Abstract
Many biomarkers and clinical estimators of ovarian reserve exist. Some of these include a patient's age, antral follicle count, day 3 basal follicle-stimulating hormone (FSH) level, and Anti-Müllerian hormone (AMH) [1]. Some of these have been validated over time (i.e., day 3 FSH levels) versus newer estimates of ovarian reserve (i.e., AMH), which are slowly gaining favor [2–5]. The benefit of laboratory-derived markers of ovarian reserve include reproducibility, decreased element of human error and widespread availability.
Traditionally, day 3 basal FSH levels > 12 IU/l have been used as a predictor of diminished ovarian reserve and as a marker for decreased response to assisted reproduction technologies [6]. Recently, the use of AMH has been validated, and has the added benefit of being cycle day independent. However, the cost associated with obtaining AMH levels is substantially higher than day 3 basal FSH. Nonetheless, the antral follicle count (AFC) and AMH levels are the current standard of care for assessing ovarian reserve.
The answer to predicting ovarian reserve probably requires multiple clinical and laboratory estimates. The goal when evaluating infertility is to achieve the best objective data while maintaining low cost for the patient. Laing
To date, the literature remains inconclusive on the usefulness of the FSH/LH ratio as a laboratory predictor of response to controlled ovarian stimulation [10]. However, more recent work has renewed interest in this measure as a valuable tool for prediction of response to controlled ovarian stimulation [11,12]. The objective of this study is to provide further evidence in validation of this measure of ovarian reserve both by increasing sample size, and by further identifying the patient population that would benefit from this particular assessment.
Materials & methods
We performed a retrospective cohort study of all women undergoing
Women were included if they met the following criteria: age <40 years and women undergoing IVF (no distinction between intra-cytoplasmic sperm injection [ICSI] versus non-ICSI cycles was made). Women were also included only if their cycles were completed or cancelled secondary to poor ovarian response. Criteria used at our center for poor response is a follicle count of four or less, with those follicles measuring at least 10 mm (Table 1).
Patient and cycle characteristics.

FSH: Follicle-stimulating hormone; LH: Luteinizing hormone.
Exclusion criteria included age >40 years, protocols using an estrogen patch, estrogen at start >70 pg/ml, and FSH at start >12 IU/l. A level of 12 IU/l was used as our FSH cutoff since this has proven implications for decreased ovarian reserve and decreased response to controlled ovarian stimulation [13,14]. Estrogen level of >60–80 pg/ml was shown by Licciardi
The main outcome measure was cycle cancellation. Patients that cancelled their cycles secondary to any reason other than poor response (as indicated by mature follicle count or estrogen level) were excluded from the study. Reasons for cycle cancellation that were found and excluded from the study included: self discontinuation secondary to intolerance of medication, divorce or for reasons not indicated).
Other outcome measures included peak estrogen level, total gonadotropin dose required/given, days of stimulation, total number of oocytes retrieved and number of mature oocytes, eggs fertilized and frozen blastocysts.
A total of 676 patients were identified using the inclusion and exclusion criteria as defined above. Of these 676 patients, 198 patients had a FSH/LH >3.0 and 477 patients had a FSH/LH <3.0. This particular cutoff of 3.0 appears to be the most sensitive for predicting cycle cancellation as supported by prior studies [17,18].
The statistical analysis was performed using STATA 11.1 statistical software. Student t-test and Wilcoxon sum-rank tests were used for parametric and non-parametric values, respectively. Statistical significance was defined as p < 0.05. c2 was used in relevant sections using the public statistical software openepi.com where indicated.
Results
Between January 2010 and December 2010, 676 patients were identified who met the inclusion criteria for this particular cohort. Again, 198 patients were in Group 1 (i.e., FSH/LH >3.0) and 477 patients were in Group 2 (i.e., FSH/LH <3.0). The primary outcome of cycle cancellation was available for 198 patients in Group 1 (100%) and 477 patients in Group 2 (100%). Secondary outcomes were also available for all 676 patients within the cohort. Secondary outcomes included: total days of stimulation, total oocytes retrieved, number of mature oocytes, number of fertilized oocytes and number of blastocysts frozen.
There was no significant difference in age observed between Group 1 and 2 (36.9 and 35.9 years old, respectively; p < 0.05). The average estrogen level at start for Group 1 was 27.719 pg/ml and for Group 2 29.275, which was also not significant. Antral follicle count for Group 1 was 10.803 and for Group 2 was 12.487, which was statistically significant.
In terms of cycle cancellation Group 1 (FSH/LH >3.0) was more likely to have their cycle cancelled than Group 2 (FSH/LH <3.0; 15 vs 5.24%; p < 0.0001) (Tab le 2). The relative risk of cycle cancellation was calculated to be 3.0 (95% CI: 1.81–5.03). The total gonadotropin dosage was greater in the higher ratio versus lower ratio group (2636 IU [CI: 2447–2825] vs 2242 IU [CI: 2136–2347]), which was also a significant finding. In addition peak estrogen level was significantly lower in Group 1 (FSH/LH >3.0) versus Group 2 (FSH/LH <3.0; peak estradiol [E2]: 1635 pg/ml, CI: 1524–1746 vs peak E2: 1942 pg/ml, CI: 1868–2016) (Table 2). However, this lower peak E2 did not translate into significant difference in overall cycle outcome, as demonstrated by the similar total number of oocytes retrieved, mature and fertilized (Table 3). Furthermore, the number of frozen blastocysts was similar between the two groups.
