Abstract
Obesity is a worldwide health problem and its prevalence is reaching epidemic proportions. As obesity does not spare women of childbearing age, obstetric anesthesiologists will increasingly be exposed to the challenges of anesthesia in this population. The purpose of this article is to give the reader a thorough understanding of the anesthetic implications of obesity relating to cesarean deliveries. Obesity is associated with hypertension, diabetes, obstructive sleep apnea and other comorbidities. It increases the risk of cesarean delivery, postpartum wound infections and deep venous thromboembolism. Obese parturients are prone to anesthetic complications such as aspiration of gastric contents, difficult monitoring, positioning, airway management and challenging neuraxial techniques. A thorough precesarean delivery preparation should include an evaluation by an anesthesiologist for women with a BMI over 40 kg/m2 and institution of an antacid prophylaxis protocol, thromboprophylaxis and antibiotic prophylaxis. Regional anesthesia should ideally be used in all obese parturients unless contraindicated. The goals of postpartum care include efficacious analgesia, physiotherapy and early mobilization. Monitoring and vigilance in an intensive care unit or step-down units should be considered for morbidly obese women.
Keywords
It is not surprising that the WHO designates the worldwide epidemic of obesity as ‘globesity’. In 2005, it was estimated that 1.6 billion adults were overweight and at least 400 million adults were obese [201]. The most recent statistical report from the Health and Social Care Information Center in England reported that 25% of women were obese and 32% were overweight [1]. This number indicates a marked increase in the proportion of obese women, which represented 13% of the population in 1993. Women of childbearing age are not spared by this epidemic, as approximately 12% of women in the 16–24-year-old age group and almost 25% of women aged 35–44 years in England were obese in 2008 [2]. These figures emphasize the importance for anesthesiologists involved in obstetrical practice to be knowledgeable about the implications of obesity in the obstetrical population.
Obesity poses significant challenges to obstetric anesthesiologists: increased incidence of comorbidities such as hypertension, diabetes and cardiovascular disease; higher rates of elective and emergent cesarean deliveries; and increased difficulties and risks with regional and general anesthetic procedures [3–5]. Indeed, the latest report from the Confidential Enquiry into Maternal and Child Health (CEMACH) in the UK states that obese pregnant women with a BMI above 30 are far more likely to die, and four of the six direct deaths attributable to anesthesia occurred in obese parturients [6]. Taking this into account, the UK National Collaborating Centre for Women's and Children's Health antenatal guideline (2008) recognizes obesity as one of the conditions for which additional care to baseline clinical care for pregnant women is required [7].
Before any anesthetic procedure on obese pregnant subjects can be performed, several considerations must be evaluated
Definition
The BMI is a widely used weight:height ratio calculated by dividing the subject's weight (in kilograms) by the square of the height (in meters). In the UK, the Centre for Maternal and Child Enquiries (CMACE) and the Royal College of Obstetricians and Gynaecologists (RCOG) Joint Guidelines on Management of Women with Obesity in Pregnancy defines obesity as a BMI over 30 kg/m2 at the first antenatal consultation and proposes using the WHO classification described in
WHO definition of obesity.

Physiologic changes
Respiratory system
Hormone- and anatomic-induced changes on the respiratory system in term parturients are significant and many of these changes are exacerbated by obesity
Pregnancy- and obesity-induced physiologic changes to the respiratory system.

More than one arrow represents the degree of intensity.
↓: Decreased; ↑: Increased; ↔: Unchanged; FEV1: Forced expiratory volume in 1 s; PaCO2: Partial pressure of carbon dioxide; PaO2: Partial pressure of oxygen; VC: Vital capacity; V/Q: Ratio of ventilation to perfusion.
Adapted with permission from [11] © John Wiley and Sons.
Anesthetic considerations of the obese pregnant woman
Difficult venous access
Difficult blood pressure monitoring
Inadequate equipment
Longer surgical times
Increased risks of aspiration of gastric content
Increased oxygen requirements/rapid desaturation
Increased risk of supine hypotension syndrome
Difficult airway management
Difficult neuraxial anesthetic techniques
Cardiovascular
Pregnancy- and obesity-induced physiologic changes on the cardiovascular system.
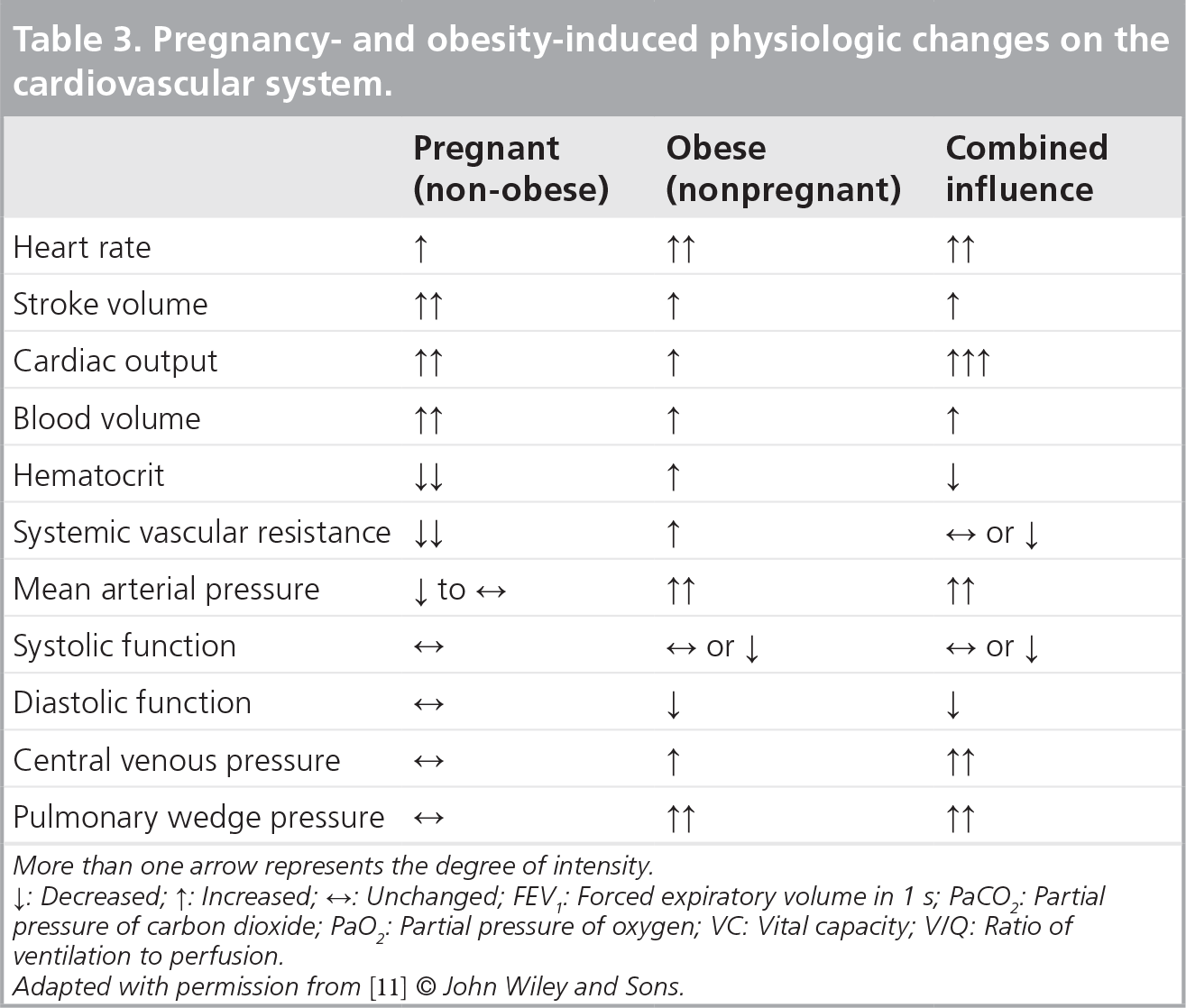
More than one arrow represents the degree of intensity
↑: Decreased; ↓: Increased; ↔: Unchanged; FEV1: Forced expiratory volume in 1 s; PaCO2: Partial pressure of carbon dioxide; PaO2: Partial pressure of oxygen; VC: Vital capacity; V/Q: Ratio of ventilation to perfusion.
Adapted with permission from [11] © John Wiley and Sons.
