Abstract
Polycystic ovary syndrome (PCOS) is the most prevalent endocrine disorder in women of reproductive age and by far the most common cause of anovulatory infertility. Lifestyle change alone, and not in combination with pharmacological ovulation induction such as clomifene citrate or metformin, is generally considered the first-line treatment for the management of infertile anovulatory women with PCOS who are overweight or obese. Clomifene citrate should be considered as a first-line pharmacological therapy to improve fertility outcomes. Second-line medical treatments may include ovulation induction with gonadotropins (in clomifene citrate-resistant or clomifene citrate failure women) or laparoscopic ovarian drilling (in clomifene citrate-resistant women) or possibly with metformin combined with clomifene citrate (in clomifene citrate-resistant women). There is currently insufficient evidence to recommend aromatase inhibitors over that of clomifene citrate in infertile anovulatory women with PCOS in general or specifically in therapy-naive or clomifene citrate-resistant women with PCOS.
Stein and Leventhal were the first to describe an association between the presence of polycystic ovaries, oligo/anovulation and hirsutism, later known as polycystic ovarian syndrome (PCOS) in 1935 [1]. PCOS, a heterogeneous condition characterized by chronic anovulation and hyperandrogenism, is the most prevalent endocrine disorder in women of reproductive age and is the most common cause of anovulatory infertility [2]. The prevalence of PCOS depends on the population studied and the diagnostic criteria used, and has been estimated to affect 4–12% of the general population, 28% of obese and 5% of lean reproductive age women [3].
Although there is continuing debate on what constitutes PCOS, the Rotterdam Consensus on Diagnostic Criteria for PCOS formulated in 2003 and published in 2004 is the most current agreed international definition
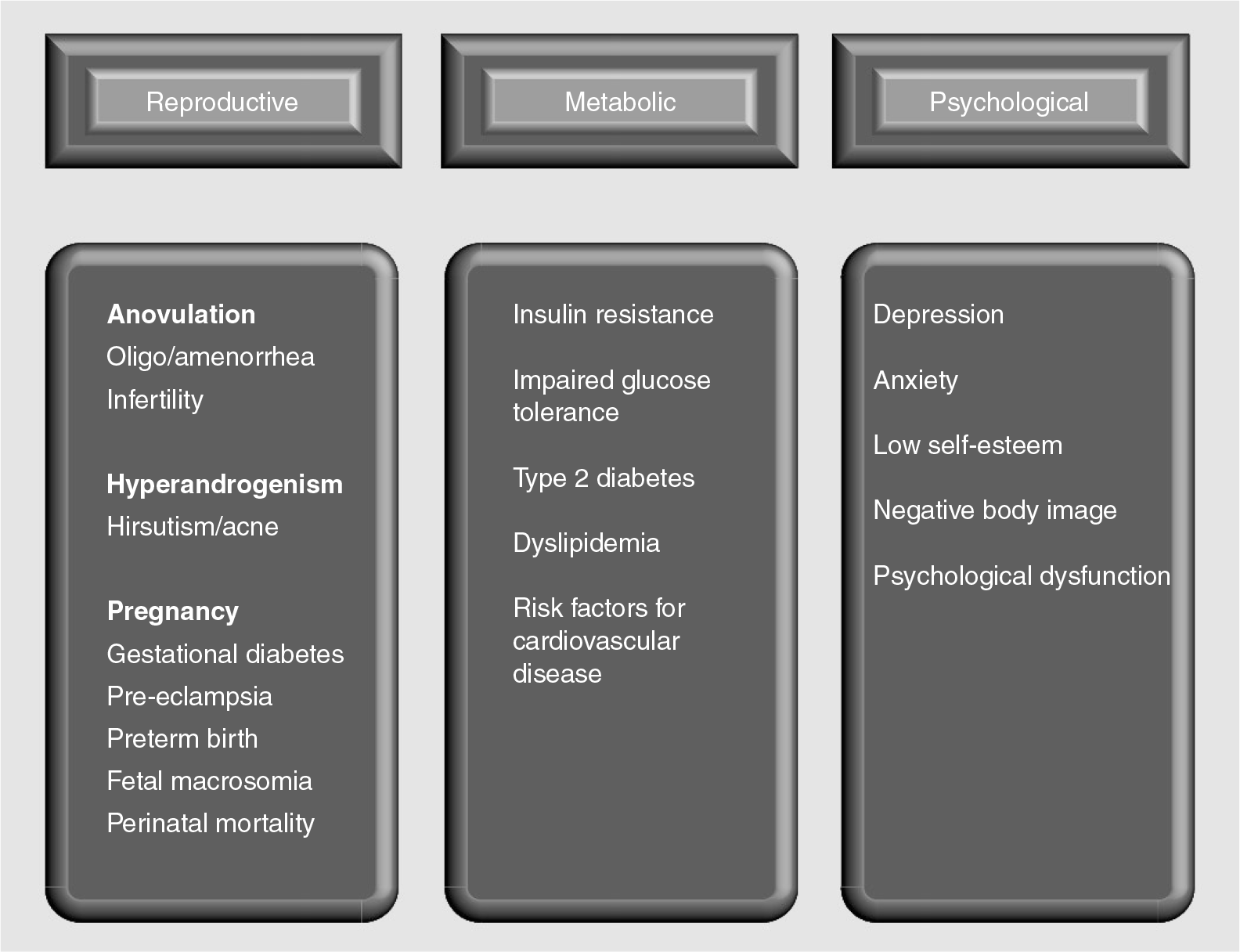
Clinical sequelae of polycystic ovary syndome.
