Abstract
Breast cancer is currently the most frequent indication for intraoperative radiotherapy with increasing numbers worldwide. Intraoperative radiotherapy can be used as a tumor bed boost followed by whole breast radiotherapy, or as a distinct form of accelerated partial breast irradiation in selected patients. This article summarizes the theoretical background including pattern of recurrence and distribution of tumor cell foci in the breast and discusses the rationale for intraoperative radiotherapy, especially using a miniature x-ray generator (Intrabeam®). The concepts of how to avoid geographic and temporal miss by giving radiotherapy during surgery to the open wound cavity are described. Experimental and clinical experience is presented based on in vitro experiments and more than 300 treated patients in a single department with mature follow-up.
Keywords
Intraoperative radiotherapy (IORT) has increased tremendously in clinical use and significance with the development of mobile radiotherapy devices (e.g., Liac, Novae 7, Mobetron and Intrabeam®) approximately 10–15 years ago. Breast cancer has become the most frequent indication for IORT over the last decade. Meanwhile, more than 100 centers worldwide use a miniature x-ray generator (e.g., Intrabeam®, Carl Zeiss Surgical, Oberkochen, Germany) (

Miniature x-ray generator (Intrabeam®, Carl Zeiss Surgical, Oberkochen, Germany) in preparation for intraoperative radiotherapy during breast-conserving surgery.
Theoretical background
BCS followed by external beam radiotherapy (EBRT) to the whole breast has almost completely replaced radical mastectomy in suitable patients. EBRT following BCS using a dose of 50 Gy in standard fractions improves local control, breast cancer-specific survival and overall survival in these patients compared with BCS alone [4]. At present, local recurrence rates of less than 5% after 5 years are typically achievable (

The local recurrence rate in all 337 breast cancer cases undergoing external beam radiotherapy after standard breast-conserving surgery in the Department of Radiation Oncology, University Medical Centre Mannheim (Germany) in the years 2005 and 2006.

For high-risk patients (i.e., younger patients, larger tumor and node positivity), it was shown that an additional dose to the tumor bed could improve local control. Therefore, the tumor bed boost is widely accepted as standard of care [8].
For low-risk patients, the situation is still controversial. It was speculated that radiotherapy may be omitted completely in selected patients. However, all randomized studies have shown significantly poorer local control rates when EBRT after BCS was omitted even in low-risk patients (Table 1) [9–12]. At present, several randomized studies are investigating the concept of a limited radiotherapy dose to the tumor bed and its immediate surroundings (e.g., TARGIT [102], ELIOT [13], GEC-ESTRO [103], NSABP B-39 [104]).
Can we do without external beam radiotherapy after breast-conserving surgery?
All randomized trials comparing TAM alone with TAM + external beam radiotherapy in highly selected patients showed significantly inferior local control rates.
AI: Aromatase inhibitor; IBTR: In breast tumor recurrence; RT: Radiotherapy; TAM: Tamoxifen.
Rationale for IORT
The elimination of geographic and temporal miss are the two most important advantages of delivering radiotherapy during BCS. Geographic miss is a relevant challenge in day-to-day clinical practice, especially for the localization of the tumor bed boost when radiotherapy is given after chemotherapy, which may last several months, or when oncoplastic reconstruction methods have been used during BCS [14]. Targeting the tumor bed during surgery before oncoplastic reconstruction with the surgeon and radiation oncologist both present in the operating room eliminates this source of reduced local control.
The concept of temporal miss is based on the fact that a delay of radiotherapy by several weeks reduces local control significantly [15] and that the IORT boost reduces overall treatment time by 1–2 weeks compared with a percutaneous fractionated boost. Furthermore, there is no tumor cell proliferation before and/or during radiotherapy, which can be observed before and during fractionated radiotherapy which is protracted over several weeks and is started several weeks to months after surgery. In addition, there is the theoretical concept of local cure by the IORT boost, which is depicted and explained in Figure 4 [16].