The supine hypotension syndrome is a well-known phenomenon caused by compression of the inferior vena cava by the gravid uterus. The resulting impaired venous return leads to reduced cardiac output, a sudden drop in maternal blood pressure, reflex tachycardia and potentially cardiovascular collapse. This situation may be prevented by slightly tilting the mother's abdomen to the left to reduce the compression. The supine hypotension syndrome has also been observed in obese patients, and in fact can be exacerbated in obese parturients, where the large abdomen may further compress the great vessels.
Gastrointestinal
As a result of hormone influences on the relaxation of the lower esophageal sphincter and gastric transit time, term pregnant women are prone to gastroesophageal reflux disease and aspiration of gastric contents during general anesthesia [9–11,14]. The gravid uterus increases the intra-abdominal pressure, hence further predisposing the parturients to gastroesophageal reflux disease. Gastroesophageal reflux disease is exacerbated in the obese population, owing to a further increase in the intra-abdominal pressure resulting from the large abdominal panniculus, and a high incidence of hiatus hernia. However, Wong et al. demonstrated that gastric emptying is not delayed in the obese nonlaboring parturient and it is suggested that fasting guidelines should not be modified from those applying for lean pregnant women scheduled for elective cesarean delivery [16]. One should bear in mind, however, that obesity is a significant risk factor for diabetes [7], which may cause gastroparesia and delayed gastric transit. Labor, on the other hand, delays gastric emptying, particularly in the overweight and obese population [17]. Considering all these risk factors for the aspiration of gastric contents, it is recommended that second and third trimester parturients, whether in labor or not, should be considered to have ‘full stomachs’ and their anesthesia managed accordingly [18].
Presurgical evaluation
Since obesity is frequently associated with several concomitant diseases
Obstructive sleep apnea
Although pregnant women may suffer from disordered sleep breathing patterns, the prevalence of obstructive sleep apnea (OSA) in this population is unknown [21]. Pregnancy may confer protection against OSA, secondary to the hormone-induced increase in respiratory drive, a preference for sleep in the lateral decubitus position and other changes in the mother's respiratory nocturnal pattern [21]. On the other hand, association between obesity and sleep disorders such as OSA are well recognized [22]. OSA should be suspected in patients with a BMI over 35 kg/m2, a neck circumference of 16 inches, who demonstrate pauses in breathing during sleep and who awaken from sleep with choking sensations [23]. OSA is also associated with chronic hypertension, congestive heart failure, myocardial infarction, cerebrovascular accidents, increased mortality, diabetes and metabolic syndrome [24]. Therefore, screening for OSA, preoperative polysomnography and continuous positive airway pressure treatment may be advisable during the antenatal assessment [24,25]. Further elements relating to postoperative care of patients with OSA are discussed in the postoperative care section.
Antacid & antireflux drug prophylaxis
It is generally agreed that a gastric pH below 2.5 and gastric volume above 25 ml are risks factors for the development of Mendelson's syndrome in the event of aspiration of gastric contents [17,26]. These numbers have often been debated, yet antacid prophylaxis is recommended in modern obstetric anesthesia [27]. In a recent meta-analysis on interventions to reduce risks of aspiration pneumonitis in parturients undergoing cesarean delivery, Paranjothy et al. concluded that H2 antagonists in combination with nonparticulate antacids (such as sodium citrate) significantly reduced the risk of intragastric pHs below 2.5 at intubation compared with placebo [26]. The literature investigating the efficacy of metoclopramide included in this meta-analysis could not provide evidence for its routine use as an antireflux prophylactic agent. There is no formal guideline with regards to preoperative antacid and aspiration prophylaxis, and practice may significantly vary from one institution to another. A national survey on the subject in the UK revealed that ranitidine 150 mg administered orally every 6 hours was the most frequent antacid agent given during labor [28]. Prokinetic drugs (e.g., metoclopramide) and proton pump inhibitors were less likely to be used. A total of 61% of the obstetric units stratified the risk of aspiration during cesarean delivery (obesity was considered a risk factor in this survey) before deciding to administer prophylaxis, whereas it was given routinely in 32% of the surveyed units. An example of an antacid prophylaxis regimen for elective and emergency cesarean deliveries is provided in
Antibiotic prophylaxis
Postoperative wound infection is a frequent incidence after cesarean delivery, which is exacerbated in the obese population, and narrow-spectrum antibiotic prophylaxis (e.g., cefazolin) is recommended [29]. Although traditionally administered after cord clamping, recent data suggested that antibiotics administered 30 min before skin incision may reduce postcesarean wound infection by up to 50% [29,30].
