Abstract
Arthritis is the leading cause of physical disability in the USA and osteoarthritis (OA), the most common form, affects nearly 27 million people. Women have a higher prevalence of OA than men, but the underlying causes for the increased susceptibility of women to OA are not fully understood. This review discusses the associations of sex hormones, obesity and physical activity with the incidence and progression of OA in women. Although many studies have explored the relationships of estrogen and reproductive history with the risk of OA or joint replacement, there is no consensus on the role of these factors. Obesity clearly increases risk for OA, but whether obesity affects women and men differently is uncertain. Moderate levels of physical activity do not appear to increase the incidence or progression of OA and may even have a weak protective effect. Future investigations should focus on sex-specific mechanisms for the development of OA and determine whether there are modifiable factors (e.g., estrogen, obesity, physical activity) that can be targeted through prevention and treatment strategies to mitigate the increased incidence and prevalence of OA in women.
Background on osteoarthritis
Arthritis is the leading cause of physical disability in the USA. The most common form of arthritis is osteoarthritis (OA), which affects nearly 27 million people in the USA [1]. OA causes deterioration of the joint cartilage and formation of reactive new bone at the margins and subchondral areas of the joints. This degeneration results from a breakdown of chondrocyte function, most commonly in the distal and proximal interphalangeal joints, but also in the hip and knee joints [2,3]. More recently, it has become apparent that OA affects not only articular cartilage, but also the entire joint. In addition to erosion of articular cartilage, there are alterations in subchondral bone, degeneration of menisci, inflammation of the synovium, and bone and cartilage overgrowth (i.e., osteophytes) [4]. More detail regarding the pathophysiology of OA has been reviewed recently by Hunter [4].
Clinical signs of symptomatic OA include slowly developing joint pain, stiffness and enlargement with limitation of motion. Radiographic signs include osteophytes, joint space narrowing and sclerosis [3]. Pain symptoms are not always present with OA, even when extensive radiographic damage is evident [5–7]. Felson and others reported that only 40–80% of people with radiographic evidence of OA have clinical symptoms [6,7]. Radiographic OA of the knee is present in more than 33% of people over 60 years of age, whereas 10–15% of people over 60 years of age have symptomatic knee OA, defined as knee pain on most days [7].
Although the precise etiology is unknown, OA probably results from a combination of metabolic, genetic and mechanical factors (Box 1) [7–11]. Some of the prominent risk factors for the development of OA include older age, female sex, obesity, previous knee trauma, occupational load, ethnicity and genetics [4,10,12,13]. Other risk factors for OA incidence that are more controversial include sex hormones (described further later), biomechanical malalignment, joint laxity and quadriceps muscle weakness [7,9,14–16]. It has been suggested that the incidence and progression of symptomatic and radiographic knee OA involve different processes [17,18]. If true, risk factors for the progression of OA may be different than those for the development OA. A systematic review of prognostic factors for progression of OA found strong evidence that both higher serum hyaluronic acid levels and generalized OA were predictive of knee OA progression [19], whereas sex, knee pain, radiologic severity, knee injury, quadriceps strength and regular sport activities were not [19]. The review also found conflicting evidence as to whether body mass index (i.e., obesity) and age influence the progression of OA [19].
The factors that influence the development and progression of OA are not fully understood. The intent of this review was to discuss whether sex hormones, obesity and physical activity influence the incidence and progression of OA in a sex-specific manner. After careful review of the literature, we have chosen to emphasize some studies over others in an effort to summarize findings, while providing a fair and balanced summary of the existing literature.
Known and suspected risk factors for developing osteoarthritis.
Age
Female sex
Obesity
Previous knee trauma
Repeated overuse, especially occupational
Knee bending or lifting (knee osteoarthritis)
Genetics
Ethnicity
Estrogen therapy (protective?)
Early hysterectomy/surgical menopause
Osteoporosis (protective)
Biomechanical malalignment
Muscle weakness
Associations of OA with sex hormone factors
Women have a higher prevalence of OA than men, especially in the knee (Table 1) [20–22]. OA not only occurs more frequently in women than in men, but also with greater severity [23,24]. Women often have more joints affected and report greater morning stiffness and joint swelling than men [24]. Women waiting for knee or hip replacement typically report higher levels of pain than men [25,26]. An investigation of the relationship between symptoms and radiographic grades of knee OA recently suggested that women have more severe symptoms and symptom progression than men presenting with the same radiographic grade of OA [23].
