Abstract
Keywords
The transdermal application of steroids using patches containing an estrogen alone or in combination with a progestin is well established as a method of hormonal therapy in postmenopausal women [1]. A transdermal patch for the purposes of contraception has also been developed and, in 2002, European medical regulators approved a weekly transdermal contraceptive patch containing 0.06-mg ethinyl estradiol (EE) and 6-mg norelgestromin in a monophasic 21-day regimen per 28-day cycle [101]. In the USA, a slightly different formulation was approved by the US FDA in November 2001 [102].
A transparent once-a-week transdermal contraceptive patch has recently been developed that delivers low doses of EE and gestodene (GSD). Use of this patch over a 7-day application period results in the same systemic exposure, in terms of bioavailability, as observed after oral administration of a combined oral contraceptive (COC) containing 0.02-mg EE and 0.06-mg GSD [Bayer Pharma AG, Unpublished Data].
The use of daily COCs represents the most common means of birth control in the developed world [103]. Although daily COCs are highly effective, they have been associated with poor user compliance and, thus, reduced efficacy [2]. Additionally, the use of daily COCs is associated with the tendency for serum hormone concentrations to fluctuate rapidly and to a significant degree [3], with large intra- and inter-individual pharmacokinetic variations in serum hormone levels [4], as well as a limited bioavailability of EE (38–48%) [5].
Transdermal delivery of hormones for contraceptive purposes is an effective alternative to daily COCs, offering several advantages over the oral administration of hormones. For example, it offers the convenience of weekly patch application and avoids reductions in estrogen/progestin bioavailability due to hepatic metabolism and enzymatic degradation in the GI tract, while also resulting in relatively constant exposure to these hormones due to continuous administrations of the drug over the application period [3,6,7]. Transdermal contraceptive patches therefore provide a further contraceptive option to women and may increase the level of compliance.
Both EE and GSD are well absorbed through the skin and are therefore appropriate for use in combination for transdermal contraceptive delivery [3,7]. At present, EE is the most potent estrogen agonist available for clinical use [8]. Both EE and GSD are well established as contraceptive agents, having been in use for decades [7–11]. GSD has good skin absorption properties [7] and requires a low absolute dose [12], which allows for a small patch size [Bayer Pharma AG, Unpublished Data].
The efficacy of combined estrogen/progestin contraceptives is based on the interaction of various factors, the most important of which are the inhibition of ovulation and changes in cervical mucus. The increased levels of estrogen and progestin resulting from COC use act on the hypothalamo–pituitary axis in order to reduce the secretion of gonadotropin-releasing hormone, follicle-stimulating hormone and luteinizing hormone [13]. This, in turn, suppresses ovarian follicular development, preventing ovulation. The progestin component also acts on the endometrium [14] and, in doing so, can cause increases in endometrial vascularity that lead to abnormal bleeding; however, there is evidence that the progestin component can also induce endometrial atrophy [15], thus limiting the effect on vascularity.
Evaluation of endometrial changes is an important aspect of contraceptive efficacy studies and is specified in guidance from the EMA [104]. Hence, the aim of the present study was to investigate the endometrial effects after 1 year (i.e., 13 cycles of 28 days each) of this EE/GSD contraceptive patch. Additional evaluations included contraceptive efficacy, aspects of safety and compliance.
Materials & methods
Objectives
The primary objective of the study was to investigate the effects on the endometrium of a transdermal contraceptive patch containing 0.55-mg EE and 2.1-mg GSD. Secondary objectives were to evaluate the contraceptive efficacy of the patch and aspects of its safety profile. In addition, this study also investigated compliance.
Study design & participants
The study (EudraCT: 2010-021255-81) was an open-label, uncontrolled, Phase IIb study that enrolled 92 female volunteers at a single center in Germany. Participants were healthy women aged between 18 and 35 years (no older than 30 years if a current smoker) with a normal cervical smear at screening or within the previous 6 months, a history of regular, cyclic menstrual periods and an endometrial biopsy devoid of abnormality at screening. A negative pregnancy test was required before application of the first patch. A urine human chorionic gonadotropin test (home pregnancy test) was to be performed by the subject before the first patch application. In addition, urine pregnancy tests were conducted at the site during visits 1a, 1b, 6 (before biopsy) and 7.
Key exclusion criteria included: hypersensitivity to any ingredient of the study drug; significant skin reactions to transdermal preparations or sensitivity to surgical/medical tape; pregnancy or lactation (i.e., fewer than three menstrual cycles between delivery, abortion or lactation and start of treatment); and obesity (BMI >30 kg/m2).