In addition, a thorough anesthetic evaluation will include an examination of the airway
Technical aspects
Sometimes even a straightforward procedure can become a real challenge in obese patients. Noninvasive blood pressure measurements must be taken with an appropriate-sized blood pressure cuff in order to avoid overestimation of the true blood pressure. Even then, the cuff may sometimes slide from its position when inflating against an obese patient's funnel-shaped arm and it may help to apply the cuff around the forearm instead. In selected cases, invasive monitoring of blood pressure with an arterial line may be indicated [6].
Obtaining appropriate peripheral venous access may also prove considerably difficult. No anesthetic procedure should be initiated before a reliable venous cannulae is inserted, hence it might be necessary to insert a central venous line. Ultrasound (US)-guided insertion of a central venous line is now the standard of care in the UK and an ultrasonographic device may increase the success rate and reduce the risks associated with central venous cannulation in this population [31].
Equipment adapted to the size and weight of morbidly obese pregnant women must also be available. It is imperative that the dimensions and the weight-bearing capacity of the operation table fit the size and weight of the parturient. In addition, retractors, bars and other tools may be required to retract the abdominal pannus in order to achieve adequate surgical exposure. In morbidly obese parturients where abdomen retraction may impair proper ventilation and oxygenation, it is proposed that a vertical and cephalad suspension of the panniculus may alleviate maternal hypoxia and hypotension

Angled suspension of abdominal pannus.
Obesity-associated comorbidities
Chronic hypertension
Coronary artery disease
Cerebrovascular disease
Diabetes mellitus
Gastroesophageal reflux disease
Asthma
Obstructive sleep apnea
Nonalcoholic fatty liver disease
Thromboembolic disease
Depression
Obesity-related risk in pregnancy
Maternal death
Maternal morbidity
Gestational hypertension
Pre-eclampsia/eclampsia
Gestational diabetes
Thromboembolism
Instrumental delivery
Cesarean delivery
Postcesarean wound infection
Postpartum hemorrhage
Macrosomia
Stillbirth
Protocol for antiacid prophylaxis prior to cesarean delivery used in the authors’ institution
All pregnant women from 20 weeks of gestation to 48 h after delivery should have antacid prophylaxis
All patients for elective cesarean section must receive oral antacid premedication
This includes: ranitidine 150 mg p.o. the night before surgery (22:00); ranitidine 150 mg + metoclopromide 10 mg p.o., 1 h before surgery; sodium citrate 0.3 M, 30 ml p.o. just before induction if general anesthetic
If the patient did not receive any oral antacid premedication during labor:
– Metoclopromide 10 mg iv.; ranitidine 50 mg iv.; sodium citrate 0.3 M, 30 ml p.o.
– If general anesthetic, consider emptying the stomach with a large orogastric tube with the mother still anesthetised at the end of the operation before extubating. Avoid nasogastric route to prevent epistaxis
iv.: Intravenous; p.o.: Orally.
Warnings of difficult laryngoscopy during airway examination
Long upper incisors that are significantly anterior (protuberant) to the lower incisors during normal jaw closure
Incapacity of patients to bring mandible incisors in front of maxillary incisors and bite the upper lip
Limited mouth opening with distance of less than 3–4 cm between upper and lower incisors
Receding mandible, especially if thyromental distance is less than 5 cm (three large finger breadths)
Impaired visualization of the uvula and faucial pillars in seated patients with protruded tongue
Large tongue in relation to the oral cavity
High-arched palate associated with a narrow and long mouth
Short, thick and/or muscular neck
Limited range of motion – particularly extension – of head and neck
Regional anesthesia
Single-shot spinal, epidural or combined spinal–epidural anesthesia
Single-shot spinal (SSS) anesthesia is the most popular anesthetic approach to provide adequate anesthesia to women undergoing elective cesarean delivery. It provides a reliable, rapid-onset and dense block. An acceptable block is characterized by a loss-of-touch sensation to the T5 dermatome [33]. However, as obesity may increase surgery time, SSS anesthesia, which is of limited duration, may expose the mother to unacceptable intraoperative pain. By inserting an epidural catheter into the epidural space, the anesthesiologist may then use incremental doses of local anesthetic to prevent intraoperative breakthrough pain. In a systematic review published in 2004, Ng et al. compared SSS to epidural anesthesia for cesarean delivery in non-obese parturients [34]. Failure rate, need of intraoperative analgesia, maternal satisfaction and need for conversion to general anesthesia were similar with the two techniques. As expected, the onset of surgical block was faster, and the need for treatment of hypotension was greater with SSS. An increasingly popular alternative to the SSS and epidural anesthesia is to perform a combined spinal–epidural (CSE) technique. A CSE provides the reliability of a spinal block and the flexibility of epidural anesthesia. A description of the ‘needle-through-needle’ technique is provided in

Combined spinal–epidural ‘needle-through-needle’ technique.
Technical aspects of neuraxial approaches
Neuraxial techniques in the obese pregnant population may prove particularly difficult and yield a higher risk of failure and complications than in their non-obese counterparts. Inability to palpate the spinous process and to identify the midline due to fat tissue accumulation in the lumbar area explain in part these observations [35]. Deeper skin-to-epidural space distance and false positives when using the loss-of-resistance technique due to fat pockets and hormonal softening of the interspinous and flaval ligaments also increase the difficulty in finding the epidural space and increase the risk of accidental dural puncture. The anesthesiologist may partially overcome these difficulties by: using longer spinal and epidural needles; asking the patient if she feels that the puncture is performed in the midline [36]; ‘probing’ the subcutaneous fat with a needle to determine the position of a spinous process lying above and below a lumbar intervertebral space; and using ultrasonographic imaging [37]. US imaging is becoming an increasingly popular aid in performing neuraxial blockade. It may help to identify the midline, localize the epidural space and measure the skin-to-epidural space distance [38]. Using US imaging, Grau et al. observed several pregnancy-induced changes in the spinal anatomy of term parturients: smaller optimal puncture site on the skin, narrowing of the interspinous channel and epidural space, and epidural space deformation caused by soft-tissue changes [39]. Although the visibility of several anatomical landmarks was reduced during pregnancy, the authors concluded that US imaging may provide invaluable prepuncture information and that this technique will become useful clinically in the future.
