Abstract
The technical development of instruments for endoscopic surgery started in the field of gynecology. In the early 1970s, with the improvement of optics and instruments for laparoscopic surgery, gyne-endoscopic surgery developed and set milestones for all other surgical fields. However, the general surgeons propagated the advantages of 2D or 3D imaging surgery much better than the conservative gynecologists. Surgery on a 2D screen without direct vision is regarded as more advantageous than open surgery and has achieved wide acceptance. Several schools of gynecologic endoscopy in Europe (in Kiel, Giessen, Clermont Ferrand and Strasbourg) have set guidelines for gyne-endoscopic surgery. Our catalog of indications in the areas of gyne-endoscopic surgery, published in 2002, reveals the broad application of these techniques today. 3D vision, robotic instruments and systems, such as the da Vinci® Surgical System from Intuitive Surgical, Inc. (CA, USA), round up the picture of endoscopic surgery. The advantages of endoscopic surgery over open surgery (more precision, less trauma, less postoperative pain, shorter hospital stays and a faster recovery period) are becoming more accepted. The present healthcare systems and hospital administrations understand the challenges of imaging in surgery, particularly in endoscopic surgery.
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Future Medicine Ltd. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this Journal-based CME activity for a maximum of
All other clinicians completing this activity will be issued a certificate of participation. To participate in this journal CME activity: (1) review the learning objectives and author disclosures; (2) study the education content; (3) take the post-test and/or complete the evaluation at www.medscape.org/journal/wh; (4) view/print certificate.
Learning objectives
Upon completion of this activity, participants should be able to:
Describe the da Vinci® Surgical System
Evaluate the practice of fertiloscopy
Distinguish relative contraindications to magnetic resonance-guided focused ultrasound surgery
Analyze imaging techniques during fertility procedures
Diagnostic imaging by x-ray, ultrasound technologies, computed tomography and MRI have been followed by therapeutic surgical imaging. Surgical imaging can be used in the planning and in the implementation of surgery. When used for planning a surgical procedure, no damage can yet occur; however, direct surgical steps performed under imaging control are irreversible. Surgical procedures performed under 2D or 3D vision are a reality of today's surgery in gynecology and obstetrics as well as in other medical fields.
Different surgical fields have developed depending upon whether the view of the surgeon is direct, indirect via ultrasound, or via 2D or 3D laparoscopic images with instruments with multiple degrees of liberty or robotic transmission of movements. Two examples are given below:
The endoscopic historical technical development of improved imaging with superior technology, from Georg Kelling to Kurt Semm to the da Vinci® Surgical System (Intuitive Surgical, Inc., CA, USA) [1–3];
MRI-guided focused ultrasound.
MRI revolutionized diagnosis 25 years ago. Today, MRI-guided focused ultrasound promises to do the same with surgery.
Currently, focused ultrasound has US FDA approval for the treatment of uterine fibroids; however, research has begun to determine its safety and efficacy in the treatment of brain, breast, prostate, bone and liver tumors and conditions such as epilepsy, stroke, chronic pain and Parkinson's disease.
In focused ultrasound surgery, treatment is conducted within a specialized resonance imaging (MRI) scanner. High-frequency and high-energy soundwaves are directed through a source (gel pad) to destroy uterine fibroids.
Technical achievements
Technical developments and a better understanding of a combination of surgical and imaging techniques revolutionized the 20th century. The first laparoscopic surgery in the world was performed by Georg Kelling from Dresden (Germany), when he performed an endoscopy on a dog. Since 2000, robotic endoscopic surgery with the da Vinci Surgical System and an increasing number of new instruments with multiple degrees of liberty, articulation and new hemostatic effects have enriched surgical endoscopic possibilities. Cameras with a range of settings from 0° to 120° (Endocameleon™, Karl Storz, Tuttlingen, Germany) and improved optical systems (high-definition television; HDTV) give brilliant pictures. Today we differentiate the following techniques for imaging surgery in gynecology:
Laparoscopy (conservative operative, robotic), natural orifice surgery, natural orifice transluminal endoscopic surgery and single port entries (laparoendoscopic single port surgery, single-incision laparoscopic surgery);
Hysteroscopy: endoscopy inside the uterus;
Fertiloscopy: transvaginal hydrolaparoscopy;
Transvaginal pelvic tumor biopsy under ultrasound control;
Magnetic resonance-guided focused ultrasound surgery (MRgFUS);
Uterine arterial embolization (UAE) for fibroids;
Follicular puncture for IVF, intracytoplasmatic sperm injection (ICSI) and embryo transfer (ET);
Transvaginal ovarian cyst or peritoneal cyst puncture: aspiration;
Endometrial ablation under ultrasound control;
Saline perfusion hysterosonography for fibroid location (better than vaginal sonographic evaluation alone);
Hysterosalpingo contrast sonography.