Prevalence of knee osteoarthritis.

Although OA affects other joints similarly, the largest percentage of OA occurs in the knee; therefore prevalence data are presented for the knee by sex, age and BMI.
OA: Osteoarthritis.
Data taken from [20].
Above the age of 50 years (which approximates the age of menopause), the incidence of OA rises more steeply in women than in men [27], which makes it tempting to postulate that ovarian hormones play a protective role in joint health [3,14,28,29]. The observations that articular chondrocytes have functional estrogen receptors and that estrogen can upregulate proteoglycan synthesis [30,31], bolster the scientific plausibility for direct benefits of estrogens on articular cartilage. However, although many studies have examined female sex hormones as determinants of OA risk, there is no clear consensus opinion on their mechanistic role [31,32]. Recent systematic reviews evaluated multiple aspects of hormone exposure as potential determinants of hip, knee and hand OA, including endogenous hormone levels, age at menarche and menopause, duration of fertile period, menopausal status, years since menopause, and oophorectomy [14,28]; some of these factors will be discussed further below. The main conclusion was that the postulated beneficial effects of ovarian hormones on joint health were not supported by the available evidence. Much of the evidence pointed toward no association between OA and exposure to female sex hormones.
One of the largest investigations of the associations of estrogens and reproductive history with advanced OA (i.e., joint replacement) was conducted as part of the Million Women Study through the National Health Service breast screening centers in England and Scotland [29]. Information was collected from 1,306,081 women at study entry on age at menarche, parity, age at menopause, menopausal status, and use of sex hormone therapies. Women were followed an average of 6.1 years, over which time the incidence rates of total knee and total hip replacement were evaluated. The major findings from this large prospective cohort study were:
There was a linear trend for increased parity to be associated with increased risk for joint replacement, and the effect was more pronounced for the hip than the knee. The relative risks (RR) for hip and knee replacement increased by 2 and 8 %, respectively, per birth;
Early age of menarche (aged 11 years or younger) was associated with an increased RR of hip (RR: 1.09; 95% CI: 1.03–1.16) and knee (RR: 1.15; 95% CI: 1.08–1.22) replacement;
When compared with women who had neverused menopausal hormone therapy (HT), current use was associated with an increased RR of hip (RR: 1.38; 95% CI: 1.30–1.46) and knee replacement (RR: 1.58; 95% CI: 1.48–1.69). RR was lower in past users of menopausal HT, but still significantly elevated (hip RR: 1.13; 95% CI: 1.06–1.21; knee RR: 1.39; 95% CI: 1.29–1.49). Both estrogen-only (hip RR: 1.21; 95% CI: 1.10–1.33; knee RR: 1.48; 95% CI: 1.35–1.63) and estrogen plus progestin therapies (hip RR: 1.51; 95% CI: 1.41–1.62; knee RR: 1.68; 95% CI: 1.54–1.83) were associated with increased risk for joint replacement. Past use of hormone contraceptive therapy was not associated with increased risk.
From a very broad perspective, this Million Woman Study suggested that increased exposure to sex hormones is associated with increased risk for joint replacement, but alternative interpretations were also discussed. For example, although the association of parity with joint replacement risk was adjusted for BMI at the time of study entry, the authors acknowledged that the association could have been influenced by increased weight-bearing during pregnancy, rather than by the sex hormone environment. They also suggested that the associations of HT with joint replacement risk could have reflected nonbiological factors, because women on therapy may have had better access to healthcare services.
Estrogen-based hormone therapy
Many epidemiological studies have investigated the association of menopausal estrogen-based HT with OA. Some studies [33–39], but not all [29,40], found a lower incidence or prevalence of knee and hip OA in HT users, supporting the concept that estrogens have a protective effect on joints. In the prospective Chingford study, 715 middle-aged women were followed for 4 years to determine the incidence of knee OA, defined by the development of osteophytes and joint space narrowing, and whether age, weight and HT are risk factors for OA [34]. Age and obesity were the strongest determinants of the incidence of knee OA; current HT use conferred a nonsignificant, protective effect (overall response [OR]: 0.41; 95% CI: 0.12–1.42) [34].
