Abstract
Monitoring bone turnover of the adult and aging skeleton is essential for optimal treatment of bone metabolic diseases, such as postmenopausal osteoporosis. Diagnosis of osteoporosis is based solely on dual-emission x-ray absorptiometry-based measurements of bone mineral density. However, within the last 20 years, biochemical markers of bone turnover have been implemented to a larger degree, and especially within the field of drug development. Numerous clinical studies have underscored that the markers have promise in terms of predicting patients at high risk of losing bone, future fracture events and importantly also the fracture efficacy of drugs in development. Furthermore, while classical methods often require years to monitor the changes, the bone turnover markers do so within a shorter time span. The aims of this article are to provide an update on the different biochemical markers of bone turnover, and to give an overview of their applications in epidemiological and clinical research especially in women. The main emphasis will be on their utility in clinical trials testing the efficacy of drugs for the treatment of osteoporosis, and their ability to supplement bone mass measurements. Finally, recent evidence suggests that biochemical markers may provide information on bone age that may indirectly relate to bone quality, and this is discussed together with future possibilities for measuring bone quality using bone turnover markers. In summary, a more targeted use of biomarkers could assist in the identification of high-risk patients, the process of drug discovery and monitoring of the efficacy of osteoporosis treatment in clinical settings.
Introduction to bone remodeling & osteoporosis
Maintenance of a healthy skeleton depends on bone resorption and bone formation. Under normal circumstances resorption and formation are tightly coupled, and resorption is always followed by the same amount of formation leading to complete replacement of the removed bone [1,2]. Loss of regulation of this process leads to pathologies, such as osteoporosis (OP) [2,3].
Osteoporosis is a large health problem, and it is estimated that roughly 10 million Americans >50 years of age have OP and another 34 million Americans are osteopenic [4]. All in all this accounts for approximately 1.5 million osteoporotic fractures in the USA each year and the relative numbers of OP and osteopenia are thought to be similar in the UK [4]. With respect to gender differences it is estimated that in persons >50 years 50% of all women and 20% of all men will experience an osteoporotic fracture [4].
The main cause of OP is loss of sex steroid production, which both in women and men is associated with increased degradation of the bones, although the process occurs more rapidly in women than men [5,6]. Additional causes of OP are numerous, and these include genetic predisposition, ethnicity, aging, smoking, prolonged bed rest, alcohol intake and drugs, such as glucocorticoids [7].
Osteoporosis is a systemic skeletal disease characterized by low bone mass and microarchitectural reduction of bone tissue, leading to enhanced bone fragility and a consequent increase in fracture risk [5].
The diagnosis of OP is inherently linked to low bone mass [2]. Bone mineral density (BMD) is noninvasively quantified by dual-energy x-ray absorptiometry; however, low BMD is only a moderate predictor of fractures, and while initiatives, such as the FRAX® tool are used, the ability to predict fractures is still somewhat limited [8]. The FRAX tool takes a series of parameters relevant for fracture prediction, such as age, gender, prior fractures, BMD, family history of fractures, BMI, smoking, alcohol, rheumatoid arthritis and glucocorticoid use into account [8]. However, FRAX does not include any biochemical marker, a finding based on the high natural variation in these, highlighting the need for more markers predicting fracture risk, and the potential for fracture reduction by treatments [9]; however, inclusion of biochemical markers of bone turnover (BTMs) into FRAX is of relevance, and should be considered carefully.
Treatments for OP are numerous, and include antiresorptives, such as bisphosphonates, estrogen, selective estrogen receptor modulators (SERMs), denosumab and calcitonin, as well as anabolic molecules, such as parathyroid hormone (PTH)1-34 and PTH1-84. In addition, new entities, such as cathepsin K and ClC-7 inhibitors, as well as anti-sclerostin antibodies, which all work through novel modes of action, are at various stages of development, and extensively use biochemical markers to underscore their modes of action [3].
The aim of this article is to provide an up-to-date summary of recent developments in the field of bone turnover markers, while providing a detailed discussion of their ability to predict fractures, as well as their application in drug development.
Rationale for BTMs
Osteoporosis is a disease characterized by slow progression, and hence the changes in BMD are small, and when performing clinical studies the small BMD changes and low numbers of fractures necessitate large numbers of study participants [10].