Imaging & surgical gynecological procedures
Let us focus on six of these 11 imaging surgical procedures: laparoscopy, hysteroscopy, fertiloscopy, uterine arterial embolization, MRgFUS, and follicular puncture for IVF, ICSI and ET.
Laparoscopy
Kelling performed the first ‘laparoscopy’, named ‘coelioscopy’ by him in 1901 on a dog using Nitze's cystoscope and an air insufflation mechanism in Hamburg, Germany at an Assembly of German Natural Scientists. This technique was developed further by Raoul Palmer, Hans Frangenheim, and after 1950 mainly by Kurt Semm. Semm initiated diagnostic and operative laparoscopy for gynecologists and operative endoscopic surgery for many other medical specialities [1].
Work on ‘imaging’ progressed from camera and light source development to the rodlens invention, from one chip to three chip, to HDTV and Endocameleon cameras as well as from direct view to a 2D or 3D view on the TV monitor. Apparatus and instrument development, in continuous cooperation with the medical technical industry, brought new features for coagulation, suturing and robotics. Instruments went from two to multiple degrees of liberty, from straight to rotating, multifunctional, articulated and robotic forms.
Precision surgery by means of an enlarged visual image and small or even single port entries for an instrument set with multiple degrees of liberty has become a reality. Imaging surgery on the video screen is the technique of the future and is already used for most benign and some malignant diseases in the field of gynecology [2,3]. It is applied for vaginal natural orifice surgery and can replace laparotomy in approximately 80% of cases. Cesarean sections, hysterectomies for large uteri and some cancer surgery are still performed by laparotomy.

Myomectomy with SprayShield™ application.

Classic intrafascial subtotal hysterectomy.

Total laparoscopic hysterectomy.
Robotic laparoscopic surgery
The growing popularity of laparoscopic surgery focused new attention on the need for both improved laparoscopic camera control and instrument range in terms of motion and dexterity.
The first robotic camera assistant used in endoscopic surgery was the ‘automated endoscopic system for optimal positioning’ (AESOP; Computer Motion, CA, USA). This hand-, foot- or voice-controlled arm allows the surgeon to perform complex laparoscopic surgery faster than with an assistant holding the camera [4]. The next surgical robot was a voice-controlled robot, ZEUS (Computer Motion), that consists of an AESOP to hold the camera and two additional AESOP-like units, which have been modified to hold the surgical instruments. The modern robot generation named da Vinci Surgical System is based on the technologies of Computer Motion, which have been developed further by Intuitive Surgical (CA, USA). The da Vinci Surgical System was approved by the FDA in May 2005 for clinical use in gynecology and was first used in reproductive gynecology for tubal surgery [5] and later in oncologic surgery [6,7].
There are four main components of the da Vinci S Surgical System:
Surgeon's console: the surgeon sits viewing a magnified 3D image of the surgical field
Patient side-cart: this system consists of three instrument arms and one endoscope arm
Detachable instruments (endowrist instruments and intuitive masters): these detachable instruments allow the robotic arms to maneuver in ways that simulate fine human movements. There are seven degrees-of-freedom, which offer considerable choice of rotation in full circles. The surgeon is able to control the amount of force applied, which varies from a fraction of a gram to several kilos. Tremor and scale movements are filtered out. The movements of the surgeon's hand can be translated into smaller ones by the robotic device
3D vision system: the camera unit or endoscope arm provides enhanced 3D images with the result that the surgeon knows the exact position of all instruments in relation to the anatomical structures

Da Vinci® S Surgical System.
The da Vinci Surgical System has captured the imagination of the surgical community worldwide; however, the costs are very high and remain at the same level for each new surgical procedure. On the other hand, smaller innovative robotic systems with instruments with up to seven degrees of liberty are coming onto the market. New HDTV systems allow the surgeon to work in what is almost a 3D field.