Infertility is a prevalent presenting feature of PCOS with approximately 75% of these women suffering infertility due to anovulation [2]. Infertile women with PCOS should undergo a thorough infertility work-up to look for causes of infertility other than anovulation. Where the only cause is anovulation, a number of infertility treatments have been proposed

Treatments for anovulation in polycystic ovary syndrome.
Methodology
A broad-ranging systematic search for terms related to PCOS and infertility was used. This PCOS search string was then combined with specific searches tailored for each of the lifestyle and pharmacological treatments for infertility in women with PCOS. Outcomes were rates of ovulation, pregnancy, live birth, multiple pregnancies, miscarriage and adverse events. The search strategy was limited to English language articles and there were no limits on the year of publication. The literature was searched dating up to July 2011. The following electronic databases were employed to identify relevant literature: CINAHL, Medline (OVID), EMBASE, The Cochrane Library, Cochrane Database of Systematic Reviews (Cochrane Reviews), Database of Abstracts of Reviews of Effects (Other Reviews) and Cochrane Central Register of Controlled Trials (Clinical Trials). We also searched the bibliographies of relevant studies identified by the search strategy and relevant reviews/meta-analyses for identification of additional studies.
Rotterdam criteria for diagnosis of polycystic ovary syndrome.
Oligo-ovulation and/or anovulation
Clinical or biochemical evidence of hyperandrogenism
Polycystic ovaries on ultrasound assessment (≥12 small antral follicles 2–9 mm in at least one ovary and/or ovarian volume >10 ml)
With the exclusion of other causes of oligo-anovulation or hyperandrogenism such as congenital adrenal hyperplasia, androgen-secreting tumors or Cushing's syndrome
During the assessment of the relevant studies, the characteristics or type of PCOS women were noted in terms of weight (normal [BMI 20–25 kg/m2], overweight [BMI 25–30 kg/m2], obese [BMI >30 kg/m2] and morbidly obese [BMI >35 kg/m2]) and previous exposure and/or response to ovulation induction therapy (therapy naive [no previous exposure], clomifene citrate resistant [CCR; anouvlatory to clomifene citrate], clomifene citrate failure [CCF; ovulatory to clomifene citrate but did not conceive] or a mixture of patients).
Preconception counseling
Preconception counseling should be provided before commencing treatment in order to optimize health before pregnancy, with emphasis being placed on lifestyle modification, cessation of smoking and reduction of alcohol intake [7]. Counseling, where appropriate, should also consider the adverse effects of obesity on reproduction. These include chronic anovulation, failure of ovulation induction, miscarriage and later pregnancy complications including preeclampsia, gestational diabetes, increased risk of cesarean section, fetal macrosomia and venous thromboembolism, and in some series, congenital defects [6,8–10]. A recent prospective observational study comparing 97 pregnant women with PCOS and 73 pregnant control women found that adverse obstetric and neo natal outcomes were restricted to those with both oligoanovulation and clinical or biochemical hyperandrogenism [11].
Lifestyle change
Lifestyle change is the first-line treatment for women with PCOS who are overweight or obese, with as little as 5–10% weight loss having significant clinical benefits in improving psychological outcomes, reproductive features (menstrual cyclicity, ovulation and fertility) and metabolic features (insulin resistance and risk factors for cardiovascular disease and Type 2 diabetes mellitus) [3]. A Cochrane systematic review published in 2011, comparing lifestyle interventions (diet, exercise, behavioral management techniques for modifying diet or exercise, or a combination of these) versus minimal intervention in women with PCOS, located six randomized controlled trials (RCTs), of which no studies reported on live birth, two studies reported on pregnancy data, and three RCTs reported on ovulation [12]. Of the two RCTs reporting on pregnancy, only two out of 20 women [13] and zero out of 51 women [14] were actively seeking pregnancy, making any comparison of pregnancy rates between the two interventions meaningless [15].
Three RCTs reported on ovulation [13,16,17], but unfortunately the data was in a form inappropriate for meta-analysis [15]. Guzick et al. randomized 12 obese women with PCOS to either 12 weeks of diet and behavioral modification or no treatment, and found that four out of six (67%) women were ovulatory in the treatment group compared with one out of six (17%) women in the control group (odds ratio [OR]: 6.59; 95% CI: 0.73–59.34; p = 0.09) [16]. In a RCT of 20 overweight (BMI >25 kg/m2) women with PCOS comparing diet plus exercise plus behavioral modification with no lifestyle intervention, the mean number of ovulations per woman were 6.0 versus 2.8 (mean difference: 3.2; 95% CI: −1.02–7.42; p = 0.14), respectively, over a treatment duration of 48 weeks [13]. Finally, the same group of investigators compared diet plus exercise plus behavioral modification with standard office advice on diet and exercise in 22 obese (BMI >95th percentile) women with PCOS over a 24-week period and reported 60 versus 50% ovulatory cycles between the treatment and control group (no statistical comparison reported), respectively [15,17].
An expert international panel of PCOS researchers appointed by the Androgen Excess and PCOS Society recently reviewed the literature on the lifestyle management (dietary, exercise or behavioral interventions) of PCOS and concluded that lifestyle management should be the primary therapy for the treatment of metabolic complications, and may improve ovulatory function and pregnancy in overweight/obese women with PCOS. The latter conclusion on reproductive outcomes was based on 13 uncontrolled observational studies and a single RCT showing a nonsignificant increase in ovulation with lifestyle treatment. There were no studies identified concerning the modification of dietary macronutrient composition in terms of menstrual function, ovulation, pregnancy or live birth [18].
