins and normetanephrins, dopamine metabolites (for the diagnosis of paranganglioma and malignant disease) or plasma catecholamines. Evaluation for other causes of secondary hypertension will depend on clinical suspicion or presence of risk factors for the secondary causes. The Task Force suggests referral to a physician with expertise in treating hypertension if secondary hypertension is suspected.
The presence of proteinuria and the rate of increase of proteinuria may predict future deterioration of renal function in these patients. Rate of decline of renal function is also affected by level of BP control. Renal function is more likely to decline in African–American patients and those with advanced maternal age or pre-existing renal disease [32].
Medications that are known to have adverse fetal outcomes and have been prescribed for BP control should be discontinued prior to conception.
Monitoring BP
BP is checked on a monthly basis in all pregnant women as part of standard prenatal care. Good clinical practice recommends increased monitoring for women with BPs above the target range. Task force recommendation for women with chronic hypertension is to use home BP monitoring.
Fetal surveillance for pregnant women with chronic hypertension
The risk of fetal growth restriction is higher in pregnant women with chronic hypertension 8–15% [33]. Per Task Force recommendations the use of ultrasonography for screening fetal growth restriction is suggested in women with chronic hypertension. If evidence of fetal growth restriction is found in pregnant women with chronic hypertension, assessment of the fetoplacental status is recommended using umbilical artery velocimetry (Figure 2). Antenatal surveillance with either NST or BPP may be associated with decreased perinatal morbidity and mortality in these women [34]. Task Force therefore recommends antenatal fetal testing in women with chronic hypertension with need for medications, underlying medical conditions, any evidence of fetal growth restriction and superimposed pre-eclampsia.

Umbilical artery reversed end-diastolic flow.
Treatment
The goals of treatment in chronic hypertension include minimizing fetal risks attributable to hypertension, development of vascular disease and possible harmful effects of antihypertensive medications causing decreased uteroplacental perfusion or potential harmful fetal effects. Commonly used oral antihypertensives are reviewed in Table 1.
Oral antihypertensives commonly used in pregnancy.

Nonpharmacologic treatment
It is suggested by the Task Force that weight loss and extremely low sodium diets (less than 100 mEq daily) not be used for management of chronic hypertension in pregnancy. It is also suggested that moderate exercise be continued during pregnancy for women who are accustomed to exercising.
Prevention of superimposed pre-eclampsia
Systematic reviews demonstrate that the use of anti-platelet agents and low dose aspirin is associated with a significant 17% reduction in developing pre-eclampsia [35]. Task Force therefore recommends initiating the administration of daily low-dose aspirin (60–80 mg) beginning in the late first trimester for women with chronic hypertension at greatly increased risk of adverse pregnancy outcomes. These risks include early onset pre-eclampsia and preterm delivery at less than 34 weeks of gestation or pre-eclampsia in more than one prior pregnancy.
NICE recommends use of daily 75 mg of aspirin from 12 weeks gestation throughout pregnancy to all women at increased risk for development of pre-eclampsia. These include women with hypertensive disease during a previous pregnancy, chronic kidney disease autoimmune disease such as systemic lupus erythematosis or antiphospholipid syndrome, Type 1 or 2 diabetes and chronic hypertension [36].
Intrapartum management & timing of delivery
Expectant management is recommended in patients with mild gestational hypertension or pre-eclampsia without severe features and no other indications for deli delivery less than 37 weeks of gestation [37]. This is due to the fact that immediate delivery is associated with increased admission rates to the neonatal intensive care unit, neonatal respiratory complications and the increased risk in neonatal death.
Delivery is suggested for women with gestational hypertension or pre-eclampsia without severe features at or beyond 37 weeks of gestation, 38 weeks for chronic hypertension. The mode of delivery does not need to be cesarean section. It should be determined by fetal gestational age, fetal presentation and maternal–fetal conditions. Vaginal delivery is usually less likely successful with decreasing gestational age and the likelihood of cesarean delivery increases [38–40].