The Framingham Osteoarthritis Study found that postmenopausal women who used HT had lower odds of developing radiographic knee OA and the protective effect was greater with increased duration of HT treatment [33,38]. In this study, 551 women were categorized according to HT use (349 never-users; 162 past users; 40 current users) [38]. Over 8 years of follow-up, 17.4% of never-users had knee radiographic scores that worsened by 1 grade and 5.8% by 2 or 3 grades. Among current HT users, only 11.7% of knee radiographic scores worsened by 1 grade and none worsened by more than 1 grade. With adjustments for age and other potential confounding factors, the RR of incident radiographic knee OA, in comparison with never-users of HT, was 0.8 (95% CI: 0.5–1.4) in past users and 0.4 (95% CI: 0.1–3.0) in current users. Current use of HT also showed a nonsignificant decrease in risk of progressive knee OA compared with never-use (OR: 0.5; 95% CI: 0.1–2.9). When both incident and progressive radiographic knee OA cases were combined, current HT use was associated with a nonsignificant 60% decreased risk compared with never-use (OR: 0.4; 95% CI: 0.1–1.5). These findings are seemingly in direct contrast with the Million Women Study (discussed above), which found that the use of menopausal HT was associated with an increased incidence of joint replacement [29].
Few randomized controlled trials (RCTs) have evaluated the effects of HT on OA from an intervention perspective, and none have evaluated OA risk as a primary outcome [41,42]. In the intention-to-treat analyses of data from the Women's Health Initiative (WHI) intervention trials, women randomized to estrogen-only therapy had a lower rate of joint replacement than women on placebo therapy (hazard ratio [HR]: 0.84; 95% CI: 0.70–1.00); there were trends for benefits for both hip (HR: 0.73; 95% CI: 0.52–1.03) and knee replacement (HR: 0.87; 95% CI: 0.71–1.07) [41]. When censored for nonadherence to assigned HT, women on estrogen therapy had significantly lower risks for any joint replacement (HR: 0.73; 95% CI: 0.58–0.93) and hip replacement (HR: 0.55; 95% CI: 0.35–0.88), but not knee replacement (HR: 0.80; 95% CI: 0.61–1.05). By contrast, women randomized to estrogen plus progestin therapy in the WHI trial did not have a change in incidence of joint replacement (HR: 0.99; 95% CI: 0.82–1.20), and censoring for adherence to therapy did not alter the results [41]. These findings suggest that postmenopausal estrogen therapy has beneficial effects on joint health, but that estrogen plus progestin therapy does not. The Heart and Estrogen/Progestin Replacement Study (HERS) also concluded that there was no significant effect of estrogen plus progestin therapy on knee pain or related disability when compared with placebo therapy [42].
A number of factors may explain the lack of consensus for the effects of HT on OA. The highest level of evidence (i.e., RCTs) is lacking, because only two large RCTs have been performed and neither had joint replacement or incident OA as a primary outcome [41,42]. The prospective observational studies of incident OA in HT-users versus non-users may have been influenced by confounding factors, such as access to healthcare services, different levels of physical activity or body weight [29,41,42]. It is also possible that HT has different effects on clinical knee symptoms than on the structural radiographic evidence of OA [3,42]. Studies that have examined the association of HT with symptomatic or clinical OA are less consistent than studies of radiographic OA in suggesting a protective role for HT [3,42]. HT may slow certain radiographic changes of OA, but has little or no effect on symptoms. Investigations that focus on both symptoms and radiographic evidence of OA will be necessary to explore these potential differential effects. Finally, the WHI intervention trials suggested that the beneficial effects of estrogens on joint health are countered by progestins [41]. Typically, prospective observational studies have not examined the independent effects of effects of estrogen therapy versus estrogen plus progestin therapy. Therefore, the type of HT and duration of use may confound inferences from observational studies regarding the role of HT in the incidence and progression of OA.
Previous hormonal contraceptive use
A systematic review of the effects of hormonal contraception on OA of the hand, hip and knee found that there was no significant association of the use or duration of use with the development of OA [28]. The Million Women Study also found that previous oral contraceptive use was not associated with the incidence of hip or knee replacement (RR: 1.02; 95% CI: 0.98–1.06; RR: 1.00; 95% CI: 0.96–1.04, respectively) [29]. Thus, the available evidence suggests that use of hormonal contraception does not influence the risk of developing OA.
