Abstract
Vaccination is one of the most effective strategies employed to prevent morbidity and mortality from infectious diseases. Pregnancy is considered to be a time when women have consistent contact with their healthcare providers and it presents an opportunity for providers to review their immunization status and to advocate for appropriate vaccination antepartum and in the immediate postpartum period. All forms of immunization, with the exception of live viral or live bacterial vaccines are generally considered to be safe for administration during pregnancy. It is important that healthcare providers counsel pregnant women about the benefits of receiving the vaccines that are recommended during pregnancy as well as the potential risks to the developing fetus. It is imperative that obstetricians and primary care providers are aware of and implement the vaccination guidelines for women, both during pregnancy and in the postpartum period.
Vaccine-preventable diseases remain major causes of illness and death in less developed nations, while most of these diseases have been reduced to rare occurrences in the USA and developed countries. Vaccination is one of the most cost-effective interventions to have been implemented in the field of medicine in over 2 centuries. As a primary prevention tool, it has had a remarkable impact on decreasing infectious diseases in the USA as evidenced by a 100% decrease in indigenous poliomyelitis and diphtheria, a greater than 99% decrease in measles, mumps and rubella, and a 95% decrease in whooping cough
Immunization during pregnancy
Ideally, all women should have their immunization status up to date prior to conception. However, owing to suboptimal vaccine administration in all adult women
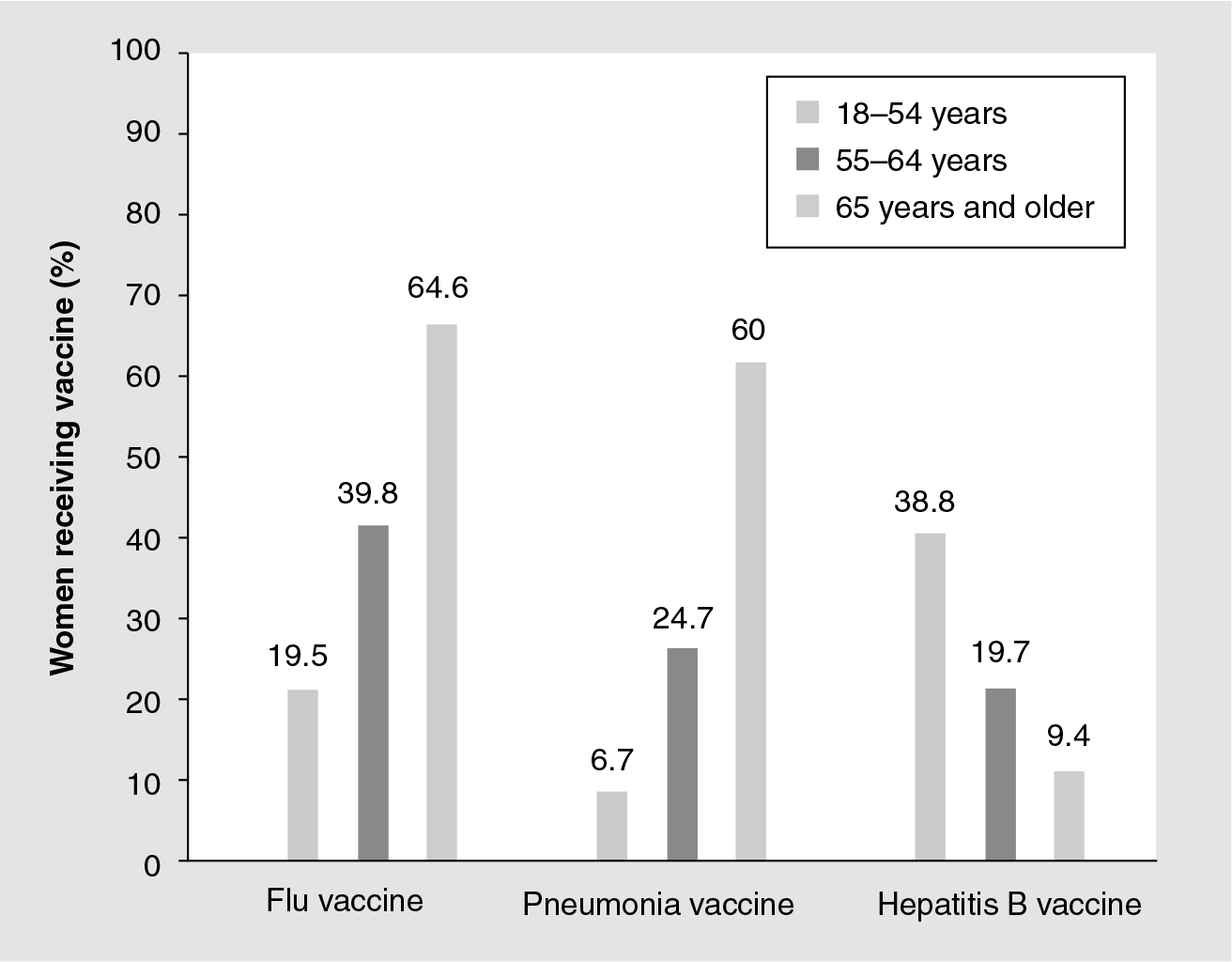
Receipt of selected vaccinations among women aged 18 years and older.
Vaccine pharmacology & theoretical risks in pregnancy
Immunization may be passive through immune globulin preparations or active through toxoids, antigenic components, inactivated virus vaccines and live-attenuated vaccines. All but the live viral or live bacterial vaccines are generally considered safe for administration during pregnancy. Of note, one of the vaccines against typhoid is the only live bacterial vaccine available. The risk of fetal infection associated with maternal immunization with a live vaccine is theoretical. To date, there is no evidence to demonstrate teratogenicity of any of the live vaccines currently available (e.g., measles, mumps, varicella, polio and rubella) [4,5]. Therefore, inadvertent immunization with a live vaccine during pregnancy is not an indication for pregnancy termination. Owing to the theoretical concern for fetal harm, however, live vaccines should not be administered during pregnancy unless the potential risks are outweighed by clear benefits. Furthermore, nonpregnant women immunized with live vaccines should be counseled to delay pregnancy for at least 1 month [101].
