Abstract
In the USA, family planning clinics are primary providers of reproductive healthcare to young women and their male partners and have long provided quality sexually transmitted infection (STI) care and prevention. Chlamydia, an easily treatable STI that can lead to serious adverse outcomes if untreated, is the most common bacterial STI in the USA, and annual chlamydia screening is recommended for sexually active women aged ≤25 years. As early adopters of routine screening, family planning clinics screen >50% of all care-seeking eligible women for chlamydia, performing better than private sector healthcare plans. To achieve high levels of quality care, family planning clinics have been leaders in implementing evidence-based care delivery and developing prevention innovations. As national healthcare reform is implemented in the USA and categorical STI clinics close, public-sector demand on family planning clinics will increase.
Keywords
An estimated 2.8 million Chlamydia trachomatis infections occur annually in the USA [1]. However, chlamydia infections are largely asymptomatic, and many are not detected and treated [2]. Despite this, chlamydia is still the most commonly reported nationally notifiable disease [3]. In 2010, over 1.3 million cases were reported; four-times more chlamydia cases were reported than gonorrhea cases, the next most frequently reported notifiable disease [4]. Chlamydia disproportionately affects racial/ethnic minorities [5].
Chlamydia, a sexually transmitted infection (STI), may lead to serious adverse outcomes among women, including pelvic inflammatory disease (PID) and related sequelae such as ectopic pregnancy, tubal-factor infertility and chronic pelvic pain. Among men, chlamydia may result in urethritis, prostatitis and epididymitis. The frequency of occurrence, asymptomatic nature of infection and the possibility of adverse outcomes prompted the development of widespread screening recommendations for women in 1993 by the Centers for Disease Control and Prevention (CDC). Currently, CDC recommends annual chlamydia screening of all sexually active women aged 25 years or younger [6]. Similarly, the US Preventive Services Task Force (USPSTF) recommends the screening of sexually active women aged 24 years or younger [7]. Pregnant women with chlamydia can transmit the infection to their newborns; thus, chlamydia screening during pregnancy is also recommended [6,7].
Chlamydia screening has been recognized by the National Commission on Prevention Priorities as one of the most beneficial and cost-effective preventive services among those recommended by USPSTF or the Advisory Committee on Immunization Practices, based on a standardized analysis evaluating and comparing these services [8]. The National Commission on Prevention Priorities also identified chlamydia screening as one of the most underutilized preventive services. Primary prevention strategies applicable to the prevention of all STIs include promoting consistent, correct condom use and increasing access to condoms; encouraging use of dual birth control methods; comprehensive sex education; and other general strategies focusing on a healthy sex life. Chlamydia screening, partner services (notification, testing and/or treatment of partners of infected individuals), and rescreening of previously infected individuals are all effective, recommended secondary prevention strategies [6].
In the era of healthcare reform in the USA, there is an excellent opportunity to improve chlamydia control. Implementation of the Patient Protection and Affordable Care Act (ACA) promises continuous insurance coverage for many Americans. Furthermore, the ACA reinforces the concept of the patient-centered medical home, where clinical and prevention services are coordinated to optimize primary care and reproductive health outcomes. Improving access to and reinforcing family planning clinical services within the broader context of preconception care is consistent with ACA goals. Importantly, chlamydia screening will be a fully covered preventive service, as will other preventive reproductive health services such as screening for pregnancy intentions, provision of contraception and referral to/coordination of primary care services during interconception periods [9]. Although the challenges to implementing such integrated services across the USA's fragmented healthcare system seem vast, there are lessons to be learned from other publicly funded systems of care, such as family planning clinics.
In the USA, family planning clinics are major primary providers of reproductive healthcare to adolescent and young women and their male partners, merging STI prevention with unplanned pregnancy prevention. Most family planning clinics in the USA are at least partially publicly funded as part of the federally funded Title X program created over 40 years ago to “provide individuals with comprehensive family planning and related preventive health services” [101]. At least one Title X-participating family planning clinic is located in approximately three-quarters of all US counties (>4500 community-based clinics). Although provision of family planning services is their primary function, family planning clinics have been on the forefront of chlamydia prevention for more than two decades. In addition to promoting primary prevention strategies, family planning clinics have provided high-quality secondary chlamydia prevention services including screening and treatment, rescreening and partner management [10].
Early efforts to address chlamydia in US family planning clinics
From 1959, when C. trachomatis was first isolated from the genital tract [11], until the early 1980s, chlamydia diagnoses were made on the basis of a culture test, whose optimal performance was based on narrow specimen transport and storage conditions. Early prevalence studies based on culture, which were conducted in family planning populations, consistently showed that prevalence was highest among young women [12]. The introduction of nonculture tests, such as the enzyme immunoassay and nucleic acid hybridization tests enabled further expansion of screening [13]. In 1988, a pilot project assessing the impact of a large-scale chlamydia screening and treatment program in family planning clinics was started in US Public Health Service Region X (Alaska, Idaho, Oregon and Washington). This project recruited young women for screening who were enrolled in a large health plan (similar to a registry-based approach). At the beginning of the campaign, in 1988, chlamydia positivity among women aged 15–24 years who were attending 150 participating family planning clinics was 11.1% (

Creation of the infertility prevention project
In 1992, based on the findings of the 1988 chlamydia screening pilot, the Infertility Prevention Project (IPP) was created through the passage of H.R. 3635, ‘Preventive Health Amendments of 1992’ [15] with the goal to reduce infertility through screening and treating young, sexually active women for chlamydia and gonorrhea. IPP was ultimately expanded in 1993 and 1995 to include all ten US Public Health Service regions, administered through collaboration between CDC and the Office of Population Affairs. IPP was also expanded to include other sexual health partners, most notably sexually transmitted disease (STD) clinics.
