Abstract
Endometrial cancer is the most common gynecologic cancer in the USA and the second most common worldwide after cervical cancer. While common symptomatology of endometrial cancer leads to early diagnosis and favorable 5-year survival in most cases, there is a subset of cancers that have a poorer prognosis. The clinical and pathologic prognostic factors for endometrial cancer are well known and instrumental in determining the need for adjuvant therapy. Recently, research has been focused on the identification of molecular changes leading to different histologic subtypes to improve classification of endometrial cancer. The identification of novel mutations and molecular profiles should enhance our ability to personalize adjuvant treatment with genome-guided targeted therapy.
Background
Approximately four decades ago endometrial cancer became the leading gynecologic cancer in the USA [ 1 ]. While the incidence of endometrial cancer in the US reached a plateau for a few decades, it has been steadily increasing over the last 5 years from 39,080 estimated new cases in 2007 to 49,560 in 2013 [ 2 , 3 ]. In addition, the incidence of endometrial cancer worldwide has increased since 2000 and it is now the second most common gynecologic malignancy [ 4 , 5 ]. There has also been a concomitant increase in mortality from endometrial cancer. The incidence is comparable in developing and developed countries; however, the mortality rate is higher in developing countries [ 5 ] with a 5-year survival rate of 75% [ 6 ] compared with the US rate of 82% for all stages [ 3 ]. Clearly the burden of endometrial cancer is increasing in the US and worldwide, hence increasing the need to investigate its causes and improve prevention, early diagnosis and treatment.
The escalation in the number of women entering menopause in addition to risk factors, such as obesity and diabetes, may explain a fraction of the increased incidence of endometrial cancer [ 7 – 9 ]. The median age at diagnosis is 61 years with approximately 85% of the cases being diagnosed after 50 years of age [ 10 ]. Consequently, this is generally a disease of postmenopausal women and most cases are diagnosed in early stages owing to the clinical symptoms of postmenopausal bleeding and abnormal discharge. The 5-year survival for early-stage disease is as high as 95% but it rapidly declines to 67% for stage III and 16% for stage IV disease ( Figure 1 ) [ 3 ]. According to the Surveillance, Epidemiology and End Results (SEER) data collected from 2003–2009, 68% of endometrial cancer cases were diagnosed at an early stage, but a considerable number (28%) were diagnosed after regional or distant spread [ 11 ]. While some of the advanced cases may be attributable to delay in diagnosis, a subset of is comprised of a different histologic subtype that is more aggressive with a higher propensity of metastasis. The two histologic subtypes of type I and II are morphologically distinct and their heterogeneity in clinical outcomes and molecular markers is described in further detail throughout this review. Although type II histology represents a small proportion of endometrial cancers, it accounts for a relatively high recurrence and cancer-related death rate [ 12 ]. In order to improve overall survival and decrease recurrence, we need to identify the tumors with poor prognostic behaviors and treat them appropriately with adjuvant and targeted therapy ( Box 1 ).
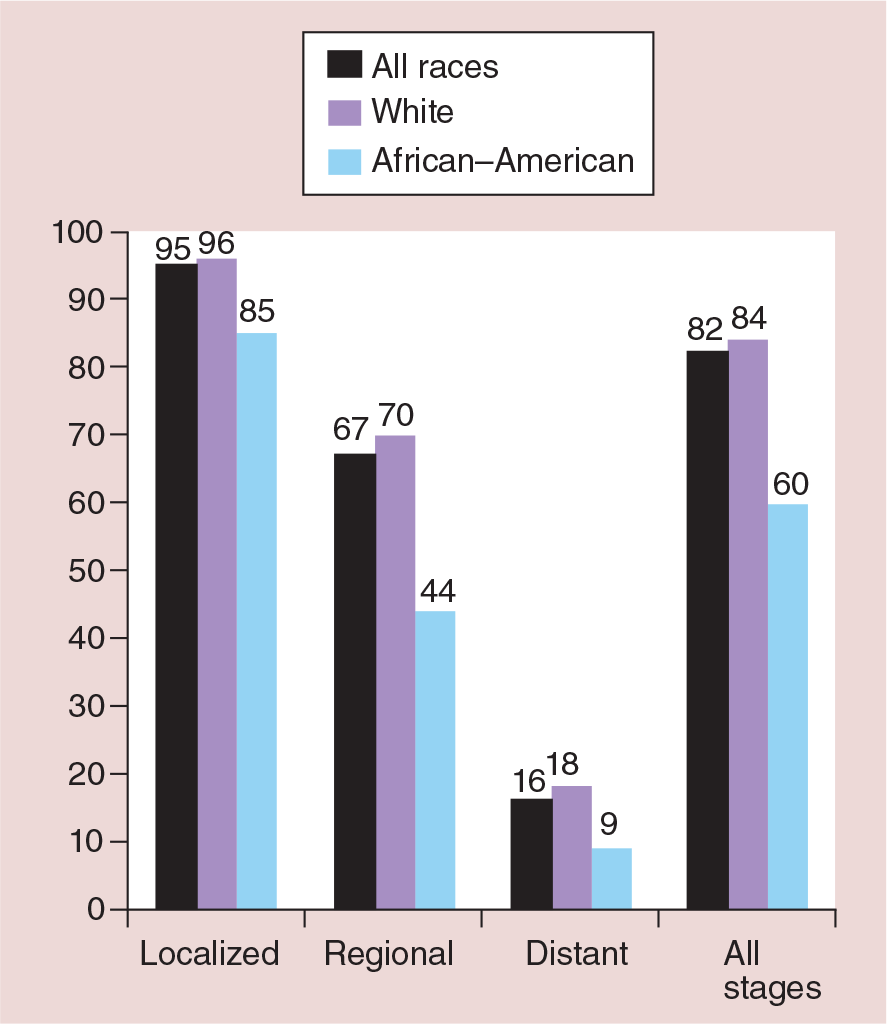
Five-year survival by race for each stage at diagnosis.
Prognostic factors for endometrial cancer.
Age
Race
Histologic subtype
FIGO stage
Histologic grade
Lymphovascular space invasion
Hormone receptor; cellular expression
PTEN; loss-of-function mutation
P13K/AKT/mTOR; pathway aberrations
TP53; loss-of-function mutation