Despite maternal immunologic adaptations to pregnancy, in which pregnancy is considered an immunoattenuated state, the immunization of pregnant women appears to be as effective as in nonpregnant women [6,7]. Currently, there are no recommendations suggesting antibody testing or additional doses of vaccine after immunization during pregnancy for any of the currently available vaccines. However, when live-attenuated vaccines are administered during pregnancy, there is the theoretical risk of insufficient control of replication of the vaccine strain, but this has not been proven.
Another controversial subject in regard to immunization in pregnancy is thimerosal, a mercury-containing compound used as a preservative since the 1930s in biological products including vaccines. Concerns first came to public attention in the 1970s owing to an increasing awareness of the neurotoxicity of organomercurials. Subsequent studies have shown that ethylmercury, a breakdown product of thimerosal, does not accumulate and does not therefore cause harm to the fetal brain like methylmercury, and there does not appear to be an increase in neurodevelopmental disorders from exposure to thimerosal-containing vaccines [8–11]. The US Public Health Service and the US FDA have recommended that efforts should be made to eliminate or reduce the thimerosal content in vaccines as part of a strategy to reduce mercury exposure from all sources [12,102]. Currently, all vaccines have a thimerosal-free alternative, with the exception of the Japanese encephalitis vaccine. In the case of vaccinations recommended during pregnancy (e.g., influenza), limitations in the availability of a thimerosal-free alternative should not preclude administration of the vaccine [13].
Immunization & lactation
Vaccines have been studied for their excretion in breast milk and subsequent transmission from mother to infant. In the case of the rubella vaccine, the vaccine virus is frequently excreted in breast milk but has not been reported to cause clinical illness or any other adverse effects in the infant [14]. Varicella vaccine, another live-attenuated vaccine administered to postpartum women, has not been shown to be excreted in breast milk [15]. Therefore, children born to women lacking immunity to varicella should be considered susceptible to varicella regardless of whether or not their mothers are vaccinated while breastfeeding, since it must be assumed that no passive transfer of immunity occurred as a result of vaccination of their mothers [15]. Inactivated, recombinant, subunit, polysaccharide, conjugate vaccines and toxoids pose no risk for mothers who are breastfeeding or for their infants [16].