Each of the ten regional IPPs worked closely with federal, state and local partners (family planning and STD programs, public health laboratories and other key stakeholders) to disseminate scientific and programmatic data and provide technical assistance through regional advisory committees. These committees provided leadership in the prevention of chlamydia and gonorrhea infection and their sequelae through application of evidence-based prevention strategies and support of innovative research advancing the field of program science [16]. IPP successfully implemented sound practices, encouraged use of performance measurement throughout implementation and served as a model for forming collaborative partnerships across diverse groups. Such models will be of increasing importance in the future, as the emphasis on using evidence-based practices, creating sustainable systems and providing continuity of patient care grows in the context of healthcare reform in the USA.
Trends in chlamydia screening & treatment
Screening young women for chlamydial infections has been the cornerstone of national chlamydia control strategies for decades, and family planning clinics have been leaders in increasing the proportion of young women screened. In 1999, the National Committee for Quality Assurance added chlamydia screening coverage among women enrolled in private health plans and Medicaid as a measure in the Healthcare Effectiveness Data and Information Set (HEDIS) [17]. In 2010, the emphasis was further increased when the National Committee for Quality Assurance added the chlamydia screening measure to the accreditation dataset [18], which is used to accredit health plans [19]. The development of HEDIS measures has not only acted as a structural level intervention to improve access to chlamydia screening, but has provided critical data on the level of screening coverage in managed care settings. In public settings, chlamydia screening coverage in family planning clinics has been monitored since 2005, as part of the Family Planning Annual Report (FPAR). FPAR submission is required of all Title X family planning clinics.
Chlamydia screening coverage in managed care, as measured by HEDIS, has increased steadily over time. From 2001–2010, screening coverage among healthcare-seeking sexually active young women aged 16–24 years enrolled in a commercial health plan increased substantially from 23.1 to 43.1% [20]. However, many experts have suggested that HEDIS overestimates the proportion of eligible women screened. Women who do not seek care in the measurement year and women who are not determined to be sexually active (but may have had a visit for another reason, such as an acute illness) are not included in the denominator. Another estimate puts screening coverage at 34.2% for 3.2 million women aged 15–25 years who were enrolled in a commercial healthcare plan and had a reproductive health visit in 2008 [21]. By contrast, 50% of the 2.8 million women aged <25 years seen in the Title X family planning system were screened for chlamydia in 2005 [22]. By 2010, FPAR data showed that 57% of women aged <25 years were screened [23].
A critical component to chlamydia screening programs is the availability and provision of treatment. Chlamydia is easily treated with a single dose (1 g) of azithromycin. In the first half of 2011, IPP family planning clinics reported treating 73.0% of individuals with chlamydia within 14 days of diagnosis, increasing to 85.8% within 30 days [24]. These treatment rates are similar to those reported by STD clinics (73.3% within 14 days and 83.7% within 30 days).
Lessons learned #1: increasing chlamydia screening & treatment among adolescents & young women in family planning settings
Maximizing adherence to national screening recommendations is a key strategy to identify and treat asymptomatic chlamydial infections. Family planning settings present opportunities for identifying young women at risk for chlamydia during routine and urgent care visits related to contraception and other reproductive healthcare concerns. As noted above, despite increases in screening among family planning clients, coverage continues to be suboptimal. As in other clinical settings, several provider barriers have been associated with lower rates of recommended chlamydia screening, including awareness of screening recommendations, capacity to collect appropriate specimens for available diagnostic tests, lack of capacity and time to conduct sexual risk assessments, and lack of information on provider-specific screening performance.
Early interventions to address clinic work flow barriers showed that linking chlamydia testing to Pap testing by sequentially collecting the cervical specimens needed for each test during a pelvic examination was associated with increases in chlamydia screening [25]. Given the volume of Pap testing conducted in family planning clinics and the population served, family planning clinics rapidly adopted the practice of pairing the two tests, thus mitigating the need for the provider to be aware of chlamydia screening recommendations and specifically order the test.
While pairing the Pap and chlamydia tests was effective, chlamydia test technology has substantially changed over time and testing is no longer limited to specimen collection during a pelvic examination. The current optimal test technology utilized to detect chlamydia infections is a nucleic acid amplification test (NAAT) [26], with an estimated sensitivity of greater than 95% and specificity levels of approximately 99%. In 2000, 24.5% of all chlamydia tests conducted in surveyed public health laboratories in the USA were NAATs [27]; by 2007, this proportion had increased to 81.6% [28]. NAATs can now be performed using several types of specimens: cervical, urine, and self- and clinician-collected vaginal specimens. Increases in the proportion of chlamydia tests conducted with NAATs are likely related to the ability to test with urine [28,29]. With the development of NAATs that could be performed on noninvasive specimens, the capacity to increase screening has significantly grown, especially during family planning visits where pelvic examinations are not necessary [30,31].