Age of menopause
The increased incidence of OA around the time of menopause suggests that the development of OA is linked with the menopausal withdrawal of ovarian hormones [3]. However, attempts to establish a temporal relationship between the onset of OA and menopause have been inconclusive. Several studies have found that neither age at menopause nor menopausal status are independently related to incidence of OA [14,28,29,39,43]. By contrast, a large epidemiological study in Italy found that women experiencing surgical or natural menopause were at increased risk of OA (OR: 1.13; 95% CI: 1.07–1.21; OR: 1.18; 95% CI: 1.08–1.28) when compared with premenopausal women, yet there was no clear relationship of age at menopause with risk of OA [39]. It has been postulated that the perimenopausal decrease in progesterone levels results in a transient period of exposure to unopposed estrogens that increases risk of OA [44]. However, this hypothesis is seemingly in contrast with the finding from the WHI RCT that unopposed estrogen therapy had favorable effects on joint health. It remains unclear how short-term hormonal imbalances or permanent declines in hormone concentrations contribute to the surge in OA that appears to occur around the age of menopause.
Age of menarche
Only a few studies have investigated the relationship between age of menarche and OA risk [45–47]. The Million Women study reported that a younger age at menarche (age 11 years or less) was related to a greater risk of hip and knee replacement when compared with menarche at age 12 years (RR: 1.09; 95% CI: 1.03–1.16; RR: 1.15; 95% CI: 1.08–1.22 for hip and knee replacement, respectively) [29]. Young age at menarche was associated with increased risk for hand OA in one study (n = 745) [46], but not in another (n = 348) [47]. However, the latter study did find that the total number of years of menstruation was directly related to the presence of hand OA [47]. These findings suggest that increased exposure to sex hormones promotes the development of OA. However, because early age of menarche has been associated with increased risk for other age-related chronic diseases [48], it is possible that factors other than sex hormones influence the association with OA.
Parity
Several studies have evaluated the effects of parity and the risk for OA [8,29,36,40,43,47,49–51]. The Million Women Study found that parity was associated with a small (2–8 %), but significant, increase in RR of knee and hip joint replacement [29]. Another study found increased risk of hand OA with increasing number of births [47], suggesting that greater body weight during or after pregnancy does not fully account for the increased OA risk in weight-bearing joints. Nevertheless, despite the findings of these two studies, the majority of investigations have found no relationship between parity and risk for OA [8,36,40,43,49–51].
Summary of associations of OA with sex hormone factors
Although a number of studies have examined the relationships of sex hormone factors with the risk of OA or joint replacement, there is currently no clear consensus on what roles these factors play. This is likely because the mechanisms by which sex hormones may affect the development and/or progression of OA are not yet understood; there are limited hormone data available for evaluation. Secondary analyses of data from large RCTs of HT suggest that estrogen therapy, but not estrogen plus progestin therapy, reduces the incidence of joint replacement in postmenopausal women [41,42]. It is unlikely that there will be a large RCT of the effects of estrogen versus estrogen plus progestin therapy on joint health. Therefore, prospective cohort studies of the association between use of sex HT and OA risk should include a focus on the type of HT.
BMI & OA
Although the cause of knee OA is almost certainly multifactorial, obesity is a primary risk factor [17,34,52–56]. Obesity is also the most important modifiable risk factor for the development and progression of OA [17,52,53]. Increased body weight is especially related to developing OA of the knee, and it has been speculated that this is because of increased mechanical loading at the joint [54–56]. However, the observation that obesity is associated with hand OA suggests mechanisms other than, or in addition to, increased joint loading [24,57]. One possible explanation is that obesity is connected to an array of systemic and hormonal factors responsible for bone and cartilage metabolism, especially in women [24,56,58]. Such factors could alter the density of subchondral bone or exert metabolic actions that have systemic effects on joints throughout the body [24]. For example, leptin, which is secreted primarily from adipocytes, may play an important role in the onset and progression of OA [59]. Other adipocyte-derived factors, such as IL-6 and C-reactive protein, appear to be procatabolic for chondrocytes [60]. Further research is necessary to evaluate whether leptin or other adipokines are important systemic or local factors to explain the link between obesity and OA. Furthermore, because women at any given BMI have a greater total body adiposity (fat mass, percent body fat) than men [61], adipocyte-derived systemic and hormonal factors may explain, in part, the higher incidence of OA in women.
In light of the potential role of systemic factors influenced by obesity, it is difficult to isolate the biomechanical effects of BMI on the development and progression of OA. Many obese patients exhibit abnormal lower extremity alignment (discussed later), which results in increased joint-reactive forces in the medial or lateral compartment of the knee. Such alterations in joint geometry may interfere with nutrition of the cartilage or alter load distribution, either of which could change the biochemical composition of the cartilage [62].