Contraindications to regional anesthesia
Refusal of patient
Coagulopathy
Sepsis
Infection at puncture site
Hypovolemia
Intracranial hypertension
Spinal and other CNS lesions (relative)
Local anesthetic allergy
The lateral decubitus position for insertion of the epidural catheter in obese patients is often advocated. In laboring obese parturients receiving epidural analgesia, Bahar et al. showed that the Trendelenburg lateral decubitus position reduced the incidence of accidental epidural venous cannulation, compared with the normal lateral decubitus and sitting positions [40]. Moreover, a change in position from sitting to lateral decubitus position after having fixed the catheter to the skin may cause a significant outward movement of the catheter (potentially out of the epidural space) and may lead to failure of the epidural component of the CSE to provide adequate analgesia or anesthesia [41]. However, it is unadvisable, especially in the morbidly obese parturient, to inject an anesthetic dose of local anesthetic in the intrathecal space while the patient is in a prolonged Trendelenburg position, as it may result in an undesired cephalad extension of the block. We suggest that, when performing a CSE, the patients should be either sitting or lying in a lateral decubitus position and, as proposed by other authors [41,42], the catheter should be threaded more than 4 cm in the epidural space. This would minimize the risks of the tip of the epidural catheter moving outside the epidural space.
Reliability of epidural catheters
A concern related to the epidural catheter used as part of a CSE resides in the fact that its efficacy is not verified prior to initiating the surgical procedure. As the cesarean delivery of an obese mother may last longer than expected, the chances for ‘top-up’ requirements through this unverified catheter may be higher. Failure to provide adequate anesthetic block would negate all the benefits for which a CSE was ultimately performed; that is, it may force the anesthesiologist to induce general anesthesia. In a study comparing epidural anesthesia with CSE anesthesia for elective cesarean delivery in a non-obese obstetrical population, Davies et al. demonstrated that a CSE technique provided lower intraoperative pain scores and better maternal satisfaction [43]. Several large retrospective studies have confirmed that an epidural catheter placed as part of a CSE technique for labor analgesia has a high rate of success [44]. Norris et al. showed that the efficacy of an epidural catheter at the time of cesarean delivery was not altered by the technique used (stand-alone epidural vs CSE) to provide labor analgesia [45]. It is probable that when a CSE is performed with the needle-through-needle technique, the presence of cerebrospinal fluid (CSF) in the spinal needle confirms the adequate placement of the tip of the epidural needle in the epidural space, thus increasing the chances of appropriate insertion of the catheter.
Local anesthetic dose for spinal & CSE anesthesia
In the non-obese obstetric population, the 95% effective dose for intrathecal bupivacaine is estimated to lie between 9.8 and 13 mg (factors such as baricity of the local anesthetic and addition of adjuvants such as fentanyl may explain this variability) [46,47]. In pregnant subjects, it is believed that this effective dose may be lower than that of the nonpregnant population. Several factors are proposed to explain the lower requirement for local anesthetic in the obstetric population: spinal cord neurotransmission and permeability of the neural sheath may be altered by hormonal influences, the endogenous analgesic system may be potentiated in pregnancy and local anesthetic spread may be enhanced by the mechanical effect of epidural venous engorgement [9,11].
Whether this dose should be further reduced in the obese parturient is controversial. Both the pregnant state and obesity reduce the CSF volume, as demonstrated by MRI [48]. Epidural venous plexus engorgement, secondary to the compression of the vena cava by the gravid uterus and abdominal panniculus, results in an increase in the epidural space volume. Excessive soft tissue may also protrude through the intervertebral foramen and further increase epidural space volume and pressure, thus decreasing CSF volume [48]. In addition, when the patient is in the lateral or supine position, fat tissue accumulation around the buttock may place the vertebral column in a slight head-down position, thus potentially favoring the cephalad spread of a hyperbaric local anesthetic mixture [49].
This fear of extended cephalad spread in obese parturients does not seem to be substantiated in the literature. In a letter to the editor, Emett and Gowrie-Mohan reported a case series of 62 consecutive CSEs in morbidly obese patients, among which 20 were parturients undergoing cesarean delivery [50]. All patients received 0.5% hyperbaric bupivacaine 2–2.5 ml with fentanyl 10–15 μg intrathecally. The authors reported no difference in block height or anesthetic complications compared with non-obese counterparts. In a study examining the requirements for hyperbaric bupivacaine in 40 obese and normal-weight term parturients undergoing cesarean delivery, Lee et al. demonstrated that the 95% effective dose of bupivacaine was similar between the groups [51]. None of the subjects receiving a dose of 11.25 mg or greater required epidural supplementation and the authors did not describe any excessive spread of local anesthetic within their sample.
In nonlaboring parturients undergoing elective cesarean delivery, Ithnin et al. demonstrated that for an identical dose of intrathecal local anesthetic mixture, the block level obtained when the injection was performed as part of a CSE was significantly higher than when it was injected as a SSS (median level of block C6 vs T2, respectively) [52]. It is hypothesized that the conduit between the atmosphere and epidural space created by the introduction of a large gauge epidural needle may counterbalance the negative pressure in the epidural compartment. This may result in thecal compression, reduced dural sac volume and an extended cephalad spread of a spinal dose of local anesthetic. However, Horstman et al. failed to demonstrate a difference in CSF pressures in patients receiving a SSS or CSE anesthesia, and did not observe any difference in block extent between the groups [53]. In a population of laboring parturients undergoing emergency cesarean delivery, Lim et al. showed similar block level in mothers receiving a SSS compared with a CSE technique [54]. We conclude that for surgical delivery, the spinal dose requirements of local anesthetic, administered either as part of a SSS or CSE anesthesia, in term obese parturients is not reduced compared with their normal-weight counterparts.