Another recently published systematic review evaluating the effect of exercise as an independent intervention on clinical outcomes in PCOS, identified two studies (one RCT and one cohort study) that reported results on reproductive function (ovulation and/or pregnancy rate) in a total of 134 pooled obese women with PCOS, of whom 83 received exercise intervention over a 20–24-week duration [19]. Both studies reported an improvement in ovulation with exercise compared with baseline in the RCT [20] and dietary therapy in the cohort study [21], with the latter study also reporting a 35% pregnancy rate with exercise versus 10% with diet (p = 0.058). The authors of this review recommended that women with PCOS should be advised to engage in at least 90 min of aerobic activity per week at moderate intensity to achieve improved reproductive and cardiometabolic outcomes [19]. The investigator group of the above cohort study subsequently published a RCT demonstrating that 6 weeks of structured exercise training and hypocaloric diet increases the ovulation rate with clomifene citrate in overweight and obese CCR PCOS women [22]. In addition to improving ovulation, structured exercise training has been demonstrated in a RCT to improve cardiopulmonary functional capacity in young overweight PCOS women [23].
In summary, with regards to lifestyle treatment of women with PCOS with infertility, the limited randomized evidence to date shows a trend in improvement in ovulation in all three published RCTs. Unfortunately there are no RCTs assessing lifestyle treatment in women with PCOS that report on pregnancy or live-birth rates. More evidence is required in order to assess the effect of lifestyle interventions on fertility outcomes in women with PCOS. In the meantime, an international position statement by an expert panel has recommended lifestyle management as first-line treatment for infertile overweight/obese women with PCOS [18].
Lifestyle &/or pharmaceutical treatment
There are a number of published RCTs that compare the effectiveness of lifestyle treatment with ovulation induction medications (clomifene citrate and metformin) in overweight/obese women with PCOS that have reported outcomes relevant to fertility including menstrual function, ovulation and pregnancy rates [13,17,24,25]. These four RCTs had varying sample sizes (20, 21, 46 and 343 women [four intervention groups in this largest study]) and compared lifestyle treatment (diet in one RCT, diet plus exercise in two RCTs and diet plus exercise plus behavioral modification in one RCT) versus metformin alone (four RCTs), clomifene citrate alone (one RCT) or metformin combined with clomifene citrate (one RCT) with treatment durations ranging from 24 to 48 weeks. The largest RCT compared four treatment groups: lifestyle, metformin alone, clomifene alone and metformin combined with clomifene citrate [24].
No significant differences were found between lifestyle and pharmaceutical (clomifene citrate or metformin, alone or combined) treatments in terms of menstrual function [13,17,24,25], ovulation [13,17,24,25] or pregnancy rates [24,25] in these trials. None of the RCTs reported on live-birth data. It is important to note that there were no sample size or power calculations reported for any of these studies suggesting that the negative study findings may be attributed to lack of study power given the overall small sample sizes.
There are also a number of published RCTs comparing lifestyle treatment alone with ovulation induction medications combined together with lifestyle therapy in overweight/obese women with PCOS that have reported fertility outcomes [13,17,26–30]. Again, the majority of the seven trials had small sample sizes (20 for three studies, 30, 36, 40 and 143 women) and compared various lifestyle treatments (diet in four RCTs, diet plus exercise in one RCT and diet plus exercise plus behavioral modification in two RCTs) versus combined lifestyle treatment and metformin (all seven RCTs) with treatment durations ranging from 16 to 52 weeks. All seven studies used a placebo pill with the lifestyle treatment alone groups. Only one of the seven RCTs reported sample size or power calculations and was powered to detect an 83% improvement in menstrual regularity [30].
Only two of the seven RCTs found a significant difference in any of the fertility outcomes evaluated [27,29]. The other five RCTs found no significant differences between the treatment groups in terms of menstrual function [13,17,26,28,30], ovulation [13,17,18] and pregnancy rates [30]. None of the RCTs reported on live-birth rate. Gambineri and colleagues compared menstrual function only between metformin plus diet versus placebo plus diet treatments in 40 women with PCOS with a BMI >28 kg/m2, and found a significantly higher frequency of menstruation at 6 months (4.3 vs 3.2, respectively; p = 0.017) and at 12 months (4.6 vs 3.2, respectively; p < 0.001) [27].
In the study by Pasquali et al., 143 obese women with PCOS were randomized to 28 weeks of treatment with either metformin plus diet or placebo plus diet, with a significant improvement in menstrual frequency with the former treatment (4.7 vs 3.2 menses; p < 0.05) but no difference in pregnancy rate [29].
In summary, the randomized evidence to date does not support treatment with ovulation induction medications such as clomifene citrate or metformin ahead of lifestyle treatment alone or starting lifestyle treatment in combination with such ovulation induction agents compared with lifestyle treatment alone in infertile anovular overweight/obese women with PCOS.