Regardless of the exact mechanisms by which obesity contributes to greater OA risk, a number of investigations have found that increased body weight contributes to the incidence of OA [11,17,63–66]. Cross-sectional data from the Framingham Osteoarthritis Study indicated that obese individuals (BMI > 30.0 kg/m2) are four-times more likely to have knee OA than those with a BMI of 25.0 kg/m2 [63]. Furthermore, the RR for OA of the knee among overweight individuals is higher in women (OR: 1.8; 95% CI: 1.2–2.6) than men (OR:1.0; 95% CI: 0.5–2.1) [11,64]. Cooper et al. reported that obesity was a strong predictor of knee OA after adjusting for age and sex; obesity was also a significant, albeit weaker, predictor of OA progression [17].
Weight loss reduces symptoms of knee OA [52], and weight loss and exercise are recommended for obese patients with knee OA by both the American College of Rheumatology and the European League Against Rheumatism (EULAR) [67,68]. Observational data from the Framingham Knee OA Study showed that a reduction in weight of 5.1 kg decreased the risk of developing knee OA by 50% in women with a baseline BMI over 25.0 kg/m2 [52]. Intervention studies support this finding, with a moderate amount of weight loss (5% of body weight) over the course of 18 months resulting in better self-reported physical function and decreased disability, less pain and stiffness, as well as faster walking speed and stair climb time compared with a weight-stable control group [69]. More intensive weight loss (10% weight loss goal) resulted in similar improvements in self-reported function and walking speed [70].
Weight loss also results in reduced compressive knee-joint loads, as demonstrated after an 18- month clinical trial of diet and exercise [71]. In fact, the force reduction at the knee joint was larger than would be predicted for the weight loss. For every 1 pound of weight loss, there was a 4-pound reduction in knee-joint load per step. The theoretical accumulated reduction in knee load for a 1-pound loss in weight would be more than 4800 pounds per mile walked (assuming 1200 strides/mile). With a 10-pound weight loss, each knee would be subjected to 48,000 pounds less in compressive loading per mile walked. A reduction of this magnitude would likely be clinically relevant. Indeed, Felson et al. found that a weight loss of 11.2 pounds over a 10-year period was associated with a 50% reduction in the risk of developing knee OA [52]. Although such observational studies highlight the potential benefit of weight loss on joint health, long-term RCTs are needed to better isolate the role of weight loss and exercise on joint protection.
The Framingham study also examined whether lower extremity limb alignment was related to BMI and OA progression in women and men [72]. Among the 394 knees examined, 90 (22.8%) demonstrated some disease progression. Limb alignment was strongly associated with OA progression risk; the proportion of knees showing OA progression was greater with poorer alignment (9.2% of neutrally aligned limbs [0–2°]; 22.3% of limbs with 3–6° of malalignment; 48.7% of limbs with severe malalignment [>7°]). The association of alignment with progression was similar between men and women [72], although in general women tend to have worse lower extremity alignment than men [73]. BMI was also associated with OA progression, but surprisingly, the effect of BMI on progression was limited to knees in which there was moderate malalignment (OR per 2-unit increase in BMI: 1.23; 95% CI: 1.05–1.45). In neutrally aligned limbs and severely malaligned limbs, body weight had no effect on risk of knee OA progression in either sex. It may seem paradoxical that increased body weight did not augment the progression of OA when severe malalignment was present. However, because malalignment is a very strong risk factor for structural progression, it is possible that the stresses placed on cartilage with severe malalignment already exceeded thresholds for cartilage loss and were not further worsened by excessive body weight [58,74,75].
A limitation of studies examining the impact of obesity on OA is that many have not differentiated between fat and fat-free mass. A small study (n = 22) of overweight adults with knee OA evaluated whether interventions geared toward decreasing body fat and increasing physical activity improved symptoms of OA [76]. Investigators found that decreasing body fat and increasing physical activity were more important in producing symptomatic relief of knee OA than other indices of obesity, such as weight loss.
In summary, obesity is a primary risk factor for developing OA, but it is less clear how obesity influences the progression of OA. The strong relationship between obesity and incident OA could be secondary to increased mechanical loading at the joint, which could be further exacerbated by joint malalignment. Alternatively, systemic and hormonal factors associated with obesity may adversely affect joints throughout the body, and make it difficult to isolate the biomechanical effects of obesity on OA. Regardless of the mechanisms for obesity-related OA, several studies suggest that weight loss reduces symptoms of knee OA.
