Abstract
Many important issues regarding HRT and the management of postmenopausal women are unresolved and require ongoing consideration and some explanation. HRT is only one of many factors affecting the risk of breast cancer and the risk of breast cancer is not increased in past users of HRT. There is seemingly a paradox in the effect of body weight and HRT on breast cancer risk. It is unclear whether the benefit of giving progestin with estrogen to prevent endometrial cancer in HRT outweighs the increased risk of breast cancer. Sequential HRT may not fully prevent endometrial cancer, and continuous combined regimes are to be preferred. The prevention of the consequences of premature ovarian insuffiency, both immediate and long term, has been neglected, and a strong case can be made for estrogen replacement therapy in all women with premature ovarian insuffiency, particularly following bilateral oophorectomy.
There are a number of important unresolved issues regarding the benefits and risk of HRT and the management of postmenopausal women that require ongoing consideration and some explanation. The unresolved issues include breast cancer, endometrial cancer and the management of women with premature ovarian insufficiency (POI).
Breast cancer
HRT is only one of many factors affecting the risk of breast cancer
In the Nurses' Health Study (NHS) the three main factors affecting the relative risk (RR) of breast cancer, namely family history of breast cancer, history of benign breast disease and HRT (assumed to be used continuously for 10 years from the age of 50 to 60 years) were of equal importance (
Main risk factors for breast cancer from the Nurses Health Study 1970–1994 with cumulative incidence and relative risk of breast cancer women aged 30–70 years.
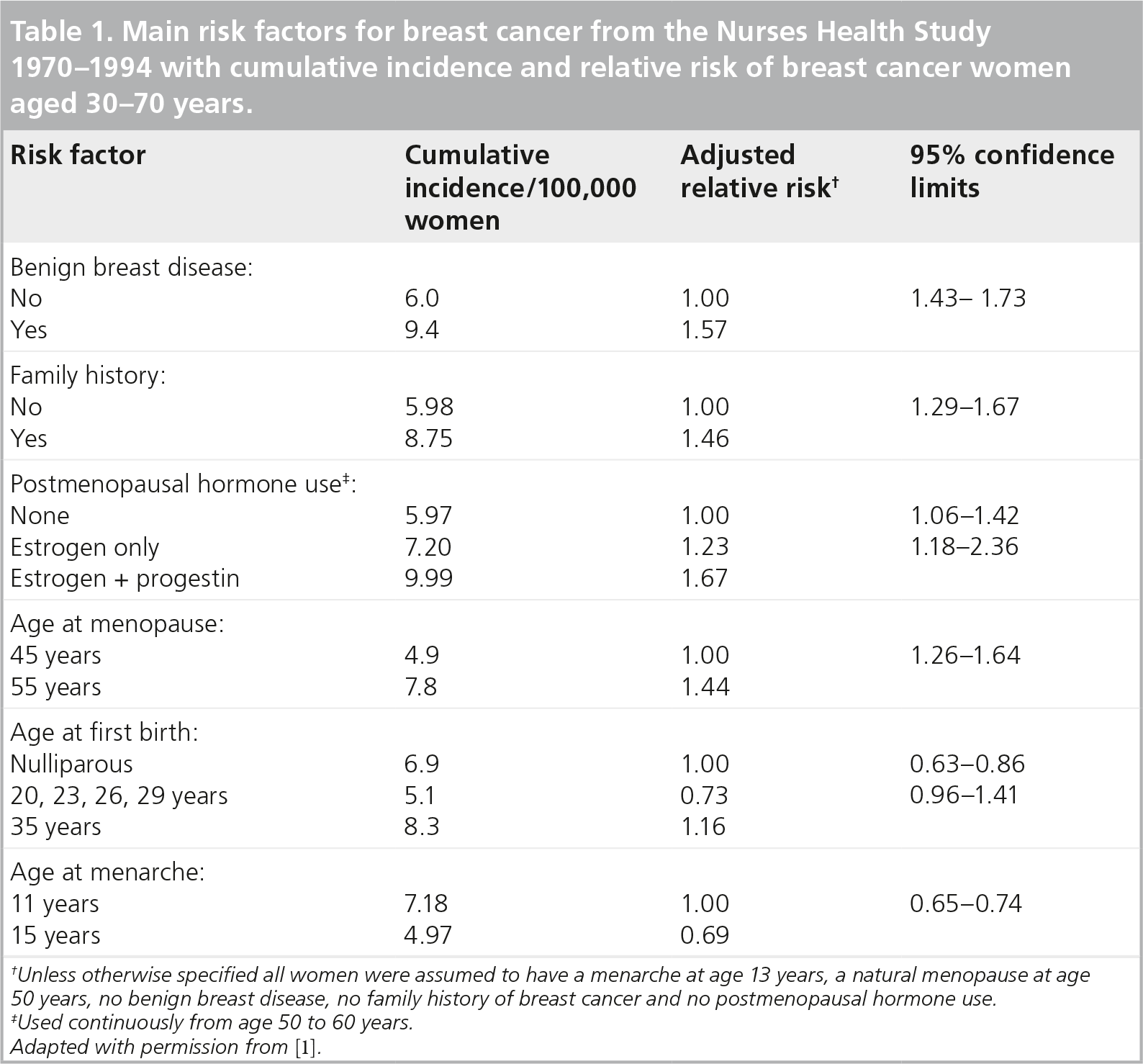
Unless otherwise specified all women were assumed to have a menarche at age 13 years, a natural menopause at age 50 years, no benign breast disease, no family history of breast cancer and no postmenopausal hormone use.
Used continuously from age 50 to 60 years.
Adapted with permission from [1].
The nature of HRT also influences breast cancer risk including: the composition of HRT (estrogen plus progestin carries more risk than estrogen alone); duration of use (the longer the exposure the greater the risk); the interval between menopause and starting HRT (the longer the interval the lower the risk); and BMI (the greater the BMI the less the effect of HRT) [2]. The risk of breast cancer may be significantly less with micronized progesterone than with synthetic progestins [3–5]. In the lay literature and in counseling women on the risks and benefits of HRT it is often assumed, at least tacitly, that HRT is the only factor affecting breast cancer risk. The risk in each woman should be put into perspective and it needs to be stressed that HRT is only one of several factors influencing the risk of breast cancer.