Magnesium sulfate is not recommended for seizure prophylaxis, with systolic BP less than 160 mmHg and diastolic BP of less than 110 mmHg (Table 2).
Magnesium sulfate: dosages, serum levels and associated findings.

DsLR: Dextrose 5% with lactated ringer; DsW: Dextrose 5% with water; iv.: Intravenous.
Postpartum management & follow-up
BP needs to be monitored for at least 48 h in high-risk chronic hypertension patients since they are more likely to have postpartum complications such as renal failure, pulmonary edema and hypertensive encephalopathy [3,9,22].
Even in the absence of intrapartum treatment, postpartum antihypertensive therapy is usually indicated in women with chronic hypertension before and during pregnancy. During this time period, special attention must be paid to prescribe medications with minimal adverse effects for breastfeeding. The goal of medical management during the postpartum period is to maintain BP levels less than severe ranges (systolic BP less than 160 and diastolic BP less than 100 mmHg).
Many antihypertensive medications could be found in low concentrations in breast milk. Water soluble medications usually have lower concentrations compared with lipid soluble ones. Methyldopa and calcium channel blocking agents are known to have the lowest concentration in breast milk and are therefore considered to be safe drugs during breastfeeding. Among β-blocking agents, metoprolol and atenolol have high concentrations in breast milk, as opposed to labetalol and propranolol that have low concentrations. Angiotensin-converting-enzyme (ACE) inhibitors such as captopril are known to result in minimal levels in breast milk and are therefore considered to be safe during breast feeding by many experts. Diuretics lower chances of successful breastfeeding and breast milk production.
Nonsteroidal anti-inflammatory agents should not be used in the postpartum period in women with chronic hypertension and particularly superimposed pre-eclampsia, as these medications can increase BP and sodium retention.
In cases of severe persistent hypertension resistant to treatment with two antihypertensive medications, referral should be made for further evaluation of secondary hypertension.
Hypertensive disorders in pregnancy with severe features
In the presence of severe features, more aggressive management approach is recommended. Severe features (Box 2) are associated with higher pregnancy complications (Box 1) and thus are consistent with severe disease regardless of the diagnosis of hypertension (gestational, pre-eclampsia or superimposed pre-eclampsia).