Physical activity & incidence of OA
Physical activity and OA have a potentially complex association because a certain level of mechanical joint stress is essential for good joint health [77,78], but excessive joint stress may promote the development of OA [77,79–81]. In the absence of major joint injury, there is no evidence that regular moderate-to-vigorous physical activity (in amounts that are commonly recommended for general health benefits) increases the risk of developing OA [13]. Figure 1 illustrates that the risk for OA does not appear to increase or decrease with varying levels of physical activity. In addition, there is limited, weak evidence from observational and animal studies that low-to-moderate levels of physical activity, particularly walking, may provide protection against the development of hip and knee OA [13]. Whether exceeding some threshold of physical activity promotes the development of OA is unclear, but some level of physical activity is likely essential for optimal joint health [13].

Felson et al. examined the role of physical activity in the development of OA in the Framingham cohort (n = 1279) [82]. Participants who did not have OA at the beginning of the study were asked on a regular basis about their recreational activities and knee pain. Over an average follow-up time of 9 years, new cases of OA were defined three ways: radiographic OA (progression of the Kellgren and Lawrence [K/L] grade to at least 2); tibiofemoral joint space loss (1-grade K/L change on either lateral or anterior-posterior views); and symptomatic OA (a knee that did not have the combination of symptoms and radiographic disease at baseline, but developed that combination by follow-up). Results suggested that neither recreational walking, jogging, working up a sweat, nor high activity levels was associated with a decrease or increase in risk of OA compared with sedentary individuals. When compared with participants who reported no walking activity, the RR for symptomatic OA was 0.96 (95% CI: 0.57–1.62) in those who reported walking less than 6 miles per week and 0.78 (95% CI 0.49–1.24) in those who reported walking more than 6 miles per week. Similarly, neither the risk of incident radiographic OA nor joint space loss was substantially affected by walking for exercise. Only a few subjects (n = 68) reported jogging or running regularly for exercise, but they also did not appear to be at increased risk of developing knee OA. Importantly, this investigation also found that people with a BMI above the median level (27.7 kg/m2 for men and 25.7 kg/m2 for women) who were physically active were at no greater risk for OA than those who were sedentary [82]. Therefore, while physical activity did not protect against the development of OA, it also did not increase the incidence of OA, even in individuals who were overweight or obese.
Another longitudinal study of women found some evidence for a protective effect of exercise on incidence of OA [34]. Individuals were followed over 4 years (n = 715) for the incidence of radiographic OA. Women who walked more than 5 miles per week had significantly less joint space narrowing (OR: 0.38; 95% CI: 0.15–0.93) than women who walked less than 5 miles per week (OR: 0.60; 95% CI: 0.22–1.71). Although evidence is weak, other cohort and case-control investigations suggest that low-impact activity may have a small protective effect on the development of OA [83,84].
The Musculoskeletal Health subcommittee of the Federal Advisory Committee, who prepared the Physical Activity Guidelines Advisory Committee Report (2008), examined the scientific evidence from observational epidemiologic studies assessing physical activity exposure in terms of walking and OA incidence (Table 2) [13]. Less information is available regarding higher level activities such as running and sports participation. The report suggested that the weak protective effect of physical activity may be stronger among women than men [13]. Two studies found that low and high levels of physical activity provided some protection from OA for women (not all were statistically significant due to small sample sizes), but only high levels of activity offered protection for men [83,84].
Studies examining the association between participation in walking and risk of hip/knee osteoarthritis from the Physical Activity Guidelines Advisory Committee report.

No details were provided on the question used to determine walking in Hart et al. However, another published paper from the same cohort described the walking variable as less than versus greater than 5 miles per week.
Ci: Confidence interval; OA: Osteoarthritis; OR: Odds ratio.
Adapted from the Physical Activity Guidelines Advisory Committee Report [13].
Participation in certain sports has been associated with an increased risk of OA [13]. These sports include football, soccer, track and field, basketball, boxing, ice hockey, orienteering, running, wrestling, tennis, ballet and handball. One reason these sports are associated with increased risk for OA may be that joint injuries are common in these sports and joint injury is a strong risk factor for incident OA [9,11,85]. Recent studies have shown a significantly increased incidence of knee injuries in female athletes when compared with their male counterparts [86,87]. As participation in women's recreational, high school, collegiate and professional sports continues to increase, sex differences for incidence of OA may widen even further [87].