In addition, subjects were excluded if they had laboratory values outside the reference range. Laboratory values included: measures of general serum plasma chemistry (potassium, sodium, creatinine, total protein and albumin); hematology (leukocytes, erythrocytes, hemoglobin, hematocrit and platelets); liver enzymes (glutamic pyruvic transaminase/alanine transaminase, glutamic oxaloacetic transaminase/aspartate transaminase, γ-glutamyltransferase, alkaline phosphatase, cholinesterase and total bilirubin); carbohydrate metabolism (glycosylated hemoglobin); and lipids (total cholesterol, high-density lipoprotein, low-density lipoprotein and triglycerides).
Subjects were also excluded if they had diseases or conditions that could affect the pharmacokinetics of the study drug or that may worsen during hormonal treatment; if they had diseases or conditions that could lead to altered absorption, excessive accumulation, impaired metabolism or altered excretion of the study drug (e.g., known skin disease with suspected alteration of dermal absorption or poor adherence of the patch, such as psoriasis); or if they had cardiovascular conditions, liver conditions, or a history of medical conditions in these areas.
Metabolic diseases; undiagnosed abnormal genital bleeding; abuse of alcohol, drugs or medicines; or use of any other contraceptive method (oral, vaginal or transdermal hormonal contraception, sterilization, intrauterine devices with or without hormone release, implants, or long-acting preparations within three-times the length of the injection interval prior to the start of treatment) were also criteria for exclusion.
The EE/GSD patch (BAY 86-5016) is a colorless, translucent, matrix-type patch with a size of 11 cm2. It consists of five layers: a polyethylene backing layer, a polyisobutylene adhesive layer containing an ultraviolet absorber, a polyethylene terephthalate intermediate layer and a polyisobutylene layer containing EE and GSD. Before use, the drug-containing adhesive layer is protected by a polyethylene terephthalate release liner.
The study consisted of 13 treatment cycles. All women received the EE/GSD patch, given in a 21-day regimen per 28-day cycle (one patch every 7 days for 21 days followed by a 7-day patch-free interval). A second endometrial biopsy was performed at cycle 13 (on days 7–21) in order to assess the primary treatment effect. Secondary and safety outcomes were assessed during scheduled study visits on days 7 and 21 in cycles 3, 7, 10 and 13. For the assessment of compliance, women were required to keep diary cards until the follow-up visit, and completed diary cards were collected at visits 3–7, with missing data obtained by direct questioning. The study was conducted in accordance with the ethical principles of the Declaration of Helsinki and the International Conference on Harmonization. The protocol was approved by the appropriate independent ethics committee or institutional review board (Landesamt für Gesundheit und Soziales, Germany), and all participants provided written, informed consent.
Assessments
The primary variable in the study was the effect of an EE/GSD contraceptive patch on the endometrium at cycle 13. Endometrial biopsies were taken at screening and between days 7 and 21 of cycle 13 using an aspiration technique consistent with current clinical practice at the center. Based on histologic analysis by a pathologist at the central laboratory, biopsies were categorized as atrophic, inactive, proliferative (weakly, active or disordered), secretory (cyclic or progestational) or menstrual. At screening, biopsy classifications of women in the full analysis set were as follows: 16 (18.0%) were ‘atrophic’; 19 (21.3%) were ‘inactive’; one (1.1%) was ‘menstrual’; 20 (22.5%) were ‘proliferative (active)’; three (3.4%) were ‘proliferative (weakly)’; 18 (20.2%) were ‘secretory (cyclic)’; and 12 (13.5%) were ‘secretory (progestational)’. Women who prematurely discontinued the study, but had been treated for at least 6 months, were asked to have an endometrial biopsy performed at the time of discontinuation.
Efficacy variables included the number of unintended pregnancies while receiving treatment (up to 7 days after removal of the last patch; participants were also followed-up for 3 months after discontinuation) and treatment compliance. Mean treatment compliance was calculated as a percentage of the number of actual patch-wearing days recorded versus per-protocol patch-wearing days using the following formula:
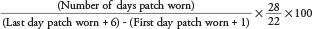
Adverse events (AEs) were documented throughout the study and were coded using the Medical Dictionary for Regulatory Activities (MedDRA) version 14.0. Pregnancy was considered to be an efficacy parameter and was thus not considered to be an AE. This is in contrast to the outcome of pregnancy. Any abnormal pregnancy outcome (i.e., other than spontaneous full-term delivery) was considered an AE due to the standard operating procedures of the sponsor.