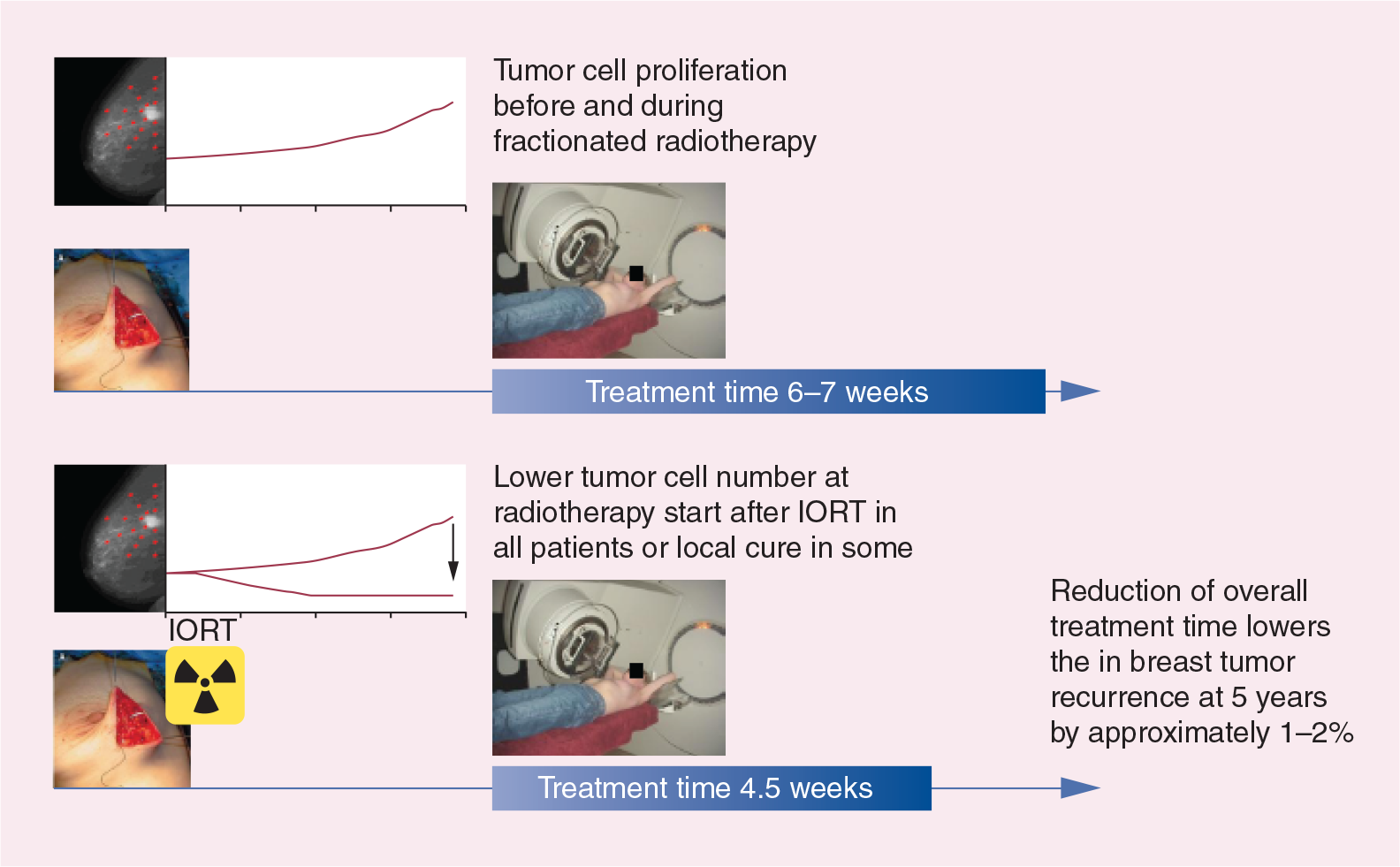
Rationale for Intrabeam
There is an ongoing controversial debate based on the considerable uncertainty regarding the optimal target volume concepts and the target volume definition for partial breast radiotherapy [17–21]. There are some widely accepted facts, based on systematic pathologic studies, about tumor cell foci outside of the macroscopic tumor [16]. Approximately 40% of all breast cancer patients have no additional disseminated tumor cell foci in the ipsilateral breast. Around 20% have tumor cell foci within a radius of 2 cm and approximately 40% of the patients have tumor cell foci disseminated in the whole breast (Figure 5A). However, this number is certainly lower in patients with small ductal invasive tumors lacking an extensive intraductal component. Regarding the most common histological subtype (i.e., ductal invasive carcinoma), it is reasonable to assume that due to the ductal growth and distribution pattern, there is an exponential decline of tumor cell density outside of the macroscopic tumor (
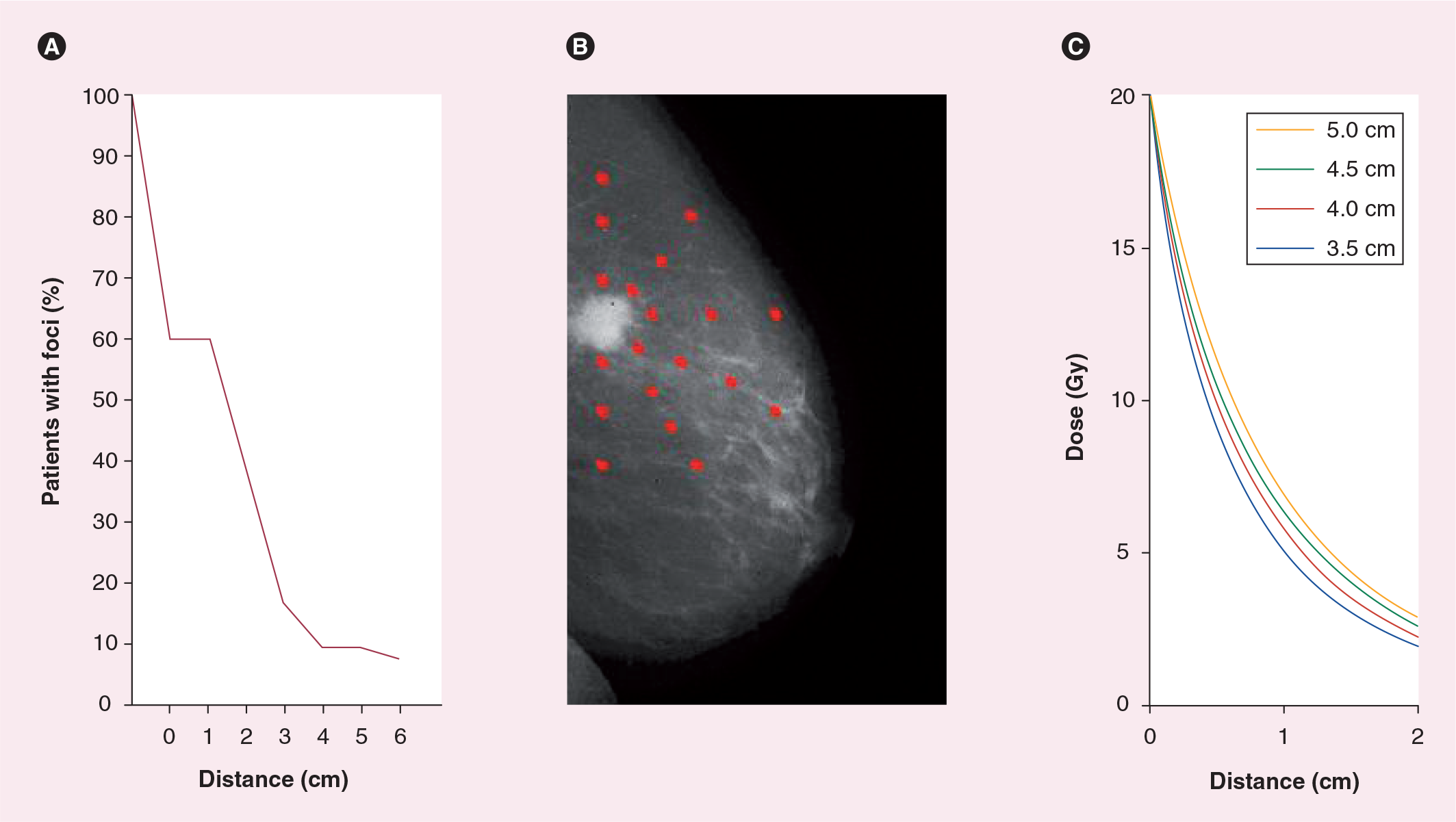
Experimental background
Traditionally, radiation oncologists are reluctant to apply high single doses, especially to late-reacting tissues such as brain, spinal cord or breast. However, clinical experience from radio-surgery and stereotactic ablative radiotherapy has demonstrated that, as long as the volume is small, high single doses are highly efficient in sterilizing even macroscopic tumors while being well tolerated.
There is a special radiobiological situation in IORT with the Intrabeam system regarding the expected normal tissue toxicity [22–24]:
A high single dose is delivered with a low dose rate (20–50 min) allowing significant repair in normal tissues during IORT;
The dose is limited to a small volume due to the steep dose drop off of 50 kV x-rays;
The relative biological effectiveness of 50 kV x-rays is higher compared with standard 6 MV x-rays from linear accelerators.
After extensive radiobiological modeling, these results have been experimentally validated [22,23]. One may assume that the chest wall thickness completely shields the lung and heart from relevant radiation doses, and that the skin should be kept at least 5–10 mm away from the applicator to avoid the induction of fibrosis.