HER2/neu; activating mutation
MSI; MMR gene defects
Angiogenesis markers; mediates VEGF
MEK/ERK; activating mutation
K/Ras; activating mutation
DNA ploidy
Metformin use
FIGO: International Federation of Gynecology and Obstetrics
MSI: Microsatellite instability.
Clinical prognostic factors
Age
Overall, older women have a worse prognosis and a lower 5-year survival than younger women. It is important to evaluate whether this difference is purely based on age or other poor prognostic features associated with age. In a study involving data from 15,471 endometrial cancer patients in the SEER database, Lee
Using data from a GOG study of clinical stage I and II endometrial adenocarcinoma patients with type I or type II histology, Zaino
Both these studies show that age at diagnosis is an independent prognostic factor for survival. In addition to a lower 5-year disease-specific survival rate of 68% in patients aged over 75 years compared with 86% in patients aged 50–75 years (p < 0.0001), Lurain
Race
Compared to the US white population, the lifetime risk of being diagnosed with endometrial cancer is lower for African–Americans, Hispanics, Asian Pacific Islanders and Native Americans/Alaska Natives in decreasing order [ 11 ]. The risk of dying from cancer is lowest amongst American Indian/Alaska natives at 0.32 and comparable between white patients, Hispanics and Pacific Islanders at 0.52, 0.50 and 0.45, respectively. The topic of interest and further research in ethnic and racial disparities has been the almost twofold higher likelihood of death in African–American women when compared with white women.
African–American women have a 6% lower incidence rate of all cancers when compared with white women; however, the death rate is 16% higher [
3
]. This disparity is more evident in endometrial cancer with a 12% decrease in incidence rate and an 86% increase in death rate of African–Americans when compared with white women [
11
]. Several population-based and large institutional studies have shown that African–American women are more likely to present with advanced stage, unfavorable histologic subtypes (serous, clear cell, sarcoma and carcinosarcomas) and higher-grade tumors [
17
–
19
]. The 5-year survival of African–American women is lower than white women for every stage of diagnosis (
Figure 1
); however, this is probably because they have higher grades and more aggressive tumors even when comparing patients with the same stage at diagnosis [
20
]. Two studies show conflicting data about the presence of survival differences between the two races when adjusting for histologic type and grade [
18
,
19
]. Smotkin
Given the significant racial disparity with regards to histologic subtype, molecular and mutation analysis of endometrial cancer specimens from African–Americans and white patients has been performed in an attempt to identify potential genetic differences that can explain the magnitude of this disparity. As expected, in a study of advanced endometrial cancers, white women were more likely to have
Differences in biologic features and the disproportionate distribution of poor histology between the two races are the principal factors leading to the disparity in endometrial cancer survival. To account for the rest, review articles make a strong argument that socioeconomic status plays an important role in explaining survival differences across races since African–Americans are more likely to live below the poverty level and in underserved areas [ 17 , 26 ]. Rationally, a low socioeconomic status would seem to affect access to appointments, time to diagnosis and access to treatment and follow-up; however, studies presented in the review articles mentioned above are still conflicting on whether it is an independent prognostic factor in endometrial cancer.