Hysteroscopy
Although the first exploration of the uterine cavity dates back to Bozzini, the modern CO2 liquid, office and operative hysteroscopy (resectoscopy) was developed by Hans Lindemann (1972 in Hamburg) and improved and modified by Loffer, Cooper, Gallinat and Bettocchi. Recently, vaginoscopy has again been applied to guide the diagnostic hysteroscope from the vagina into the uterine cervix without any traction of a cervical tenaculum. With saline solution as the distension medium and under exact pressure and flow control, the visualization of the uterine cavity and its pathology are visible, including the synecchia, septum, endometrium, cervical canal, uterine cavity with uterine ostia, polyps and fibroids [8–12].
Fertiloscopy: transvaginal hydrolaparoscopy
Fertiloscopy has been developed as an alternative primary screening procedure for infertility that makes it possible to evaluate tubal blockage, mucosal damage and both adhesions and endometriosis. There are contraindications to its use in patients with retroverted uterus, severe endometriosis, known ovarian, uterine or tubal pathology and in preoperated patients. Fertiloscopy can almost always be carried out under local anesthetic. The benefits are the following:
It is quick and ergonomically efficient as both the hysteroscopy and transvaginal hydrolaparoscopy component can be done in one seated position, unlike traditional laparoscopic surgery and hysteroscopic surgery in the operating room;
The surgeon has a good view of the reproductive organs and can therefore pinpoint more accurately and completely the diagnosis of unexplained infertility;
The view obtained with fertiloscopy is more ‘physiological’ than with conventional laparoscopy, providing a view of the upper reproductive tract that reflects well its normal functioning. For example, there is an enhanced view of the ovaries and tubes and their apposition, enabling clear assessment of the ‘egg pick-up’ mechanism;
It is possible to perform salpingoscopy; that is, detailed assessment of the outer third of the tubal lumen to make or exclude the diagnosis of previous tubal infection.
Another advantage of fertiloscopy as an office procedure (but only when carried out under light general anesthesia) is that polycystic ovaries can be treated as well as at laparoscopy, while limited treatment of endometriosis and adhesions may be possible.
Such treatment can still be completed in less than 20 min. This allows the surgeon's time to be used efficiently, avoids a second anesthetic and is convenient for the patient.
The key argument for fertiloscopy is that it will yield information in respect of the causes of infertility equivalent to a combination of hysteroscopy, hysterosalpingography or hysterosalpingo contrast sonography. If necessary, in approximately 30% of cases, laparoscopy and salpingoscopy/microsalpingoscopy have to be done in a second step. Moreover, being much less traumatic it is more likely to be recommended to – and accepted by – patients as part of the initial infertility work up.
Fertiloscopy has been in use for around 10 years as an alternative to standard laparoscopy in infertility. Results of an international multicenter prospective trial, The Fertiloscopy–Laparoscopy (FLY) study, showed that fertiloscopy findings correlated highly with those of diagnostic laparoscopy. Of all tubal patency testing techniques without anesthesia, fertiloscopy gives the best information of tubal patency and reveals some pathologies [13].
UAE
Uterine arterial embolization is used for uterine fibroid shrinking and to stop major obstetric hemorrhage. Polyvinylglycol particles are injected into the femoral artery under MRI control with the purpose of connecting to both uterine arteries: the embolization does not impede later fertilization [14–16]. In a Dutch nationwide study by Zwart et al. the rate of obstetric hemorrhage that necessitated hysterectomy after arterial embolization was 5.7 per 1000 deliveries [17]. Fertility was preserved in 46% of women by successful arterial embolization.
Uterine arterial embolization is a minimally invasive, image-guided therapy, in which the uterine fibroid blood supply is blocked by catheterization and the insertion of embolic particles that cause the ischemic necrosis of the fibroids. The embolic particles are usually composed of polyvinyl alcohol, tris-acryl or gelatin sponge.
MRgFUS
Surgical treatments for uterine fibroids include hysterectomy and myomectomy via laparotomy or today via laparoscopy in all its variants [18]. Minimally or noninvasive treatments include UAE, MRgFUS and hormonal therapy [19,20]. Each of these treatment options, which require minimal or no hospitalization, enable women to preserve their uterus and usually minimize complications, recovery time and treatment costs [21].
Magnetic resonance-guided focused ultrasound surgery is a noninvasive treatment in which ultrasound energy, focused on the fibroid in multiple focal spots, raises the temperature of tissue within the focal zone and causes coagulative necrosis. MRI guides and monitors the procedure, thus providing a closed-loop anatomical and thermal feedback. MRI is used to identify tumors or fibroids in the body before they are destroyed by ultrasound. Therapeutic ultrasound is a minimally invasive or a noninvasive method to deposit acoustic energy into tissue. Applications include tissue ablation (high-intensity focused ultrasound) for tumor treatments and fibroid destruction, hyperthermia treatments (low-level heating combined with radiation or chemotherapy) or the activation or enhanced delivery of drugs.