The risk of breast cancer is not increased in past users of HRT
The risk of breast cancer is increased with current use of estrogen plus progestin HRT but is not increased in past users of HRT [6,7]. In the Women's Health Initiative (WHI) the incidence of breast cancer was increased in the women who currently used estrogen plus progestin HRT but the incidence progressively decreased after stopping HRT. In the WHI, the incidence of breast cancer was reduced in women with estrogen-only HRT compared with placebo and the incidence remained low for 10.7 years after stopping the HRT [8,9]. In subgroup analyses of the effect of prior hormone therapy on the incidence of invasive breast cancer in the estrogen plus progestin arm of the WHI, the incidence of breast cancer in the placebo arm was significantly decreased in women with any prior use compared with those with no prior use [10]. In the NHS, the multivariate adjusted risk of breast cancer was RR: 1.44 (95% confidence limits [CL]: 0.99–2.08) in women who stopped taking HRT less than 2 years earlier, RR: 0.80 (95% CL: 0.55–1.06) in those who stopped 2–4 years earlier and RR: 0.95 (95% CL: 0.74–1.25) in those >5 years after stopping HRT. The RR of dying from breast cancer, adjusted for family history and history of benign breast disease, was 1.14 (95% CL: 0.85–1.51) in current users and 0.80 (95% CL: 0.60–1.07) in women who had used HRT at any time in the past [11].
These findings suggest that there must be a ‘compensatory’ decrease in the risk of breast cancer for a period after stopping HRT. In an autopsy study of women who died accidentally, small occult neoplastic breast tumors were found in 39% of women aged 40–50 years [12]. It has been hypothesized that a small but increasing proportion of these occult cancers become invasive over time and that the administration of estrogen and progestin in postmenopausal women accelerates the growth of pre-existing occult tumors but does not initiate malignant change. After a period of estrogen and progestin hormone replacement therapy (EPHRT) this may leave a relative deficit of occult tumors sensitive to estrogen and progestin. The observed decrease in the incidence of breast cancer for a period after stopping HRT could be a direct consequence of the increased incidence during HRT. It is suggested that the decrease in the number of tumors after stopping EPHRT balances out the increase in the number of occult tumors that become invasive during EPHRT. The total combined number of occult tumors becoming invasive both during and after stopping EPHRT would then not be increased and there would be no long-term increase in the risk of breast cancer in past users of EPHRT compared with never users.
