Abstract
Rheumatoid arthritis (RA) is a chronic, autoimmune, systemic, inflammatory disorder primarily involving the joints and is associated with increased risk of cardiovascular disease and osteoporosis. The etiology of the disease, although not fully understood, involves a complex interplay of environmental and genetic factors [1]. RA affects 1–1.5% of the population worldwide [2]. It is predominant in women and the female:male ratio is approximately 2–4:1 [3]. Although the peak age of onset in women is in the sixth decade, it can occur at any age.
Many RA sufferers use some form of complementary or alternative (self-prescribed) therapies, including dietary supplements [4]. It has been reported that 33–75% of RA patients believe that food plays an important role in their symptom severity, [5] and 20–50% have attempted dietary manipulation as a means of relieving symptoms [6].
Dietary risk factors have also been considered. It appears that diets rich in fish, olive oil, and cooked vegetables confer a protective effect against the development of RA, whereas red meat, dairy products, and cereals are often claimed to aggravate symptoms of RA. However, no foods or food groups have been consistently identified as a cause, trigger or aggravating factor in RA patients [7,8].
Chronic diseases such as RA are known to be associated to some degree with impairment of nutritional status. Rheumatoid cachexia is a common finding among the RA patients. According to Rall and colleagues, two thirds of RA patients have some evidence of cachexia, probably because of overproduction of TNF-α and IL-1 [9]. Poor nutritional status in diagnosed RA patients has been reported in several observational studies [10,11]. In fact, some studies have shown that the nutritional status of RA patients worsens despite adequate intake [12]. However, owing to the difficulties of assessing some nutritional parameters in the rheumatic patients, evidence is not sufficiently strong to indicate a significant change in the nutritional status of RA patients. For example, a recent study could not provide strong evidence of malnutrition in women with rheumatoid arthritis by clinical anthropometrical measurements or nutritional evaluation tools [13].
The present study aimed to determine nutritional status of women with RA in Iran and to compare intake of essential micronutrients with the standard dietary reference intake (DRI) values.
Materials & methods
In this cross-sectional study, 90 women with RA were randomly recruited from an original sample of 200 patients who were referred to the Rheumatology Center affiliated to the Tabriz University of Medical Science. The eligibility of participants was verified according to the American Rheumatism Association (ARA) criteria [14], an age range of 20–70 years and stable pharmacological treatment during the past 2 months. To minimize confounding effects, subjects with diabetes mellitus, evidence of other metabolic or inherited diseases, such as nephrotic syndrome, liver disease, gastrointestinal disorders and Cushing's syndrome, medication with β-blockers, oral contraceptives and angiotensin converting enzyme inhibitors were excluded from the study.
Information regarding lifestyle, such as physical activity and smoking habits, was obtained at the time of clinical examination using a validated questionnaire [15]. Height without shoes was determined in each patient using a stadiometer (Holtain Ltd, Crymych, UK), and weight was measured in light clothing without shoes using a physician's balance scale (Seca, Hamburg, Germany). BMI of the patients was determined using Quetelet's index (BMI = weight [kg]/height [m2]). The clinical examination was done by a rheumatologist consultant and disease activity score (DAS-28) was calculated using the number of tender and swollen joints, global assessment of pain by the patient using visual analog scale (VAS) and serum testing of high sensitivity C-reactive protein (CRP). Fasting blood samples were obtained from all patients. Serum CRP was measured by a two-point immunoturbidimetric method using the kit produced by Parsazmun, (Iran; Lot. No.83001). Serum total antioxidant (TA) of the patients was estimated using a Randox Total Anti Oxidant Status test in serum (Randox Laboratories Ltd, UK, Cat. No. NX 2332, Lot. No.: 023195). ABTS® (2,2′-Azino-di-[3-ethylbenzthiazoline sulfate]) was incubated with a peroxidase (metmyoglobin) and H2O2 to produce the radical cation ABTS. This has a relatively stable blue–green color, which is measured at 600 nm. Antioxidants in the added sample cause suppression of this color production to a degree that is proportional to their concentration. Malondialdehyde (MDA) concentration was measured using MDA reaction with thiobarbituric acid then extraction with butanol. The optic density of the aqueous extract, measured spectrophotometrically at a wavelength of 532 nm, is compared with a standard curve [16].
Dietary information was provided from a 3-day, 24-h recall (2 weekdays and 1 weekend day). A trained interviewer conducted the 24-h dietary recalls on each patient using standard food models. A validated food frequency questionnaire (FFQ) was also used to determine dietary habits during the previous year [17].
The recalls were analyzed for total energy intake (kcal), macronutrients and micronutrients using computerized nutritional analysis software (Nutritionist III software, version 7.0; N-Squared computing, Salem, OR, USA), which was modified for Iranian foods, and the levels were compared with DRI values [18].