Figure 3 summarizes management of hypertensive disorders in pregnancy associated with severe features. Beyond 34 weeks, delivery is indicated for women with severe pre-eclampsia, and those with unstable maternal–fetal conditions. Stabilization of maternal status with magnesium sulfate and antihypertensive therapy is recommended.
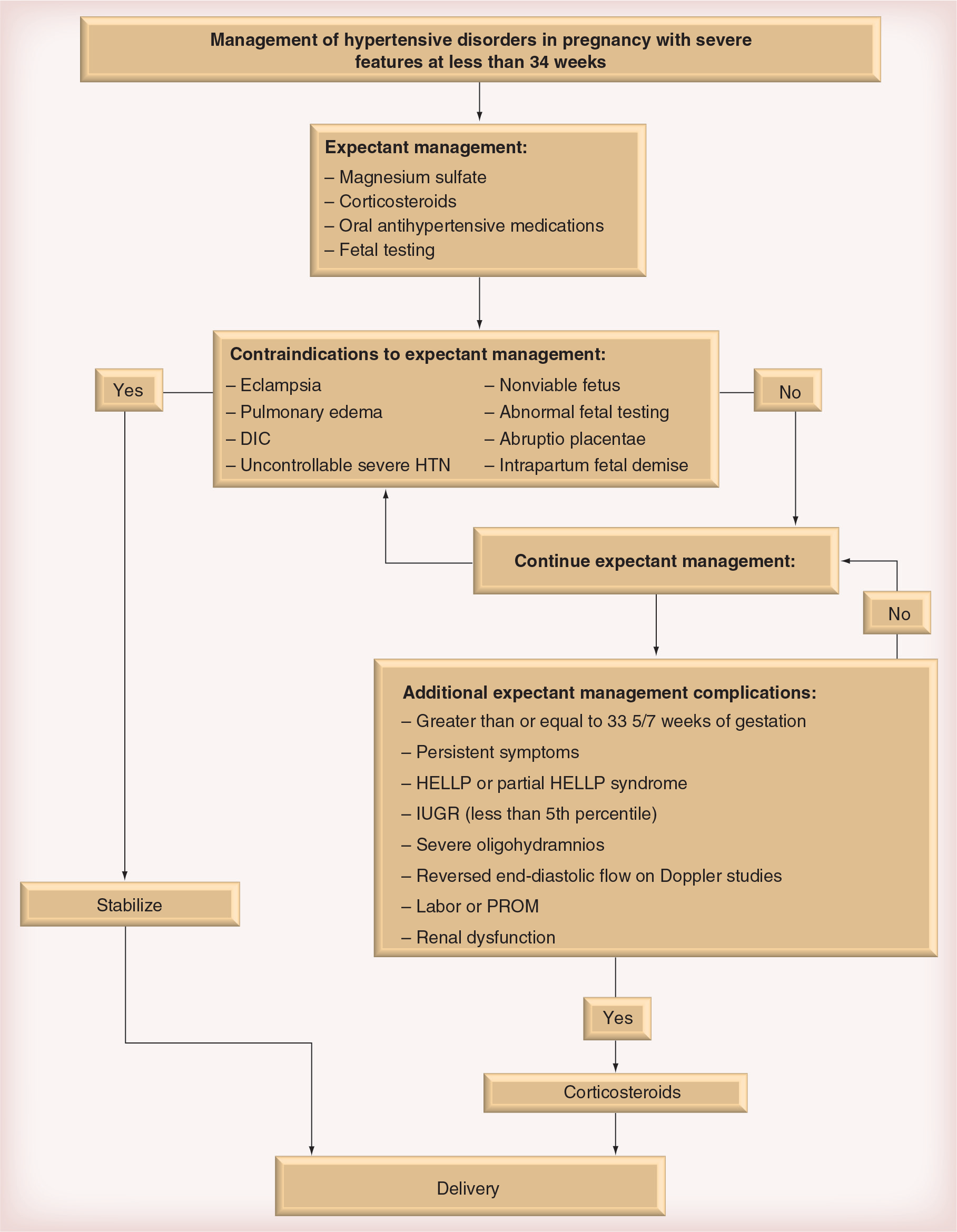
Management of hypertensive disorders in pregnancy with severe features at less than 34 weeks.
For women with severe pre-eclampsia at 34 weeks or less, the administration of corticosteroids for fetal lung maturity is recommended. At less than 34 weeks of gestation with stable feto–maternal conditions, expectant management is recommended only at facilities with adequate maternal and neonatal intensive care units. Task Force recommends delivery after maternal stabilization for women with severe pre-eclampsia before fetal viability. Expectant management is not recommended in these cases.
In women with severe pre-eclampsia and a viable fetus at 33 6/7 weeks or less, it is suggested that corticosteroids be used and delivery deferred for 48 h, without the presence of any contraindications to expectant management or any associated complications. These include pregnancy complications like preterm premature rupture of membranes, labor and oligohydramnios. Laboratory abnormalities (thrombocytopenia, persistent elevation of liver enzymes and creatinine) as well as abnormal fetal growth and testing mainly reversed end diastolic flow on umbilical artery Doppler's (Figure 2).
In the same group of patients, it is recommended that delivery not be delayed regardless of gestational age if maternal condition is complicated by uncontrollable severe hypertension, eclampsia, pulmonary edema, abruptio placentae, disseminated intravascular coagulation, evidence of nonreassuring fetal status or fetal demise [13].
Since pre-eclampsia with severe features or hypertension may develop in the postpartum period either as a new event or as an exacerbation of a mild form of the hypertensive disorder of pregnancy, all women should be educated about the signs and symptoms of severe hypertension or pre-eclampsia. Magnesium sulfate is indicated in the presence of signs and symptoms of persistent or new onset of superimposed pre-eclampsia. Eclampsia and cerebrovascular events associated with elevated BPs most commonly occur during the postpartum period [41]. If BP in the postpartum period continues to remain in the severe ranges despite optimal treatment with a combination of antihypertensive medications, the patient should be referred to a specialist to rule out secondary causes of hypertension.