Several measures are used to assess the efficacy of these minimally or noninvasive treatments; amongst them are a uterine fibroids symptoms quality-of-life assessment questionnaire, fibroid shrinkage and patient satisfaction. As with any fibroid treatment, besides hysterectomy, symptoms can recur following these less invasive approaches. Consequently, referral to an alternative treatment after a particular modality has been pursued is also a measure of the treatment efficacy.
Different patient selection criteria are established for UAE and MRgFUS treatments. For UAE, submucosal and pedunculated fibroids may be considered relative contraindications, as are previous internal iliac or uterine artery occlusion or recent gonadotrophin-releasing hormone analogue administration. In addition, these are insufficient data to advocate UAE as a means of preserving fertility. For MRgFUS, hyper-intense fibroids and multiple fibroids may be considered relative contraindications as they are difficult to treat. In addition, in cases where the ultrasound beam is interrupted by anatomical structures, such as bowels, bones or nerves, MRgFUS treatment may be impossible without successful mitigation techniques [22].
Follicular puncture for IVF/ICSI & ET
The main indications today for IVF-ET and ICSI are tubal sterility, idiopathic infertility and male infertility.
The first aspirations of human follicles in the ovary were performed laparoscopically under direct vision (1975–1985) and the oocytes were detected under the microscope [23,24].
However, after the development of video laparoscopy between 1984 and 1986, the follicles were aspirated laparoscopically using this imaging technology. Later, transvesical aspirations under ultrasound control followed and only after 1987 was the transvaginal ultrasound-controlled follicular puncture developed. Since then, this technique has been introduced into all IVF and ET programs, including ICSI and blastocyst transfer [25].
Follicle retrieval is performed via the vagina under ultrasound guidance. The vaginal ultrasound probe is equipped with a fixed needle guide through which the needle for oocyte retrieval can be inserted and manipulated
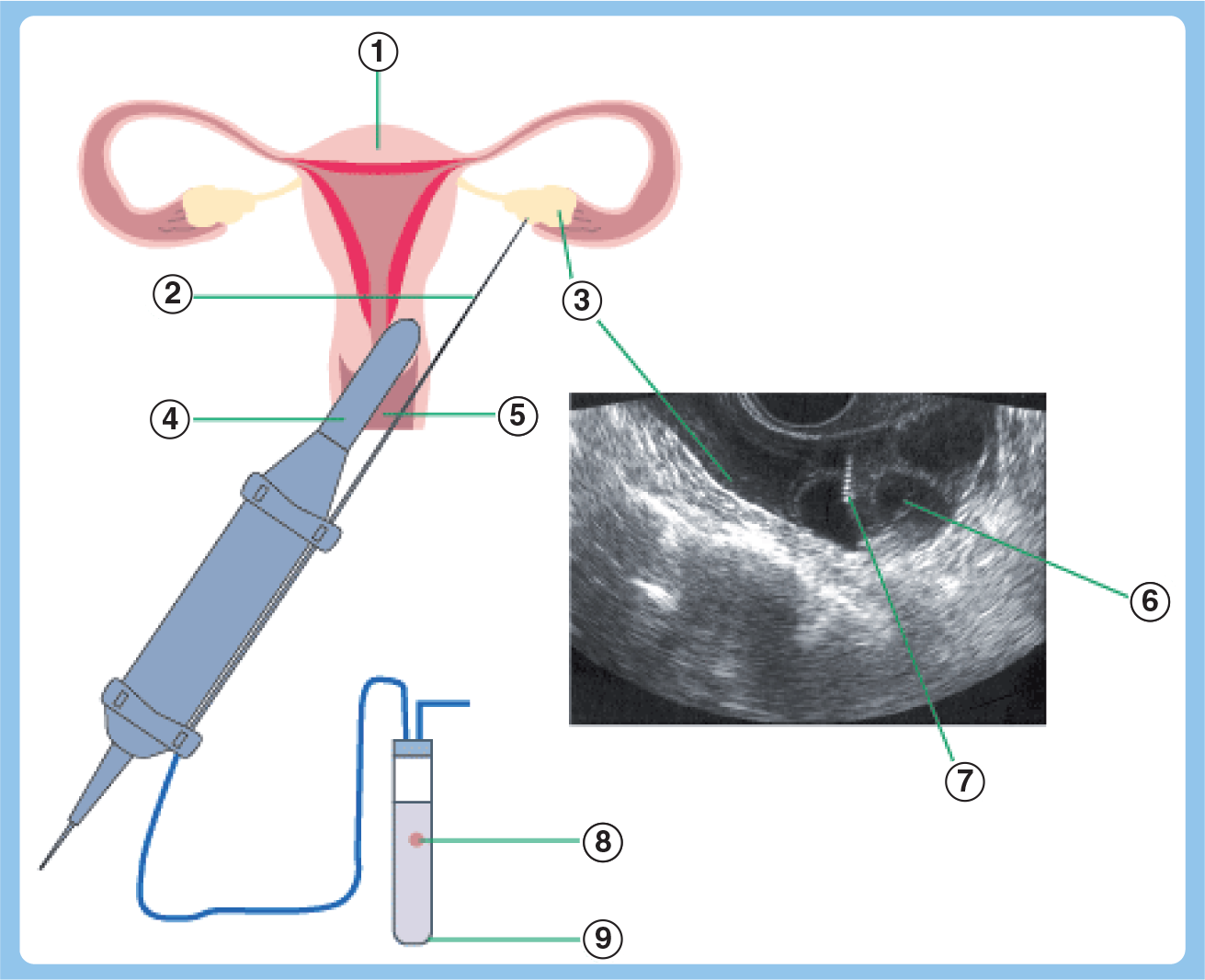
Scheme for follicular puncture.
The ultrasound picture shows the individual follicles
The procedure can be performed under local or general anesthesia. In our clinic we prefer a simple sedation. The patient can visualize the detected oocytes on the video screen – another imaging technique. After the procedure minor pain and small vaginal bleeding may be observed. This technique is performed as an outpatient procedure.
The embryo transfer is optimally performed under abdominal ultrasound control. The gynecologist can visualize the transfer catheter, which is inserted through the cervix into the uterus without touching the uterine wall. The deposition of the culture medium containing the embryos can also be seen.
The early embryonic development in the uterine cavity becomes visible within the fifth week of pregnancy.
Conclusion & future perspective
