Abstract
Estradiol valerate and dienogest have been combined to create a novel four-phasic oral contraceptive pill effective for both pregnancy prevention and treatment of heavy menstrual bleeding. This formulation represents the only oral contraceptive pill available in the USA containing an estrogen component that is biologically active as the endogenous estrogen 17β-estradiol. This medication was developed out of efforts to replace the most common estrogen in contraceptive pills, ethinyl estradiol, which is known to be a potent inducer of hepatic protein synthesis. Estradiol valerate has been available since the 1970s in oral and injectable forms indicated for the treatment of menopausal climacteric symptoms. Dienogest has been used in other oral contraceptive pills for over 10 years. Previous attempts to develop an oral contraceptive pill with natural estradiol or estradiol valerate were unsuccessful due to poor cycle control. A novel dynamic-dosing regimen was devised to improve the bleeding pattern. This medication has been shown in several clinical trials to have good contraceptive efficacy and cycle control. Recent studies have also demonstrated that this medication is effective for the treatment of heavy menstrual bleeding. However, compared with other oral contraceptive pills, this medication is associated with a higher frequency of absent withdrawal bleeding. Furthermore, the dynamic dosing regimen requires relatively complex instructions for users who miss pills.
The first combined oral contraceptive pill was approved in the USA in 1959, and consisted of the progestin norethynodrel and the estrogen mestranol. Mestranol is biologically inactive, but undergoes demethylation in the liver to yield active ethinyl estradiol (EE) [1]. The stability and high potency of EE have established this compound as the estrogen component in nearly all currently available combined oral contraceptive pills in the USA.
Combined estrogen and progestin contraceptives, now available in oral, vaginal and transdermal preparations, are known to be safe and well tolerated, and to have various noncontraceptive benefits [2]. Despite this, some women who take the pill experience adverse hormonal effects. Furthermore, oral contraceptive pills are known to affect metabolic and hemostatic parameters, and are associated with an increased risk of arterial and venous thrombosis [3]. The EE component is thought to be primarily responsible for these risks, as it is known to be a potent inducer of hepatic protein synthesis [4].
The undesired side effects and risks associated with this common form of contraception have led to the development of modern formulations, which contain either lower doses of EE, or utilize newer types of estrogens and progestins. Contraceptives containing very low doses of EE, from 10–20 μg, seem to be equally efficacious for pregnancy prevention and have a lower incidence of various side effects such as breast tenderness and bloating compared with higher-dose pills. However, they are associated with increased breakthrough bleeding, particularly in the first few cycles of use [5,6]. Lower-dose EE pills have not been shown to have a lesser impact on hemostatic parameters [7,8].
Initial attempts to develop a natural estradiol (E2)-containing contraceptive pill date back to the 1970s, but were unsuccessful due to poor cycle control [9–12]. Multiple studies of contraceptive pills containing E2, including micronized E2 and estradiol valerate (E2V) as the estrogen component failed to demonstrate tolerable bleeding profiles [13,14]. Several decades after these initial trials, however, it was determined that a specific progestin component and dynamic dosing regimen would together provide a solution to this problem. Dienogest (DNG) was chosen as the progestin to best accompany E2V, as it has particularly potent effects on the endometrium [15].
This E2V/DNG pill represents the first oral contraceptive pill in the USA with an estrogen component that is identical in its active form to the most potent endogenous estrogen, 17β-estradiol. Although the use of natural estradiol may be appealing to patients or clinicians who are concerned about the hepatic effects of EE on metabolic and hemostatic parameters [8], large epidemiologic studies designed to assess whether this is a safer pill have not yet been completed.
Overview of the market
The oral contraceptive pill is used by an estimated 100 million women worldwide and 12 million women in the USA [16]. It is currently the leading form of contraception in the USA [16], where 80% of women will use the pill at some point during their reproductive years [17].
This new E2V/DNG formulation is packaged as a 28-day cycle with a short, 2-day hormone-free interval. This combination of hormones, along with a dynamic dosing regimen, is associated with a satisfactory bleeding profile and low incidence of side effects. This represents a reasonable choice for many new users of the pill who would prefer a natural form of estrogen, or for women who are dissatisfied with other pills. However, E2V/DNG carries the same contraindications as all other combined oral contraceptive pills [101].
Oral contraceptive pills are commonly used as a treatment for heavy menstrual bleeding, as they are known to decrease the amount of bleeding in women with normal cycles [18]. While US FDA-approval of E2V/DNG for the indication of heavy menstrual bleeding has not yet been obtained, the results of two large placebo-controlled randomized trials support its use for this purpose, and provide evidence that it may be more efficacious than other oral contraceptive pills [19,20]. These data have led to the approval of E2V/DNG for the treatment of heavy menstrual bleeding in Europe, the UK, Australia and Brazil. There are few reliable estimates of the prevalence of heavy menstrual bleeding; however, this is known to account for significant morbidity among women, particularly in the later reproductive years [21].