In an effort to further engage family planning providers in improving adherence to screening guidelines as part of continuous quality improvement, ongoing provider-specific feedback has been used to monitor baseline performance and changes over time in the quality of care delivered in family planning settings. In California's Family Planning, Access, Care, Treatment (PACT) program, providers were sent information on individual screening rates with comparison to average program performance and an informational letter based on level of adherence to screening guidelines. Providers who screened <50% of young family planning female clients significantly increased screening rates compared with providers with higher baseline screening rates [32]. As a result, this program was expanded to provide semi-annual provider-specific data feedback on this and other clinical quality measures (Provider Profiles) [33] and ongoing increases in chlamydia screening since initiation have been observed.
The most effective interventions to improve screening rates should systematically address as many provider barriers as possible. One such holistic approach is based on rapid cycle continuous quality improvement or ‘Plan-Do-Study-Act’ in which the key aspects include engagement of providers, team-building, redesign of clinical practice and sustaining the gain. This systems approach facilitated chlamydia screening through streamlining clinic protocols for risk assessment and universal urine collection and maintained ongoing data feedback within clinic teams for further changes in clinic protocols, resulting in significant increases in chlamydia screening among providers serving adolescents in managed care settings [34]. Within family planning settings, a similar approach has been used in the Family PACT program to coordinate program- and provider-level interventions, including development of standardized sexual risk assessment forms, provider-level data feedback (Provider Profiles) and technical assistance through webinars, and clinical practice alerts to reinforce screening recommendations for young women and maintain consistently high screening rates that exceed those reported for managed care plans [35].
Lessons learned #2: reducing over-screening of women aged 26 years & older
In this era of cost-effective interventions, family planning clinics have worked actively to reduce unnecessary chlamydia screening. National chlamydia prevalence monitoring data have consistently shown that positivity among young females tested in family planning settings is above the 3% threshold for cost-effective screening [4,36]. Both CDC and USPSTF recommend chlamydia screening for older women if they have risk factors, but these risk factors are ill-defined [7,14]. Screening coverage data from large family planning programs show that a substantial proportion of older women aged >25 years are routinely tested for chlamydia [23,35]. Analyses of selective screening among older women with sexual risk behaviors indicate that targeted screening can increase case-finding yield while simultaneously screening fewer women [37,38]. Given the poor compliance with the collection of comprehensive sexual histories and risk behavior, it may be difficult to operationalize highly specific screening guidelines for women over 25 years of age without dramatically reducing the cost–effectiveness of screening activities.
One strategy that has been examined in family planning settings is targeted chlamydia screening for only specific established risk factors. Among women aged 25–29 years who were attending STD clinics in Region X, chlamydia exposure, a new sex partner and one or more clinical findings were associated with a positive chlamydia test [39]. Another strong predictor identified using data from women tested for chlamydia in California and Region IX family planning clinics was a partner concurrency measure assessing whether a client felt her primary partner was likely to have another concurrent partner [37,40].
Another strategy that reduced over-screening of older women in family planning settings was one in which tests were not paid for unless certain risk factors were marked on the laboratory requisition form. The San Francisco STD Program developed an intervention in Title X-funded family planning clinics whereby chlamydia test laboratory requisitions for these clients were required to have a supporting clinical or behavioral reason for testing; otherwise, specimens were not tested. Analysis of pre- and post-implementation of the conditional testing policy was associated with a 24% reduction in chlamydia test volume and a nonsignificant change in test positivity [41]. Similar laboratory- and billing-based approaches to limiting testing among women aged ≥25 years by managing test kit allotments also demonstrated reductions in testing while shifting resources to higher priority young females [42,43].
Ongoing efforts to implement cost-efficient practices in screening older female clients can ultimately strengthen family planning program resources through shifting testing resources to young women with higher chlamydia prevalence. A recent analysis demonstrated that reallocating resources in family planning settings to the youngest group of women among whom routine chlamydia screening is recommended, adolescents aged 10–19 years, could improve case-finding by 33% [44].