Pathologic prognostic factors
Histologic type
Bokhman first described the concept of two pathogenetic types of endometrial cancer arising in two clinically diverse subsets of patients in 1983 [ 27 ]. He prospectively observed 366 patients with endometrial cancer and compared prognostic factors between the two proposed histologic subtypes. He noticed that the subtype currently designated as type II histology was found in 35% of endometrial cancers and was associated with high nuclear grade, poor tumor differentiation, deep myometrial invasion, high frequency of lymph node metastasis, decreased sensitivity to progestins and poor 5-year survival rate (58.8%). This type is found in women who tend to be thin and lack the endocrine and metabolic disturbances leading to obesity, hyperlipidemia and a hyperestrogenic state. Type I histology was more common (65%), arose in obese women with endocrine and metabolic disturbances, were highly sensitive to progestins, and had favorable surgical–pathologic prognostic factors and 5-year survival rate (85.6%). In subsequent studies, type I histology is also referred to as endometrioid and type II referred to as nonendometrioid, papillary serous or serous, clear-cell and undifferentiated carcinomas ( Table 1 ). Currently, the term papillary is no longer being used to describe serous carcinomas.
Clinical-pathologic characteristics of type I and type II endometrial cancer.

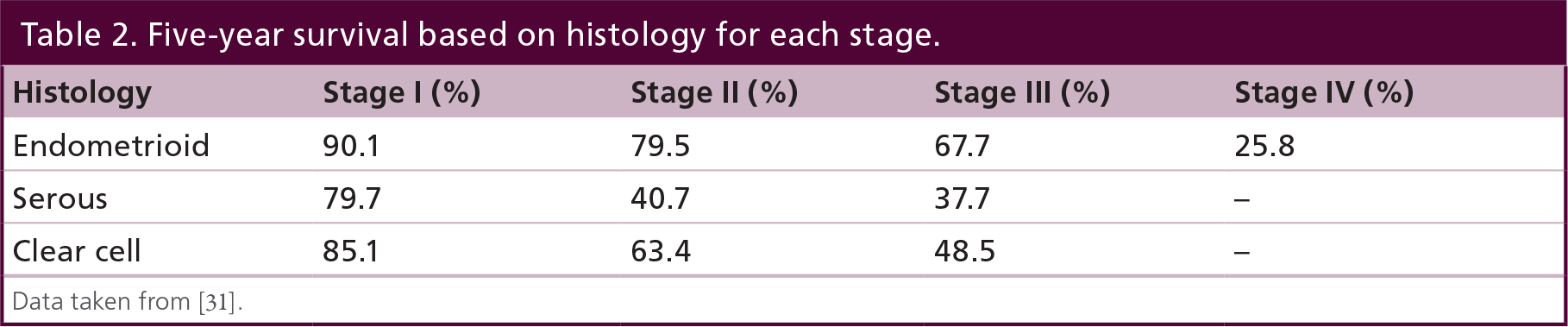
Previously mentioned studies of clinical stage I endometrial cancer found that the 5-year survival for serous and clear cell histology was significantly lower than endometrioid with and without squamous differentiation [ 15 , 16 ]. A retrospective review shows that a majority of tumors with this histology have a higher extent of extrauterine disease and a higher frequency of recurrence despite adjuvant radiation treatment [ 28 ]. Even in surgical stage IA cancers with papillary serous and clear cell histology, the 5–year survival was 57% compared with >90% for endometrioid counterparts [ 29 ]. Adjusting for stage of disease, this continues to be the trend ( Table 2 ). Interestingly though, if poor histology is isolated to endometrial curettings without any evidence in hysterectomy specimen, this may not affect prognosis [ 30 ].