The compound & its chemistry
The four-phasic dynamic-dosing regimen of E2V/DNG provides early estrogen dominance in order to allow endometrial proliferation and progesterone sensitivity. Combined E2V/DNG pills are then given in an estrogen step-down/progestin step-up approach. In the mid-to-late cycle, DNG provides endometrial stability to prevent unscheduled breakthrough bleeding [22].
The currently marketed formulation of this medication is a 28-day cycle consisting of E2V 3 mg on days 1–2, E2V 2 mg and DNG 2 mg on days 3–7, E2V 2 mg and DNG 3 mg on days 8–24, E2V 1 mg on days 25–26, and placebo on days 27–28
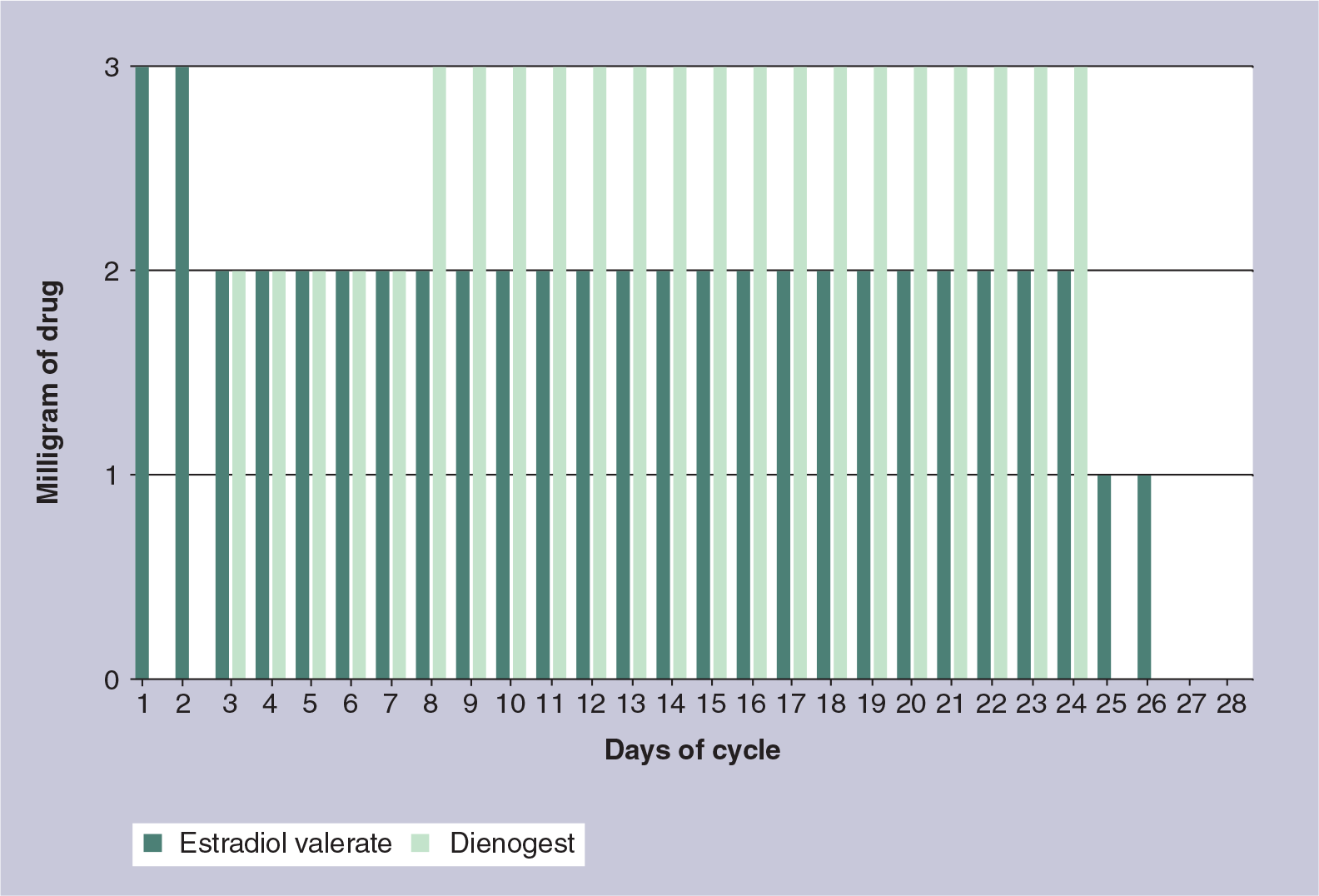
Dynamic dosing regimen of estradiol valerate and dienogest.