Pharmaceutical or medical ovulation induction treatments
Clomifene citrate
Clomifene citrate, a selective estrogen receptor modulator with both estrogenic and anti-estrogenic properties [31], was first approved for use in women with anovulation in 1967 and, therefore, has been used as the first-line medical ovulation induction agent for over 40 years [32]. Acting as an anti-estrogen, clomifene citrate competitively inhibits the binding of estradiol to its receptors in the hypothalamus and pituitary, which in turn blocks the negative feedback effect of endogenous estrogens, including estradiol. This release of the hypothalamus from negative inhibition results in an increased secretion of pulsatile gonadotropin-releasing hormone secretion leading to an increase in follicle-stimulating hormone (FSH) and luteinizing hormone (LH) production and secretion from the pituitary gland. This increase in FSH secretion stimulates follicular growth and estradiol production with the final occurrence of a midcycle LH surge and ovulation [33].
It is usual practice to administer clomifene citrate for 5 days beginning on day 2–5 of the menstrual cycle, starting with 50 mg/day and increasing to 150 mg/day. If ovulation cannot be achieved with clomifene citrate administration at doses of 150 mg/day, then the patient is said to be CCR. If pregnancy cannot be achieved after six ovulatory cycles with clomifene citrate, then the patient is described as having CCF [34].
Studies with clomifene citrate have shown an ovulation rate of 60–85% and a pregnancy rate of 30–50% after six ovulatory cycles. This apparent discrepancy between ovulation and pregnancy rates has been attributed to the anti-estrogenic effects of clomifene citrate on the endometrium and cervical mucus. The rates of twin pregnancy and triplets with clomifene citrate are 5.0–7.0 and 0.3%, respectively. The incidence of ovarian hyperstimulation syndrome is less than 1% [35]. Although more studies are required, it may be advisable to limit a patient's lifetime exposure to clomifene citrate to 12 treatment cycles, as additional cycles may place the patient at increased risk of borderline ovarian tumors [36], although more recent studies have not supported this association [37,38].
The best available evidence supporting the use of clomifene citrate as an ovulation induction agent in PCOS comes from a systematic review and meta-analysis of three RCTs comparing clomifene citrate with placebo showing that clomifene citrate improves ovulation rate per patient (OR: 7.5; 95% CI: 3.24–7.23; p < 0.00001 [three trials, 133 women]) and pregnancy rate per patient (OR: 5.8; 95% CI: 1.55–21.48; p < 0.009 [three trials, 133 women]). There were no published RCTs comparing clomifene citrate with placebo or no treatment examining live-birth rate as an outcome measure [39].
The addition of an ovulatory trigger dose of human chorionic gonadotropin (hCG) to clomifene citrate ovulation induction therapy does not improve reproductive outcomes, according to a single multicenter RCT of 125 therapy-naive women with WHO class 2 anovulation as the sole cause for infertility randomized to a single treatment cycle with either clomifene citrate with a hCG trigger or clomifene citrate alone, and timed intercourse [39,40]. There were no differences between the two treatment groups in terms of ovulation, pregnancy, twin pregnancy or miscarriage rates.
Therefore, clomifene citrate without the use of a hCG trigger should be used as the first-line pharmacological therapy to improve fertility in anovulatory women with PCOS without other infertility factors.
Metformin
Metfomin is a biguanide that is used as an oral antihyperglycemic agent in the treatment of Type 2 diabetes mellitus [41]. The rationale for the use of insulin-sensitizing agents such as metformin to restore ovulation in PCOS is based on the observation that insulin resistance with compensatory hyperinsulinemia is a prominent feature of PCOS [42], affecting approximately 65–70% of such women [43]. Hyperinsulinemia results in increased ovarian androgen biosynthesis in vivo and in vitro and decreased sex hormone-binding globulin synthesis from the liver, leading to increased bioavailability of free androgens. This excess in local ovarian androgen production augmented by hyperinsulinemia causes premature follicular atresia and anovulation [44].
Of the insulin-sensitizing drugs, metformin has been the most widely studied in PCOS, and has the most reassuring safety profile [45]. The first published report on the use of metformin as a treatment for PCOS was in 1994 [46]. The early studies examining the reproductive system effects of metformin in women with PCOS showed promising results but most of the studies were observational in design and had relatively small sample sizes [44].
Therapeutic regimens of metformin administration in PCOS are not well standardized in clinical practice, and various protocols have been used in the studies available in the literature with an extremely variable target dose of 1500–2550 mg per day having been proposed [45]. The most frequent adverse reactions to metformin are gastrointestinal symptoms including diarrhea, nausea, vomiting, abdominal pain and loss of appetite.
This section of the review addresses four sets of comparisons: metformin versus placebo/no treatment, metformin versus clomifene citrate, metformin plus clomifene citrate versus clomifene citrate, and metformin plus clomifene citrate versus metformin.
Metformin versus placebo or no treatment
The highest level of evidence on the use of metformin is evident in the Cochrane systematic review by Tang et al. who performed a meta-analysis of RCTs comparing metformin with placebo or no treatment in women with PCOS, which showed that metformin improves ovulation rate (OR: 2.12; 95% CI: 1.5–3.0; 13 RCTs; 875 women) and clinical pregnancy rate (OR: 3.86; 95% CI: 2.18–6.84; six RCTs; 479 women), but not live-birth rate (OR: 1.0; 95% CI: 0.16–6.39; two RCTs; 50 women) per woman [47].
Metformin versus clomifene citrate
Tang et al. also compared metformin versus clomifene citrate in women with PCOS and found a reduced ovulation rate per cycle (OR: 0.48; 95% CI: 0.41–0.57; three RCTs; 2470 cycles) and a reduced clinical pregnancy rate per woman (OR: 0.63; 95% CI: 0.43–0.92; three RCTs; 600 women), but no difference in live-birth rate per woman (OR: 0.67; 95% CI: 0.44–1.02; three RCTs; 600 women) with metformin in women with PCOS in general [47]. However, there was significant statistical heterogeneity seen in all three outcomes and therefore the results need to be interpreted with caution.