The Task Force also recommends that in all patients with gestational hypertension, pre-eclampsia and superimposed pre-eclampsia, BP should be monitored in the hospital or equivalent outpatient surveillance should be performed for at least 72 h postpartum and again 7–10 days after delivery or earlier in women with symptoms.
Pharmacologic treatment
In the absence of strong evidence supporting the use of antihypertensive therapy for mild-to-moderate chronic hypertension during pregnancy, initiation of pharmacological therapy is not suggested unless BP approaches severe range. Per the Task Force Recommendations, antihypertensive therapy is indicated for pregnant women with persistent severe chronic hypertension, with a systolic BP of 160 mmHg or higher or diastolic BP of 105 mmHg or higher. NICE recommends maintaining systolic BP level less than 150 mmHg and diastolic BP less than 100 mmHg in pregnant women with uncomplicated chronic hypertension [36]. Aggressive BP lowering is discouraged due to concerns about uteroplacental blood flow compromise caused by pharmacologically induced hypotension. Available data suggest that there is potential maternal benefit of antihypertensive treatment by reducing the risk of developing severe hypertension with no direct fetal benefit or improvement in perinatal outcomes in these patients [42]. Treatment also does not prevent pre-eclampsia or placental abruption [43,44].
For women with evidence of end-organ damage, such as chronic kidney disease, diabetes and cardiac disease, BP goals are systolic BP less than 140 mmHg and diastolic BP less than 90 mmHg [45,46] with an objective to maintain systolic BP <130, Diastolic BP <80. Normotensive BP ranges are <140 for systolic and <90 for diastolic BP in the settings of left ventricular hypertrophy and renal disease.
The treatment of severe hypertension fulfills the therapeutic objective of maternal cerebrovascular accidents prevention and congestive heart failure without cerebral perfusion compromise or uteroplacental blood flow reduction.
Antihypertensive agent selection
When choosing an antihypertensive medication for treatment of chronic hypertension in pregnancy, the goals of therapy are either: acute lowering of severe hypertension (Table 3); or long-term treatment of BP in the outpatient settings (Table 1). The choice of antihypertensive medication should be based on the potential adverse effects, as well as clinician's individual experience and familiarity with a particular medication [47].
Acute treatment of hypertension.

iv: Intravenous; p.o.: Per os; prn: Pro re nata (as needed).
Intravenous labetalol, intravenous hydralazine and oral nifedipine are first-line agents for lowering BP in the acute hospital settings (Table 3).
Drugs for long-term management: oral antihypertensive agents
Oral antihypertensive agents are used for treatment of pregnant women with chronic hypertension in the long-term [48]. Commonly used oral antihypertensive medications are listed in Table 1.
α- & β-blockers
Labetalol (an α- and β-blocking agent) is probably the most commonly used antihypertensive agent in pregnancy. It may be given orally or intravenously, hence, may be used for routine BP control with easy conversion to parenteral route in case of severe hypertension, hypertensive crisis or in patients who are unable to take oral medications. Adverse effects include fatigue, lethargy and bronchoconstriction.
Other β-blocking agents including atenolol and propranolol could increase the risk of intrauterine growth retardation and are generally avoided in pregnancy [49].
Pure α-blockers are not used in pregnancy and may have limited role in the nonpregnant population since the recent publication of the ALHAT study. An arm of the study comparing doxazosin (α-blocker) and chlorthalidone (diuretic) in the treatment of hypertension was terminated because doxazosin doubled the risk of heart failure [50].
Central-acting agents
The central-acting agents include α-methyl dopa (methyldopa) and clonidine. Until recently, methyldopa was the first-line agent for treatment of hypertension in pregnancy as it was probably the most studied antihypertensive medication with a well-documented safety profile. There are no adverse effects on the uteroplacental or fetal hemodynamics or fetal wellbeing [51,52]. However, recent evidence indicates that it is no longer the drug of choice for BP control in both pregnant and nonpregnant patients. Side effects of methyldopa include abnormal liver transaminases that might be difficult to differentiate from elevated liver enzymes secondary to pre-eclampsia, hepatic dysfunction and necrosis and hemolytic anemia.