Standard oral contraceptive pill regimens contain 21 active and seven placebo pills. Many newer formulations have decreased (e.g., to 4 days) or eliminated the hormone-free interval. Studies of these regimens demonstrate a reduction in cycle-related symptoms such as headaches and breast tenderness [23–25].
As with other combined oral contraceptive pills, E2V/DNG prevents pregnancy mainly due to the suppression of gonadotropins and ovulation inhibition [26]. The estrogen component primarily suppresses follicle-stimulating hormone, while the progestin component suppresses luteinizing hormone [27]. Thickening of the cervical mucus represents another important contraceptive effect attributable to the progestin component. This effectively blocks ascension of sperm into the uterus and fallopian tubes, preventing fertilization even if ovulation does occur [26].
Endometrial changes vary slightly according to the specific estrogen and progestin components of oral contraceptive pills, but are generally associated with decreased menstrual bleeding and decreased dysmenorrhea [2]. A recent study of 218 women who underwent endometrial biopsy after 20 cycles of use of E2V/DNG demonstrated no cases of endometrial hyperplasia or malignancy [28]. Over 80% of study subjects had atrophic, inactive or secretory endometrium.
Combined oral contraceptive pills are associated with a 50% decreased risk of endometrial cancer and a 40% decreased risk of ovarian cancer [1]. While there are no data specifically regarding E2V or new progestins such as DNG, this formulation is expected to have the same protective effect as other pills.
Metabolism & pharmacokinetics
Estradiol valerate is the esterified form of 17β-estradiol
Estradiol valerate and dienogest.
A 1 mg dose of E2V contains 0.76 mg of E2 based on molecular weight [30]. E2 is widely distributed and circulates bound to sex hormone-binding globulin (38%) and albumin (60%); only 2–3% circulates in free form [31]. A significant portion of oral E2 undergoes first-pass hepatic metabolism, with conversion of E2 to estrone and estriol, and metabolism to nonestrogens including sulfate and glucuronide conjugates. The CYP3A family of cytochrome P450 enzymes is the most important pathway of hepatic metabolism for E2 [32]. Most of the oral dose of E2 is metabolized by the GI tract and liver prior to entering the systemic circulation [33].
Dienogest is a 19-nortestosterone derivative that exhibits antiandrogenic activity
Estradiol metabolites including the sulfate and glucuronide conjugates are mainly excreted by the kidneys. Approximately 10% is excreted through the feces [29]. DNG metabolites are excreted mainly by the kidneys [15].
The pharmacokinetic parameters of E2V/DNG were evaluated as part of a Phase I clinical trial [34]. For this study, healthy women aged 18–50 years were treated with the current dosing regimen.
Summary of Phase I pharmacokinetic study, n = 15 (day 24 of cycle, fasting).

Cmax, AUC0–24 and T1/2 presented as mean (standard deviation); Tmax presented as median (range). AUC0–24: Area under the concentration–time curve from 0 to 24 h after administration; Cmax: Maximum serum concentration; NA: Data not available; T1/2: Elimination half-life; Tmax: Time to maximum serum concentration. Data taken from [34].
Studies in postmenopausal women have shown significant interactions between E2V/DNG and CYP3A inducers (rifampin) and inhibitors (ketoconazole and erythromycin) [35].
Pharmacodynamics
The development of an E2-containing contraceptive pill was sought in order to achieve a more favorable metabolic profile compared with the standard EE-containing pills. EE is a potent inducer of hepatic enzymes and globulins such as sex hormone-binding globulin, even after the first-pass effect [4]. Thus transdermal and transvaginal EE have similar metabolic effects as oral preparations. While E2V has not yet been shown to be associated with a decrease in estrogen-related adverse events such as thrombosis, it appears to have a smaller effect on hemostatic parameters and markers of carbohydrate metabolism compared with EE at doses of equal potency [7]. Large epidemiologic studies will be needed in the future to determine if these differences in laboratory markers are clinically relevant. Thromboembolic events occur infrequently and differences in incidence cannot be assessed in Phase III trials.