Vaccination guidelines
In the USA, the The Advisory Committee on Immunization Practices (ACIP) establishes recommendations for vaccine administration in consultation with experts and governmental agencies. These vaccination guidelines are periodically updated and are endorsed by professional organizations, including the American College of Obstetricians and Gynecologists (ACOG) [17]. ACIP recommendations for vaccination against specific infections that may affect pregnant women and their newborn infants are discussed.
Hepatitis A
Hepatitis A is a RNA virus that is transmitted via the fecal–oral route. Infection with hepatitis A may manifest as symptoms such as fever, malaise, anorexia, nausea, abdominal discomfort and jaundice [18]. Symptoms typically resolve within 2 months; however, a small percentage of individuals may be affected by a prolonged or relapsing course. Infection with hepatitis A during pregnancy may be associated with serious complications such as preterm labor [19]. Vertical transmission of hepatitis A infection also has been described [20].
Although data on the safety of the hepatitis A vaccination during pregnancy are limited, pregnancy is not a contraindication to vaccination against hepatitis A. The vaccine is produced from an inactivated virus; therefore, the theoretical risk associated with vaccination during pregnancy is expected to be low [18]. If a woman is anticipated to be at high risk for contracting hepatitis A (e.g., travel to an area where hepatitis A is endemic, such as Central and South America, Africa, Middle East, Asia and Western Pacific), the vaccine is typically recommended.
The hepatitis A vaccine is available as both a single-antigen vaccine and as a combination vaccine containing both hepatitis A virus (HAV) and hepatitis B virus antigens. Both vaccines use inactivated HAV and the hepatitis B virus component is a recombinant protein nonviral antigen. There are currently two available HAV vaccines that are given in two doses, either 6–12 months apart or 6–18 months apart. The combination vaccine is given in three doses at 0, 1 and 6 months [21]. Immune globulin remains available for postexposure prophylaxis, although primary vaccine-based prevention is preferred [21,22].
Hepatitis B
Hepatitis B is a bloodborne DNA virus. Symptoms, when present, may include anorexia, malaise, nausea, vomiting, abdominal pain and jaundice. Those chronically infected are most severely affected and are the primary reservoir for hepatitis B transmission. Unfortunately, 90% of infants infected with hepatitis B become chronically infected [23]. Perinatal transmission from infected mothers and infection from household contacts are the primary sources of infection in early infancy.
Vaccination to prevent hepatitis B infection is extremely important. Ideally, women should be vaccinated prior to pregnancy. However, if a woman did not receive prior vaccination, the hepatitis B vaccine is composed of a noninfectious viral particle and should cause no harm to the developing fetus [24,25]. Therefore, the ACIP recommends counseling pregnant women who may be at high risk for acquiring hepatitis B about vaccination during pregnancy [23]. Those at high risk include women with multiple sex partners or a recent sexually transmitted disease, current injection drug users, or women who have a partner infected with hepatitis B. Nearly 80% of new infections are associated with high-risk sexual behavior or injection drug use [17].
Influenza
Influenza is caused by a group of RNA viruses that belong to the family
The inactivated influenza vaccine has been administered during pregnancy in the USA since the 1960s [28]. As the influenza vaccine has minimal immunogenicity prior to 6 months of age, maternal vaccination during pregnancy has the potential to decrease neonatal influenza. In fact, in one study, immunization during pregnancy was shown to reduce the incidence of laboratory-confirmed influenza in infants up to 6 months of age by 63% as well as lessen febrile influenza-like illness by approximately a third in both young infants and mothers [29]. Only five women, therefore, would need to be vaccinated during pregnancy to prevent a single case of febrile influenza-like illness in a mother or an infant.