Recent reviews state that since uterine papillary serous carcinoma is morphologically, genetically and clinically different than endometrioid carcinoma, there may be more heterogeneity between the two than originally thought [ 32 , 33 ]. Recent research on the genomics of endometrial cancer provides evidence that the differences between type I and type II histologic subtypes may be reflected in their genetic and molecular profiles [ 34 ]. Identifying the distinct genetic mutations that affect cell signaling pathways is the next step to make advancements in targeted therapies.
Tumor stage
The purpose of staging in malignancies is to classify tumors based on size and spread of disease in order to accurately assess prognosis. Prior to 1988, endometrial cancer was staged based on size of the uterus and clinical extent of disease. Since reports in the 1970s showed that survival of clinical early-stage endometrial cancer was not as high as expected, multiple studies were performed looking at pathologic prognostic factors for recurrence and survival. These studies noted a substantial discrepancy between clinical stage prediction and the actual extent of disease based on pathologic review of specimens from surgical staging [ 15 , 16 , 35 – 37 ]. In 1988, International Federation of Gynecology and Obstetrics (FIGO) classification changed from clinical to surgical staging for endometrial cancer. The staging classification was revised by FIGO in 2009 by combining stage IA and IB, removing stage IIA, and incorporating important prognostic variables regarding paramentrial involvement and lymph node status into stage III.
Surgical stage is the most significant prognostic variable for endometrial cancer. Data from GOG 33 looked at surgical and pathologic factors that were associated with extrauterine disease, lymph node involvement and recurrence in clinical early-stage disease. In a multivariate analysis, depth of myometrial invasion and extrauterine disease was associated with lymph node metastasis [ 36 ]. Degree of myometrial invasion was one of the most important prognostic factors related to recurrence in patients with early-stage endometrial cancer [ 15 , 16 , 38 , 39 ]. Recurrence was also highly dependent on pelvic and para-aortic lymph node status, which in turn was associated with extrauterine metastasis to the cervix, adnexa and peritoneum. Implications from these findings were important in confirming the importance of surgical staging and classifying patients in prognostic groups in order to provide adequate adjuvant treatment.
Histologic grade
Data from the GOG 33 protocol showed that poor tumor differentiation was associated with a higher depth of invasion [ 36 ]. In a multivariate analysis, grade of tumor was independently and significantly related to lymph node metastasis. Long-term data from the same patients showed that for patients with surgically confirmed stage I disease, the greatest determinant of recurrence was grade 3 histology with the relative risk of grade 3 being three-times greater than grade 1 [ 39 ].
The FIGO annual report of patients treated from 1999–2001 showed that the 5-year survival decreases with increasing grade for every stage of disease ( Table 3 ) [ 31 ]. Grade of tumor differentiation is a marker of prognosis and is associated with other prognostic factors such as myometrial invasion, extrauterine disease and lymph node status. It is included while reporting the FIGO stage assignment of endometrial cancer.
Lymphovascular space invasion
Similar to histologic grade, the presence of malignant cells in the capillary-like space increases the risk of pelvic and para-aortic lymph node involvement by four- and six-times, respectively [ 36 ]. The relative risk of recurrence for patients with lymphovascular space invasion (LVSI) compared to without was 2.4 [ 39 ]. A retrospective review of 757 endometrioid endometrial cancers shows that the presence of LVSI is highly associated with lymph node metastasis in a univariate analysis (odds ratio: 11, p < 0.0001) and a significantly reduced progression-free survival and overall survival in a multivariate analysis [ 40 ].
In further support of this data, a more recent retrospective study looking at early stage endometrioid tumors from a single institution showed that LVSI was the only consistent prognostic factor affecting recurrence and survival [ 41 ]. In the multivariate analysis, after adjusting for other important prognostic factors such as age, myometrial invasion, grade and adjuvant therapy, LVSI was an independent risk factor with a 2.6 higher odds of recurrence. The hazard ratio for overall survival and disease-specific survival was 2.8 and 7.0, respectively. This pathologic factor is specifically important in early stage patients where the benefit of adjuvant therapy is debatable.