Statistical analyses were carried out using SPSS software (version 17). Quantitative variables were reported as mean ± standard deviation (SD). Pearson and Spearman's correlational analyses were performed to identify relations among variables. All tests were two-tailed, and p < 0.05 was the significance threshold.
This research project was approved by the ethical committee of Tabriz University of Medical Sciences (No. 871).
Results
The mean age, duration of the disease and anthropometric measures of patients, such as weight, height and BMI are shown in
Demographic, anthropometric, clinical and biochemical characteristics of the patients.

BMI: Body mass index; CRP: C-reactive protein; DAS: Disease activity scale; MDA: Malondialdehyde; SD: Standard deviation; TA: Total antioxidant; VAS: Visual analog scale.
All of the patients in the present study had a low active (72%) or sedentary (28%) lifestyle and none of them were smoking.
Intake of total energy, fat (polyunsaturated fatty acids [PUFA], monounsaturated fatty acids [MUFA] and saturated fatty acids [SFA]), protein, vitamins and minerals are presented in
Nutrient intake in women with rheumatoid arthritis.
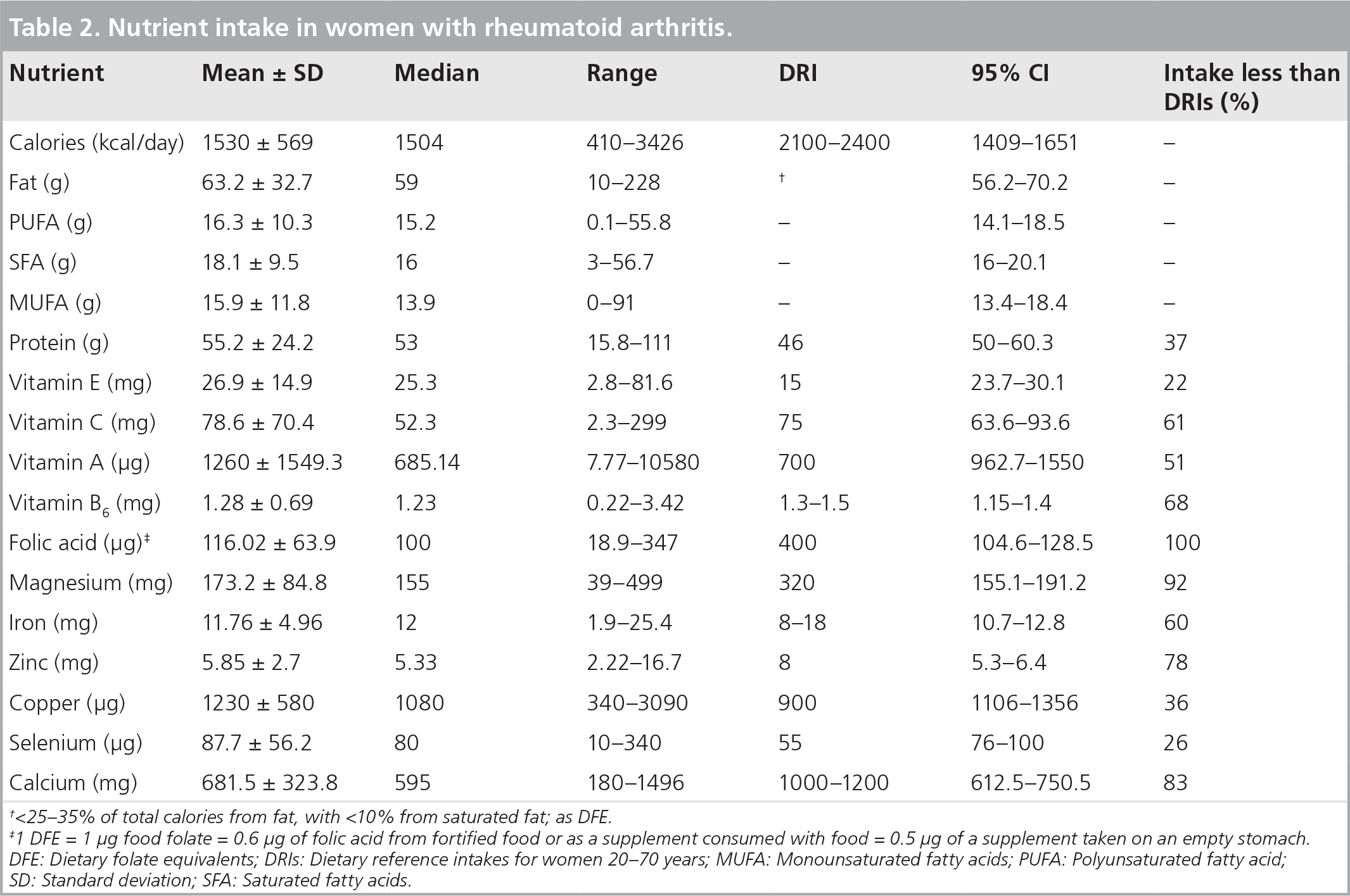
<25–35% of total calories from fat, with <10% from saturated fat; as DFE.