Measuring the impact of screening on disease trends
Increasing chlamydia screening coverage has likely had a substantial impact on trends in reported cases, since as more women are screened, more prevalent cases are detected (
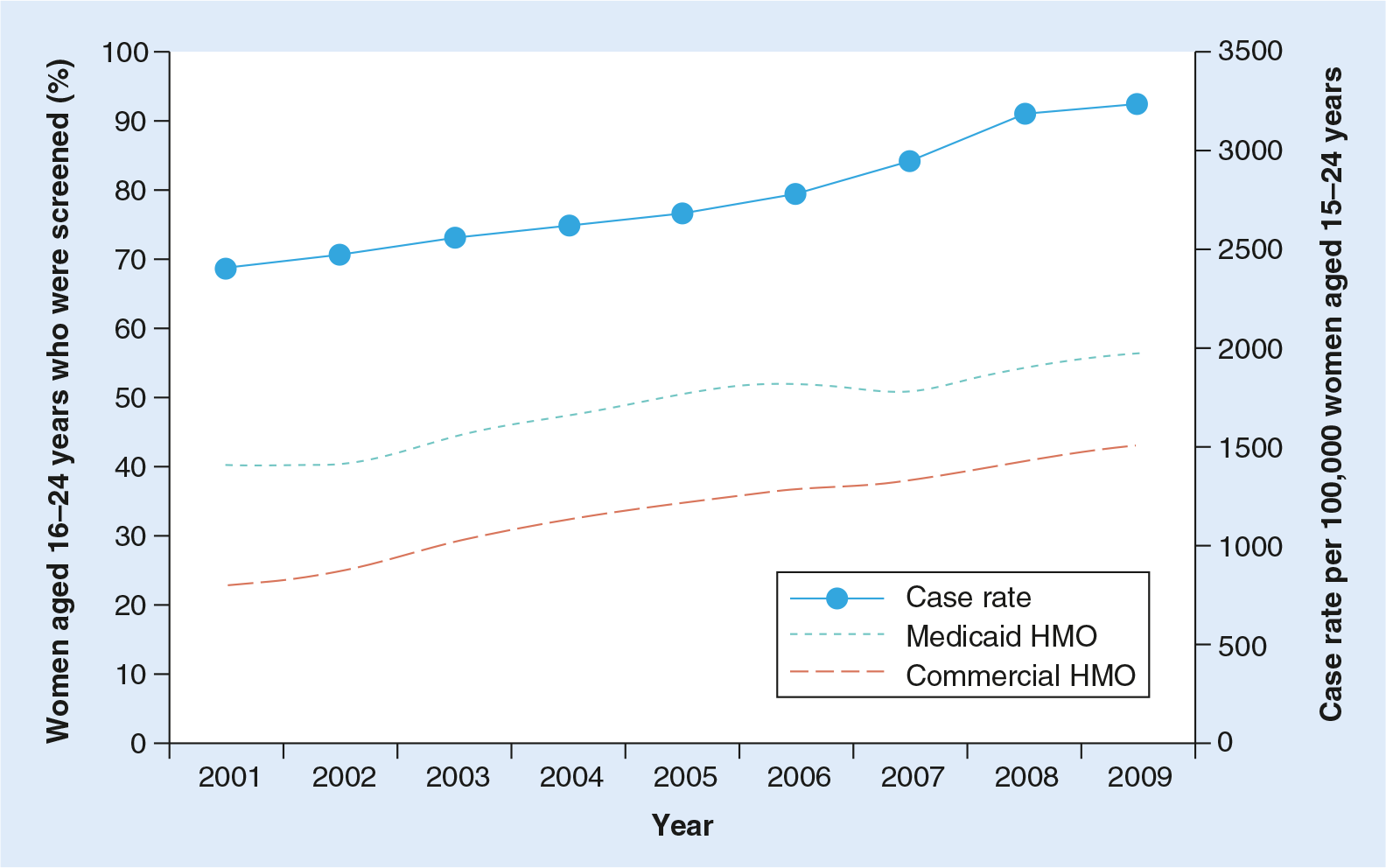
More comprehensive trend analyses show that while chlamydia prevalence is not increasing in the USA, prevalence is not conclusively decreasing either. Based on nationally-representative data from the National Health and Nutrition Examination Survey (NHANES), chlamydia prevalence among the general population of US men and women aged 14–39 years significantly decreased from 1999 to 2008 [5]. However, prevalence among young women aged 15–24 years, the group targeted by screening efforts, did not significantly decline. Additionally, chlamydia positivity among women aged 15–24 years who attended family planning clinics remained unchanged from 2004 to 2008 [45]. Encouragingly, among women aged 15–24 years tested in prenatal clinics, in whom testing is routinely recommended and thus less likely to be influenced by perceived STD risk, positivity did decline from 2004–2009 [46].
Despite increases, chlamydia screening remains suboptimal and there has been no apparent decline in chlamydia prevalence in the population targeted for screening. However, ecologic analyses of PID incidence do show decreasing trends, which may suggest an impact from expanded chlamydia screening efforts [47,48]. Mathematical modeling has suggested that a shift in the emphasis and mix of prevention strategies employed may be more effective in further decreasing chlamydia burden and adverse sequelae [49]. In particular, at the current level of chlamydia screening, increasing the proportion of partners treated may have a collective synergistic impact on disease transmission and reduce prevalence more than increasing screening coverage alone.
Lessons learned #3: chlamydia partner notification & treatment
Repeat infections, which increase the risk of complications, occur in nearly 15% of women and men within 6 months after treatment [50]. To prevent repeat infections, reduce complications in individuals and reduce further transmission of infections in the community, sex partners of chlamydia cases must be provided timely and appropriate antibiotic treatment. CDC recommends that all sexual contacts within the previous 60 days from the onset of symptoms or diagnostic test results be treated with appropriate antibiotics [6]. If the last sexual contact was over 2 months prior to the diagnosis, the most recent sex partner should be offered testing and treatment.