A study of four different oral contraceptive regimens was carried out to evaluate the effect of partial replacement of EE with E2V on hemostatic parameters [8]. Subjects were followed over six cycles. While all of the regimens contained EE, 25 of the 100 subjects were randomized to receive a pill containing E2V with a low (10 μg) dose of EE. The effects on total and free protein S, tissue plasminogen activator and plasminogen activator inhibitor were lower for subjects on the E2V/EE/DNG regimen than those on the EE/DNG regimens (containing at least 20 μg EE), demonstrating that the impact of 2 mg E2V on some hemostatic parameters is less than that of 10 μg of EE. Levels of protein C activity increased 10–15% during use of all regimens and were not significantly different among the four groups.
Another study compared the hemostatic effects of E2V/DNG versus a monophasic combined oral contraceptive containing EE 30 μg and levonorgestrel (LNG) 150 μg [36]. A total of 32 women aged 18 to 50 years were randomized in a crossover design for three cycles, with two washout cycles between treatments. Intra-individual absolute increase in
In a published abstract by Parke et al. [37], 58 subjects aged 18–50 years were randomized to receive the four-phasic E2V/DNG regimen or a tri-phasic EE/LNG regimen. Individual-specific changes in high-density lipoprotein and low-density lipoprotein cholesterol levels were assessed from baseline to cycle seven. Hemostatic characteristics were also measured. While none of these changes was statistically significant, the overall direction of all measures suggests a lower impact with E2V/DNG. The observed change in high-density lipoprotein levels was +7.9% (±21.8%) for those using E2V/DNG and −2.3 (±14.4%) for those using EE/LNG, and this difference approached statistical significance (p = 0.055). For low-density lipoprotein levels, the observed change was −6.5 (±15.9%) for E2V/DNG and −3.0 (±17.4%) for EE/LNG (p = 0.458). Changes in hemostatic characteristics were reportedly less pronounced with E2V/DNG than with EE/LNG. Sex hormone binding globulin levels increased by 62.7% (±50.5%) for E2V/DNG, compared with 111.6% (±48.0%) for EE/LNG. No clinically relevant metabolic changes were observed in either group.
Contraceptive efficacy
The optimal dosing regimen for effective ovulation inhibition was determined by two Phase II, randomized, open-label studies by Endrikat et al. [22]. The first study was designed to evaluate the proper length of treatment with DNG during a 28-day cycle. The second study was then used to determine the most effective dose of DNG. The primary efficacy end point was the proportion of women with Hoogland score of 5 (luteinized unruptured follicle on transvaginal ultrasound) or 6 (ruptured follicle >13 mm on transvaginal ultrasound accompanied by serum progesterone >5 nmol/l and E2 of >0.1 nmol/l). Results of the second study demonstrated that the four-phasic regimen (E2V 3 mg on days 1–2, E2V 2 mg/DNG 2 mg on days 3–7, E2V 2 mg/DNG 3 mg on days 8–24, E2V 1 mg on days 25–26, placebo on days 27–28) provided the lowest effective dose for ovulation inhibition; only three of 96 subjects (3.13%; 95% CI: 0.2–6.05%) had a Hoogland score of 5 or 6, similar to other commonly used contraceptives [38].
Contraceptive efficacy of the four-phasic E2V/DNG regimen was established by two large-scale Phase III studies, demonstrating results similar to that of other recently approved low-dose EE-containing oral contraceptive pills
Summary of contraceptive efficacy studies.

During study treatment, 13 pregnancies occurred over 23,368 cycles. Unadjusted Pearl index was calculated as 0.73 (upper limit of 95% CI: 1.24). A total of six of the pregnancies were attributed to method failure, resulting in an adjusted Pearl Index of 0.34 (upper limit of 95% CI: 0.73). As expected, the majority of pregnancies occurred in younger women; 12 of the 13 pregnancies occurred over 16,608 cycles in the subset of women aged 18–35 years, giving an unadjusted Pearl Index of 0.95 (upper limit of 95% CI: 1.65); five of the 12 were attributed to method failure, resulting in an adjusted Pearl Index of 0.40 (upper limit of 95% CI: 0.92). Kaplan–Meier estimates for the cumulative failure rate over 20 treatment cycles was 0.0109 (95% CI: 0.0063–0060.0188).
A second Phase III study was conducted in the USA and Canada [41]. In this multicenter noncomparison study, 490 healthy women between the ages of 18 and 35 years were given E2V/DNG for up to 28 cycles. Exclusion criteria included BMI >30 and other contraindications to oral contraceptives. The initial efficacy data were obtained from the first 13 cycles; the remaining cycles were used to obtain additional safety data. Six pregnancies occurred during the treatment period. The study was not powered for a separate Pearl Index calculation.