Advisory Committee on Immunization Practices and ACOG recommend administration of the trivalent-inactivated influenza vaccine to women who will be pregnant during the influenza season (October through to May) regardless of gestational age [30,31]. Immunization is especially important for women who will be in their third trimester or who will have infants under the age of 6 months during the influenza season [28]. Immunization with the live-attenuated influenza vaccine is not advised during pregnancy [25]. Although these recommendations have been put forth, a suboptimal number of pregnant women ultimately receive the influenza vaccine
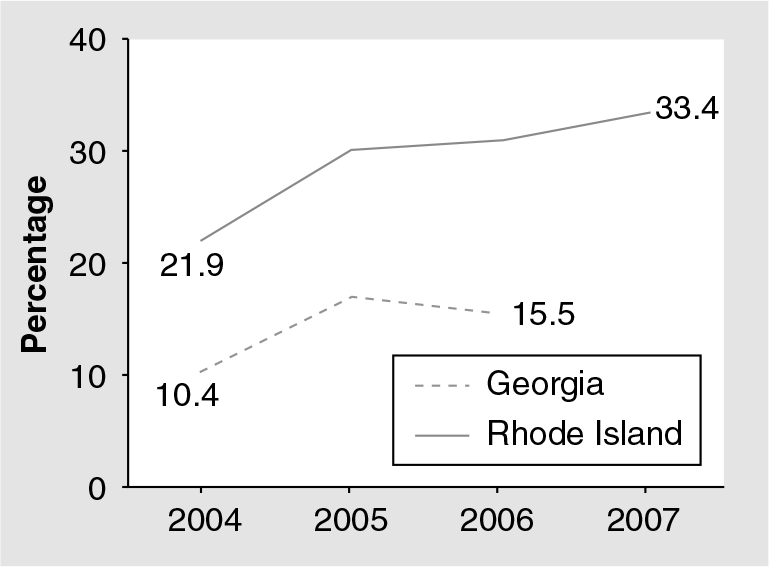
Percentage of women with live births who reported receiving the influenza vaccine during pregnancy in Georgia and Rhode Island (USA).
The H1N1 virus is a specific subtype of influenza A that was determined to be responsible for a worldwide influenza pandemic that began in 2009. The viral strain responsible for the pandemic originated from a reassortment of several swine strains, a human strain and an avian strain [26]. The genetic shift involved in the emergence of the novel H1N1 strain limits the ability of the immune system to recognize and destroy the virus. As with seasonal influenza, cough, fever, headache, sore throat, rhinorrhea, chills and muscle aches are the most common symptoms [33]. Pregnant women are at high risk for influenza-related complications [34]. Furthermore, the mortality rate appears to be higher in pregnant women, particularly if infection occurs in the third trimester [35].
Vaccines for H1N1 became available in 2009 in both live-attenuated and inactivated formulations, and pregnant women were one of the initial target groups for immunization with the inactivated vaccine [33,36]. The USA 2010–2011 influenza vaccine will protect against an H3N2 virus, an influenza B virus and the 2009 H1N1 influenza [103].
Pneumococcus
In addition to preventing pneumococcal infection in pregnancy, immunization during pregnancy is a strategy that can provide young infants protection from these pathogens. Administration of pneumococcal polysaccharide vaccine to women in the third trimester of pregnancy was safe and well tolerated [38]. Efficient transplacental passage of vaccine-induced pneumococcal antibodies was documented with concentrations of pneumococcal antibodies in infants at birth similar to those in their mothers [38].
Increased concentrations of IgA antibodies have been found in breast milk after maternal immunization with the pneumococcal vaccine [39], demonstrating that immunity may be conferred to the newborn infant through this route as well. Women who are at high risk for contracting pneumococcus (e.g., those who are immunosuppressed, have had a splenectomy, or who have sickle-cell disease) and who have not previously received the pneumococcal vaccine are candidates for vaccination during pregnancy [25].