Calcium-channel blockers
Calcium-channel blockers (nifedipine, diltiazem and verapamil) have a very good safety profile in pregnancy and also have a renoprotective effect that may be useful in diabetic patients.
A study by Sibai
Verapamil is another calcium channel blocker that could be used for treating hypertension in pregnant patients with cardiac disease. It is also used in management of arrhythmias and cardiac disease in pregnancy. Diltiazem may also be used in pregnant cardiac patients.
Diuretics
Pre-eclampsia patients are frequently noted to have intravascular volume depletion, even though they may appear to be edematous and fluid overloaded. Many physicians therefore tend not to use diuretic agents in patients with pre-eclampsia to avoid further decrease in intravascular volume, and prevent any negative effects on fetal growth. Therefore, diuretics are considered as second-line agents for treatment of hypertension in pregnancy [5]. Although that may be a concern in the intrapartum period, there is no reason for diuretics not to be used in the postpartum period especially if there is presence of pulmonary edema or evidence of fluid overload.
Among the diuretics, thiazides and loop diuretics are used in specific situations during the postpartum period. One of the adverse effects of thiazide diuretics is hyperglycemia, which is usually not significant when used for a short duration of time. The loop diuretics may cause hypokalemia; therefore, the serum level of potassium needs to be measured if the woman is receiving the drug for more than a few days.
ACE inhibitors & angiotensin receptor blockers
Angiotensinogen is converted to angiotensin I by rennin while angiotensin I is converted to angiotensin II by ACE. Angiotensin II has two main receptor sites; type 1 and 2 receptor sites. The ACE inhibitors block conversion of angiotensin I to angiotensin II while the angiotensin-receptor blockers (ARBs) block primarily the type 1 receptor sites. The type 1 receptors are highly expressed in the first trimester of pregnancy in the sheep placenta and may play a role in placental function [57].
ACE inhibitors and ARBs, may cause fetal anomalies, such as fetal renal insufficiency, oligohydramnios, growth restriction, pulmonary hypoplasia, cranial anomalies and severe fetal hypotension especially in the second and third trimesters and therefore should be avoided in pregnancy [58,59]. The Task Force recommends discontinuation of ACE inhibitors and ARBs, and any associated classes of medications such as rennin inhibitors during pregnancy [60]. Women on ACE inhibitors are advised to stop the medication prior to conception. However, if discovered in the first trimester, they may stop the medications without significant damage to the fetus [61,62].
The use of ARBs (losartan, valsartan, irbesartan or candesartan) in pregnancy is still in the embryonic stage. They have similar indications as ACE inhibitors but are preferred in cases where the patient cannot tolerate ACE inhibitors owing to cough [63]. The ARBs and ACE inhibitors can cause life-threatening angioedema and significant fetal toxicity. They may be used in the postpartum period especially in diabetic patients because of their renoprotective effect. It has been suggested that combination of ARB and ACE inhibitors may be more efficient in reducing BP than each agent alone [64].
In conclusion, some of the common antihypertensive medications used outside of pregnancy that including ARBs, ACE inhibitors and some diuretics are contraindicated in pregnancy. Providers caring for women in the reproductive age group should be cautious about the use of these medications, patient's reproductive plans and potential risks, including awareness of potential side effects and teratogenesis.
Management of women with prior pre-eclampsia
The goals of management of patients with prior history of pre-eclampsia are to optimize maternal health conditions prior to conception, detect potential adverse outcomes and to achieve optimal perinatal outcomes in subsequent pregnancy. Box 5 summarizes the evaluation of women at risk for pre-eclampsia recurrence, including their preconception counseling, early prenatal care and regular monitoring of the maternal and fetal wellbeing.
Evaluation and management of women at risk of pre-eclampsia recurrence.