Comparison of cycle response.
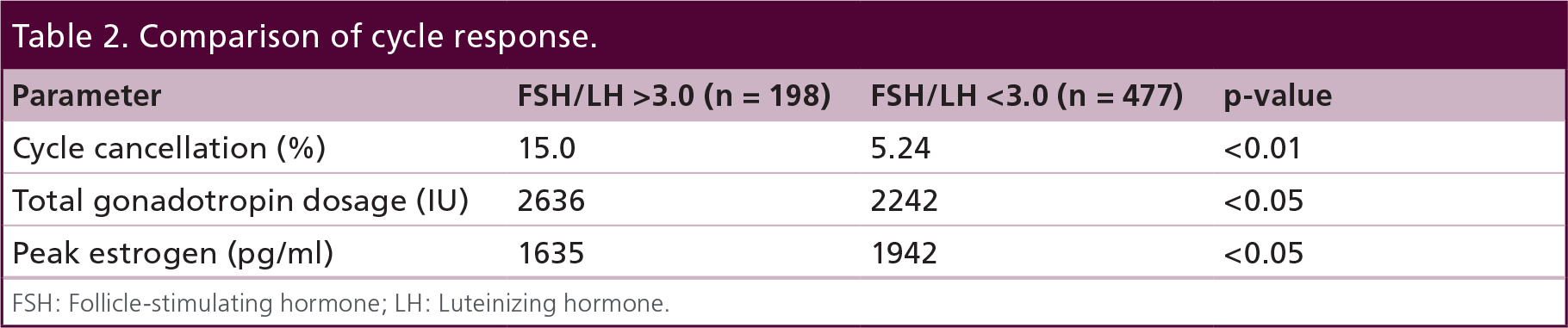
FSH: Follicle-stimulating hormone; LH: Luteinizing hormone.
Cycle outcomes.

FSH: Follicle-stimulating hormone; LH: Luteinizing hormone.
Other secondary outcomes included total days of stimulation. The average number of days of stimulation was 9.28 and 9.38 in Group 1 and Group 2, respectively (p = 0.48) (Table 3).
Discussion
Clinical/biochemical markers that can be used for predictors of ovarian reserve and response to controlled ovarian stimulation should have certain characteristics. The test should be low cost, reproducible and universally available. One of the pitfalls of previous attempts to determine the best marker for ovarian reserve is the failure to realize that the most accurate assessment of ovarian reserve is likely a combination of multiple clinical and laboratory factors [19–22].
Although compelling evidence is provided for the utilization of the FSH/LH ratio as a predictor for cycle response, it is important to note that certain limitations cannot be avoided in a cohort study of this design. However, patient characteristics were similar between the two study groups and other measures of ovarian reserve (i.e., AFC) were statistically significant, which was expected and provides further validation to the use of this ratio.
The benefit of the FSH/LH ratio is that it uses already standardized and universally obtained day 3 laboratory values and allows for risk stratification. In our study we showed a very significant result and tendency for patients with FSH/LH >3.0 to have higher cycle cancellation rates secondary to poor response.
Another potential benefit of this particular ratio is the finding that patients with an elevated FSH/LH ratio require higher total doses of gonadotropins (2636 vs 2242 IU). This allows the clinician to potentially start a patient on a higher dose of gonadotropins than they otherwise would have and lead to a more desirable cycle outcome.
Of the patients who did not have cycle cancellation in the elevated ratio group, the overall cycle outcomes as measured by total oocytes, total mature oocytes and total fertilized oocytes were not statistically significant. Again, this is likely secondary to the higher gonadotropin doses given to Group 1 (FSH/LH >3.0) versus Group 2 (FSH/LH <3.0). Although we have no evidence, it is reasonable to expect that lower gonadotropin doses in Group 1 would have led to significant differences in secondary outcomes.
Overall, this particular study shows a different method to interpret already standardized day 3 laboratory values. In particular it allows the clinician to stratify his/her patients into low versus high responders and tailor the individual treatments to allow for greater cycle success.
Future perspective
Future application and standardization of the FSH/LH ratio will likely require a direct comparison to our current standard predictor of ovarian reserve: AMH and AFC. However, the application of known markers in different ways will increase our knowledge of ovarian reserve.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
Executive summary
Follicle-stimulating hormone (FSH)/luteinizing hormone (LH) ratio is a relatively inexpensive test that can provide clinically useful information for individualized patient response to controlled ovarian stimulation.
The FSH/LH ratio can be used in conjuction with day 3 FSH and cycle independent anti-Müllerian hormone values to provide more information to the physician and patient.
An elevated FSH/LH ratio (i.e., >3) predicts cycle cancellation due to poor ovarian response to gonadotropins. This may be overcome by initiation of higher dose gonadotropins from the beginning of the ovarian stimulation protocol.
Patient protocols are not static and gonadotropin dosage can be adjusted leading to similar outcomes between groups with high and low ratios as shown in this article.