The authors subgrouped the RCTs, comparing metformin versus clomifene citrate according to BMI (BMI >30 kg/m2 or <30 kg/m2) and the statistical heterogeneity disappeared for all three outcome measures. In women with PCOS with a BMI >30 kg/m2, there was a lower ovulation rate (OR: 0.43; 95% CI: 0.36–0.51; two RCTs; 2044 cycles), clinical pregnancy rate (OR: 0.34; 95% CI: 0.21–0.55; two RCTs; 500 women) and live-birth rate (OR: 0.30; 95% CI: 0.17–0.52; two RCTs; 500 women) with metformin. However, in women with PCOS with a BMI <30 kg/m2, there was no difference in ovulation rate (OR: 0.84; 95% CI: 0.56–1.25; one RCT; 426 cycles), but a higher pregnancy rate (OR: 3–47; 95% CI: 1.52-7.90; one RCT; 100 women) and live-birth rate (OR: 4.94; 95% CI: 1.99–12.26; one RCT; 100 women) with metformin based on a single RCT [47].
Two RCTs comparing metformin versus Clomifene citrate in women with PCOS were published after Tangs Cochrane review [24,48]. The first RCT in 180 women with PCOS with a BMI between 25 and 30 kg/m2 found no difference in pregnancy rate per patient between metformin and clomifene citrate treatment over 3–6 months [24]. The second RCT in 71 women with PCOS with a BMI ≤32 kg/m2, found no difference between metformin and clomifene citrate treatment over 6 months in terms of ovulation, pregnancy or live-birth rate per patient [48].
Randomized controlled trials comparing clomifene citrate with metformin in polycystic ovary syndrome women.
Other causes of infertility excluded.
↑: Increased; ↓: Decreased; : No difference; b.i.d.: Twice daily; C: Cycle; CC: Clomifene citrate; CCR: Clomifene citrate-resistant; LBR: Live-birth rate; M: Metformin; MCR: Miscarriage rate; MPR: Multiple pregnancy rate; NR: Not reported; OR: Ovulation rate; P: Placebo; Pa: Patient; PCOS: Polycystic ovary syndrome; PR: Pregnancy rate; preg: Pregnancy; Rx: Treatment; TN: Therapy naive.
Therefore, in summary, Clomifene citrate is superior to metformin overall for ovulation induction in women with PCOS, and for women with PCOS with a BMI >30 kg/m2 while the RCT data are conflicting in women with PCOS with a BMI <30 kg/m2. A recent meta-analysis of RCTs comparing metformin versus Clomifene citrate in nonobese women (BMI <30–32 kg/m2) with PCOS demonstrated no difference in clinical pregnancy rate per patient (relative risk [RR]: 0.91 favoring clomifene citrate; 95%) CI: 0.35–2.35; three RCTs; 285 patients) or live-birth rate per patient (RR: 0.83 favoring clomifene citrate; 95% CI: 0.22–3.24; three RCTs; 285 patients) between the two treatments. However, these results should be interpreted with caution as the results are based on highly statistical heterogeneous data. In addition, this meta-analysis is unable to exclude an important difference between either metformin or clomifene citrate as the results include wide confidence intervals indicating insufficient power [49].
Metformin plus clomifene citrate versus clomifene citrate alone
The most recent systematic reviews and meta-analyses of RCTs that have compared the combination of metformin plus clomifene citrate versus clomifene citrate alone in various classes of women with PCOS included ‘overall’ women with PCOS, those with a BMI <30 kg/m2 or >30 kg/m2 and those who were clomifene citrate-naive or -resistant [47,50,51]. Metformin combined with clomifene citrate had a higher ovulation, pregnancy and live-birth rate without statistical heterogeneity compared with clomifene citrate alone only in CCR PCOS. Therefore, the addition of metformin to clomifene citrate is more beneficial in CCR women with PCOS than persisting with more cycles of clomifene citrate alone.
Metformin plus clomifene citrate versus metformin alone
The most current systematic review and meta-analyses of RCTs comparing the combination of metformin plus clomifene citrate versus metformin alone in PCOS demonstrated a higher ovulation rate per patient (OR: 0.23; 95% CI: 0.15–0.34; two RCTs; 493 women), pregnancy rate per patient (OR: 0.23; 95% CI: 0.14–0.37; two RCTs; 492 women) and live-birth rate per patient (OR: 0.23; 95% CI: 0.13–0.40; two RCTs; 492 women) with metformin combined with clomifene citrate [52]. The two RCTs included in this meta-analysis included women with PCOS with a mean BMI of 36 kg/m2, of whom approximately half had been exposed to previous infertility treatment [53] and therapy-naive women with PCOS with a mean BMI of 33 kg/m2 [54]. Therefore, if one is considering ovulation induction treatment with metformin alone in infertile women with PCOS who are obese (BMI >30 kg/m2), the evidence supports the addition of clomifene citrate to improve fertility outcome.