1 DFE = 1 μg food folate = 0.6 μg of folic acid from fortified food or as a supplement consumed with food = 0.5 μg of a supplement taken on an empty stomach. DFE: Dietary folate equivalents; DRIs: Dietary reference intakes for women 20–70 years; MUFA: Monounsaturated fatty acids; PUFA: Polyunsaturated fatty acid; SD: Standard deviation; SFA: Saturated fatty acids.
Approximately 49% of energy of the patients was obtained from carbohydrate, 36% from fat and 14% from protein, and protein intake of them per kilogram was 0.73 ± 0.4. On average, the patients consumed approximately 8 servings from the bread and cereal group, 1.2 servings from vegetables, five servings of fruits, one serving from the milk group, four servings from the meat group and approximately ten servings of fat daily.
There was a negative association between disease activity and calorie intake, however, this association did not reach statistical significance (r = −0.218, p = 0.053). There was no significant relationship between the intakes of different nutrients (antioxidant or pro-oxidant nutrients) and total antioxidant capacity of serum (TA), lipid peroxidation (MDA) and the inflammatory marker CRP.
Discussion
This study is the first one conducted in Iran that assesses the nutritional status of RA patients. Lifestyle aspects of the patients, such as physical activity, smoking habits and supplement intakes were investigated. The biochemical markers of inflammation (CRP), lipid peroxidation (MDA) and total antioxidant capacity of the plasma were also assessed.
Anthropometric measurements of the subjects showed that the average BMI of the patients was in the normal range (20–25 kg/m2); none of the patients had a BMI of less than 18.5, which is the cutoff value for malnutrition. The reported BMI of women with RA in Elkan's study was also in the normal range (24.4 kg/m2), and only 12% of the patients had a BMI of less than 18.5 kg/m2 [13]. The researchers concluded that BMI and nutritional assessment are not appropriate tools for detecting malnutrition in RA patients. One possible reason for these findings is the fact that in RA patients muscle wasting is usually compensated for with an increase in fat mass, and this means that losing weight or BMI would not be likely [19]. A serum albumin level of 3.4 g/dl was used as the malnutrition cut-off point in another study of rheumatic women [20]. According to their findings, approximately 24% of the patients with RA suffered from malnutrition; and there was a positive correlation between malnutrition and the activity of disease. However, since albumin is one of the negative acute-phase respondent proteins, it may not be a reliable indicator of malnutrition in inflammatory conditions such as RA. It seems that newer and better methods are needed for malnutrition assessment of RA patients. Recently, the use of new techniques such as DEXA or whole-body MRI provided more accurate information for diagnosing rheumatoid cachexia [21].
In this study, the average calorie intake of the patients was 1530 kcal, which is very close to the findings of Woolf [22], who assessed the nutritional status of postmenopausal women with RA in the USA, where the mean energy intake was 1570 kcal. In the present study a 3-day, 24-h recall was used to assess dietary intake of the patients, as previously recommended [23]. Some people may underestimate or under-report their intake. However, 3-day, 24-h recall is still the most applicable tool, especially in populations like Iran where illiteracy remains among the older population. Other research findings have indicated that there is a positive correlation between an impaired nutritional status and inadequate intakes of energy and nutrients in patients with RA [22,24].
Several sociological and physiological factors may be involved in this correlation, such as limited ability to shop or prepare meals, medication use related to the disease and the pathology of the disease itself, both of which can cause anorexia and reduced appetite [25]. Another reason for malnutrition in RA patients can be adherence to fad diets. Patients with RA often claim that their symptoms are alleviated by special diets or by simply eliminating certain foods. According to some researchers, this type of symptom alleviation can be explained by food-related antigens, predominantly from protein sources that may provoke hypersensitivity responses and, thus, may increase symptoms of RA [26].
Although intake of nutrients from certain food groups like milk or meat was near the recommended rates, most of our patients believed that some foods, such as chicken or yogurt, aggravate symptoms of their disease when interviewed regarding eating habits. We couldn't find any scientific rationale for this belief.