Polio
Poliomyelitis is caused by one of three serotypes of the poliovirus. The majority of poliovirus infections are asymptomatic. In those who are symptomatic, fever may be followed by aseptic meningitis or paralytic disease [40]. If a woman is infected during pregnancy, neonatal mortality approaches 40% [25]. Fortunately, poliomyelitis has been nearly eradicated in the western world with the development of vaccines against polio in the 1950s. However, polio remains endemic in several countries (Nigeria, India, Pakistan and Afghanistan) [41].
There are two polio vaccines available: the inactivated vaccine and the live-attenuated oral vaccine. The live-attenuated oral vaccine is reserved for the control of polio outbreaks and endemic communities. The live-attenuated polio vaccine has been studied in pregnancy and has proven to be a safe alternative to the inactivated vaccine to be given to unimmunized or partially immunized pregnant individuals [42]. Although no adverse outcomes have been associated with the inactivated or live-attenuated oral polio vaccines when given during pregnancy, vaccination should be avoided on theoretical grounds unless necessary [25,40]. Therefore, if a pregnant woman requires immediate protection, the inactivated or live-attenuated polio vaccine may be considered, with preference given to the nonlive vaccine. If the woman is aged 18 years or older, the inactivated polio vaccine should be given in a three-dose series – the second dose 4–8 weeks after the first and the third dose 6–12 months after the second [40].
Measles, mumps & rubella
Infection with the measles virus typically leads to fever, cough, rhinorrhea and a generalized rash. Complications include diarrhea, bronchopneumonia and encephalitis, which may result in permanent brain damage. Measles infection during pregnancy is associated with miscarriage, premature labor and low infant birth weight [43].
Painful swelling of the salivary glands, classically the parotid glands, is the presenting symptom in the majority of infections with the mumps virus. Swelling of the salivary glands may precede fever, headache, malaise, myalgia and anorexia. Although serious complications, such as meningoencephalitis, may occur, mumps is typically a mild and self-limited illness. Mumps infection in the first trimester of pregnancy is associated with an increased risk of fetal death [43].
Infection with the rubella virus typically manifests as fever and malaise followed by a rash that begins on the face and spreads to the trunk and extremities. Complications such as arthralgia, encephalitis and thrombocytopenic pupura may result, particularly in adults [44]. Congenital rubella is a severe condition that is associated with deafness, cataracts, cardiac defects, microcephaly, mental retardation and hepatosplenomegaly. Congenital rubella may affect up to 85% of fetuses infected in the first trimester [44].
Before vaccination against rubella became available in the USA, approximately 20,000 cases of congenital rubella syndrome occurred during a rubella epidemic from 1964 to 1965 [43]. Due to the potential for congenital rubella syndrome, vaccination against rubella prior to pregnancy is particularly important. As the rubella vaccine given alone or in combination with measles and mumps is a live vaccine, pregnancy should be delayed for 1–3 months after immunization. Furthermore, immunization is contraindicated during pregnancy. Inadvertent vaccination during pregnancy, however, is not an indication for termination as no deleterious effects on the developing fetus have been confirmed [44]. Women who are discovered not to be immune to rubella during pregnancy should be vaccinated in the immediate postpartum period. Breastfeeding is not a contraindication to vaccination.
Tetanus, diptheria & pertussis
Tetanus occurs when
Diphtheria is a severe respiratory infection caused by
Pertussis is a prolonged respiratory infection caused by the Gram-negative coccobacillus
In 2005, two tetanus-reduced diphtheria and acellular pertussis (Tdap) vaccines were licensed for use in the USA. These vaccines provide added coverage for pertussis and are designed for single-dose use in place of the next dose of the tetanus and diphtheria toxoids vaccine (Td). Administration of Tdap during routine wellness visits, when indicated, is the preferred strategy to ensure protection from tetanus, diphtheria and pertussis. Pregnancy need not be delayed after vaccination with Tdap as the vaccine only contains toxoids and purified bacterial components [45]. Pregnancy is not a contraindication to Tdap vaccine and in special situations, Tdap may be administered during pregnancy (i.e., exposure to a pertussis outbreak). However, women must be counseled that there is a lack of data on the safety of Tdap in pregnancy and administration is generally delayed until after the first trimester despite there being no data demonstrating an adverse effect in early pregnancy. Women who have not previously received the Tdap vaccine should be counseled for immunization in the immediate postpartum period, and it may be administered as soon as 2 years after the most recent Td vaccination [45]. Any adults or adolescents who will be in contact with an infant should receive Tdap as well.