Recognition of risk factors (i.e., Type 2 diabetes mellitus, obesity, hypertension and family history)
Review outcome of previous pregnancy (abruptio placentae, fetal death, fetal growth restriction, and gestational age at delivery)
Baseline metabolic profile and urinalysis
Optimization of maternal health status
Supplementation with folic acid
Ultrasound for assessment of gestational age and fetal number
Baseline metabolic profile and complete blood count
Baseline urinalysis
Folic acid supplementation
Offer first trimester combined screening
For women with prior pre-eclampsia resulting in delivery before 34 weeks or pre-eclampsia occurring in more than one pregnancy, offer low-dose aspirin late in the first trimester and discuss the risks and the benefits
Counsel patient about sign and symptoms of pre-eclampsia
Monitor for signs and symptoms of pre-eclampsia
Monitor blood pressure at prenatal visits, or at home
Perform ultrasonography at 18–22 weeks for fetal anatomy evaluation and to rule out molar gestation
Perform uterine artery Doppler studies
Hospitalize for severe gestational hypertension, fetal growth restriction, or recurrent pre-eclampsia
Monitor for sign and symptoms of pre-eclampsia
Monitor blood pressure at prenatal visits, or at home
Perform laboratory testing, serial ultrasonography for fetal growth and amniotic fluid assessment, umbilical artery Doppler with nonstress test, biophysical profile, as indicated by clinical situation
Hospitalize for severe gestational hypertension, fetal growth restriction, or recurrent pre-eclampsia
Adapted with permission from [68].
Prevention & prediction of pre-eclampsia
It is thought that pre-eclampsia can develop secondary to alterations in systemic prostacyclin–thromboxane balance. There is also increased inflammation [65]. Therefore, low-dose aspirin (81 mg or less), an anti-inflammatory agent blocking thromboxane synthesis, has been shown to be effective for prevention of pre-eclampsia. For women with a history of early-onset pre-eclampsia and preterm delivery at less than 34 weeks of gestation, or pre-eclampsia in more than one prior pregnancy, it is recommended to initiate daily low dose aspirin beginning in the late first trimester by the Task Force.
The administration of vitamin C or vitamin E has not been proven to be of any effect in prevention of pre-eclampsia. It is recommended that dietary salt not be restricted during pregnancy for the prevention of pre-eclampsia. Bed rest or physical activity restriction is not recommended for the prevention of pre-eclampsia and its complications.
Multiple efforts have been made to predict pre-eclampsia including uterine artery Doppler studies and serum biomarkers. Uterine artery Doppler studies were found to be superior in predicting pre-eclampsia in an early gestational age compared with term. Increased resistance to flow within the uterine artery results in an abnormal waveform pattern represented by either an increased resistance or by the persistence of a unilateral or bilateral diastolic notch (Figure 4). Currently, however, the best screening to predict pre-eclampsia is still obtaining a comprehensive medical history.

Uterine artery notching.
Controversies & futures considerations
Despite the recent improvement in understanding the pathophysiology of pre-eclampsia, many unanswered questions remain to be investigated. Research areas suggested in the Task Force statement cover etiology, management and prevention. The list of proposed research recommendations by the Task Force is extensive [17].
Etiology-related research is suggested to focus on placentation, immunological and angiogenic abnormalities associated with pre-eclampsia during pregnancy. In addition to that, genetic factors, molecular and cellular mechanisms involved in the development of pre-eclampsia should be studied.
Regulation of sFlt-1 production and its inhibitors is one particularly interesting research field that has a lot of potential to further contribute to our understanding. sFlt-1 is a substance that is released from the placental villi in response to reduced oxygenation. It appears to play an important role in the pathogenesis of pre-eclampsia. Therapeutic interventions hypothetically would arise from its extraction from maternal circulation, resulting in possible reversal of the development of pre-eclampsia [66].
Biomarkers that can predict pregnancy complications and perinatal mortality are currently being studied. Further understanding of these biomarkers can be the applied clinically to modify management approach in an objective to reduce the risks of such morbidity. Other proposed research covering important clinical questions that relate to direct pregnancy and postpartum management is also addressed.