In the present study, intake of calcium, folic acid, zinc, magnesium and vitamin B6 was below the DRIs, which is inadequate. This is aggravating because even at sufficient levels of intake, most of the medications used by these patients interact with these micronutrients and impair their absorption and metabolism. For example, long-term use of corticosteroids affects calcium metabolism. Methotrexate and sulfasalazine are antagonists of folate metabolism and
In agreement with our study, other researchers have found an inadequate intake of some micronutrients such as calcium, magnesium, zinc, iron, folic acid and vitamin B6 in the diets of women with RA [28,29].
Some biochemical markers of the patients, including MDA, an end product of lipid peroxidation, were also investigated. There are many types of reactive oxygen species (ROS) in the cellular environment of RA patients, which can lead to lipid peroxidation and the production of MDA. As reported elsewhere [30], plasma MDA concentrations have been shown to respond to alterations in antioxidant nutrient status. For example, plasma MDA concentrations in patients with cystic fibrosis are affected by dietary carotenoid alterations or supplementation [31]. However, other studies involving healthy individuals have reported no effects of supplemental β-carotene on erythrocyte MDA [32]. We did not find any significant correlation between MDA concentration and intake of different nutrients. Other studies assessing the nutritional status of RA patients did not examine the lipid peroxidation or total antioxidant capacity of the patients [22,24,27,29]. Although we could not find any relationship between intake of several antioxidant nutrients and serum total antioxidant capacity, antioxidant intake must increase to neutralize the increased oxidative stress in RA patients.
An elevated plasma concentration of CRP is a significant marker of underlying systemic inflammation as well as a sensitive and independent marker of malnutrition. Some previous studies have shown a positive or negative correlation between some micronutrients and CRP concentration. For example, Chiang and colleagues have found a negative correlation between CRP and serum levels of vitamin B6 and concluded that the low vitamin B6 status in RA patients is not caused by lower intake or excessive catabolism of vitamin B6, and is likely to result from the inflammatory process [33]. No significant relationship was observed between intake of different nutrients and CRP concentration, probably owing to the impaired utilization of some nutrients because of the disease process.
This study also had some limitations. It should be mentioned that there was no healthy control group in the present study and therefore nutrient intakes of patients were compared with DRIs. In our previous study to investigate the nutritional status of postmenopausal osteoporotic women in the north-west of Iran [34], we showed a lower intake of calcium and folic acid despite increased BMI compared to the present study. Hence, in order to determine whether these deficiencies are observed only in these patients or the healthy women in this area have also such deficiencies, it is suggested to have an investigation that also includes healthy control group.
Conclusion
As a conclusion, although patients with RA in the present study had a normal BMI, intake of energy and micronutrients such as folic acid, calcium, zinc and vitamin B6 were significantly lower than the recommended values. Inadequate intake of micro- and macro-nutrients is particularly important in these patients owing to the fact that many RA medications adversely interact with these nutrients.
Executive summary
The BMI of the patients in the present study was within the normal range. However, according to the literature, BMI or albumin is not an appropriate tool for assessing nutritional status of rheumatoid arthritis patients and more efficient methods should be developed.
There was a considerable deficiency in the intake of some micronutrients such as folic acid, calcium, magnesium and vitamin B6. This deficient intake is particularly important in these patients owing to the fact that many rheumatoid arthritis medications adversely interact with these nutrients.
Although calorie consumption of the patients was relatively low, intake of protein and some micronutrients, such as selenium was in an acceptable range for most of the patients.
There was an adequate intake of different food groups among the patients however most of the patients eliminate some food items because they believe that these foods (e.g., chicken and yogurt) may aggravate their disease.
There was a negative association between disease activity and calorie intake, however, this association was not statistically significant. There was no relationship between intake of different nutrients and disease activity.
There was no significant relationship between the intakes of different nutrients (antioxidant or pro-oxidant nutrients) and total antioxidant capacity of serum (total antioxidant), lipid peroxidation (malondialdehyde) and the inflammatory marker C-reactive protein.
Future perspective
In the last few years, identification of abnormal regulation of cytokines, particularly TNF-α and IL-1, has provided a rationale for dietary manipulation; because anti-inflammatory diets can potentially retaliate the inflammatory cytokine cascade [7].
It has been long known that plant-based diets can have beneficial effects in some RA patients. However, it is difficult to determine what aspect of the diet is responsible for the observed effects on RA symptoms. Recently some mechanisms have been suggested, such as gut flora alteration by vegetarian diets [35]. In addition a role for some micronutrients such as potassium in alteration of hormonal environment of RA patients has been hypothesized [36].
It seems there is still long way to find the appropriate diet for RA patients and large-scale randomized clinical trials are needed to assess different suggested diets. What we know is an anti-inflammatory diet with low quantities of meat and plenty of fish, whole grains, vegetables and fruits (which are rich in folic acid, magnesium, complex carbohydrates and fiber, vitamin C, E and other antioxidants) have been shown to offer distinct advantages.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