If more than 10 years have elapsed since a woman's last tetanus and diphtheria booster, Td is recommended during pregnancy. However, administration of Td may be substituted with Tdap in the immediate postpartum period if sufficient protection from tetanus and diphtheria will be present until delivery. Protection is likely if a pregnant woman has received complete childhood immunization and one or more Td boosters if less than 31 years old or two or more Td boosters if 31 years or older, if the level of serum tetanus antitoxin is 0.1 IU/ml or greater by ELISA [45].
Typhoid
Typhoid, characterized by fever, gastroenteritis and nonbloody diarrhea, occurs upon ingestion of food or water contaminated with the bacteria
The typhoid vaccine is not contraindicated during pregnancy and women who are anticipating travel to endemic areas (e.g., Latin America, Africa and Asia) should be counseled about vaccination. Several typhoid vaccines are available for use in the USA – an oral live-attenuated vaccine, a parenteral heat-phenolinactivated vaccine and a parenteral capsular polysaccharide vaccine [47]. Although limited data exist on the safety of these vaccines during pregnancy, use of the capsular vaccine has been recommended by certain experts [25].
Varicella
Varicella infection, although uncommon in adults, may result in significant maternal and fetal morbidity. Nearly 30% of pregnant women affected by varicella will succumb to pneumonia [44]. Furthermore, infection during pregnancy may result in congenital varicella syndrome, neonatal varicella or herpes zoster during infancy [48]. Congenital varicella infection may be manifested by cerebral cortical atrophy, microcephaly, low birth weight, cutaneous scaring and limb hypoplasia.
Immunity to varicella should be determined during pregnancy. Evidence of immunity to varicella includes the documentation of age-appropriate vaccination, laboratory evidence of immunity, or verification of a history of varicella or herpes zoster by a healthcare provider [48]. In women who are susceptible to varicella, a two-part vaccination series should be initiated postpartum, with the second dose given 4–8 weeks after the first [17]. Breastfeeding is not a contraindication to vaccination against varicella [48]. As the varicella vaccine is a live-attenuated vaccine, it should not be administered during pregnancy and pregnancy should be delayed for at least 4 weeks after immunization. However, unintentional vaccination during pregnancy is not a reason for termination.
New developments
Exciting advancements in the creation of vaccines to prevent other infections that may affect pregnant women and their newborn infants are underway. For example, congenital cytomegalovirus (CMV) infection may result in permanent hearing, cognitive and motor impairments in affected infants. A CMV vaccine composed of recombinant CMV envelope glycoprotein B has recently undergone a Phase II placebo-controlled randomized double-blinded trial in nonpregnant subjects with favorable results suggesting that it may have the potential to prevent CMV infection in young women and congenital CMV in infants [49].
On another front, vaccines against malaria are currently being developed. Pregnant women are more susceptible to malaria, particularly in the first and second trimesters [50]. Furthermore, malaria infection during pregnancy confers greater maternal as well as neonatal risk, including anemia and a reduction in birth weight [51]. Vaccines targeting the dominant variant surface antigen, VAR2CSA, as well as other
Group B Streptococcus (GBS) infection is another target for potential immunization. Despite a substantial increase in antibiotic prophylaxis during delivery, the incidence of late-onset disease in infants has not decreased [52]. Approximately 25% of infants with late-onset GBS present with meningitis, and those affected by meningitis are at risk for permanent neurological impairment, including cerebral palsy, hydrocephalus and mental retardation. GBS conjugate vaccines have undergone Phase 1 and II testing, including a Phase 1 randomized double-blinded controlled trial conducted in pregnant women at 30–32 weeks gestation [52]. Immunization against GBS has the potential to improve both maternal and neonatal outcomes.