Vital signs and body weight were assessed at each visit. In addition, a cervical smear and gynecologic examination were carried out during cycle 7 and at the final examination during cycle 13.
Statistical analysis
As analysis of the primary variable – endometrial biopsy result at cycle 13 – was purely descriptive, a stringent sample size calculation based on statistical analyses could not be performed. Based on suggestions from regulatory authorities, the decision was made to enroll 80 volunteers, based on screening of approximately 100 women, with the aim of obtaining approximately 60 evaluable biopsies at cycle 13.
The full analysis set included all women who used at least one patch during the study and for whom at least one observation after admission to treatment was available. A restricted full analysis set was defined as the main population for the primary target variable, containing women who received study treatment and provided at least one postscreening endometrial biopsy during treatment between cycle days 7 and 21. A per-protocol set was also defined, and included all women in the full analysis set with no protocol deviations that may have affected the primary target variable.
All analyses were carried out using SAS® software version 9.1 or higher (SAS Institute, Inc., NC, USA). Variables measured on metric scales were summarized by use of descriptive statistics. Variables measured on ordinal or nominal scales were summarized by use of frequency tables showing the number and percentage of women.
Results
Of the 148 women screened, 92 were enrolled in the study (Figure 1). Of these, three did not receive the study treatment (i.e., did not start to apply the patch) and were excluded from the analyses. The full analysis set therefore consisted of 89 women. The majority of subjects in the full analysis set had never given birth (84.3%) and had never undergone an abortion (82.0%). The mean time since the last birth or abortion was 1635.9 ± 1906.8 days. Baseline characteristics of the full analysis set and per-protocol set are shown in Table 1. In total, 57 women (64%) completed the study. The most frequent reason for discontinuation was an AE (18 subjects; 20.2%). The restricted full analysis set and per-protocol set contained 53 and 49 women, respectively.
Baseline characteristics of the full analysis set and per-protocol set†.
No demographic data are available for the restricted full analysis set. The restricted full analysis set and per-protocol set differed by only four subjects; thus, any differences between these evaluation sets are negligible.
SD: Standard deviation.
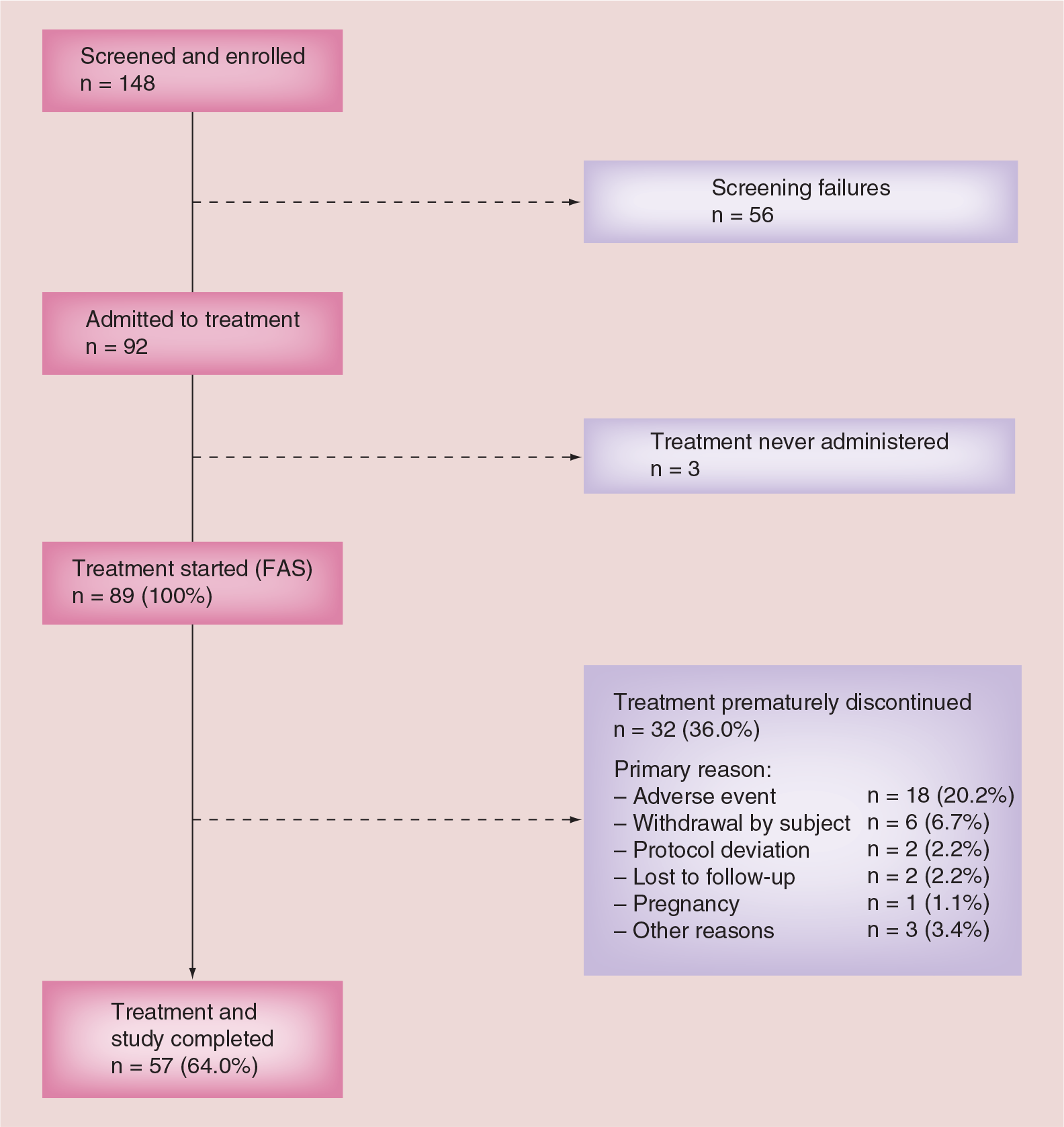
Study flow diagram.
Endometrial biopsy