Effect of body weight & HRT on breast cancer risk: a paradox
The effect of body weight and of HRT on the risk of breast cancer is sometimes misunderstood. There is a seeming paradox that the risk of breast cancer increases with increasing body weight but the risk is not increased further by HRT in overweight and obese women. Most studies have found that the incidence of breast cancer increases with increasing body weight and BMI. In the collaborative analysis of 51 studies, the incidence of breast cancer in postmenopausal women not receiving HRT was increased by 3.1% per each kg/m2 increase of BMI [6]. The RR of breast cancer was RR: 1.52 (SE: 0.083) in women with a BMI of <25.0 kg/m2 and RR: 1.02 (SE: 0.103) in women with a BMI of >25.0 kg/m2 in women who had used HRT for 5 years or more and who last used HRT less than 5 years previously. In the Million Women Study, the risk of breast cancer in current users of estrogen plus progestin compared with nonusers was RR: 2.5 (95% CL: 2.12–2.53) in women with a BMI of <25.0 kg/m2 and RR: 1.78 (95% CL: 1.64–1.96) in women with a BMI of >25.0 kg/m2 [7]. The increased risk of breast cancer associated with HRT was significantly greater in ‘thin’ women (BMI: <25.0 kg/m2) than in ‘overweight and obese’ women (BMI: >25.0 kg/m2). It is noteworthy that in the WHI over 80% of the women were already overweight or obese and no association was found between BMI and the risk of breast cancer [13,14]. In overweight and obese women the risk of breast cancer is significantly increased but the risk is not increased further by HRT.
Overweight and obese women have higher levels of circulating estrogens than thin women and it has been proposed that the increased risk of breast cancer is a result of the increased endogenous estrogens [15]. There appears, however, to be an upper limit at which the risk of breast cancer is not increased further by either increased endogenous or exogenous estrogens. It is suggested that there is a limit to the rate at which occult neoplastic breast tumors become invasive and a ‘ceiling’ above which no further increase in the incidence of breast cancer occurs with increased levels of circulating sex hormones. Overweight and obese women should be counseled that although the risk of breast cancer may not be increased further by HRT they are at increased risk and require appropriate surveillance.