In contrast to imaging techniques, BTMs obtained in serum or urine samples show markedly larger changes compared with the imprecision of the assay (<8–10%). Typical magnitudes of effects are decreases of 50–80% or increases of 100–200% shortly (days to weeks) after initiation of treatment with antiresorptive or anabolic drugs, respectively [11]. By contrast, the changes in BMD range from 6 to 7% after 2 years of bisphosphonate therapy [12] to 2–3% or less for SERMs agents and calcitonin [13,14], changes which are fairly small taking into account the assay coefficient of variation of 1–2%.
Hence, biochemical markers provide a superior signal-to-noise ratio, and therefore can be used to shorten trials and to decrease study cohorts in size, although the variability of these prevents them from being used at the individual level, as opposed to imaging-based techniques, such as dual-emission x-ray absorptiometry [15,16].
Another important point in the use of BTMs is the ability to measure both the formation and the degradation of any given matrix at a specific time point, hence providing information about the turnover of the tissue, rather than a static image of volume or density (
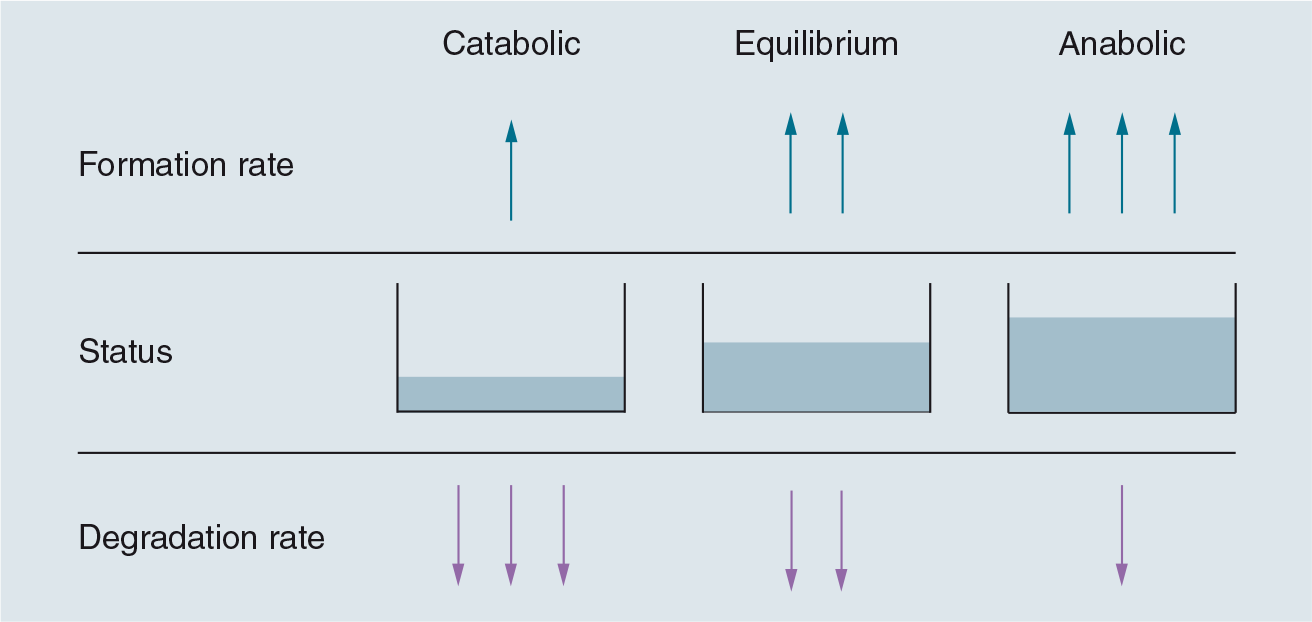
Tissue condition measured by bone mineral density.
Biochemical markers of bone turnover
The bone field is one of the few fields where several types of well-validated BTMs exist. The markers of bone turnover can be divided into three categories:
Bone resorption markers
Markers that reflect osteoclast number
Bone formation markers
Bone resorption markers are released from the bone matrix during osteoclast-mediated bone resorption. During bone resorption cathepsin K is secreted by the osteoclasts, which leads to massive proteolysis resulting in the release of collagen fragments from bone. The two most commonly used bone resorption markers are the C-terminal crosslinked telopeptide of type I collagen (CTX-I) fragment and the N-terminal telopeptide of type I collagen fragment [15,16].