At their last visit, endometrial biopsies were available from 59 of 61 women in the full analysis set (97%) who completed at least 6 months of treatment; two subjects declined the procedure and, thus, samples were not obtained from these participants. Of the 59 biopsies that were conducted, all were considered to be histologically consistent with treatment and devoid of any abnormality. Of the 59 biopsies, five were performed during days 22–28 of cycle 6 (n = 3) and cycle 7 (n = 2) due to subjects' premature discontinuation from the study. Across all populations, approximately half of the women had endometrial biopsies categorized as ‘inactive’ (full analysis set: 44.3%; restricted full analysis set: 49.1%; per-protocol set: 51.0%). A further 16–18% of women (full analysis set: 16.4%; restricted full analysis set: 17.0%; per-protocol set: 18.4%) had a biopsy categorized as ‘atrophic’. Histologic descriptions for endometrial biopsies, for all populations, are shown in Table 2.
Postscreening biopsy results and histologic descriptions of endometrial biopsies.
All data are shown as n (%).
Subjects for whom a last postscreening visit to obtain a biopsy sample was recorded.
Contraceptive efficacy
One woman in the full analysis set (1.1%) became pregnant during the study treatment (i.e., between the first patch application and 7 days after the last patch removal). The positive pregnancy test result was obtained on day 100 (cycle 4), after which the woman immediately stopped patch use. In the absence of evidence to indicate that the study drug was used incorrectly, the pregnancy was deemed a ‘method failure’.
Compliance
Mean (± standard deviation) overall compliance was 97.9 ± 6.5% (range: 57–114%) for the full analysis set and 99.7 ± 1.0% (range: 97–102%) for the per-protocol set; median (interquartile range) compliance rates were 100% (97.4–100%) and 100% (99.5–100%), respectively.
Safety
In total, 80 of the 89 women in the full analysis set (89.9%) experienced at least one treatment-emergent AE, most of which were mild or moderate in intensity, and typical of a young female population using a transdermal contraceptive product (Table 3). Treatment-emergent AEs considered by the investigator to be related to the study treatment occurred in 65 women (73.0%) and, again, were those that could be expected with a transdermal contraceptive patch (Table 3). In total, 18 women (20.2%) discontinued treatment as a result of a treatment-emergent AE. Serious AEs occurred in four women (4.5%), and these were lower abdominal pain and salpingo-oophoritis, appendicitis, induced abortion (the sponsor required that any pregnancy that did not end in a spontaneous term delivery was to be recorded as a serious AE; the pregnancy itself was recorded as an efficacy, not safety, parameter) and pyelonephritis. All resolved without sequelae, and none were considered to be related to the study medication. There were no deaths during the study.
Most common treatment-emergent adverse events and drug-related treatment-emergent adverse events occurring in >10 and >5% of participants using the ethinyl estradiol/gestodene patch, respectively (full analysis set; n = 89).