Individuals with occupations that require excessive knee bending/kneeling or involve lifting and carrying heavy loads, and who also participate in moderate or vigorous recreational activity, may have increased risk for lower-extremity OA because of additive effects over time [8,88–90]. Data from the US National Health and Nutrition Examination Survey and the Framingham cohort reported that women with jobs requiring repetitive knee bending had triple the rate of knee OA compared with those with jobs not requiring such activity (men had double the rate), even after controlling for known risk factors [8,90].
In summary, exercise at moderate levels (e.g., walking) does not appear to increase the risk of developing OA. Instead, moderate exercise may confer a weak protective effect, possibly favoring women over men. Certain sports may increase the risk of OA, possibly because of greater likelihood for joint injury. Similarly, occupations with high physical demands may predispose individuals to OA. Finally, individuals with higher BMIs can safely participate in moderate-intensity physical activity without increased risk for knee OA.
Physical activity & progression of OA
Considerable attention has focused on how physical activity affects disease progression once OA is present. Exercise intervention trials have evaluated whether endurance and/or resistance exercise training slows the progression of OA (i.e., radiographic changes) and whether these exercise strategies improve or exacerbate OA symptoms and quality of life [13,91–94].
The Physical Activity Guidelines Advisory Committee Report (2008) concluded that there is strong evidence that endurance, resistance and combined endurance and resistance exercise programs reduce symptomatic OA by decreasing pain and improving function, quality of life and mental health, as well as delaying the onset of disability with activities of daily living [13,91,92]. Table 3 includes exercise programs selected for the Physical Activity Guidelines Advisory Committee Report that could be replicated in community settings (e.g., group exercise classes, home programs) with or without supervision. Most of the endurance exercise training studies in Table 3 included moderate-intensity, low-impact exercise, such as walking, water exercise, tai chi, aerobics and cycling. Of the 15 endurance exercise studies reviewed, the average exercise dose was 2.9 sessions per week and 48 min per session for a total of 137 min per week (Table 3) [13]. Most of the studies found a reduction in pain and improvements in physical function in response to exercise training, and some found that exercise improved quality of life, muscle strength and physical activity levels. Importantly, none of the studies found increases in pain, stiffness or other measures of disease progression, including radiographic progression.