Clinical experience
The clinical experience using low-energy x-rays for IORT in breast cancer has now lasted for more than 10 years, when the procedure was pioneered by the Vaidya et al. [3]. Several pilot studies, case series and the randomized Phase III TARGIT trial have been published [25–34]. In this section, we summarize our updated experience regarding the clinical indications for IORT as a boost, IORT as accelerated partial breast irradiation ‘only’ and IORT during a second BCS in patients with previous radiotherapy to the breast.
IORT as a boost
The TARGIT study group (with centers in London, Mannheim, Perth, New York and Aviano) has prospectively treated and followed approximately 300 pilot patients receiving IORT as a tumor bed boost followed by EBRT It was a group with a relatively high-risk for local recurrence (~30% younger than 50 years of age; ~30% T2 tumors; and ~30% node positive). The median follow-up was more than 5 years. The 5-year local recurrence rate was only approximately 2% [29,31–33], demonstrating the efficacy of this approach. We now have a single center experience of more than 200 patients with a median follow-up of 48 months (
Rates of ipsilateral breast recurrences after intraoperative radiotherapy during breast-conserving surgery.
Rates are in more than 300 patients with a follow-up of more than 18 months (median follow-up of 48 months).
BCS: Breast-conserving surgery; EBRT: External beam radiotherapy; IBTR: In breast tumor recurrence; IORT: Intraoperative radiotherapy; RT: Radiotherapy.
IORT as accelerated partial breast irradiation ‘only’
TARGIT was initially given during a pilot phase as a boost to the tumor bed to assess its safety and feasibility, and immediately following this the TARGIT A trial was launched in March 2000 [29,31]. IORT alone was initially given in parallel to 80 patients in whom no EBRT was possible [34]. This included patients with comorbidities such as collagen vascular disease or metastases at first presentation. After a follow-up of more than 3 years, the local recurrence rate in these patients was 1.5%.
The international prospectively randomized Phase III trial (TARGIT A) recruited more than 2200 patients over 10 years [30]. Older patients with small, bioptically verified ductal invasive breast cancer that appeared unifocal on imaging studies were entered. Patients were randomized into the standard arm (BCS followed by EBRT and systemic therapy) or into the risk-adapted experimental arm. These patients received IORT during BCS and when the final histopathological report revealed additional risk factors that were not known before surgery, EBRT was added as per protocol. The first analysis of the trial was published in June 2010 and demonstrated a 4-year local recurrence rate of approximately 1% in both arms [30]. The noninferiority criteria for the novel risk-adapted approach were met. In Germany, the inclusion criteria were stricter as compared with other countries, and we have not seen any recurrence in our single-center experience (
IORT during a second BCS
Patients suffering from recurrent breast cancer in a previously irradiated breast (e.g., from previous breast cancer or Hodgkin's disease), are typically recommended to undergo mastectomy even when the second cancer is detected at a very small size. This is based on the expected toxicity of a second course of whole breast radiotherapy. We have offered a second, breast-conserving approach with IORT to 22 patients and could demonstrate that conservation of the breast can be achieved in 90% of the cases at 4-year follow-up (
Conclusion
Although there are already several consensus statements of national and international societies regarding the use of accelerated partial breast irradiation ‘only’ outside of clinical studies (e.g., ASTRO, ASBS, ABR, GEC-ESTRO and DEGRO), the TARGIT study group is currently still offering the possibility to enter patients in the trials to prolong follow-up and to fill additional subprotocols. At the end of May 2011, there have been almost 3000 patients randomized in the TARGIT A trial. There will be a sub analysis of the German modification (≥50 years, ≤2 cm), and elderly patients can be entered in the single-arm TARGIT E trial (NCT01299987) (

Future perspective
Accelerated partial breast irradiation ‘only’ using IORT with low-energy photons will become the standard of care for selected patients with early breast cancer, assuming that the noninferiority to EBRT will be demonstrated with mature follow-up.
Executive summary
Intraoperative radiotherapy as a tumor bed boost: the rationale is the lack of geographic and temporal miss. Clinical experience shows very low local recurrence rates with an acceptable rate of side effects.
Intraoperative radiotherapy as accelerated partial breast irradiation ‘only‘: the rationale is the predominance of local recurrences at the initial tumor bed in selected patients. First results from randomized trials demonstrated noninferiority to the standard approach. Further studies are ongoing.
Footnotes
Carl Zeiss Surgical/Oberkochen supports radiobiologial research at the University Medical Centre Mannheim (F Wenz). The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