Metformin coadministration with other fertility therapies
A number of systematic reviews and meta-analyses of RCTs have found no evidence for a benefit when metformin is added to laparoscopic ovarian drilling (one RCT; 42 CCR women with PCOS randomized), gonadotropin ovulation induction with timed intercourse (three RCTs; 77 CCR women with PCOS randomized) or IVF/intracytoplasmic sperm injection (ICSI) treatment (fve RCTs; 426 women with PCOS randomized) in terms of live-birth rate, pregnancy rate or miscarriage rate, although metformin coadministration with IVF results in a 70–80% lower risk of ovarian hyperstimulation syndrome [50,55,56]. A recently published RCT assessing the use of metformin starting 3 months prior to and continuing during IVF/ICSI treatment in 150 randomized nonobese women with PCOS also found no benefit in terms of clinical pregnancy or live-birth rates per woman when the outcomes of the IVF/ICSI population were analyzed separately [57].
Gonadotropins
Use of intramuscular gonadotropins derived from the purified urine of postmenopausal women and containing both FSH and LH began in the 1960s. Over the years, urinary products containing mainly FSH were developed, followed by the introduction of recombinant human FSH over the last decade. The use of gonadotropins for ovulation induction in PCOS is based on the physiological concept that a transient increase in serum FSH above a threshold level for a sufficient duration will initiate and maintain ovarian follicular growth. Gonadotropin therapy is often used as second-line therapy in infertile anovulatory women with PCOS who are resistant to ovulation induction with anti-estrogen treatment such as clomifene citrate (CCR) or who have ovulated but failed to conceive with clomifene citrate (CCF) [6,58].
Women with PCOS are particularly sensitive to gonadotropin therapy and have a significant chance of multiple follicular development leading to an increased risk of multiple pregnancy, ovarian hyperstimulation syndrome and cycle cancellation [59]. The two common regimens of ovulation induction with gonadotropins are the ‘low-dose step-up’ protocol (starting treatment with a low dose of 50–75 IU daily for 7–14 days, and increasing the dose with small increments every week until follicular development) [60] and the ‘step-down’ protocol (commencing treatment with a standard dose of 150 IU daily, which is then reduced with dominant follicle development) [6]. The chance of pregnancy is comparable between the two protocols with the ‘low-dose step-up’ approach being safer with regard to the induction of monofollicular ovulation and potentially easier to monitor. Low-dose gonadotropin ovulation induction in PCOS results in an approximately 70% monofollicular ovulation rate, 15–20% pregnancy rate per ovulatory cycle, cumulative pregnancy rate of 55–70%, multiple pregnancy rate of 4–18% and ovarian hyperstimulation syndrome rate of 1% [6,60].
Unlike the interventions for infertility in PCOS discussed above (lifestyle, lifestyle and/or pharmaceutical treatment, clomifene citrate and metformin), there are no RCTs comparing gonadotropin ovulation induction versus placebo/no treatment in either therapy-naive women with PCOS or CCR/CCF women with PCOS. However, there is a large body of observational evidence supporting the use of gonadotropin ovulation induction in women with PCOS with CCR or CCF [34,60]. Two separate Cochrane systematic reviews and meta-analyses of RCTs have shown no difference between recombinant FSH and urinary FSH ovulation induction [61] or urinary FSH and human menopausal gonadotropin ovulation induction [59] in terms of ovulation rate, pregnancy rate, multiple pregnancy rate and miscarriage rate in CCR women with PCOS.
There are two published RCTs comparing recombinant FSH ovulation induction with clomifene citrate ovulation induction in women with PCOS who are therapy naive [62,63]. The first single-center RCT compared up to three cycles of a chronic low-dose step-up regimen of FSH ovulation induction with up to three cycles of clomifene citrate in 76 therapy-naive women with PCOS and found a trend towards an improvement in cumulative pregnancy rate at 3 months (42 vs 24%, respectively; p = 0.09 or RR: 1.78; 95% CI: 0.92–3.54) and no difference in cumulative live-birth rate (29 vs 16%, respectively; p = 0.17 or RR: 1.83; 95% CI: 0.79–4.40), ovulation rate per woman (92 vs 79%, respectively; p = 0.10 or RR: 1.17; 95% CI: 0.97–1.46), multiple birth rate per pregnancy (19 vs 11%, respectively; p = 1.0) or miscarriage rate per pregnancy (31 vs 33%, respectively; p = 1.0). This RCT was stopped early owing to an inability to recruit the desired number of study participants (the original required total number of study participants was 304, which was calculated by a priori power calculations based on the end point of cumulative pregnancy rate) [62].
The second and more recent multicenter RCT also compared the chronic low-dose step-up regimen of FSH ovulation induction with clomifene citrate over three treatment cycles in 255 therapy-naive women with PCOS and found a higher clinical pregnancy rate per patient (58 vs 44%, respectively; p = 0.03) and live-birth rate per patient (52 vs 39%, respectively; p = 0.04) with FSH treatment. The authors concluded that although FSH is more effective than clomifene citrate ovulation induction, the differences in cost and convenience may limit the choice of FSH as a first-line treatment [63].
Therefore, the use of gonadotropins for ovulation induction in women with PCOS with CCR or CCF is supported by a large volume of observational evidence with recent randomized evidence suggesting higher efficacy compared with clomifene citrate in therapy-naive women with PCOS.
Aromatase inhibitors
Aromatase inhibitors were first proposed as ovulation-inducing drugs in anovulatory women (with an inadequate response to clomifene citrate) in 2001 [64]. Letrozole and anastrozole are the most commonly used aromatase inhibitors in ovulation induction, with letrozole being the more widely used [41].