C-terminal crosslinked telopeptide of type I collagen is a rather unique biomarker in that it combines a series of processing steps occurring in type I collagen. First, and as mentioned previously, CTX-I is a cathepsin K-generated fragment and the antibody specifically recognizes this small fragment. Second, the epitope contains crosslinks between the lysines in the sequences, and third, the DG motif within the CTX-I sequence (1207EKAHDGGR1214) isomerizes with increasing age of the collagen fibers [17], and all of these changes are monitored in the CTX-I assay [10]. Two forms of CTX-I can be measured, one with no isomerization called α-CTX-I, which reflects resorption of young bone, and one with isomerization called β-CTX-I, which reflects resorption of old bone [10].
Deoxypyridinoline, pyridinoline and hydroxyproline levels also reflect bone resorption; however, they are less sensitive than CTX-I and N-terminal telopeptide of type I collagen [18]. Furthermore, hydroxyprolines are released from all types of collagen, and hence are markedly less specific [19,20].
The matrix metalloproteinase-generated C-terminal telopeptide of type I collagen fragment has been used to monitor bone resorption under certain circumstances, such as the resorption induced by bone metastasis and rheumatoid arthritis; however, it does not reflect the increase in resorption seen in OP, potentially indicating that it is not directly derived from bone resorption, but from the inflammation occurring under these circumstances [21].
Markers of osteoclast number, rather than function, are tartrate resistant acid phosphatase (TRACP) 5b and, potentially, cathepsin K. These enzymes are produced and released into the circulation by the osteoclasts [22].
Tartrate resistant acid phosphatase 5b, which is the osteoclast-specific isoform of total TRACP, is the most commonly used marker of osteoclast number. Cathepsin K is still undergoing evaluation as an osteoclast marker [22,23] and further analysis will show whether it correlates with resorptive activity or osteoclast number.
The bone formation markers are related to two aspects of osteoblast function, namely differentiation and bone formation. The markers reflecting differentiation are molecules, such as bone-specific alkaline phosphatase and osteocalcin [15,16], while the markers reflecting bone formation directly are fragments of procollagen released during collagen incorporation into the newly formed bone matrix. These include the procollagen type I N-terminal propeptide (PINP) and procollagen type I C-terminal propeptide (PICP) [15,16]. Of importance is that a recent study showed that PINP is also derived from the liver [24], and hence this information should be included when analyzing data from this marker.
In addition to these classical markers of bone turnover, a couple of new markers have been investigated, and these are soluble inhibitors of the Wnt signaling system, such as DKK1 and sclerostin. Early studies of DKK1 indicate that it shows an inverse correlation with BMD at different sites, in alignment with its role as a bone formation inhibitor [16]. By contrast, sclerostin levels are positively correlated to BMD under some circumstances [25], while they are suppressed by PTH treatment [26]. These data underscore that more data are needed on these two markers before their pathological relevance becomes clear [27,28].
Monitoring disease status & prognosis in women
Biochemical markers of bone turnover levels can be measured in either urine or serum or both and importantly have been shown to be increased in women after menopause, correlating well with the acceleration of bone turnover seen in this group [15,16].
Furthermore, in postmenopausal women bone turnover rates account for >50% of the variation in bone mass, while in premenopausal women this percentage is between 0 and 10% of the variation in bone mass, supporting the idea that BTMs provide information in addition to the BMD measurements [29].
Clinical investigations support an inverse correlation between BTMs and BMD, and a series of prospective studies demonstrated correlations between levels of markers and rates of bone loss assessed by serial BMD or bone mineral content measurements conducted at different skeletal sites over a time period spanning 13 years [30]. This again supports the idea that BTMs provide additional information to BMD measurements. The inverse association of BTMs to BMD becomes stronger with advancing age, especially in postmenopausal women and importantly it is markedly stronger for resorption markers than for formation markers [31], likely due to the fact that the acceleration of bone resorption exceeds the acceleration of bone formation [32].
Of importance is that the inverse correlation between BTMs and BMD does not seem to apply to all skeletal sites. In groups of untreated postmenopausal women, a significant correlation between baseline measures of bone turnover markers and the subsequent rate of bone loss at the hip or wrist was demonstrated [33,34], while this did not apply to the lumbar spine [30].