Does the benefit of giving progestin with estrogen to prevent endometrial cancer in HRT outweigh the increased risk of breast cancer?
This is still an open issue. In the Million Women Study of breast cancer and hormone replacement therapy, the cumulative incidence of both breast and endometrial cancer in 1000 women between the age of 50 and 65 years was estimated in three groups of women: those who had never used HRT; those who had used estrogen-only HRT; and those who had used estrogen–progestin HRT [7]. With estrogen–progestin HRT the estimated increased risk of breast cancer was three times greater than with estrogen-only HRT. The estimated excess number of cases of breast cancer was 19 per 1000 women and five per 1000 women respectively for 10 years use of estrogen–progestin and estrogen-only HRT compared with never users. Breast cancer is much more common than endometrial cancer and with estrogen-only HRT, the estimated increased incidence of breast cancer was much greater than the increase incidence of endometrial cancer (
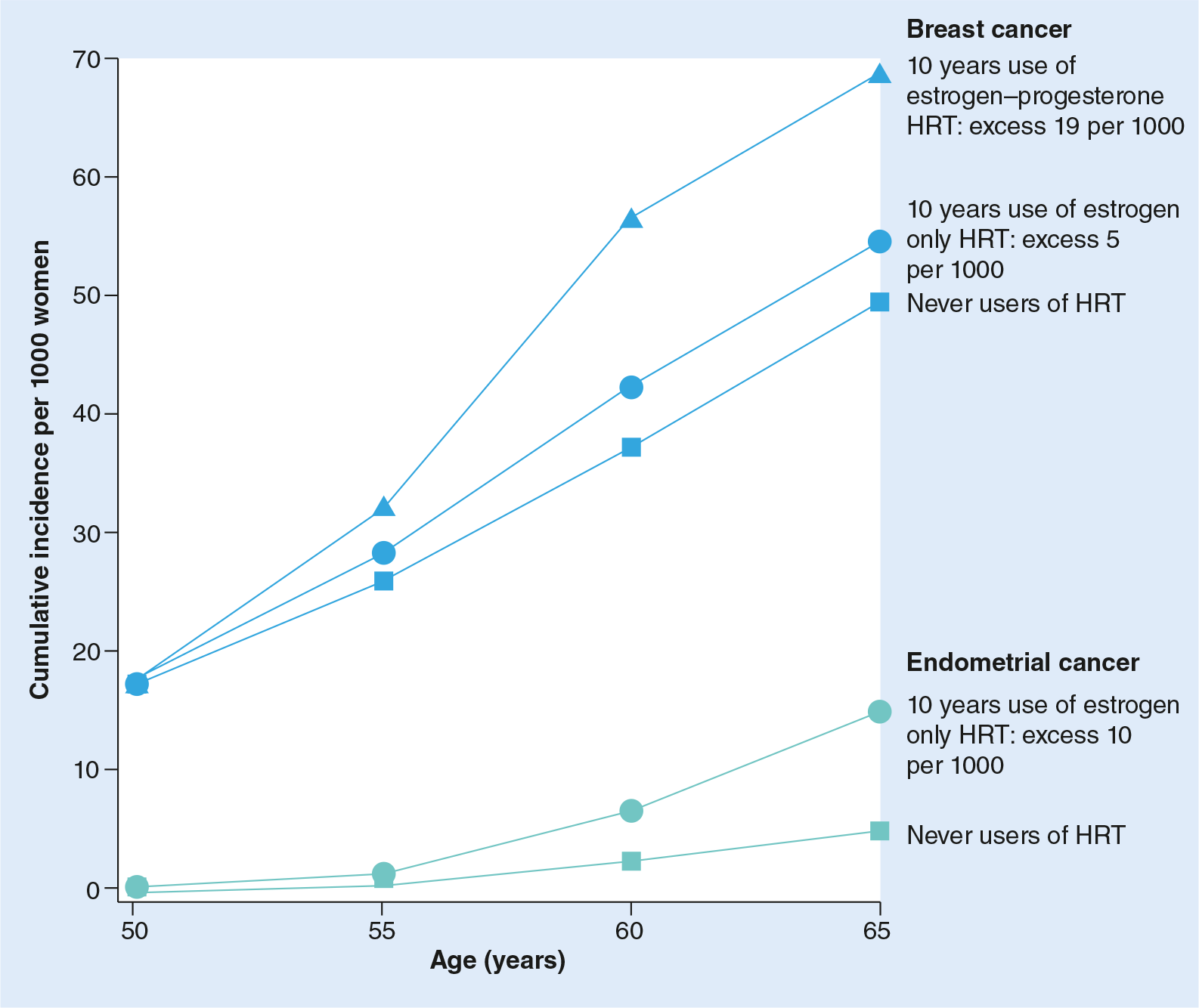
Endometrial cancer
Sequential HRT regimes may not fully prevent endometrial cancer and continuous combined estrogen–progestin regimes are to be preferred. Administration of unopposed estrogen (without progestin) in peri- and post-menopausal women with an intact uterus results in an approximate 50% increase in the incidence of endometrial hyperplasia and carcinoma within 3 years [16]. Many trials including the WHI have reported that with sequential and continuous combined estrogen–progestin HRT, the incidence of endometrial hyperplasia and carcinoma in women with an intact uterus is not increased [13,14]. In a nationwide Finnish study of 224,015 women aged 50 years or more who had used estradiol–progestin therapy between 1994 and 2006, 1364 women who had developed type I and type II endometrial cancers were identified by the Finnish Cancer Registry [17]. The use of sequential estrogen–progestin therapy was associated with a significant 69% increase in the incidence of endometrial carcinoma if progestin was added monthly and 276% if it was added trimonthly. By contrast, the use of continuous combined estrogen–progestin therapy was associated with a 76% decrease compared with never users.