A third study provides efficacy data for 399 women who were randomized to receive E2V/DNG for seven cycles [42]. This double-blinded study, conducted in 34 centers in Europe, compared E2V/DNG to an oral contraceptive containing EE 20 μg/LNG 100 μg (399 women in each arm, 798 total). Primary outcome was bleeding pattern. No pregnancies occurred in the E2V/DNG group. One pregnancy occurred in the EE/LNG group.
The efficacy data from all three efficacy studies were pooled in a report presented at the American College of Obstetricians and Gynecologists Annual Clinical Meeting in 2009 [41]. A total of 2266 women provided 2513 women-years of treatment exposure. The subset of 1687 women aged 18–35 years provided 1267 women-years of exposure. A total of 16 pregnancies occurred during the first 13 cycles (including the 14-day period after discontinuing study treatment) giving a calculated Pearl Index of 1.27 (upper limit of 95% CI: 2.06). Using only the seven pregnancies attributed to method failure gives an adjusted Pearl Index of 0.72 (upper limit of 95% CI: 1.37). Of note, in the FDA-approved product labeling, the Pearl Index calculated from the European study is 1.04, and from the North American study is 1.64 [101].
Cycle control
Previous studies of oral contraceptives containing natural estradiol demonstrated effective ovulation inhibition but unsatisfactory cycle control, with frequent episodes of unscheduled bleeding [10,11]. Both monophasic and biphasic regimens were associated with various menstrual abnormalities including prolonged bleeding episodes and an increase in spotting. This early challenge led to the selection of DNG as the progestin component, and the development of the four-phasic dosing regimen [9].
Results of a large trial indicate that the current formulation provides excellent cycle control [42]. For this prospective, double-blinded study, conducted at 34 centers in Europe from 2005 to 2006, 798 healthy women aged 18–50 years were randomized 1:1 to receive either the current regimen of E2V/DNG or a monophasic oral contraceptive pill with 21 days of EE 20 μg/LNG 100 μg and 7 days of placebo. Smokers over the age of 30 years and those with BMI >30 were excluded. Primary outcomes were bleeding pattern and cycle control parameters: total number of bleeding/spotting days and number and length of bleeding/spotting episodes (bounded by at least 2 days without bleeding or spotting). Study treatment was for seven cycles, and bleeding parameters were evaluated during two 90-day reference periods as recommended by the WHO [43]. Scheduled bleeding, calculated from the last day of progestin intake, was also assessed. Bleeding intensity for each instance was recorded on a 5-point ordinal scale.
Women randomized to the E2V/DNG experienced significantly fewer total bleeding/spotting days than women in the EE/LNG group for both 90-day reference periods (p < 0.001). Within the first 90-day period, the mean number of days for the E2V/DNG group was 17.3 (median 16.0, standard deviation [SD] 10.4), and for the EE/LNG group was 21.5 (median 21.0, SD 8.6). In the second 90-day reference period the mean number of days for the E2V/DNG group was 13.4 (median 12.0, SD 9.3) and for the EE/LNG group was 15.9 (median 15.0, SD 7.1).
In terms of scheduled withdrawal bleeding, the median duration was shorter for women in the E2V/DNG group compared with the EE/LNG group (median 4.0 vs 5.0 days, respectively; p < 0.05). The maximum intensity of scheduled bleeding episodes was significantly lighter for the E2V/DNG group (median = light bleeding vs median = normal bleeding).
The proportion of women experiencing unscheduled intracyclic bleeding was not significantly different between the two groups (14% per cycle in the E2V/DNG group vs 12% per cycle in the EE/LNG group; p > 0.05). The maximum intensity of unscheduled bleeding was also not significantly different. The highest rates of unscheduled bleeding occurred during the first cycle for both groups.
Significantly more women in the E2V/DNG group experienced absent withdrawal bleeding (mean 19.4 vs 7.7% per cycle). Amenorrhea, defined as absence of unscheduled intracyclic and scheduled withdrawal bleeding, occurred in significantly more cycles for the E2V/DNG group (15.4 vs 4.5% of cycles). Few women in either group did not have any scheduled bleeding episodes during the entire seven-cycle treatment duration (3.3% for E2V/DNG vs 0.5% for EE/LNG). Absent withdrawal bleeding is safe, and may be considered by some women to be favorable, although for others this may lead to concern regarding possible pregnancy [1].
Information on bleeding patterns from the two Phase III efficacy trials were not published separately [40], but the data were pooled with results from the above European comparison study and analyzed over 13 treatment cycles [41].
The median length of scheduled bleeding episodes was 4.0–4.6 days. Scheduled bleeding was absent in 19.0–24.0% of women. The most frequently recorded maximum intensity of scheduled and unscheduled intracyclic bleeding was light or spotting.