The enzyme aromatase catalyzes the conversion of androgens to estrogens, specifically the conversion of testosterone to estradiol and androstenedione to estrone in the ovary. Aromatase inhibitors inhibit estrogen production and therefore release the hypothalamo–pituitary axis from estrogenic negative feedback resulting in increased secretion of FSH by the pituitary, which in turn stimulates ovarian follicular growth and development. In addition, androgens that would normally be converted to estrogens accumulate in the ovary, and these androgens increase follicular sensitivity to FSH [65].
The rationale for developing aromatase inhibitors as agents for ovulation induction was to avoid some of the adverse peripheral anti-estrogenic effects of clomifene citrate on the endometrium and cervical mucus [66] and also the increased risk of multiple pregnancy seen with clomifene citrate [67]. Unlike clomifene citrate, aromatase inhibitors do not thin the endometrial lining or deplete estrogen receptors centrally [65]. This lack of central depletion of estrogen receptors allows normal negative feedback on FSH production by estrogen secreted by the multiple developing ovarian follicles, which in turn leads to atresia of the follicles that are smaller than the dominant follicle resulting, in most cases, in monofollicular ovulation [67].
Letrozole is usually administered on days 3–7 of the menstrual cycle at doses of 2.5–7.5 mg per day in 2.5 mg increments [32]. Reported adverse effects include gastrointestinal disturbances, asthenia, hot flushes, headache and back pain [65]. Also, there are concerns regarding the potential teratogenic effect of letrozole, first raised at an American Society for Reproductive Medicine meeting in 2005 where an oral abstract presentation suggested that the use of letrozole for infertility treatment may be associated with a higher risk of congenital cardiac and ‘bone’ malformations in newborns [68]. However, two subsequent publications on letrozole use for ovulation induction did not find any increased risks of fetal anomalies [69,70].
A single RCT has compared aromatase inhibitors with placebo in women with PCOS [71]. This RCT compared one treatment cycle of letrozole or placebo in 36 CCR women with PCOS and found a higher ovulation rate per patient with letrozole (33 vs 0%; p = 0.006) but no difference in pregnancy or live-birth rate per patient. There are no published RCTs comparing aromatase inhibitors with placebo or no treatment in therapy-naive women with PCOS.
There are conflicting data on letrozole compared with anastrozole for ovulation induction in women with PCOS, as evidenced by two RCTs comparing these two medications in CCR women with PCOS. One RCT found a higher ovulation and pregnancy rate per cycle with letrozole [72], while no difference in the same outcomes was observed in the other RCT [73]. A recent RCT comparing two different doses of letrozole (5 vs 7.5 mg daily for 5 days) in 67 therapy-naive PCOS women found no difference in ovulation, pregnancy, multiple pregnancy or miscarriage rates [74]. Another RCT compared letrozole 5 mg daily for 5 days with letrozole 2.5 mg daily for 10 days in 218 CCR women with PCOS (444 cycles) and demonstrated a higher pregnancy rate per cycle with the 10-day treatment course (17.4 vs 12.4%; p = 0.03) with no difference in ovulation, miscarriage or multiple pregnancy rates [75].
Letrozole has been compared with clomifene citrate in six RCTs in women with PCOS who were either therapy naive [76,77], CCR [78], or where the type of patient with PCOS was not reported [79–81]. In the therapy-naive women, letrozole was found to have either a higher ovulation rate per cycle (60 vs 32%; p = 0.009) [77] or no difference in ovulation and pregnancy rates compared with clomifene citrate [76,77]. Letrozole was also found to have a higher ovulation rate per patient (62 vs 37%; p < 0.05), but no difference in pregnancy rate per patient in CCR women with PCOS [78].
In the three RCTs where the type of woman with PCOS were not reported, letrozole was demonstrated to have a higher ovulation (82 vs 64%; p = 0.016) and pregnancy (22 vs 9%; p = 0.037) rate per patient in one of the trials [79], but the other two trials showed no difference in ovulation or pregnancy rates per cycle [81] or patient [80]. None of these six RCTs comparing letrozole with clomifene citrate were a priori powered to detect a difference in ovulation or pregnancy rates. A recently published systematic review and meta-analysis comparing letrozole with clomifene citrate in PCOS identified six RCTs (841 patients who were a mixture of therapy naive, or type of patient with PCOS was not reported) and found no difference in ovulation, pregnancy and miscarriage rates between the two treatments. However, the 95% CIs for these outcome measures were wide and, therefore, an important difference between these two treaments is not able to be excluded [82].
Letrozole has also been compared with combined clomifene citrate and metformin treatment with no differences being demonstrated in ovulation or pregnancy rates per cycle in a RCT of 250 CCR women with PCOS over three treatment cycles [83]. Lastly, two RCTs have assessed letrozole versus laparoscopic ovarian drilling over 6 months in 260 and 147 CCR women with PCOS, respectively, and found no difference in pregnancy or live-birth rates [84,85].
Therefore, there is insufficient evidence to date to recommend letrozole over clomifene citrate in the treatment of infertile anovulatory women with PCOS in general, or specifically in therapy-naive or CCR women with PCOS.
Future perspective
The best evidence to date on reproductive outcomes in PCOS women supports lifestyle treatment as the primary therapy in overweight/obese women with PCOS and clomifene citrate as first-line pharmacological therapy.
The evidence supporting lifestyle interventions (diet, exercise, behavioral management techniques for modifying diet or exercise, or a combination of these) comes predominantly from observational studies with very few and small RCTs comparing reproductive outcomes in infertile overweight/obese PCOS women. Further quality research of lifestyle interventions in PCOS is needed in the form of well-designed, adequately powered RCTs of sufficient long-term duration that assess and report on reproductive outcomes such as ovulation, pregnancy, miscarriage and live birth.