Gynecologic surgery in the 21st century is based in many ways on ‘imaging technology’. True imaging surgery as performed in laparoscopy, hysteroscopy, fertiloscopy, UAE, MRgFUS and follicular puncture has taken over from previous conventional technologies. Open-access procedures only have preference if imaging technologies are not available.
Imaging surgery with new optical systems has nearly reached 3D standards. Articulated and robotic instruments are already replacing the expert, but often trembling, fingers of the conventional surgeon. Surgical precision in knowing hands, under imaging control, meets the demands of today and ensures the best outcome.
Areas for future development in this field include high-precision computerized robotic surgery with a wide database. Progress is based on technology and the field of medicine must integrate technological advances. For pilots, training on simulators is an accepted part of their training. For surgeons, however, the cost of training on virtual reality simulators is often regarded as too high. This way of thinking must be rectified.
Executive summary
The focus of this article is six imaging techniques that facilitate medical procedures in a patient-friendly and tolerable way: laparoscopy, hysteroscopy, uterine arterial embolization, magnetic resonance-guided focused ultrasound surgery and follicular puncture for IVF and embryo transfer.
At a glance, these procedures have taken away big transabdominal openings and give the patients the same results with less pain, reduced hospitalization time and faster reintegration into family and professional life. They challenge the medical profession and the medical technical industry as well as healthcare providers.
Endoscopic surgery has developed in all medical fields during the 20th century, and is proving to be the key surgery of the 21st century for all benign indications and, in our opinion, oncologic surgery. However, the oncologic field still requires more randomized studies to prove the concept. It was unthinkable 30 years ago to resect a uterus out of the female womb laparoscopically, but today this surgery is a routine procedure.
Magnetic resonance-guided focused ultrasound surgery discussed in this article for the gynecologist is only one example of the great advantages of combining imaging and medical therapy.
The field of artificial reproductive technology has developed rapidly since the first test tube baby, Louis Brown in 1978, with over 4.5 million such babies now born worldwide. Robert Edwards finally received, at the age of 86, the Nobel Prize in physiology and medicine in 2010. The great achievements of the test tube baby technique open new horizons in medicine and research. Science and ethics have to find their applications.
Footnotes
Imaging in gynecologic surgery
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiple-choice questions. To complete the questions and earn continuing medical education (CME) credit, please go to www.medscape.org/journal/wh. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.org. If you are not registered on Medscape.org, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,