Continuous spinal anesthesia
Continuous spinal anesthesia (CSA) consists of intentionally inserting a catheter (either a spinal microcatheter or an epidural catheter) through the dura into the intrathecal space and infusing or injecting successive boluses of local anesthetic directly into the CSF. It therefore potentially provides the reliability of a spinal block throughout the surgical procedure. Case reports of cauda equina syndrome following CSA with microcatheters led to their withdrawal from the commercial market in North America. However, it was never determined whether the cauda equina syndromes were caused by those small gauge catheters or rather as a result of the 5% lidocaine used in almost all of these patients. In a recent study looking at the efficacy and safety of a 28-gauge spinal catheter in pregnant women in labor, Arkoosh et al. demonstrated that analgesia was superior in parturients receiving a CSA compared with an epidural [55]. However, they reported a significantly higher failure rate in the spinal catheter group, which was largely attributed to catheter dislodgment. Alonso et al. examined the efficacy of 22- and 24-gauge spinal catheters for spinal anesthesia in parturients undergoing elective cesarean delivery [56]. The authors described a 20% rate of inadequate anesthesia requiring conversion to an alternative anesthetic technique. The possibility of spinal catheter migration in any other direction than cephalad [57], of pooling of local anesthetic in the sacral dural sac, of catheter dislodgment or that the authors had a limited experience with spinal catheters may explain this high failure rate. Moreover, the overall rate of postdural puncture headache was unacceptably high (29%). In a population of obese parturients, where regional anesthesia is particularly desirable and general anesthesia poses significant risks to the mother, the disappointing unreliability of spinal catheters described in those studies prevents us from advocating the routine use of a CSA technique for cesarean delivery. It should be noted, however, that in situations of multiple failed attempts at identifying the epidural space (for stand-alone epidural or CSE anesthesia) or of inadvertent dural puncture, the anesthesiologist may consider intentionally threading an epidural catheter into the intrathecal space. Indeed, although CSA is the less-favored neuraxial technique in this population, it may still be of benefit compared with general anesthesia.
General anesthesia
Several characteristics relating to pregnancy increase the risks of difficult intubation following induction of general anesthesia
Obesity also poses significant challenges and increases difficulty of successful airway management [64]. Excess fat deposition may cause distorted anatomy, such as enlarged tongue and redundant pharyngeal and palatal soft tissue. These may obstruct the airway and render mask ventilation difficult or even impossible. In addition, they may impair glottic visualization during direct laryngoscopy [65]. Proper positioning of the patients in order to align the oral, pharyngeal and laryngeal axis will significantly improve intubation. This factor is especially important in both the obese and pregnant population since the abdominal panniculus and/or enlarged breast may fall towards the head and impair mouth opening and adequate introduction of the laryngoscope in the mouth.
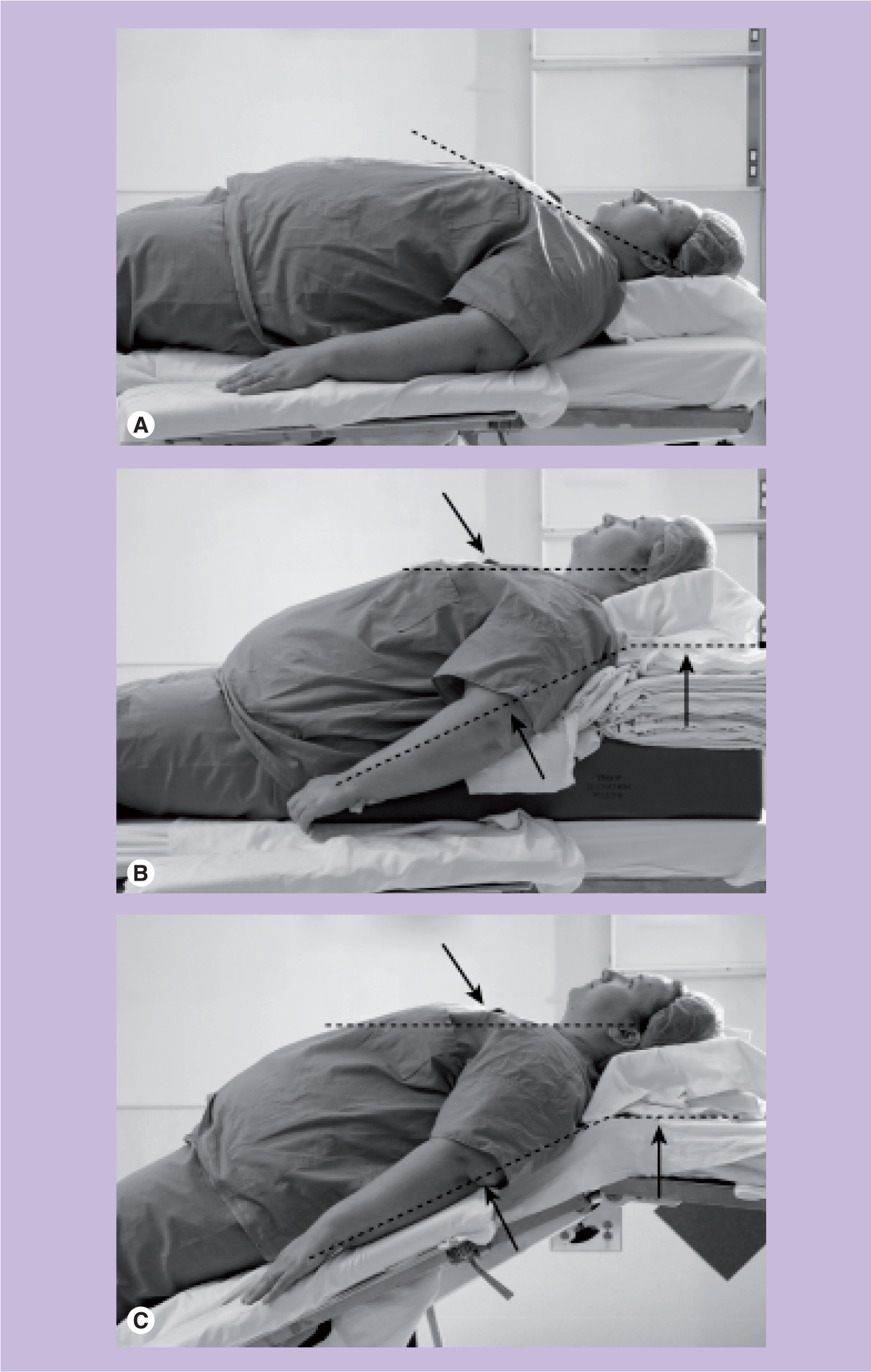
Alignment of external auditory meatus with the sternum.