Another study indicated that bone resorption markers could predict rapid bone losers (>3% loss in BMD per year) in a follow-up period of 2–12 years [35]; however, the understanding if this prediction is still somewhat limited, as it is not completely clear whether the predictive value applies to all skeletal sites [31].
Bone mineral density is the recommended and applied method for assessing fracture risk, although the FRAX algorithm is implemented on some regions. However, a large percentage (33–50%) of the fragility fractures happen in patients with T-scores in the osteopenic, and even some in the healthy, range [36,37]. In this relation, the BTMs have been shown to have predictive power independent of other parameters [29,38], and an increasing number of studies have pointed out that fracture risk is also reflected by BTMs [15,16].
Biochemical markers of bone turnover levels were shown to be an independent predictor of fracture risk in postmenopausal women, as illustrated by the Os des Femmes de Lyon (OFELY) study, with high levels of BTMs indicating a 10-year probability of 26% for fractures, a prediction that was strengthened when combined with low BMD and/or prior fractures [39].
In the Epidemiology of Osteoporosis (EPIDOS) study, markers of bone resorption were evaluated for prediction of risk for hip fracture in elderly women, and it was shown that increased levels of bone resorption markers predicted increased incidence of hip fracture, independent of initial bone mass [29]. Furthermore, combining CTX-I measurements with history of prevalent fractures was shown to perform as well as hip BMD measurements in terms of predicting the risk of future hip fractures in elderly women [40]. Furthermore, another group found that vertebral fractures could be predicted in postmenopausal osteoporotic women using BTMs [41].
Combined with BMD, age and/or prior fracture rates, BTMs contribute to an improvement in the prediction of risk of fracture at least in women, which is highly useful when designing clinical studies, as the selection of the cohort is simplified by stringent selection criteria and risk stratification [10,29,38]. However, the extent to which the fracture prediction holds is still somewhat unclear, with relatively contradicting evidence in terms of different skeletal sites [29,38,42].
Monitoring response to treatment
The largest application of BTMs is within the response to treatment category, (i.e., the efficacy markers), where the ultimate goal is the prediction of fracture prevention [10]. Classically, two types of marker profiles have been sought:
The antiresorptive profile consisting of a primary reduction in bone resorption markers followed by a secondary reduction in bone formation markers, due to the coupling between these processes [3];