Treatment for heavy menstrual bleeding
Heavy menstrual bleeding has generally been defined as total menstrual blood loss of at least 80 ml per cycle, as quantified by hemoglobin extraction from sanitary products. This is the amount of menstrual blood loss that causes women to become anemic [44]. Most clinical trials of treatment for heavy menstrual bleeding (sometimes referred to as menorrhagia, an older term) have utilized this definition, although such quantitative criteria are not used in clinical settings. Subjective definitions of heavy menstrual bleeding have been proposed by organizations such as the NICE in the UK, since women's perceptions of bleeding are what ultimately lead to presentation to a physician for evaluation and treatment [102].
Oral contraceptive pills are known to decrease the duration and intensity of normal menstrual bleeding; for this reason they are often used as a first-line treatment for heavy menstrual bleeding, despite a lack of evidence of their effectiveness for this purpose [45]. This remains an off-label use for all oral contraceptives in the USA.
Two large, Phase III, randomized, placebo-controlled trials have evaluated the efficacy and safety of the current regimen of E2V/DNG for the purpose of decreasing menstrual blood loss in women with heavy menstrual bleeding [19,20]. Inclusion criteria for both studies, evaluated during a 90-day run-in period, consisted of heavy menstrual bleeding (at least 80 ml per cycle), prolonged menstrual bleeding (at least two bleeding episodes lasting 8 days or more), frequent menstrual bleeding (more than five bleeding episodes that were collectively at least 20 days), or any combination of the three. All women underwent transvaginal ultrasound and laboratory studies prior to study treatment and were excluded if they had anatomic or coagulation abnormalities associated with increased menstrual bleeding (e.g., significant uterine leiomyomas, polyps, endometrial hyperplasia or malignancy and von Willebrand disease). Exclusion criteria also included the use of other medications used to treat heavy bleeding, including NSAIDs, tranexamic acid and sex steroids. Women with a BMI >32 and those aged 35 years and older who smoked more than ten cigarettes per day were also excluded.
The primary outcome for these studies was the proportion of women with a return to completely normal menses during the 90-day efficacy phase (the last 90 days of the 196 total treatment days), compared with the 90-day run-in phase. Other efficacy variables included measured menstrual blood loss volume, number of sanitary items used, total number of bleeding episodes and days, and laboratory measures of iron metabolism (ferritin, hemoglobin and hematocrit). Safety and tolerability were also assessed. Subjects were required to use barrier contraception throughout the treatment period, thus contraceptive efficacy was not evaluated in these studies.
In the study completed in North America, 190 women (of 1077 screened), were randomized to receive E2V/DNG (n = 120) or placebo (n = 70). Using an intention-to-treat approach, categorizing individuals with missing data as nonresponders, 29.2% of subjects in the E2V/DNG group had a complete response to treatment compared with 2.9% in the placebo group (p < 0.001). Excluding individuals with missing data, 43.8% in the E2V/DNG group and 4.2% of those in the placebo group had a complete response (p < 0.001). It is important to point out that the rigorous definition of complete response required subjects to fulfill a composite of up to eight individual criteria during the 90-day efficacy interval including an absence of baseline symptoms plus a defined improvement in the condition. Blood loss estimates are more comparable with previous literature. The mean measured menstrual blood loss reduction was 353 ml in the E2V/DNG group and 130 ml in the placebo group (p < 0.001). Among the participants with heavy bleeding as their inclusion criteria (75.8% of subjects), reduction in menstrual blood loss of 50 and 80% was reported in 80 and 45% of women, respectively. While the change in total number of bleeding episodes was not statistically significantly different, there was a significantly greater decrease in total number of bleeding days (p = 0.024) and in the number of sanitary items used (p < 0.001) for the treatment group compared with the placebo group. Mean change in hematocrit from baseline was +1.4% for the E2V/DNG group and −0.05% for the placebo group (p = 0.001). The response to treatment was rapid, with most subjects experiencing a significant decrease in measured menstrual blood loss with the first withdrawal bleed after starting E2V/DNG

Median menstrual blood loss per cycle in the North American study.
Results of the parallel study completed in Europe and Australia, designed to assess the same parameters, have recently been published [19]. In this study, 149 participants were randomized to the E2V/DNG group and 82 received placebo. Complete response rate was 40.74% in the E2V/DNG and 1.61% in the placebo group. Mean menstrual blood loss reduction was significantly greater in the treatment group. A significant improvement in the measured markers of iron metabolism was observed only in the E2V/DNG group.