Because of the higher rate of difficult airway management in the obese parturient population, videolaryngoscopes and fiberoptic intubating device should be readily available [66]. Videolaryngoscopy-assisted intubation has been shown to improve the intubation rate in the obese population, compared with traditional laryngoscopy with the Macintosh blade [67]. Alternatively, it may be necessary to intubate the trachea with the patient awake and in the head-up position [13]. An airway examination and knowledge of previous difficult intubation will help the clinician to make the appropriate decision. Awake intubation, with either a videolaryngoscope or the fiberoptic bundle device, is a well-tolerated procedure provided that a thorough step-by-step explanation of the technique is given to the patient and sufficient topical local anesthesia of the pharynx and larynx is administered. In the event of unsuccessful intubation, a failed intubation algorithm should be applied immediately and help should be called promptly [68]. Laryngeal mask airways can be lifesaving in this situation, even in obese patients [69,70]. Multiple attempts at laryngoscopy, especially with repeated doses of succinylcholine, may be detrimental as it may increase the risk of pharyngeal and laryngeal edema, obstruction of the airway and bradycardia.
Risk factors for complications of airway management in pregnant women
Difficult laryngoscopy and visualization of glottis
Full dentition
Airway edema
Enlarged breasts
Shorter time of apnea between general anesthesia induction and desaturation
Decreased functional residual capacity
Increased oxygen consumption
Increased risks of gastric content regurgitation and aspiration
Decreased lower esophageal sphincter tone
Decreased gastric emptying during labor
Weight gain
Emergency
As discussed earlier, both obese and pregnant patients are prone to regurgitation and aspiration of gastric contents. This mandates antacid and antireflux prophylaxis (vide supra) and, unless contraindicated (e.g., in the setting of expected difficult airway management), a rapid sequence induction with cricoid pressure. Cricoid pressure consists of applying a posterior force on the cricoid cartilage during induction of general anesthesia, with the aim of obstructing the upper portion of the esophagus, thus preventing reflux of gastric contents. The efficacy of this method is controversial, and a discussion of the debate surrounding this technique is beyond the scope of this article. We invite the reader to refer to an excellent editorial on the subject from Vanner, in which the author concludes that cricoid pressure is a benign, reversible technique that probably effectively prevents regurgitation of gastric content at induction of general anesthesia [71].
A rapid sequence induction technique consists of inducing general anesthesia with an hypnotic agent followed with immediate full neuromuscular blockade, with the aim of protecting the airway with tracheal intubation as rapidly as possible after the loss of protective airway reflex [72]. The cornerstone of the rapid sequence induction lies in the avoidance of mask ventilation, which may push air into the gastric cavity, resulting in gastric distension and reflux of its content into the unprotected lower airways and lungs.
As obese pregnant women are highly susceptible to oxygen desaturation, optimization of alveolar oxygen content prior to induction of apnea is of paramount importance [12]. A traditional approach consists of having the patient breathe 100% oxygen at 10 l/min through a fitted face-mask over 3 min prior to induction of anesthesia. In a trial involving non-obese term parturients, Chiron et al. studied an alternative preoxygenation technique consisting of eight vital capacity deep breaths over 1 min and demonstrated that the time of apnea without desaturation was similar to the 3-min preoxygenation approach [73]. These findings were reproduced by Rapaport et al. in a similar study examining the effect of these two preoxygenation techniques in morbidly obese parturients [74]. In the context of ‘stat’ cesarean delivery, where time is of utmost essence, the eight vital capacity breath preoxygenation technique may be an adequate alternative.
It has been demonstrated that FRC decreases in the supine compared with the head-up and full upright positions [75]. It was hypothesized that preoxygenation in the head-up position might increase the time of apnea without desaturation. Baraka investigated this question in non-obese term parturients but failed to demonstrate any benefit from the 45° head-up position on preoxygenation [76]. This is possibly explained by the presence of the gravid uterus, which impairs diaphragmatic movement, regardless of the position of the mother. On the other hand, the obese patient may benefit from a head-up position. Dixon et al. showed in a nonobstetrical morbidly obese population that a 25° head-up preoxygenation period of 3 min resulted in a time of apnea to desaturation of 201 s compared with 69 s for preoxygenation in the supine position [77]. Some experts are concerned that the semi-sitting position may increase the risk of aspiration of gastric content if regurgitation occurred, simply because of the effect of gravity. Possible difficult intubation of the trachea in this position must also be taken into account [12]. In our experience, however, the head-up position will generally free the cervical area from large breast and redundant fat tissue, facilitating the introduction of the laryngoscope into the mouth. Moreover, as stated earlier, the ‘ramp-up position’, whereby the sternum is horizontally aligned with the external auditory meatus, will greatly improve laryngoscopy [78]. Therefore, we conclude that elevation of the head and thorax should be considered when preoxygenating and inducing general anesthesia in an obese parturient.
Delay et al. showed that the use of noninvasive positive pressure ventilation (NPPV) during preoxygenation in nonpregnant morbidly obese patients speeds the time to optimal alveolar oxygenation, but does not increase the time of apnea without desaturation [79]. Moreover, patients preoxygenated with NPPV presented a modest but statistically significant gastric distension. The benefits of NPPV seem to be too limited at the present time to advocate its routine use in rapid sequence induction in obese parturients [12].
Intravenous anesthetic agents used in obese patients should be dosed on an ideal bodyweight (IBW) basis or on IBW plus some fraction of the difference between total weight and IBW. Doses calculated on a total bodyweight (TBW) basis may be excessive and result in undesired side effects [80,81]. Loss of consciousness may be induced with thiopental (4–5 mg/kg), propofol (2–2.8 mg/kg) or any other induction agent, according to the anesthesiologist's preference and clinical circumstances [9,82]. Placental transfer of these drugs may cause transitory neonatal respiratory depression; hence, personnel skilled in neonatal resuscitation should be readily available to assess and assist ventilation of the newborn.