Finally, respiratory syncytial virus (RSV) has been studied as an infection that could be reduced through maternal immunization. RSV is the most frequent cause of lower respiratory tract disease in infants worldwide. Infection typically occurs early in life and is more severe in the younger pediatric population. Low umbilical cord RSV titers are associated with the increased risk for neonatal disease, while breastfeeding is noted to be protective against infection. These latter observations led Munoz and colleagues [53] to undertake a study examining the safety and immunogenicity of an RSV purified protein subunit vaccine in women in their third trimester of pregnancy. They demonstrated that the experimental vaccine was safe, well tolerated and immunogenic in these pregnant women. Infants born to vaccine recipients did not display any adverse events related to maternal vaccination and had significant concentrations of neutralizing antibody in their bloodstream. Vaccine-specific antibodies were also detected in maternal breast milk. Given the preliminary nature of this study, clinical efficacy was not detected in infants of vaccinated mothers but the findings did support the need for further investigations in this regard.
Medicolegal & ethical considerations
Studies have not conclusively established an association between the vaccines discussed above and maternal or fetal harm. It is worth noting that, there is not a single vaccine specifically FDA-approved for use in pregnancy, primarily due to the absence of industry-sponsored trials in pregnant subjects. Therefore, critical efficacy and safety data are lacking and most clinically based vaccine recommendations extrapolate these presumptive outcomes. However, immunization during pregnancy is fraught with numerous hypothetical risks. These risks include:
Transmission of an attenuated virus to the placenta or fetus
Reproductive effects including miscarriage, congenital malformations and growth retardation
Unpredictable or idiosyncratic reactions
Ineffectiveness of the vaccine during pregnancy
Embryotoxicity of immunoglobulins produced after vaccination [54]
Since the creation of the National Vaccine Injury Act of 1986, healthcare providers are required to maintain permanent immunization records and to report any suspected adverse reactions to specific vaccines. Adverse reactions thought to be associated with a vaccine should be reported to the Vaccine Adverse Events Reporting System (VAERS), established in 1990 [17]. VAERS is managed by the US FDA and CDC [55].
Although inadvertent vaccination during pregnancy or within 3 months prior to conception are not indications for the termination of pregnancy, 80 elective terminations were reported to VAERS from 1990 to 2006 [55]. 29 of the elective terminations were associated with the varicella vaccine and 25 with the measles, mumps and rubella vaccine. Therefore, it is important for healthcare providers to enquire about the potential of a woman being pregnant at the time of vaccine administration as well as to advise that pregnancy should not be attempted for 1–3 months after vaccination with a live-attenuated virus. The expected rate of unknown early pregnancy or conception within a 4-week period is six pregnancies per 1000 reproductive-aged women [55]. Even though the risk of unintentional vaccination during pregnancy exists, advisory groups do not currently recommend routine pregnancy testing prior to administration of a live-attenuated vaccine [16].
There are complicated liability issues in the setting of vaccination during pregnancy. Not only is there a potential for adverse events concerning the mother, but there is the possibility of harm to her unborn fetus. In the USA, three types of claims may be retained when an adverse reaction is associated with a vaccination:
Failing in advising
Failing in the conception of the product
Failing in the manufacturing of the product [56]
The National Vaccine Compensation Program (VICP) was established in 1986 and provides federal, no-fault compensation for individuals injured by covered vaccines. However, this program does not address the injuries to the fetus that might have resulted from vaccination during pregnancy. Extension of this program to include vaccines recommended for administration during pregnancy could encourage the development and supply of new vaccines to improve maternal and fetal outcome [57].
Healthcare providers must remember that even though explicit recommendations exist concerning vaccination during pregnancy and the immediate postpartum period, maternal autonomy must be respected. After appropriate counseling, a woman has the right to accept or decline a vaccination, despite what may be in the best interest of her or her child. Furthermore, pregnant women are considered a vulnerable population [56]. Particular attention, therefore, must be paid to informed consent in the setting of maternal vaccination.
Executive summary
Vaccination is one of the most cost-effective interventions implemented in the field of medicine in over two centuries.