Postmarketing surveillance
A Phase IV program for this medication will assess the risks of short- and long-term use of E2V/DNG and of established oral contraceptives in a study population that is representative of actual users of the individual preparations. This includes an estimate of the absolute risk of rare serious adverse outcomes such as venous thromboembolic events, acute myocardial infarction and cerebrovascular accidents [103].
Safety & tolerability
Studies of E2V/DNG have demonstrated excellent safety and tolerability, similar to conventional EE-containing oral contraceptives. In the Phase II study by Endrikat et al. [22], five out of 104 women receiving the current dosing regimen discontinued treatment due to adverse events, which included depression, headache, worsening acne, eye irritation and furunculosis (each occurred in only one subject). There was only one serious adverse event (ovarian cyst) among the 406 subjects receiving any of the four E2V/DNG regimens.
In the European Phase III trial [40], 79.5% of subjects reported that they were satisfied or very satisfied with the study medication. The most common adverse events were nasopharyngitis (17.9%), headache (9.4%) and diarrhea (6.6%). Overall, 140 women (10.2%) chose to discontinue the study medication due to adverse events. The most common events leading to discontinuation were metrorrhagia (1.7%), acne (1.0%) and weight gain (0.9%). Five serious adverse events possibly related to the medication included once case each of ocular histoplasmosis syndrome, uterine leiomyoma, focal nodular hyperplasia of the liver, myocardial infarction, and deep venous thrombosis. The myocardial infarction occurred in a 47-year-old woman who was a smoker (in violation of the exclusion criteria). The deep venous thrombosis occurred in a 40-year-old woman subsequent to a sprained ankle, nine days after completing treatment with the study medication. She had received depot medroxyprogesterone acetate in the interim prior to diagnosis of the deep venous thrombosis.
In the North American Phase III study, 15% of subjects discontinued the medication due to adverse events [41]. Drug-related serious adverse events occurred in only two women (ruptured ovarian cyst in both).
In the European study comparing the current regimen of E2V/DNG to a monophasic 21/7 regimen of EE 20 μg/LNG 100 μg, rates of adverse events and discontinuation were not significantly different between the two groups [42]. The most frequently reported adverse events in the E2V/DNG group were breast pain (3.8%), headache (2.5%) and vaginal infection (2.5%). Discontinuation due to adverse events was 3.3% in both groups. Five serious adverse events occurred in the E2V/DNG group: one case each of ruptured ovarian cyst, autonomic nervous system imbalance, vulvar abscess, chronic tonsillitis and renal colic.
In the North American study evaluating efficacy of E2V/DNG for the treatment of heavy menstrual bleeding [20], 9.2% of subjects discontinued treatment due to adverse effects in the E2V/DNG group, compared with 6.1% of those in the placebo group. The most frequently reported adverse effects in the E2V/DNG group were acne (4.2%), breast pain (3.4%), breast tenderness (2.5%), dysmenorrhea (2.5%) and headache (2.5%). One serious adverse event occurred in the treatment group: an acute myocardial infarction (2 days after the final dose of study medication) in a 46-year-old woman with hyperlipidemia and a family history of cardiovascular disease.
Regulatory affairs
The estradiol valerate and dienogest pill was approved in 2008 as an oral contraceptive in the EU, where it is marketed as Qlaira® (Bayer HealthCare Pharmaceuticals, Berlin, Germany). FDA approval for Natazia™ (Bayer HealthCare Pharmaceuticals) was obtained on 6 May 2010. The approved indication is pregnancy prevention. It received regulatory approval from the EU for the indication of treatment of heavy menstrual bleeding on 11 October 2010. Approval has also been granted in Australia for the treatment of heavy and/or prolonged menstrual bleeding.
Conclusion
The combination of E2V with the potent progestin DNG in a four-phasic dosing regimen provides effective inhibition of ovulation and satisfactory cycle control. Some thrombotic biomarkers appear to be less impacted by E2V/DNG than by other oral contraceptive pills. This medication has also been shown to be effective for the treatment of heavy menstrual bleeding.
This pill represents an important new option for women who chose the oral contraceptive pill as their method of birth control. It may be particularly attractive to women who prefer a more ‘natural’ estrogen component, and to those who have encountered poor cycle control or breakthrough bleeding with other combined oral contraceptive pills. Women's healthcare providers may also choose this formulation for the treatment of heavy menstrual bleeding when no organic cause has been identified. Users should be informed that absent scheduled bleeding is relatively common with this pill.