The indication for metformin in the treatment of anovulatory infertile PCOS women is currently limited according to findings from RCTs and meta-analyses of RCTs. Further RCTs are needed to address whether metformin alone improves live-birth rates in overall PCOS women and whether there is a difference in efficacy between metformin and clomifene citrate in therapy-naive PCOS women with a BMI <30 kg/m2.
At present, there is insufficient evidence to support the routine use of aromatase inhibitors in either therapy-naive and CCR PCOS women and only further quality research in the form of well-designed and adequately powered RCTs for important reproductive outcomes, such as pregnancy and live-birth rates, will determine whether there is a role for aromatase inhibitors in the treatment of anovulatory PCOS.
Therefore, despite a large number of RCTs and systematic reviews of RCTs having been published assessing the effectiveness and safety of a number of lifestyle and pharmacological treatments for anovulatory infertile women with PCOS, there remains a need for a great deal of further high-quality research into these treatments, including in specific types of PCOS women with anovulation, in order to improve our management of the infertile PCOS woman.
Executive summary
Polycystic ovary syndrome (PCOS) is the most prevalent endocrine disorder in women of reproductive age and the most common cause of anovulatory infertility.
A diagnosis of PCOS according to the most current agreed international definition (Rotterdam Consensus on Diagnostic Criteria for PCOS) is based on at least two of the following three criteria: oligo-ovulation and/or anovulation; clinical or biochemical evidence of hyperandrogenism; and polycystic ovaries on ultrasound assessment with the exclusion of other causes of oligo-anovulation or hyperandrogenism.
Approximately 75% of PCOS women suffer from infertility due to anovulation.
A number of infertility treatments for PCOS have been proposed including lifestyle change, pharmacological (clomifene citrate, metformin, aromatase inhibitors and exogenous gonadotropins), surgical (laparoscopic ovarian surgery and bariatric surgery) and IVF.
Lifestyle change is generally considered the first-line treatment for the management of PCOS women who are overweight or obese and may improve ovulation and pregnancy.
Lifestyle interventions include diet, exercise, behavioral management techniques for modifying diet or exercise, or a combination of these.
In infertile overweight/obese PCOS women, the randomized evidence does not support either starting treatment with ovulation induction medications, such as clomifene citrate or metformin, ahead of lifestyle treatment alone or starting lifestyle treatment in combination with such ovulation induction agents compared with lifestyle treatment alone.
Clomifene citrate should be used as the first-line pharmacological therapy to improve fertility in anovulatory PCOS women with no other infertility factors.
If ovulation cannot be achieved with clomifene citrate, then the patient is said to have clomifene citrate resistance. If pregnancy cannot be achieved after six ovulatory cycles with clomifene citrate, then the patient is described as having clomifene citrate failure.
Studies with clomifene citrate have shown an ovulation rate of 60–85% and a pregnancy rate of 30–50% after six ovulatory cycles, with an increased risk of multiple pregnancy of approximately 5–7%.
Metformin alone has been shown to improve ovulation and clinical pregnancy rate but not live-birth rate.
Clomifene citrate is superior to metformin in all PCOS women, including PCOS women with a BMI >30 kg/m2 while the randomized controlled trial (RCT) data are conflicting in PCOS women with a BMI <30 kg/m2 and further RCTs are needed to assess whether metformin is equally effective to clomifene in this group of patients.
Metformin combined with clomifene citrate has a higher ovulation, pregnancy and live-birth rate compared with clomifene citrate alone only in clomifene citrate-resistant (CCR) PCOS.
Fertility outcomes are improved with the addition of clomifene citrate to metformin compared with metformin alone in obese PCOS women.
There is no improvement in fertility outcomes when metformin is combined with laparoscopic ovarian drilling, gonadotropin ovulation induction with timed intercourse, or IVF. However, metformin coadministration with IVF results in a 70–80% lower risk of ovarian hyperstimulation syndrome.
Gonadotropin therapy is an effective second-line therapy in infertile anovulatory PCOS women with clomifene citrate resistance or clomifene citrate failure.
Recent randomized evidence suggests that gonadotropin therapy may be more effective than clomifene citrate in therapy-naive PCOS women.
There is currently insufficient evidence to recommend letrozole over that of clomifene citrate in infertile anovulatory PCOS women in general or specifically in therapy-naive or CCR PCOS women.
There is conflicting published data on the potential teratogenic effect of letrozole when used as an ovulation induction agent.
The best evidence to date on fertility outcomes in PCOS women supports lifestyle treatment as the primary therapy in overweight/obese women with PCOS and clomifene citrate as first-line pharmacological therapy.
There remains a need for a further high-quality research in the form of well-designed, adequately powered RCTs of sufficient long-term duration that assess the effectiveness and safety of infertility treatments for anovulatory women with PCOS despite a large number of RCTs and systematic reviews of RCTs having been published.
Examples of such high quality research required include lifestyle interventions in overweight/obese PCOS women, metformin versus clomifene citrate in therapy-naive PCOS women with a BMI <30 kg/m2, and the use of aromatase inhibitors in either therapy-naive and clomifene citrate-resistant PCOS women.
Footnotes
Acknowledgements
The authors would like to thank and acknowledge N Peirce and C Leahy for their help in the preparation of the references for this manuscript.
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