The anabolic profile consisting of an induction in bone formation markers and then secondarily an increase in bone resorption markers [15]. This is often perceived as a coupling effect but is rather a consequence of the fact that the only marketed anabolic molecules are different forms of PTH, which are known to stimulate bone resorption through induction of RANKL production [3,43].
During the development of novel treatment candidates for postmenopausal OP in women, such as the cathepsin K inhibitor odanacatib, the oral formulation of salmon calcitonin, the antisclerostin antibody AMG785, glucagon-like peptide 2, the c-src inhibitor AZ0530, and inhibitors of the chloride channel ClC-7, the BTMs are used extensively, not only for assessing efficacy of the molecules, but also to profile and thereby distinguish these molecules from already existing treatments [44–50].
Biomarker changes as a function of treatment.
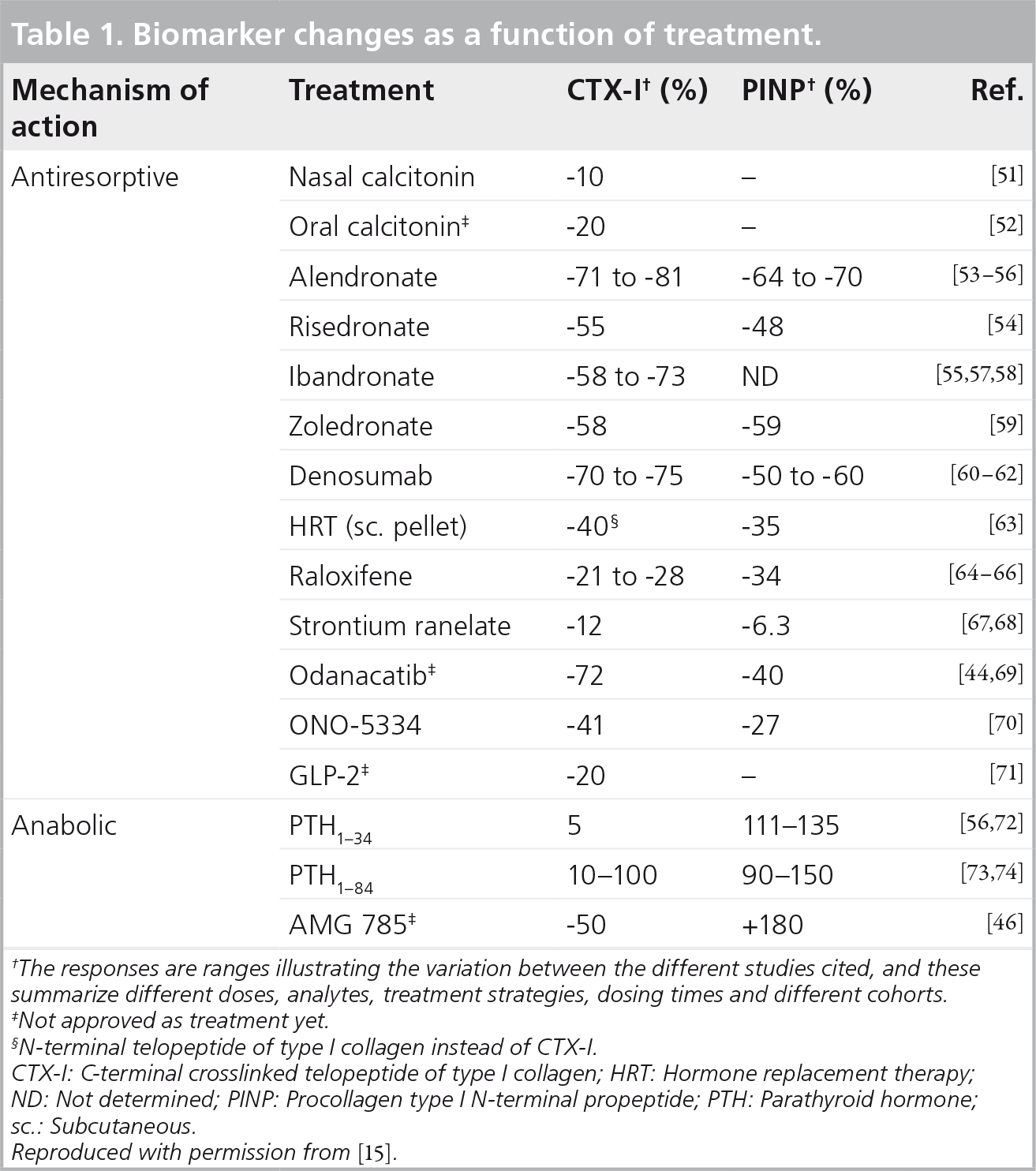
The responses are ranges illustrating the variation between the different studies cited, and these summarize different doses, analytes, treatment strategies, dosing times and different cohorts.
Not approved as treatment yet.
N-terminal telopeptide of type I collagen instead of CTX-I.
CTX-I: C-terminal crosslinked telopeptide of type I collagen; HRT: Hormone replacement therapy; ND: Not determined; PINP: Procollagen type I N-terminal propeptide; PTH: Parathyroid hormone; sc.: Subcutaneous.
Reproduced with permission from [15].
When examining the effects of the drugs on the BTMs, it quickly becomes evident that the changes in biochemical markers most often are clearly dependent on a series of parameters, such as dose of drug, type of drug, dosing frequency, dosing time, sampling frequency and analyte [15,75].
A highly relevant point is whether the BTMs are surrogate markers of BMD increases and fracture reductions as a function of treatment regimens. In this context a series of studies using different treatment strategies, such as alendronate, risedronate and raloxifene, have investigated whether these antiresorptives induce changes in CTX-I that are predictive [76–79]. In these studies the changes in resorption markers monitored within the first 6–12 months were associated with changes in BMD and fracture after 3–4 years, clearly showing that these changes are relevant surrogate markers, although reductions exceeding 50–60% failed to provide more information than reductions up to 50% [76–79]. In relation to anabolic therapy with teriparatide, the predictive value of changes was greatest when using bone formation markers, and in this case changes at 3 months correlated well with changes in BMD at 18 months [80]. Finally, in relation to strontium ranelate, for which the mode of action is still somewhat unclear, the marker with the best predictive ability was PICP, as 3-month changes in PICP predicted 3-year changes in BMD; however, none of the markers could predict fracture efficacy with strontium ranelate [81].