In a recent nationwide Danish study of 909,946 women aged 50–79 who developed different types of ovarian cancer between 1995 and 2005, the incidence of endometrioid cancer was increased in women with sequential but not with continuous combined estrogen–progestin HRT [18]. These findings require confirmation but the large size of the studies involving the whole populations of Finland and Denmark are strong evidence that continuous combined therapy should be preferred in all women with an intact uterus who receive HRT. Sequential therapy, if used at all, should be reserved for short-term use. Given the choice, the majority of peri- and post-menopausal women do not wish to have a withdrawal bleed or any form of uterine bleeding with HRT. This is a further reason why continuous combined estrogen–progestin therapy should be used in all women with an intact uterus requiring HRT.
POI (premature menopause)
The prevention of the consequences of POI, both immediate and long term, has been neglected and a strong case can be made for estrogen replacement therapy in all women with POI, particularly following bilateral oophorectomy.
There are significant differences between spontaneous POI and iatrogenic POI following bilateral oophorectomy and pelvic surgery or following chemotherapy and pelvic irradiation for malignancy. In spontaneous POI, the ovarian follicles are greatly reduced in number and are inactive but the ovarian stroma remains active and continues to produce androgens. Estradiol levels are low but sporadic increases can occur perimenopausally. Follicle-stimulating hormone is generally raised but may fluctuate, making it an unreliable diagnostic test at the menopause. The term ‘POI’ is preferred to ‘premature ovarian failure’ or ‘premature menopause’ particularly in women with premature natural menopause as the condition may resolve and menstruation may resume in a few younger women. The term also does not carry the connotation of ‘failure’, which may have important psychological consequences in younger women.
In iatrogenic POI both the ovarian follicles and the ovarian stroma are removed and estrogens or androgens are no longer secreted. Estradiol levels drop sharply, follicle-stimulating hormone levels rise abruptly, and testosterone and androgen precursors, which are the main source of estrogens in postmenopausal women, drop by 40–50% [19,20].
The immediate consequences of POI include vasomotor and other menopausal symptoms, infertility and psychological and social problems. Estrogen replacement usually relieves the vasomotor and other menopausal symptoms of POI but does not influence the psychological consequences, which can be devastating, particularly in younger women who may require continued support. Moreover, there is accumulating evidence of the long-term adverse consequences of POI, including an increased incidence of ischemic heart disease, osteoporosis and premature mortality and possibly impaired cognition and dementia [21,22].
Cardiovascular disease
In the Framingham Study, published over 30 years ago, among the 2873 women who were followed up for 24 years, the incidence of myocardial infarction and coronary heart diseases (CHDs) was more than double in the postmenopausal women under the age of 55 years, whether the menopause was natural or surgical, compared with the premenopausal women in the same age group [23]. Since then, there have been at least five major observational studies on the effects of POI and bilateral salpingo-oophorectomy (BSO) on cardiovascular disease (CVD).
In the NHS in the USA, 29,380 women who had hysterectomy for benign disease were followed-up for over 24 years; 16,345 (55.6%) of the women had hysterectomy with BSO and 13,035 (44.4%) had hysterectomy with ovarian conservation [24]. Compared with women who had ovarian conservation at the time of hysterectomy, the women who had BSO had a significantly increased risk of fatal and nonfatal CHD, lung cancer and increased all-cause mortality, although the risk of breast and ovarian cancer was decreased. The risk of CHD in women under the age of 50 years who had BSO and who had never used estrogen therapy was increased compared with women in whom the ovaries were conserved hazard ratio (HR): 1.98 (95% CL: 1.18–3.32).
In the Danish Nurse Cohort Study of 19,888 healthy women over age 44 years, 10,533 had a definable age at menopause and the risk of ischemic heart disease was significantly increased in women who had a menopause before the age of 40 years compared with women who had a menopause after the age of 45 years [25]. However, the risk of ischemic heart disease was not increased in women who had BSO before the age of 45 years and who had used HRT. Estrogen replacement reduced the risk of ischemic disease in women with a BSO but not in those with spontaneous premature ovarian failure and the benefit was greatest in current estrogen users who started treatment within 1 year after surgery.