Neuromuscular blockade will be adequately achieved within 60 s with a dose of succinylcholine 1–1.5 mg/kg. Butyrylcholinesterase levels are reduced by approximately a third in pregnancy, yet the clinical activity of succinylcholine is not prolonged. This is probably explained by an increase in volume of distribution resulting from greater total body water, plasma and blood volume [83]. In contrast to intravenous induction agents and non-depolarizing neuromuscular-blocking agents, it is suggested that succinylcholine doses should be calculated on a TBW basis rather than on an IBW basis. Lemmens et al. studied the clinical response of morbidly obese patients to succinylcholine and determined that a dose based on TBW provided excellent intubation conditions in almost every patient, whereas a third of the subjects receiving a dose based on IBW had poor intubation conditions [84]. The authors recommend that to achieve complete neuromuscular paralysis and predictable laryngoscopy conditions, a succinylcholine dose of 1 mg/kg (TBW) should be administered, bearing in mind that no dose will provide a safe duration of apnea.
Evidence suggests that rocuronium at a dose of 1.0–1.2 mg/kg (IBW), but not 0.6 mg/kg, provides equivalent intubation conditions to succinylcholine 1 mg/kg for rapid sequence induction, but at the expense of significantly prolonged curarization [85–87]. At a dose of 0.6 mg/kg, placental transfer seems to be insufficient to affect the Apgar score [88]. There is scant data with regards to placental transfer with higher doses, but we are not aware of any report of severe clinical adverse effect in neonates born from mothers who received rocuronium 0.9–1.2 mg/kg for their cesarean delivery. Sugammadex, a cyclodextrin molecule, at a dose of 12 mg/kg, will effectively reverse in 2 min a high-dose (1.2 mg/kg) rocuronium-induced curarization in surgical patients [89]. Puhringer et al. reported seven cases of successful reversal of 0.6-mg/kg rocuronium-induced neuromuscular block with sugammadex 2–4 mg/kg in women who underwent cesarean delivery (one patient had a BMI of 35.6) [90]. It has recently been marketed in the UK but, to our knowledge, the safety profile of this molecule has not been assessed in the obstetric population. Until strong data on the maternal and neonatal innocuity of sugammadex are available, we cannot advocate its routine use in the obese obstetrical population.
Maintenance of general anesthesia is provided with halogenated gases with or without nitrous oxyde (N2O). Sevoflurane and desflurane, with their steep washout curves, are associated with a slightly faster immediate recovery than with isoflurane (time to extubation: 5.6 min for desflurane vs 12.2 min for isoflurane) [91,92]. However, considering the short duration of cesarean deliveries (usually less than 2 h) and the provision of adequate immediate postoperative care, the clinical relevance of this difference is negligeable and we consider that all three agents are a good choice for obese pregnant women. N2O may be administered to lower the minimum alveolar concentration of the halogenated agent, but obese patients may not tolerate usual concentrations of N2O as they may need high FiO2 [9]. The minimum alveolar concentration (MAC) is slightly reduced in the obstetric population, but is not affected by obesity. It is suggested that the anesthesiologist should aim to provide unconsciousness by keeping the MAC close to 1.0 between intubation and delivery and then reduce it to 0.5–0.75 MAC after delivery [9]. This will reduce the risk of uterine relaxation, which may occur with higher doses of halogenated agents.
To reduce the occurrence of small airway collapse, atelectasis and hypoxia in obese patients, it is recommended that mechanical ventilation includes one or more of the following elements: use of lower inspiratory oxygen fractions (FiO2 <0.8) to avoid progressive reabsorption atelectasis; ventilation using tidal volumes lower than 10–13 ml/kg (IBW); inclusion of large, manually or automatically performed lung inflations (sighs); and application of positive end expiratory pressure after an alveolar recruitment maneuver [13,93].
Emergence from general anesthesia and extubation of patients is too often overlooked, yet the incidence of anesthesia-related adverse events is overrepresented during that step of general anesthesia. Indeed, in a review of obstetric mortality in Michigan, USA, from 1985 to 2003, Mhyre et al. reported that all maternal death related to hypoventilation or airway obstruction occurred at emergence and recovery of general anesthesia [94]. Full reversal of neuromuscular blockade and full consciousness must be confirmed before one may proceed to the extubation of the patient, which should be performed in the semi-upright position to reduce diaphragm compression. Monitoring vital signs and appropriate postoperative care in a dedicated unit by trained personnel is essential before the mother can be discharged to the ward (vide infra).
Postoperative care
Obesity increases risks of postoperative wound dehiscence and infection, deep venous thrombosis and pulmonary embolism, pulmonary morbidity (hypoxia, atelectasis and pneumonia), obstructive sleep apnea and maternal death [5,6,95]. The key elements of effective postoperative care for obese parturients will include effective analgesia, thromboprophylaxis, placing the patient in a semirecumbent position (30–45°), early mobilization, respiratory physiotherapy including incentive spirometry and the use of noninvasive continuous positive airway pressure when the PaO2/FiO2 ratio is below 300 [11,13,19]. It is proposed that parturients with class III obesity and class II obesity with medical risk factors might benefit from an admission to a surgical intensive care or step-down unit [19].
Analgesia
A multimodal analgesic approach, including regular administration of NSAID and paracetamol, may reduce the requirements for opioids [96]. Compared with systemic analgesia, neuraxial opioids provide superior analgesia for cesarean delivery, while reducing hospital length of stay, improving postoperative pulmonary complications and promoting earlier ambulation of obese patients [97–99]. Patients undergoing cesarean delivery under general anesthesia will benefit from patient-controlled analgesia, a technique whereby small doses of intravenous opioids such as morphine are administered on patient demand [100,101]. Although this allows better analgesia titratability and reduces opioid plasma concentration fluctuations, the risks of respiratory depression and hypoxia, especially in the obese population, are far from negligible, and appropriate postoperative monitoring and vigilance must be assured before such analgesic modalities may be offered to patients [102,103]. Wound infiltration and ilioinguinal nerve block with local anesthetic may also optimize postoperative analgesia in patients who cannot benefit from neuraxial analgesia [104]. Ultrasound-guided transversus abdominis plane block administered as part of a multimodal analgesic therapy for postcesarean delivery pain management has been shown to reduce patient-controlled analgesia morphine requirements [105], but does not provide superior analgesia when compared with subarachnoid morphine [106]. As the presence of the large abdominal panniculus in obese patients may render this technique highly challenging, clinicians should base their decision to perform it on the availability of other analgesic modalities, on their comfort and experience with this block and on the sonoanatomy of their patients.