In summary, the BTMs possess the ability to predict clinical efficacy in women at the level of BMD gains and also fracture prevention within a 6–12-month time frame, phenomena which may require up to 3 years to monitor using BMD measurements [15]. At the same time they also provide information about the mode of action, which cannot be detected using other methods, such as bone histomorphometry and μ-computed tomography measurements [15,82].
Monitoring responses in individuals
The clinical interest in monitoring individual responses is large, as they really could help select responders to treatment, to monitor compliance and importantly also provide a surrogate marker for the overall magnitude of the response to treatment [10].
However, BTMs are, at present, not suitable for estimating BMD, bone loss or fracture risk in individuals subject, although adding the level of CTX-I or PINP could be a relevant in relation to the FRAX tool, as they provide information independent of these parameters [8]. The major issue in relation to the use of the BTMs in individuals is the intraindividual variation, which can be substantially reduced by knowledge about the markers, their variation as a function of circadian rhythm and food intake, for example [75]. Furthermore, in a longitudinal setting it appears likely that monitoring BTMs in individuals will provide useful information, such as those in relation to compliance to treatment [15].
In summary, although it is an enticing possibility to apply the BTMs in individuals, there is a need for more work on this aspect (i.e., the establishment of the least significant change within an individual, rather than an average least significant change) [15].
Elucidating the mode of action of drugs using BTMs
Recent studies have indicated that the coupling principle, which states that bone formation always follows bone resorption, needs a revision, and that the presence of osteoclasts, independent of their resorptive activity, is involved in stimulation of bone formation [3,49]. These findings were initially based on bone histomorphometry in patients with osteoclast-rich osteopetrosis [3,49]; however, later clinical studies of drugs in development have used biomarkers extensively to profile the mode of action [3].
Using a panel of BTMs including a marker of bone resorption, a marker of osteoclast number and a marker of bone formation, these clinical studies have highlighted that classical treatments, such as bisphosphonates, estrogen and SERMs, which all cause reductions in both resorption and osteoclast number, also cause reductions in bone formation, findings which are in accordance with both the classical and the revised perception of the coupling between osteoclasts, their activity and bone formation [3]. Furthermore, the recently approved treatment denosumab has also used this triad of BTMs to clearly delineate its mode of action, namely a massive suppression of osteoclast numbers, activity and secondarily bone formation [62]. Finally, a couple of molecules that are known not to reduce osteoclast numbers, namely oral salmon calcitonin, the cathepsin K inhibitor odanacatib and the c-src kinase inhibitor saracatinib, showed less or no inhibition of bone formation [44,52,83], which is supported by preclinical studies of inhibitors of osteoclastic acid secretion, such as ClC-7 inhibitors [50].
In summary, the application of a panel of BTMs during drug development will provide unique information about the mode of action of the drug, and therefore can be used to strengthen the position of the compound.
Limitations of the BTMs
The inherent variation of the BTMs is often listed as a major limitation of these systems, and there are two types of variation that need to be included in any analysis of these.
Sources of variation that are inherent parts of conducting clinical trials within the field of bone biology include: age and gender, menopause, diseases and drugs, fractures, prolonged bed-rest and immobility, all of which are known to alter the levels of the BTMs significantly [15,75,84]. Furthermore, a couple of easily controllable parameters, namely circadian rhythm and food intake, also induce major changes in the biomarker levels [15,75]. These variations have been discussed extensively by Vasikaran et al. as part of the work of the IOF-International Federation of Clinical Chemistry and Laboratory Medicine Bone Marker Standards Working Group [15], and will not be discussed in further detail.
By contrast, an interesting aspect of the variations is that if these are well described and fully understood, these can be used to identify unique traits of drugs in development. This point is best exemplified by the studies of the oral formulation of salmon calcitonin, where knowledge about the sensitivity of CTX-I to circadian rhythm and food intake was used to delineate the optimal dosing time in terms of maximizing efficacy [85–90]. This has also been done for glucagon-like peptide 2 [71].
Hence, in terms of the variations in these markers, there is a need to know the underlying causes of variation, but when these are known these can be used to help design cohorts, pinpoint the mode of action of a drug and more.