Molecular markers associated with prognosis & targeted therapies
As discussed previously, the majority of patients with endometrial cancer present with stage I disease, which is potentially curable by surgery with or without adjuvant radiotherapy [ 31 ]. Nonetheless the reported 5-year survival for surgical stage I disease treated between 1999–2001 ranged from 79–93% when taking into account grade and histologic subtype [ 31 ]. The decreased prognosis due to high grade and type II histology should be the main focus in current research to improve the overall survival of patients diagnosed with early-stage disease. As a result, molecular markers are being evaluated in an attempt to identify the patients with early stage but high risk of recurrence, since they would benefit from adjuvant and targeted therapies.
Mortality rates increase with advancing stage of disease despite advances in radiotherapy, surgery and chemotherapeutic efforts. The median overall survival in these patients is less than 1 year [ 42 , 43 ]. By improving our understanding of the disease at a molecular level and identifying the genetic changes that give rise to the heterogeneity in this spectrum of malignancies, we will enhance our ability to develop targeted therapies to supplement our current treatment strategies.
Hormone receptors
Reports on the prognostic value of hormone receptor status on endometrial cancer have been published since the 1980s. Estrogen and progesterone receptor positivity are independent prognostic factors with a significantly improved disease-free survival [ 44 , 45 ]. Hormone therapy including progestins, aromatase inhibitors and selective estrogen receptor modulators are attractive first-line therapies for young patients wanting fertility-sparing options and supplemental therapies for patients with advanced disease since they lack adverse toxicities. A few published cases report successful response in treating young patients with early-stage type I disease; however, the median progression-free survival in patients with recurrent or advanced disease are minimal at 1–3.7 months [ 46 – 48 ]. Hormone receptor status is still an important molecular prognostic factor and hormonal therapy should always be considered as supplemental and palliative targeted therapy.
PTEN
P13K/AKT/mTOR pathway
PTEN negatively regulates the downstream pathway of AKT-mediated signals that are important for cellular growth, proliferation and survival [ 49 , 52 ]. The PI3K/AKT/mTOR pathway is the most commonly deregulated signaling pathway and is affected in more than 80% of type I endometrial cancers [ 53 ].
In addition to the previously mentioned loss of PTEN function, activating mutations in
Detecting mutations in this pathway has clinical significance as new treatment strategies targeting this pathway are being developed. The antitumor drugs have been classified as PI3K inhibitors, mTOR inhibitors, dual PI3K/mTOR inhibitors and AKT inhibitors [ 55 , 56 ]. Different mTOR inhibitors have proven benefit in other malignancies including renal cell carcinoma and sarcomas [ 57 , 58 ]. A Phase II trial conducted by the National Cancer Institute of Canada clinical trials group conveyed favorable results with the use of temsirolimus in chemotherapy naive patients and reported a 14% partial response rate and 69% stable disease rate in this subset of patients [ 59 ]. Phase II trials in recurrent endometrial cancer patients have shown disease stabilization with everolimus and significantly improved progression-free survival with ridaforolimus [ 60 ].
p53
The tumor suppressor gene
In addition to the association with type II histology,
Saffari
HER2/neu & EGFR
The HER2 protein is a transmembrane glycoprotein from the human EGFR tyrosine kinase family.
While HER2 overexpression is associated with poor surgical–pathologic prognostic factors and poor survival, it also serves as an important target for the treatment of uterine serous carcinomas. Trastuzumab and pertuzumab are two US FDA-approved humanized monoclonal antibodies targeting the extracellular domain of the HER2 receptor and are currently being evaluated in clinical trials for the treatment of endometrial cancer [ 70 ]. While single agent trastuzumab did not have a significant response in advanced stage and recurrent disease, combination treatment with standard chemotherapy is still being evaluated in a multi-institutional Phase II trial [ 70 , 71 ].
In addition to verifying that high HER2 expression is an independent prognostic factor influencing progression-free and overall survival in endometrioidtype cancers, results from Mori
EGFR overexpression has also been reported in 40–46% of type I and 34% of type II endometrial carcinomas [
72
,
75
]. In a study of uterine serous carcinoma, no
Microsatellite instability
Microsatellite instability (MSI) is the tendency to develop alterations in the number of short segments of repetitive DNA owing to DNA repair errors during cell replication. MSI occurs owing to inactivation of DNA mismatch repair genes
Studies from single institutions present conflicting data on the association of MSI with known prognostic factors, molecular markers and survival. In a prospective study of 229 endometrial cancers, white race was more common in MSI-positive tumors but advanced staged and type II histology was less frequent in MSI-positive tumors compared with microsatellite stable tumors [
79
]. Despite the significant association of MSI-positive tumors with favorable race and lower stage disease, there was no significant difference in recurrence or overall survival between the two groups. The
Microvascular proliferation
The importance of angiogenesis in the growth and metastasis of solid tumors was described over four decades ago [
83
,
84
]. Haldorsen
VEGF ligands and its receptors are the main mediators of angiogenesis, and inhibitors of these targets have shown therapeutic benefit in multiple malignancies [ 86 ]. VEGF is also the main mediator for endothelial cell proliferation in endometrial carcinoma [ 87 ]. A Phase II trial of single-agent bevacizumab, a US FDA-approved recombinant human monoclonal IgG antibody binding to VEGF, showed encouraging results in the treatment of recurrent or persistent endometrial cancer [ 88 ].