TEAE: Treatment-emergent adverse event.
As noted earlier, all endometrial biopsies taken during the study were devoid of any abnormality. At the final examination, smear test results were normal for 94.6% of the women examined. Abnormalities that were observed in the remaining four women, occurring since screening, were low-grade squamous intraepithelial lesions (two women), atypical squamous cells of undetermined significance (one woman) and a high-grade squamous intraepithelial lesion (one woman).
Discussion
COCs inhibit conception through a range of mechanisms, including inhibition of ovulation, changes in cervical mucus and effects on the endometrium [16,17]. The present study was designed to examine the effect of a contraceptive patch containing EE and GSD on the endometrium, and the results show clear suppression of endometrial proliferation after 13 cycles.
Based on biopsy samples, the endometrium was strongly suppressed in over two-thirds of women in the restricted full analysis set population: approximately half of the women had an endometrium classified as ‘inactive’ and, in a further 17%, it was classed as ‘atrophic’. These results are supported by similar data from the full analysis set and per-protocol set populations. Importantly, previous research has shown that women with atrophic or otherwise ‘regressed’ endometria are less likely than other histologic groups to experience abnormal bleeding during progestin-only contraception, despite an increase in vascular density [18]. While an increase in vascularity often leads to increased abnormal bleeding with progestin-only contraception, induction of endometrial atrophy can ameliorate this effect [15]. It seems likely, therefore, that the observed benefits of GSD over other progestins in terms of cycle control and bleeding profile [11] are mediated, at least in part, by endometrial suppression. The biopsy results in the present study are consistent with previous endometrial biopsy studies of GSD-based contraceptives [19–21]. For example, an open-label study in 27 women receiving an oral contraceptive containing 60-μg GSD and 15-μg EE found that, after six cycles, 44% of women had endometrial histology classed as ‘atrophic’ [19]. Similarly, in a study of 13 women, six cycles of orally administered treatment with 75-μg GSD and 20-μg EE resulted in an atrophic endometrium in 46% of participants [21]. As changes in the endometrium are related to the menstrual cycle, no comparison was made between baseline and follow-up biopsies, as any differences would most likely reflect the cycle phase rather than a treatment effect.
The relatively small sample size in this study potentially limits the conclusions that can be drawn regarding endometrial safety; however, it should be noted that safety was not the focus of this study and the sample size was chosen accordingly. Data were not collected during the study on endometrial bleeding and ovarian cycle control, and neither was the study powered to assess efficacy. These aspects were the focus of other studies in a comprehensive clinical development program [Bayer Pharma AG, Unpublished Data].
Conclusion
In conclusion, this study shows that the EE/GSD patch was associated with an endometrial effect that is consistent with suppression of endometrial proliferation, with more than two-thirds of women in the restricted full analysis set population having an ‘inactive’ or ‘atrophic’ endometrium after 13 treatment cycles (i.e., after 1 year of use). The safety profile of the patch revealed no safety concerns. Once-weekly application of the EE/GSD patch was associated with good efficacy and high compliance. Longer-term observation is now needed in order to confirm these promising results.
Executive summary
There was clear suppression of endometrial proliferation in response to application of the ethinyl estradiol (EE) and gestodene (GSD) patch.
Based on biopsy samples, the endometrium was strongly suppressed in over two-thirds of the women in the restricted full analysis set population.
Contraceptive efficacy was high, with only one woman becoming pregnant during the study due to ‘method failure‘. Studies powered to measure the Pearl Index with the EE/GSD patch are under preparation.
Overall compliance was high in the present study.
All endometrial biopsies taken during the study were devoid of any abnormalities.
No new safety concerns relating to use of the EE/GSD patch emerged during the course of the study.
The observed benefits of GSD over other progestins in terms of cycle control and bleeding profile appear to be mediated, at least in part, by endometrial suppression.
Longer-term studies, particularly in relation to safety, are needed in order to confirm these results and to add to the existing literature on the effects of chronic use of hormonal contraceptives when administered via the transdermal route.
Financial & competing interests disclosure
This study was supported by Bayer Pharma AG. M Merz and J Grunert are both employees of Bayer Pharma AG. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
The authors would like to thank Ogilvy 4D (UK), for providing writing assistance, which was also funded by Bayer Pharma AG.
Ethical conduct of research
The authors state that they have obtained appropriate institutional review board approval or have followed the principles outlined in the Declaration of Helsinki for all human or animal experimental investigations. In addition, for investigations involving human subjects, informed consent has been obtained from the participants involved.