Conclusion
Biochemical markers of bone turnover are here to stay, and in several cases these have provided information that normally would have taken significantly longer time and more patients. One example is the changes in CTX-I versus the changes in BMD induced by antiresorptives. In addition, they provide information, which would not have been easily and dynamically obtained using other techniques, such as bone histomorphometry on an iliac crest biopsy versus a profile of biomarkers providing information on resorption, formation and osteoclast numbers. Furthermore, while most of the data are generated in women, it appears likely that they are applicable to men as well.
However, even with the IOF guidelines on which biomarkers to use for clinical studies, there is still a way to go before they can be implemented by the general physician as prognostic/diagnostic tests, although a few OP clinics have implemented these to monitor response to treatment. On the other hand, the applicability of the BTMs in drug development is both cost and time saving, as well as indispensable in terms of understanding a given drugs mode of action.
Future perspective
The IOF recently published their recommendations for the use of bone turnover markers in clinical studies, and CTX-I and PINP were chosen as the resorption and formation markers, respectively [15]. These markers were chosen based on their validation and predictive value in terms of fractures, and the underlying rationale for these selections is discussed extensively in other articles [15]; however, one issue related to these markers is their lack of ability to monitor bone quality.
However, recent data with respect to assessing bone quality using BTMs has indicated that the ratio of isomerized versus nonisomerized CTX-I fragments may provide information about the bone quality. Type I collagen undergoes isomerization in the DG motif of the CTX-I epitope in vivo and also in vitro [17,82,91–93], and the ratio of nonisomerized to isomerized CTX-I is an indicator of bone collagen age, and hence a strong indicator of bone turnover [82]. Interestingly, antiresorptive treatments have a wide range of effects on the systemic α-CTX-I/β-CTX-I ratio, with bisphosphonates suppressing the ratio strongly, while hormone replacement therapy, raloxifene and calcitonin treatments had a markedly smaller effect, if any at all [94,95]. However, the ability of the ratio to predict overall fracture risk, as well as fracture risk as a function of treatment still remains to be investigated [82].
Another post-translational modification occurring in type I collagen is the addition of advanced glycation end products (AGEs) [96]. While AGEs are classically associated with diabetes-related pathologies [97], more recent research has indicated that the level of AGEs present in bone matrix is indicative of loss of bone quality [98,99]. Pentosidine, the most prominent AGE found in bone so far, is a cross linking AGE, as it introduces molecular bonds between two type I collagen chains [100,101]. Studies have shown that pentosidine crosslinks are numerous in aged bone samples and that these cause a deterioration of the functionality of the molecules [100,102]. These crosslinks are associated with increased stiffness of the collagen helices, which in bone is associated with increased probability of fracture [101,102]. In terms of using pentosidine as a biochemical marker, OP patients have been found to have elevated serum levels of pentosidine [103]; however, it is still somewhat controversial as some studies have indicated that pentosidine can predict fracture risk [104] whilst others have not [105]. Hence, more research on AGEs as biomarkers of bone quality is needed.
Furthermore, the markers of osteoclast number, such as TRACP 5b and cathepsin K, are not widely applied, although studies have indicated that they provide vital information on the mode of action of drugs, especially in relation to the new categories of antiresorptives that do not reduce osteoclast numbers [3,22]. Whether the lack of application of TRACP 5b and cathepsin K is because they are still novel and need more validation, because the analyte is unstable, as seen for TRACP 5b, or because the analytes measured in the assays are still somewhat obscure due to the multiple forms of either enzyme remains to be seen [23,53,106–108]. However, in this context assays specifically measuring the active forms of enzymes are of great interest.
Executive summary
Bone turnover is an essential process for the maintenance of skeletal integrity.
Bone turnover can be monitored using biochemical markers of bone turnover (BTMs).
BTMs can be assessed easily in serum/urine.
The changes in BTMs occur markedly faster than the changes in bone mineral density and fracture rates.
The bone resorption marker, C-terminal crosslinked telopeptide of type I collagen has shown some ability to predict future bone loss and fracture events.
The BTMs are highly relevant in terms of both monitoring and predicting response to treatments.
The BTMs provide a unique possibility to study the mode of action of a given study drug in a clinical situation.
The BTMs are subject to fairly large individual variation; however, this can be controlled to a large extent by careful study design.
A BTM monitoring bone quality is still missing; however, promising candidates are being studied at present.
Footnotes
All authors are employees of Nordic Bioscience. MA Karsdal and C Christiansen own stocks in Nordic Bioscience. The authors received funding from the Danish Research Foundation for this work. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