There are several strategies for notifying and managing partners in family planning settings. First-line strategies attempt to link partners with clinical services. The optimal management of sex partners involves a clinic visit that consists of a clinical evaluation, testing, treatment and referral if indicated. Clinic-based partner services provide the opportunity to observe treatment; confirm the diagnosis; examine the patient for other treatable conditions; test for other STIs and HIV; provide needed vaccinations; offer risk-reduction counseling and community referrals; and provide reproductive health services, such as pregnancy testing and contraception. These services constitute the standard of care for all partners of chlamydia cases.
Partners need to be notified of their exposure in order to seek care. Notification can be accomplished by the patient, the medical provider or the health department. Although medical providers have the option to collect the partners' contact information and notify them, there are no reimbursement mechanisms and few clinics have the resources for this activity. Furthermore, while public health efforts to notify and treat sex partners have proven successful and are considered a cornerstone of syphilis control [51], the high burden of chlamydia and limited public health resources for partner notification activities make partner notification for cases of chlamydia a low priority [52].
Thus, the default strategy for partner management for chlamydia cases has become patient referral, whereby providers counsel patients about the need for partner treatment, and the responsibility for notifying partners rests with the patient. The effectiveness of patient referral is dependent on the patient's motivation and ability to notify the partner(s), as well as the partner's motivation and ability to obtain treatment.
Lack of health insurance and limited access to medical care limit the effectiveness of partner referral. In California, males are eligible for enrollment in Family PACT, which has greatly facilitated access to STD clinical services for partners exposed to chlamydia. As of 2010, over 250,000 males were enrolled in Family PACT [35]. Yu et al. evaluated data from eight California family planning clinics that participated in a partner services evaluation from 2005–2006 [53]. Over 700 young women with chlamydia were interviewed to determine the partner service received and partner treatment outcomes. Only approximately 15% were advised to bring their partners to clinic when they returned for treatment (so-called ‘bring your own partner’ [BYOP] method); the majority of those encouraged to BYOP (nearly 80%) agreed and were successful. This approach was particularly successful for patients in committed, long-term relationships. Some studies have found that the effectiveness of traditional patient referral can be improved with better counseling and by providing printed material (booklet-enhanced patient referral) [54]. More research is needed to assess innovative strategies for linking partners to STI care, especially those in casual relationships.
When partners are unable or unwilling to seek STI services on their own, clinicians have alternative means of providing chlamydia treatment without a clinical examination. Expedited partner therapy (EPT) is the general term for the practice of treating sex partners of patients diagnosed with an STI without requiring an intervening medical evaluation [55]. Patient-delivered partner therapy is the most common type of EPT, in which the patient delivers the medication or a prescription directly to his or her sex partner(s). Other types of EPT involve alternative delivery mechanisms, such as direct provision through a pharmacy under clinical protocol [56]. Field-delivered therapy is another form of EPT in which trained public health field investigators deliver single-dose, directly observed therapy to untreated patients and their partners with uncomplicated chlamydia infections.
Several research studies, including randomized clinical trials, have demonstrated that EPT is effective in facilitating partner notification and reducing recurrent infection among index cases. A recent meta-analysis that included five clinical trials showed an overall reduced risk of repeat infection in patients with chlamydia or gonorrhea who received EPT, compared with those who received standard partner treatment methods [57].
As of August 2011 (the most recent data available), EPT for chlamydia is permissible in 31 states in the USA and potentially allowable in an additional 12 states [102]. Previous surveys have found that the general practice of EPT for chlamydia is not uncommon [58–60]. More recently, a 2007 survey of California family planning providers found that routine use of EPT was common: 73% for chlamydia, 39% for gonorrhea and 56% for trichomoniasis [61]. Optimal care for contacts to STIs includes testing for other STIs and HIV, a physical examination to rule out a complicated infection, risk-reduction counseling and referral for other needed clinical services. Ideally, partners who receive EPT will still access these clinical services.
Lessons learned #4: rescreening among women with a previous chlamydial infection
Due to the high rates of repeat infection and the consequent increased risk of adverse reproductive health outcomes, CDC recommends that all persons diagnosed with chlamydia and gonorrhea be rescreened approximately 3 months after treatment [6]. Although this recommendation has been made for a number of years, compliance has been poor [62,63]. Several challenges may contribute to low rates of rescreening following a diagnosis of chlamydia, occurring at both the provider and patient level.
While most examinations of rescreening have focused on patient level barriers, important challenges at the provider level warrant discussion. A survey of family planning providers practicing in California found that providers are knowledgeable about rescreening recommendations; however, nearly 75% of survey respondents said that patients do not return for rescreening, and less than a quarter of these providers listed rescreening for chlamydia as a high priority at their clinic [64]. Despite this, over half of providers employed some sort of flag or reminder system in an effort to improve compliance with rescreening recommendations. Prompts built into electronic medical records (EMRs) are another effective method to increase provider screening [65,66].
Patient reminder systems have been employed with varying success in improving rescreening rates among females. While many studies have examined reminder systems in the context of general STI/reproductive health appointments [66–68], fewer published reports exist on the efficacy of these systems for rescreening. A cost–effectiveness analysis found that phone calls, while resource intensive, are a cost-efficient mechanism to encourage rescreening in an STD clinic setting [69]. While some observational evaluations have suggested that reminder systems increase rates of rescreening [70,71], others have shown no impact [72]. Although many of these interventions were examined in the context of an STD clinic, it is likely they can be translated into a family planning setting with relative ease.