While there was a modest response rate of 13.5%, median progression-free survival and overall survival were 4.2 and 10.5 months, respectively, and 40.4% of patients were progression free at 6 months. The combination of bevacizumab and temsirolimus showed a slightly improved response rate of 24.5%, median progression-free survival and overall survival was 5.6 and 16.9 months, respectively, and 46.9% of patients were progression free at 6 months [ 89 ]. However, the combination treatment was associated with significant toxicity. Bevacizumab is now under further investigation in combination with chemotherapy [ 90 ].
Metformin
Metformin is an oral antidiabetic medication recommended by the American Diabetes Association as first-line therapy for Type 2 diabetes [ 91 ]. It may not seem to be a targeted therapy at first, but several studies show that metformin use is associated with a reduced risk and greater overall survival for several malignancies including breast, ovarian and pancreatic cancer [ 92 – 94 ]. Metformin targets cancer stem cells and provides a synergistic effect with chemotherapy in obstructing tumor growth [ 95 ]. A review of laboratory research shows that metformin affects multiple targets and molecular pathways leading to antitumor activity [ 96 ].
In endometrial cancer, metformin use is associated with improved clinical outcomes including recurrence-free and overall survival [ 97 ]. A retrospective cohort study of nonendometrioid endometrial cancer patients showed that overall survival was significantly improved in diabetic patients on metformin when compared with diabetic patients not using metformin and non-diabetic patients [ 96 ]. Research is ongoing to determine whether genetic polymorphisms can predict a response to metformin [ 98 ].
Conclusion
The clinical, surgical and pathologic prognostic factors for endometrial cancer have been well studied over the last three decades. Current efforts are directed towards elucidating mutation profiles and molecular markers that will lead to better histologic classification of endometrial cancer and an improved ability to identify cancers with poor prognostic behaviors. In 2012, McConechy
Future perspective
The discovery of genetic and molecular changes that give rise to the heterogeneity in endometrial cancers has allowed us to reclassify these cancers. Genomic and proteomic characterization of endometrial cancer will become commonplace in the near future and will allow for accurate diagnosis, especially in cases where histologic diagnosis by pathologists is difficult or discordant. In addition to current standard adjuvant chemotherapy and radiotherapy regimens, upcoming endometrial cancer treatment regimens will be focused on novel drug therapies targeting genomic mutations and aberrant molecular pathways identified in individual tumors. The ultimate goal is to offer personalized cancer care in order to achieve improved clinical outcomes.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Executive summary
The incidence of endometrial cancer in the USA and worldwide is increasing.
An understanding of prognostic factors affecting endometrial cancer is essential to determine the risk of recurrence and benefit of adjuvant therapy.
Clinical prognostic factors include age and race.
Surgical-pathologic factors include histologic subtype, stage, histologic grade and lymphovascular space invasion.
The identification of biologic markers and cellular pathways involved in the pathogenesis and heterogeneity of endometrial cancer is a focus of current laboratory and clinical research.
Biologic markers affecting prognosis of endometrial cancer include hormone receptors, PTEN, p53, P13K/AKT/mTOR pathway, HER2/neu, angiogenesis markers and microsatellite instability.
Novel drug therapies target mutations in molecular markers and aberrations in cellular pathways.
Combination therapy with current treatment strategies and recently discovered targeted therapies should lead us to the ultimate goal of individualized cancer care and improved clinical outcomes.
Genomic and proteomic characterization of endometrial tumors have been performed to classify different subtypes.
Genetic profiling will become routine in characterizing endometrial tumors and predicting prognosis and benefit from targeted therapies.