Obstructive sleep apnea
The American Society of Anesthesiologists considers that OSA, which may result from obesity and pregnancy, increases risks of perioperative morbidity and mortality in these patients because of potential difficulty in maintaining their airway patent. The American Society of Anesthesiologists proposes guidelines for management of patients who are judged to be at the greatest risk of OSA [23]. With regards to postoperative management, these guidelines include the following recommendations: regional analgesic techniques should be considered to reduce or eliminate the requirement for systemic opioids in patients at an increased perioperative risk from OSA; if neuraxial analgesia is planned, weigh the benefits (improved analgesia, decreased need for systemic opioids) and risks (respiratory depression from rostral spread) of using an opioid or opioid–local anesthetic mixture as compared with local anesthetic alone; if patient-controlled systemic opioids are used, continuous background infusions should be used with extreme caution or avoided entirely; supplemental oxygen should be administered continuously to all patients who are at increased perioperative risk from OSA until they are able to maintain their baseline oxygen saturation while breathing room air; if possible, patients at increased perioperative risk from OSA should be placed in nonsupine positions; and hospitalized patients who are at an increased risk of respiratory compromise from OSA should have continuous pulse oximetry monitoring after discharge from the recovery room to the postoperative wards.
Thromboprophylaxis
The risk of postoperative deep venous thrombosis (DVT) varies according to several factors (e.g., previous DVT, thrombophilia) and, as such, the American College of Obstetricians and Gynecologists (ACOG) proposes that patients should be stratified in categories ranging from low to very high risk [107]. The Royal College of Obstetricians and Gynecologists suggests that: “temporary use of intermittent compression devices, and/or unfractionated heparin or low-molecular-weight heparin (LMWH) prophylaxis might be considered, especially in the presence of obesity” [108]. LMWH dosing for thromboprophylaxis are frequently fixed doses that do not take into account bodyweight. Because obese patients may be at higher risk of DVT, and as the distribution of LMWH is bodyweight dependent, such fixed-dose regimens may lead to subtherapeutic anticoagulation [109]. It is proposed that the LMWH dose should be increased in obese patients, possibly on a TBW basis [110,111]. We recommend reading a thorough review by Dalton on the subject of antibiotic prophylaxis and thromboprophylaxis for cesarean delivery in which the issue of obesity is addressed [112]. Although there are no formal guidelines with regards to timing of thromboprophylaxis before and after central neuraxial needle/catheter insertion and withdrawal of catheters, it is recommended that neuraxial techniques should not be performed within 12 h following – and 2–4 h prior to – LMWH prophylaxis administration [113].
Conclusion & future perspective
Obesity imposes significant risks to mothers undergoing cesarean delivery, hence the need to produce clear guidelines for the anesthetic management of obese parturients. The 2007 CEMACH report and the 2010 CMACE/RCOG Joint Guideline on the management of pregnant women with obesity both emphasize the role of the anesthesiologist as an essential element of a safe medical practice in this population
Elements of a safe anesthetic practice for obese pregnant women undergoing cesarean delivery
Anesthetic resources should focus on women with class III obesity, as they are considered at highest risk
Morbidly obese women (BMI ≥40) should be referred for anesthetic assessment and advice as part of their antenatal care. Potential difficulties with venous access, airway management and intubation, regional or general anesthesia should be identified and an anesthetic management plan for cesarean delivery should be discussed and documented in the medical records
Management by consultant anesthesiologists or specialty trainees with sufficient experience is essential for the care of women with a BMI ≥40 during labor and delivery including attending any operative vaginal or abdominal delivery
The duty anesthesiologist covering labor ward should be informed when a woman with a BMI ≥40 is admitted to the labor ward if delivery or operative intervention is anticipated
Positioning the women requires sufficient manpower in the event of a requirement for induction of general anesthesia
Direct arterial pressure measurement may be useful in the morbidly obese women where noninvasive blood pressure measurement is often inaccurate
Adapted from CEMACH (2007), Box 8.4, with permission from the Centre for Maternal and Child Enquiries [6].
Executive summary
In 2005, 1.6 billion adults were overweight and 400 million adults were obese. The incidence of obesity in women has increased from 13 to 25% in England since 1993.
A thorough assessment of comorbidities, venous access, airway management, regional and general anesthesia should be performed.
Provision of antacid, thromboembolic and antibiotic prophylaxis should be assured.
Postoperative care should include a plan for effective analgesia, early mobilization and respiratory physiotherapy.
An epidural catheter inserted as part of a combined spinal–epidural technique is as efficient as a catheter inserted as part of a ‘stand-alone’ epidural approach.
The spinal dose of local anesthetic in obese parturients should probably not be reduced in comparison to non-obese pregnant women.
Strategies to improve success include:
– Using longer spinal and epidural needles
– Patient self-assessment of whether the puncture is performed in the midline or not
– ‘Probing’ the position of the lumbar intervertebral space with a needle
– Using ultrasound imaging
Significant risks include:
– A difficult airway management
– An increased risk of gastric content regurgitation and aspiration
– A shorter time of apnea without desaturation
Strategies include:
– The preoperative administration of antacid prophylaxis
– ‘Ramped-up’ positioning to free the neck from redundant tissues and ease laryngoscopy
– An appropriate preoxygenation in the head-up position
– A rapid sequence induction with cricoid pressure unless contraindicated
– Extubation of patients only after full recovery from neuromuscular blockade and full consciousness has been confirmed
Precise guidelines on the management of obese parturients undergoing cesarean delivery are needed.
Medical faculties will need more than ever to incorporate teaching objectives related to the management of obese patients.
Simulation-based teaching/training will be increasingly relevant and useful.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