In a nationwide study of all women in Sweden between 1973 and 2003, 184,441 women had a hysterectomy for benign disease. They were compared with 640,043 nonhysterectomized matched women as controls [26]. In women who were under the age of 50 years at entry to the study, hysterectomy with BSO was associated with an increased hazard ratio for CVD in women compared with the nonhysterectomized women in the same age group HR: 2.22 (95% CL: 1.01–4.83). In women over age 50 years, by contrast, there was no significant association between hysterectomy with BSO and the incidence of CVD.
In the Mayo Clinic Cohort Study of Oophorectomy and Aging, 1274 women who underwent unilateral oophorectomy (USO) and 1091 women who underwent BSO before age 45 years were compared with age-matched women from the same population [27]. The women who had a USO had a reduced mortality HR: 0.82 (95% CL: 0.67–0.99) but women who had a BSO had an increased mortality resulting from CVD, HR: 1.44 (95% CL: 1.01–1.25). In the women who had a BSO before age 45 years and were not treated with estrogens, the HR was increased HR: 1.84 (95% CL: 1.27–1.68) but in the women who were treated with estrogens the HR was decreased, HR: 0.65 (95% CL: 0.30–1.74).
In the recent Multi-Ethnic Study of Atherosclerosis of women with a menopause before the age of 46 years, the HRs for CVD, adjusted for age, ethnicity and traditional risk factors, were HR: 2.08 (95% CL: 1.17–3.70) for CHD and HR: 2.19 (95% CL: 1.11–4.32) for stroke compared with women who had a menopause in the normal age range [28]. The risk of CVD was doubled in women who had POI before the age of 46 years.
There have been no randomized controlled trials of estrogen therapy in women with POI. In a meta-analysis of 18 observational studies, the pooled stratified estimates of the RR of CVD, after controlling for age and smoking, for women who had an early menopause was RR: 1.38 (95% CL: 1.21–1.58) and for those who had an early menopause following bilateral oophorectomy the RR was RR: 4.55 (95% CL: 2.56–8.01) compared with women with a menopause at age 50 [29].
Osteoporosis & fracture risk
Women with POI have a lower bone mineral density and an increased risk of fracture than women who have a menopause in the normal range (age 45–55 years) [30]. Several studies have shown that bone loss is accelerated after the menopause and that the earlier the menopause the lower the bone density in later life [31,32]. In a long-term 34-year prospective study from Sweden, of 390 Northern European women aged 48 years at the start of the study, those who had an early menopause before the age of 47 years were compared with those who had a menopause after the age of 47 years. The women who had an early menopause had a significantly greater risk of osteoporosis RR: 1.83 (95% CL: 1.22–2.74) and fragility fracture RR: 1.68 (95% CL: 1.05–2.57) and a greater overall mortality RR: 1.59 (95% CL: 1.04–2.36) [33]. Estrogen therapy prevents bone loss and reduces fracture risk following BSO in women under age 50 and HRT increases bone mineral density in women with POI [34]. Menopause before aged 50 years is one of the commonest causes of vertebral crush fractures in women [35]. The benefit of estrogen replacement therapy in reducing the risk of osteoporosis and fragility fracture in postmenopausal women is beyond doubt and women with POI should obtain even greater benefit [36].
Cognitive function
The possibility of an increased risk of impaired cognitive function and of dementia in women who have had a POI has been raised by the Mayo Clinic Cohort Study of Oophorectomy and Aging [37]. Women who had a USO or BSO before the menopause had an increased risk of cognitive impairment or dementia of HR 1.46 (95% CL: 1.13–1.90) compared with an age-matched group of women from the same population who had not undergone oophorectomy. The risk of impaired cognition or dementia increased with decreasing age at which POI had occurred (test for linear trend p< 0.0001). The increased risk of impaired cognitive function or dementia was restricted to women who had either a USO or BSO but had not taken estrogen before 50 years of age. The women with a USO or BSO also had an increased incidence of Parkinsonism, HR 1.86 (95% CL: 1.06 −2.67) compared with the referent group of women and the incidence was increased with decreasing age at oophorectomy (test for linear trend: p = 0.01) [38].