In an effort to overcome barriers patients may have in returning to the clinic for rescreening, several groups have examined the efficacy and utility of using home-based testing (with self-collected specimens) to facilitate rescreening. In a home-testing model, patients diagnosed with chlamydia would be sent a self-collection kit (vaginal swab or urine collection kit, including instructions) as an alternative to returning to the clinic for rescreening. The specimen is then mailed to a lab. After testing is complete, results are communicated back to patients, and those found to be re-infected are asked to seek treatment at a clinical site. Xu et al. examined a home-testing intervention among females diagnosed in STD and family planning sites and found that rescreening rates were significantly improved for women randomized to home testing [73]. In a meta-analysis by Guy et al., women participating in home-based rescreening interventions were more likely to be rescreened when compared with women in other interventions or receiving standard-of-care [74].
While shown to be effective, home-based rescreening programs may be challenging to implement. Such programs require a significant amount of startup costs, including the validation of mailed specimens, development of a system for identifying patients needing a rescreening kit sent to them and development of a mechanism for returning test results (e.g., a web-based portal for returning results, letters or phone calls to the patient). Home-based rescreening programs are a relatively new concept and barriers to implementation, including reimbursement mechanisms, may be reduced in the future.
Future challenges & opportunities for chlamydia prevention in family planning clinics & beyond
Use of alternative specimens
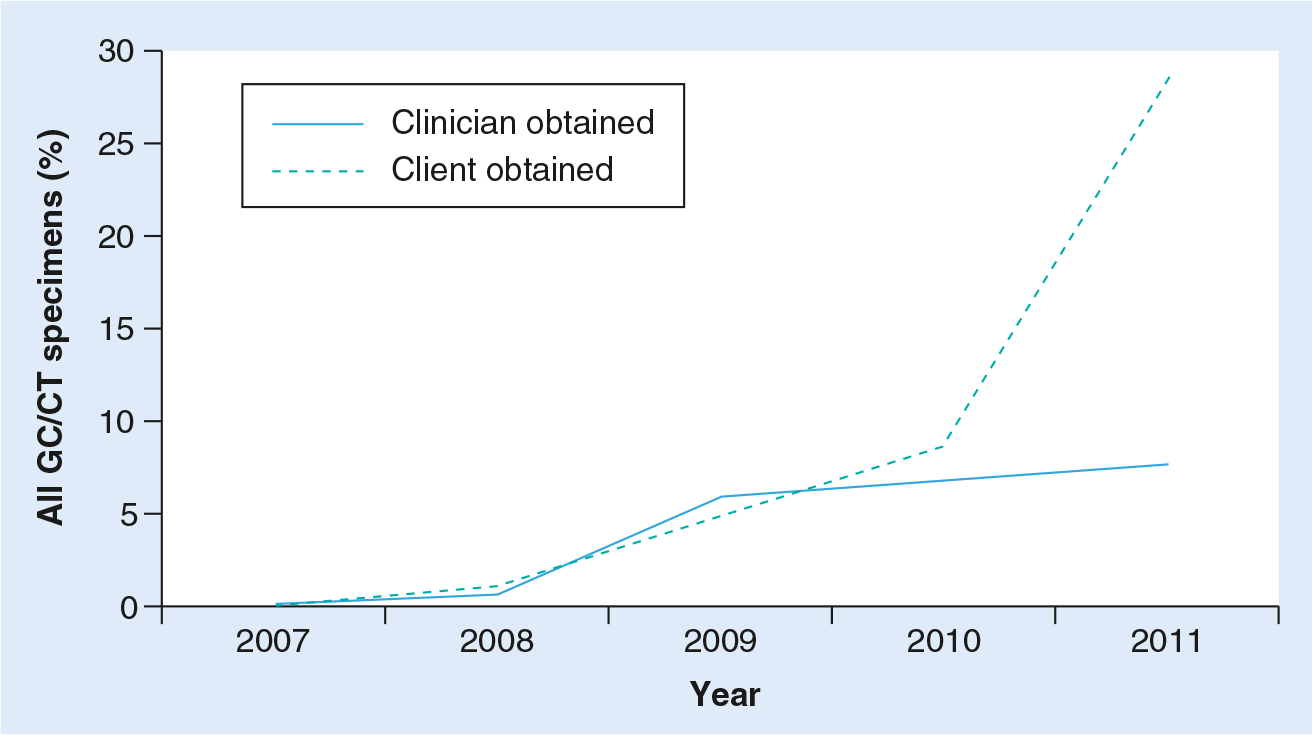
In a recent expert meeting, vaginal swabs were identified as the specimen of choice for performing chlamydia NAATs due to higher sensitivity than urine and easier collection than endocervical swabs [26]. Such endorsement should create additional opportunities for promoting the use of self-collected vaginal swabs. Many Regional IPPs have been encouraging the use of self-collected vaginal swabs. In Region X, there has been a dramatic increase in the uptake of vaginal swabs, particularly in family planning clinics. Among females in 2007, <1% of all specimens were self-collected vaginal swabs; by 2011, 28.5% of specimens collected (range: 11–36% by state) were self-collected vaginal swabs (
The availability of self-collected vaginal specimens has also given rise to newer interventions such as internet-based programs designed to improve access to screening by reducing barriers to chlamydia screening. In 2004, an internet-based program (‘I Want the Kit’) was created [76]. This website contains information about chlamydia as well as an option to request that a chlamydia test kit be mailed (women residing in the Baltimore, MD, USA area). Test results are made available after the mailed self-collected vaginal specimen swab is processed. In 2009, the Los Angeles County Department of Public Health launched the ‘Don't Think Know’ program, featuring a website where young women can order self-collected vaginal swab kits, find out their results and get information and, if necessary, referrals for treatment. The ‘I Want the Kit’ and ‘Don't Think Know’ websites have since been expanded to other geographic areas.