Fetal programming & fetal origins of adult disease
Hypertension is a systemic disease with significant impact on the micro and macro-circulation. It can lead to nephropathy, retinopathy, cardiac disease and other complications.
Barker's landmark studies, along with additional human and animal model data, shed the lights on fetal origins of adult disease [67].
Conclusion
In conclusion, hypertensive disorders in pregnancy remain a major health concern for women and their infants. Optimizing management, including prenatal care, identification of the severe features and treating appropriately, as well as close postpartum follow-up are key to reduce maternal-fetal morbidity and mortality.
Lifestyle modifications in women affected by hypertensive disorders in pregnancy might reduce their risk for cardiovascular disease at a later point in their lives. Research in this field is promising and advancements both on the basic science, as well as clinical fronts will hopefully provide further clarity to prediction, prevention and management of hypertensive disorders in pregnancy.
Future perspective
With the increasing rates of obesity and diabetes mellitus as well as advancing childbearing age, it is predicted that there will be a significant rise in the incidence of hypertensive disorders in pregnancy in the next decade.
We anticipate, however, that long-term comorbidities and mortality from this medical condition will not follow a similar pattern or may even decrease as a result of several factors. Some of these factors include further knowledge of the disease etiology, with a better understanding of its underlying cellular and molecular mechanisms and advanced screening among populations at risk, as well as optimization of management and therapeutic interventions.
Executive summary
Hypertensive disorders are the most common medical complications of pregnancy 5-10% and a common cause of maternal mortality in the USA.
Hypertension in pregnancy is associated with many complications and adverse effects. Some of these include eclampsia, abruptio placenta, preterm delivery, disseminated intravascular coagulation, hemorrhage, renal insufficiency, pulmonary edema, stroke and death.
Perinatal morbidity and mortality are also increased from preterm delivery/prematurity and intrauterine growth retardation.
Long-term maternal complications include cardiovascular disease, atherosclerosis, renal failure as well as stroke. Long-term fetal complications include cardiovascular changes predisposing to adult disease.
New diagnostic as well as management criteria for pre-eclampsia and superimposed pre-eclampsia have been implemented per the Task Force statement.
Hypertension during pregnancy consists of four categories: gestational hypertension; pre-eclampsia-eclampsia; chronic hypertension; and chronic hypertension with superimposed pre-eclampsia.
The main contribution of the Task Force is in making evidence-based recommendations to modernize the definition and management of hypertensive disorders in pregnancy.
Proteinuria was eliminated as a required criterion for diagnosis. In addition, pre-eclampsia is no longer classified as mild versus severe, but rather by having evidence of hypertensive pathology, and its severe form as defined by having severe features.
Severe features consist of symptoms including cerebral/visual symptoms, persistent right upper quadrant/epigastric pain unresponsive to treatment, or pulmonary edema. Laboratory abnormalities meeting criteria for severe features include low platelets <100,000 elevated liver enzymes (>two-times upper normal) and serum creatinine >1.1 mg (new onset).
Hypertensive disorders in pregnancy without evidence of severe features are considered consistent with mild disease. Management guidelines are thus applicable to gestational hypertension, pre-eclampsia, and superimposed pre-eclampsia as long as they are not associated with any symptoms or laboratory abnormalities consistent with severe features. Chronic hypertension falls under the same category.
In the presence of severe features, more aggressive management approach is recommended.
Research areas suggested in the Task Force statement cover etiology, management as well as prevention.
Footnotes
Management of hypertensive disorders in pregnancy
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions (with a minimum 75% passing score) and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/whe. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the “Register” link on the right hand side of the website. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider, CME@medscape. net. For technical assistance, contact CME@webmd.net. American Medical Association's Physician's Recognition Award (AMA PRA) credits are accepted in the US as evidence of participation in CME activities. For further information on this award, please refer to http://www.ama-assn.org/ama/pub/about-ama/awards/ama-physicians-recognition-award.page. The AMA has determined that physicians not licensed in the US who participate in this CME activity are eligible for