Despite availability of many different effective contraceptive options, unintended pregnancy remains a serious problem worldwide [46]. Typical-use failure rates are significantly higher than perfect-use failure rates for the oral contraceptive pill, and significantly higher than for other forms of contraception such as the intrauterine device and etonogestrel implant [1]. This is due to poor adherence with the daily dosing required for maximal contraceptive efficacy. While the bleeding profile of E2V/DNG may lead to improved compliance and decreased rates of unintended pregnancy for users of this pill, the dynamic dosing regimen requires a relatively complicated set of instructions for users who do miss pills [101]. This should factor into clinicians' decisions regarding whether to prescribe this medication for certain patients.
In evaluating this new oral contraceptive pill, one may also consider the emerging body of environmental science research concerning the presence of synthetic hormones in rivers, streams and drinking water. Hormones from combined oral contraceptives, particularly EE, are considered to be endocrine disruptors and have deleterious effects on fish and wildlife [47,48]. Though all estrogenic pollutants are potentially harmful, future studies are needed to determine whether E2V/DNG and its metabolites in human waste may represent a lesser threat to the environment.
Future perspective
The estradiol valerate and dienogest pill represents an effective oral contraceptive choice. While it is unlikely to become the first-line agent for oral contraception among all women due to its higher cost, its favorable bleeding profile may make it a better option for women with heavy or prolonged menstrual cycles. Further studies will be needed to determine whether this medication is safer or more environmentally friendly than oral contraceptive pills containing EE.
Executive summary
The estradiol valerate and dienogest pill (E2V/DNG) represents the first oral contraceptive pill with estradiol valerate as the estrogen component, which is bioactively identical to endogenous 17β-estradiol.
Most combined estrogen and progestin oral contraceptive pills contain ethinyl estradiol, a potent inducer of hepatic enzymes.
Oral contraceptives are currently used by 100 million women worldwide, and 12 million women in the USA.
Heavy menstrual bleeding causes reduced productivity and reduced quality of life for many women; oral contraceptives are frequently used to treat this condition, but no pill carries approval for this indication in the USA.
The currently available regimen of E2V/DNG includes a unique four-phasic estrogen step-up and progestin step-down with a shortened hormone-free interval of 2 days.
Estradiol valerate is rapidly cleaved in the gut to produce estradiol, identical to the most potent endogenous estrogen produced by the ovary.
Metabolic properties are similar to other oral contraceptive pills.
The estradiol valerate and dienogest pill seems to have a lesser effect on hemostatic parameters than pills containing ethinyl estradiol.
The estradiol valerate and dienogest pill has a more favorable effect on lipid profiles.
Three large-scale studies have demonstrated the efficacy of E2V/DNG for pregnancy prevention. Pooled analysis demonstrates a Pearl Index of 1.27, similar to other currently available ethinyl estradiol-containing oral contraceptives.
Previous formulations of oral contraceptives containing natural estradiol were associated with unsatisfactory bleeding profiles. This led to the development of the novel four-phasic dosing regimen of E2V/DNG.
A large randomized study comparing E2V/DNG to an ethinyl estradiol and levonorgestrel pill concluded that E2V/DNG had a favorable bleeding profile, with fewer total bleeding days and shorter scheduled menses.
Two placebo-controlled randomized controlled trials demonstrated that E2V/DNG is effective for the treatment of heavy menstrual bleeding. Women with heavy bleeding who received the study drug had large, clinically and statistically significant improvements in various bleeding parameters.
The estradiol valerate and dienogest pill has been directly compared with several other currently available oral contraceptive pills and is similarly safe and well tolerated.
Rates of adverse events with E2V/DNG are comparable to those with other oral contraceptives; the most common are headache and breast tenderness. Despite more favorable effects on hemostatic parameters, the usual exclusion criteria for oral contraceptives also apply to E2V/DNG.
Approved in the EU in 2008, and marketed as Qlaira®.
US FDA-approved in 2010. Marketed in the USA as Natazia™.
The estradiol valerate and dienogest pill represents a novel oral contraceptive and an effective treatment for heavy menstrual bleeding.
Further epidemiologic studies will be needed to determine whether or not this medication is safer than oral contraceptives containing ethinyl estradiol.
Readers are directed to the pivotal Phase III trials for contraceptive efficacy [40,41], and for the treatment of heavy menstrual bleeding [19,20]. Information on efficacy and cycle control was also provided by a large European study comparing E2V/DNG to a contraceptive pill containing ethinyl estradiol and levonorgestrel.
Footnotes
JT Jensen has received payments for consulting and giving talks for Bayer Healthcare, a company that may have a commercial interest in the results of this research and technology. This potential conflict of interest has been reviewed and managed by Oregon Health and Science University. He has also received research funding from
Wyeth, Pharmaceuticals, Warner Chilcott, M360, Abbott Pharmaceuticals, the International Committee for Contraceptive Research, and the NIH. He has also served on advisory boards for Merck. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