With advancements in test technology, some earlier effective interventions are being reconsidered. Initially, pairing a Pap test and a chlamydia test during a pelvic exam was an effective way to increase the proportion of eligible women screened for chlamydia. However, recent revised guidelines reducing the frequency of pelvic exams and Pap testing, as well as the age at which Pap testing is recommended, point to a need to restrategize this previously useful pairing. Instead of linking chlamydia testing to Pap tests and/or pelvic exams, providers should be encouraged to screen women using either urine or vaginal swabs. Associating chlamydia screening to contraception-related visits, which are not linked to pelvic exams, may also be beneficial. Future research should address linking chlamydia screening to visits where a pelvic exam is not performed.
Clinic protocols are especially needed to reduce missed opportunities for chlamydia screening among family planning clients who are being tested for pregnancy, receiving emergency contraception or receiving routine hormonal contraception, as the chlamydia risk is high in these populations [77]. The high chlamydia prevalence found among family planning clients who were seeking just a pregnancy test supports efforts to improve screening for this group in particular [78]. For instance, urine specimens already being taken for pregnancy testing could be used for both, or a self-collected vaginal swab could be obtained, thereby increasing opportunities to identify infection in patients also at risk for chlamydia.
Use of data to improve chlamydia care in family planning programs
Many family planning clinics have or are developing robust data systems that have been well-utilized to improve the delivery of care and to monitor the burden of disease in their population. The growing importance of data-driven program design and continuous quality improvement necessitate that providers of family planning services continue to collect, analyze and interpret their data, continuing to innovate practice and advance care provision in a changing healthcare environment. While family planning data collected through IPP have been used nationally, data usefulness is strongest at the local and state level. Family planning clinics should continue to demonstrate the needs of their population through thoughtful collection and presentation of these data. Such data have been and will continue to be important tools for patient and service advocacy.
Conclusion
Family planning clinics in the USA have demonstrated the development and implementation of best practices for chlamydia control, particularly around chlamydia screening and treatment, partner services and rescreening. As shown by these publicly funded clinics, cost-effective, integrated family planning and STI care can be accessible, high quality and continuously improved, providing medical homes for young women and their partners with primarily sexual and reproductive healthcare needs. While family planning clinics may not be the ideal single-setting medical home model, these clinics address many of the major clinical preventive services young women need and can provide referrals for and coordination with other needed primary care services. As healthcare reform rolls out, these services and more will be expected throughout primary care as part of a medical home for young women.
Challenges that will need to be addressed as these services are expanded further into primary care will include assurance of confidentiality and provision of sexual risk assessments, particularly with adolescents. Additionally, the development of systems facilitating reimbursement (through billing) for screening and treatment, as well as for EPT, will remain critical as the availability of public funds decreases and the insured population increases.
With further system integration and the application of health information technology for quality improvement, there will be an opportunity for better care coordination between providers and better outcome evaluation to demonstrate meaningful use of data. Holding providers and health systems accountable for the provision of required reproductive health services will be critical during this transition. Similarly, as the ACA has placed emphasis on effective interventions, the capture of better process and outcome metrics, particularly around PID and infertility, is needed.
As national healthcare reform is implemented and categorical STI clinics face closure, public sector demand on family planning clinics will likely increase. Maintaining these clinics as safety net clinics and expert providers of reproductive healthcare is critical, particularly as family planning clinics continue to be leaders in implementing evidence-based care delivery and developing prevention innovations.
Future perspective
Within the next decade, innovations to reduce the burden of chlamydia infections in young people should focus on expanding the implementation of evidence-based prevention strategies.
Primary prevention interventions
The normalization and acceptance of healthy sexual development could have enormous impacts on reducing teen pregnancy and STI acquisition, as has been seen in other developed countries [79]. Other advances in primary prevention should include the development of novel education strategies that use technology to better educate young people and allow for personalized risk assessments and even screening and referral tools [80]. Increasing condom access and, more importantly, the normalization of condom use (including readily available instruction on correct usage) should be emphasized. A more consistent and committed focus on comprehensive sexual health education may also have a wide-reaching impact on awareness and utilization of both pregnancy and STI prevention services.