These findings are in contrast to those in the Women's Health Initiative Memory Study in which it was concluded that HRT increased the risk of dementia and did not prevent cognitive impairment [39]. The women in the Women's Health Initiative Memory Study, however, were aged 65 years or older at the time of recruitment. In a review of literature and further analysis of their own data, the authors of the Mayo Clinic Study suggest that the neuroprotective effects of estrogen depend on age at the time of starting estrogen administration [40]. The authors suggest that younger postmenopausal women who have undergone a menopause before the median age of menopause (at 51 years old) may benefit, whereas older women over the age of 60 years may not benefit from HRT and that there is a ‘window of opportunity’ in younger postmenopausal women under the age of 50 years when estrogens may be neuroprotective. Estrogens, accordingly, may be neuroprotective in younger postmenopausal women who have had POI but may be deleterious in older postmenopausal women in whom neurological changes as well as cerebrovascular disease may have already occurred. If it is established that women with POI are at increased risk of cognitive impairment and of dementia in later life and that this risk may be reduced by estrogen therapy, it would be another important reason for recommending that estrogens be given to all women from the time of POI to at least the average age of the natural menopause.
There have been no long-term placebo-controlled randomized trials of estrogens in the primary prevention of the long-term consequences of POI and it seems unlikely that there will be any in the foreseeable future. The evidence of an increased risk of CVD, osteoporosis, and of impaired cognitive function and dementia in women with POI, and of a preventive effect of estrogen therapy, is considerable. Furthermore, there is no evidence of an increased risk of breast cancer with HRT in women with POI. In an authoritative review of the management of premature menopause, it was concluded that ‘there was no evidence that estrogen replacement increases the risk of breast cancer to a level greater than that in normally menstruating women’ [41]. This conclusion is supported by the absence of any evidence that the risk of breast cancer is increased with long-term HRT in women with Turner's syndrome [42]. A strong case can be made for estrogen therapy in all women with POI irrespective of symptoms. Women who have had a BSO and hysterectomy appear to be at particular risk. The addition of a progestin, which carries an increased risk of breast cancer, is not necessary in hysterectomized women and the balance of benefits and risks is influenced more favorably. It may be contended that, unless specifically contraindicated, estrogen therapy should be mandatory in all women with POI irrespective of symptoms until the mean age of the natural menopause of 50 years.
Future perspective
The management of postmenopausal women is not static and a better, more balanced understanding is being achieved. The risk of breast cancer that has dominated much of the discussion is being put in perspective. The long-term consequences of the menopause, particularly in women with POI, including CVD, osteoporosis and a possible increased incidence of Alzheimer's disease, are becoming better appreciated. It appears that there may be a ‘window of opportunity’ for hormone replacement in the 10 years or so immediately after the menopause. In women with POI, estrogen replacement from the onset of the menopause to at least to the average age of the menopause, has become an essential part of care. The results of current clinical trials of newer forms of therapy, including the use of micronized progesterone and transdermal administration of estrogen, which may convey more benefit with less possible risk should become available in the next 5–10 years. It is to be hoped that in the future appropriate HRT will become integrated into the overall care of women in the later years of life.
Financial & competing interests disclosure
Executive summary
Many important issues regarding HRT and the management of postmenopausal women are unresolved.
HRT is only one of many factors affecting the risk of breast cancer, including a family history of breast cancer and increased mammographic breast density, which carry a similar increase in risk.
The risk of breast cancer is not increased in past users of HRT, implying that there must be a ‘compensatory’ decrease in risk for a period after stopping HRT.
The risk of breast cancer increases with increasing body mass but, paradoxically, the increased risk of breast cancer associated with HRT decreases with increasing body mass.
The addition of a progestin to estrogen in HRT in women with an intact uterus reduces the risk of endometrial hyperplasia and cancer but increases the risk of breast cancer. The increase in morbidity and mortality from breast cancer associated with combined estrogen-progestin HRT may be greater than the decrease in morbidity and mortality from endometrial cancer.
Sequential HRT regimes may not fully prevent endometrial cancer and continuous combined estrogen-progestin regimes are to be preferred.
The prevention of the consequences of premature menopause, both immediate and long term, has been neglected and a strong case can be made for estrogen replacement therapy in all women with premature ovarian insufficiency, particularly following bilateral oophorectomy.