Pregnancy provides an opportunity for healthcare professionals to provide primary prevention measures.
Obstetricians and primary care providers should be aware of the vaccination guidelines published by the CDC Advisory Committee on Immunization Practices (ACIP).
Immunization may be passive through immune globulin preparations or active through toxoids, inactivated virus vaccines and live-attenuated viral and bacterial vaccines.
All forms of immunization, except the live viral or bacterial vaccines, are generally considered safe for administration during pregnancy.
Breastfeeding is not a contraindication for immunization with inactivated or live-attenuated virus vaccines, with the exception of the smallpox vaccine.
The hepatitis A vaccine is not contraindicated during pregnancy.
The hepatitis A vaccine may be given to pregnant women who are at high risk for contracting the disease (e.g., travel to an area where hepatitis A is endemic).
The hepatitis B vaccine is composed of a noninfectious viral particle.
Pregnant women who have not been vaccinated against hepatitis B and who may be at high risk for acquiring the disease should be counseled about vaccination during pregnancy (e.g., women with multiple sex partners or a recent sexually transmitted disease, current injection drug users, healthcare providers or women who have a partner infected with hepatitis B).
The inactivated influenza vaccine is recommended for all women who will be pregnant during the influenza season regardless of gestational age.
The USA 2010–2011 influenza vaccine will protect against an H3N2 virus, an influenza B virus and the 2009 H1N1 influenza.
The pneumococcal vaccine may be given to pregnant women at risk for contracting the disease (e.g., those who are immunosuppressed, have had a splenectomy or who have sickle-cell disease).
Vaccination against polio may be considered if a pregnant woman requires immediate protection against the polio virus.
Immunization with the mumps, measles, rubella vaccine is contraindicated during pregnancy.
Women who are not immune to rubella should be vaccinated in the immediate postpartum period.
Pregnancy should be delayed for at least 1 month following vaccination with the mumps, measles, rubella vaccine.
If more than 10 years have elapsed since a woman's last tetanus and diphtheria booster, tetanus and diphtheria toxoids vaccine is recommended during pregnancy.
Antepartum tetanus-reduced diphtheria and acellular pertussis administration may be delayed until the immediate postpartum period if sufficient protection from tetanus and diphtheria is anticipated during the pregnancy.
Women who have not previously received the tetanus-reduced diphtheria and acellular pertussis vaccine should be counseled for immunization in the immediate postpartum period.
Women who are anticipating travel to endemic areas should be counseled about vaccination. In these situations, a nonlive form of the vaccine should be used.
The varicella vaccine is contraindicated during pregnancy.
Immunity to varicella should be determined during pregnancy, and vaccination should be initiated postpartum for those who are susceptible.
Pregnancy should be delayed for at least 1 month after vaccination.
Future perspective
The adult vaccine platform is rapidly expanding, and in this regard the obstetrician–gynecologist must assume a more prominent role as patients advocate for vaccine protection. This implies the need for ongoing provider education to the already established adolescent and adult vaccine schedules, logistics pertaining to develop efficient office-based immunization programs and insurer recognition that the obstetrician–gynecologist carries this important mandate.
At the present time, not a single vaccine is specifically FDA approved for use in pregnancy. Future vaccine trials must include pregnant women, especially when a study vaccine may be indicated during pregnancy. This will allow for an expanded approved indication for use and will help generate critical safety data in this patient population and their progeny.
As cited in this article, some newly developed vaccines may have a specific indication in pregnancy owing to maternal risks, or equally important, to protect the fetus and neonate via passive antibody passage across the placenta. These research programs need to continue and be expanded.
Regulatory agencies must, in the future, deal head on with pregnancy-related issues as they pertain to vaccine administration. This would include the National Vaccine Compensation Program recognizing the fetus and newborn as having the potential for injury and perhaps the need for compensation through this mechanism.
Adolescent pregnancy is a common occurrence in the USA; therefore, better medico–legal definitions for emancipation should be established, so that vaccine needs can be addressed by the care provider during the course of pregnancy management in this vulnerable patient population.
Footnotes