Improving screening & treatment of young women
Every sexually active woman aged 25 years or younger should be screened for chlamydia annually, particularly as emphasis is increasingly shifted to prevention of illness rather than treatment of sequelae. To do this, every avenue should be explored including pay-for-performance on screening coverage metrics, creating consumer awareness and demand for screening services, and the expansion of more online or over-the-counter screening options. In addition, the development of sensitive and specific point-of-care tests for chlamydia will greatly improve diagnosis and treatment rates, particularly among those at highest risk for lack of follow-up. Expanding screening and treatment services in school-based health centers will also improve screening rates in adolescents. Health information technology should be better harnessed for patients and providers, providing electronic reminders for both groups.
Screening high-risk men
While routine screening of men has not been historically emphasized, the role of screening high-risk men, such as those in adolescent clinics, correctional facilities or STD clinics, is recognized as an important chlamydia prevention strategy [6]. The primary screening emphasis should still be on increasing screening among eligible women who are already seeking care; however, if high-risk men are already seeking care, screening this population should be considered, if resources permit. There are two main barriers to the wide endorsement of male chlamydia screening. The first is demonstrating the cost–effectiveness of male screening on preventing female sequelae. This will remain challenging; however, the role of chlamydia transmission between sex partners is clear. The second barrier surrounds care-seeking behaviors of men. Men seek care less frequently than women, so men are more challenging to reach. Therefore, targeting high-risk men already seeking care is an easy, straightforward approach to reducing the burden in this population.
Partner services
The majority of women diagnosed with chlamydia report only one partner. Partner services to interrupt transmission must be prioritized in the clinical setting. From a provider perspective, ensuring that their patient's partner(s) are treated is important to preserving the individual health of their patient by preventing a repeat infection and any subsequent sequelae. From a public health perspective, partner treatment can reduce transmission within core groups and communities. Family planning clinics need to take the lead in pushing partner treatment mechanisms by prescribing medication and ensuring access or providing patient-delivered partner therapy, changing the culture and stigma surrounding STI management, improving communication between partners, improving access to care for partners (especially males) and identifying mechanisms to ensure funding for partner treatment through billing and policy changes.
Retesting previously infected individuals
Even when partner treatment is successful, efforts must be made to encourage previously infected individuals to be retested approximately 3 months after treatment. Family planning clinics should adopt evidence-based or promising practices to promote retesting, such as the use of advanced health information technology that facilitates reminder systems and use home-testing kits that can be mailed to the individual without requiring a clinic visit. Identifying innovative reimbursement mechanisms for such will enhance efforts to identify repeat infections.
Financial & competing interests disclosure
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
Executive summary
Almost 3 million women aged 15–25 years attend a public family planning clinic annually.
Family planning clinics have been at the forefront of chlamydia prevention for more than two decades.
Largely asymptomatic, chlamydia may lead to serious adverse sequelae, including infertility.
An estimated 2.8 million chlamydia infections occur annually in the USA.
Annual screening of sexually active women aged <24 years is recommended; however, screening coverage is suboptimal.
Changes include an increase in persons with insurance coverage and an expanded scope of benefits, under which chlamydia screening is a fully covered preventive service.
Healthcare reform places an emphasis on the patient-centered medical home.
The Infertility Prevention Project, created in 1992, has been a model partnership, advancing sexual health through the national expansion of chlamydia screening and treatment programs.
Screening coverage has increased over time, but remains low (43.1% in commercial health maintenance organization health plans); coverage in Title X-funded family planning clinics is higher (57%).
Increases in reported chlamydia case rates are likely due to better detection through improvements in screening rates and test technology.
By contrast, prevalence studies suggest trends are flat or decreasing.
Provider and patient barriers have been addressed through electronic reminder systems and education, and providers have been encouraged to increase screening through the comparison of provider-specific screening rates to peer screening rates.
Use of nucleic acid amplification tests as the optimal test technology has increased, especially with alternative specimens, such as urine and vaginal swab specimens.
Over-screening among women aged >25 years has been reduced using payment-structured laboratory-based interventions and risk assessment questions with higher predictive value for Chlamydia.
Repeat infections are common.
Family planning clinics have implemented a number of strategies to increase partner treatment, including promoting a strategy where the patient brings their own partner in for treatment at the time they receive their treatment, and expanded the use of expedited partner therapy.
Patient reminders and novel rescreening options, such as home-based specimen collection, can increase rescreening rates.
Family planning clinics should increase the use of noncervical specimens, explore alternative methods for screening and rescreening (such as internet-based programs) and work to increase screening coverage, with particular attention given to high-risk groups, such as women seeking a pregnancy test.
Data should remain a cornerstone of local decision-making and drive quality improvement efforts.
Family planning clinics provide cost-effective, integrated family planning and sexually transmitted infection care that is high-quality and continuously improved, and can fill the medical home role for young women and their sex partners.
Family planning clinics are well-positioned to maximize the opportunities presented in an era of healthcare reform and must continue to leverage reimbursement mechanisms for sustaining services.
Family planning clinics will continue to focus on improving chlamydia screening and treatment, partner treatment and rescreening, while expanding screening to high-risk men to broaden impact on population-level chlamydia disease burden.