Summary of descriptive characteristics of the randomized controlled trials of exercise among persons with arthritis or other rheumatic conditions from the Physical Activity Guidelines Advisory Committee report.
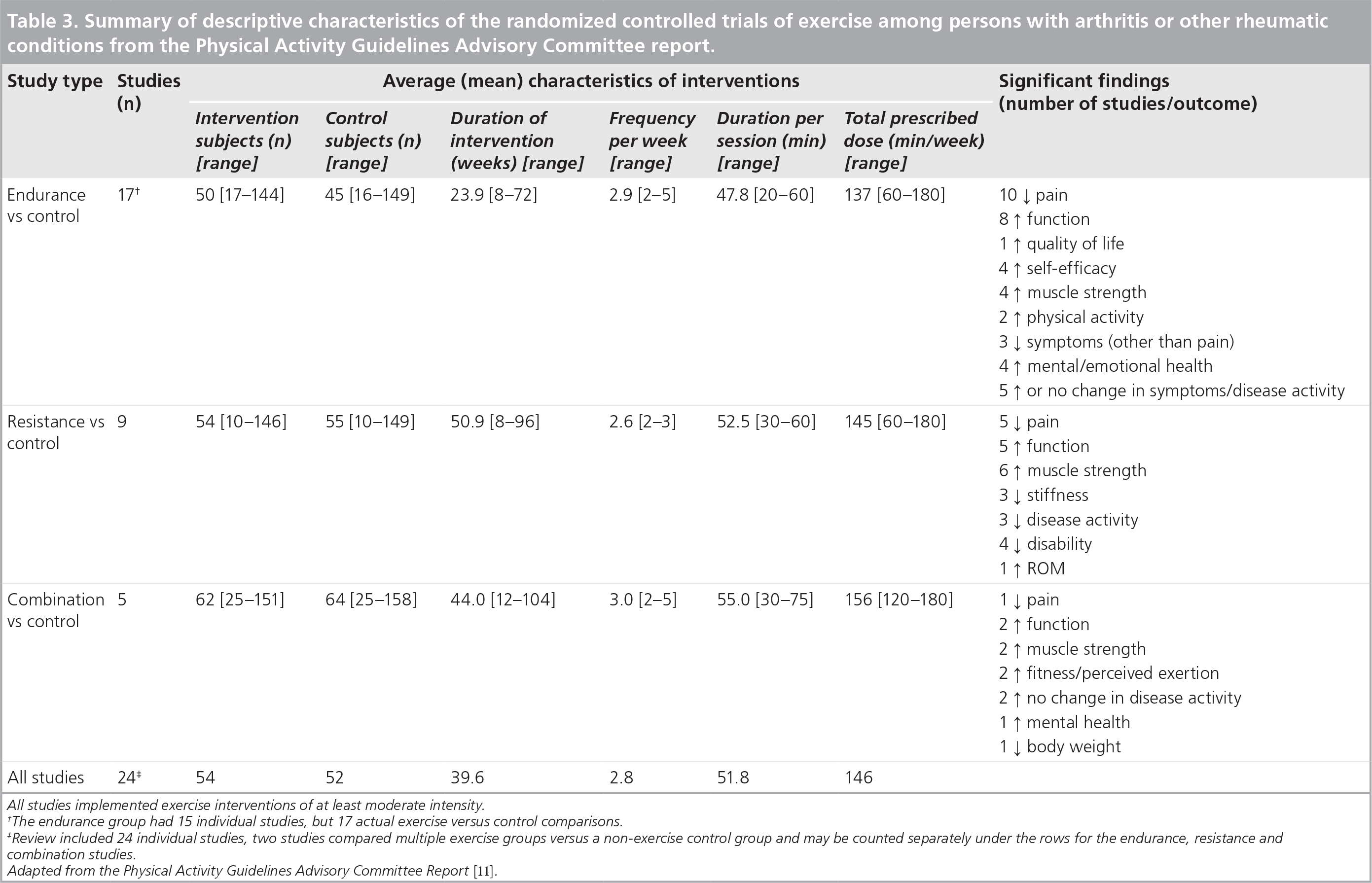
All studies implemented exercise interventions of at least moderate intensity.
The endurance group had 15 individual studies, but 17 actual exercise versus control comparisons.
Review included 24 individual studies, two studies compared multiple exercise groups versus a non-exercise control group and may be counted separately under the rows for the endurance, resistance and combination studies.
Adapted from the Physical Activity Guidelines Advisory Committee Report [11].
The Physical Activity Guidelines Advisory Committee Report also examined nine studies of resistance training that involved isotonic and/or isokinetic exercises (Table 3) [13]. Collectively, these studies demonstrated fewer symptoms of pain, stiffness and function, improved muscle strength, and provided further evidence that exercise does not increase the progression of arthritis. RCTs of combined endurance and resistance exercise training found that benefits were similar to those reported for endurance-only and resistance-only programs [13]. Thus, there is strong and consistent evidence from these relatively shortterm exercise intervention trials (up to 2 years) that exercise improves pain, function, muscle strength and mental health with no increase in disease severity.
Based on this evidence, the Physical Activity Guidelines Advisory Committee Report concluded that people with OA can experience improvements in pain and physical function by engaging in moderate-to-vigorous intensity, low-impact physical activities for approximately 150 min per week [13]. Walking was recommended as particularly appropriate. The consensus statement from the International Osteoarthritis Research Society (OARSI) is in agreement with these guidelines [95]. Of the 11 nonpharmacological OARSI recommendations, there was a strong recommendation that all patients with hip and/or knee OA should engage in endurance and resistance training and range-of-motion exercises. OARSI reported a moderate effect of exercise on pain relief (pooled effect sizes of 0.52; 95% CI: 0.34–0.70 for endurance exercise vs 0.32; 95% CI: 0.23–0.42 for resistance exercise) [95]. The American Geriatrics Association Consensus Practice Statement [96], OASIS statement [97], MOVE Consensus [98], EULAR recommendations [67,99,100], and American College of Rheumatology guidelines [67] are also in agreement with the Physical Activity Guidelines Advisory Committee Report.
In summary, there is no indication that low-to moderate-intensity exercise causes increased pain or disability in men or women with OA. In fact, there is strong evidence that endurance, resistance and combined endurance/resistance exercise programs reduce symptomatic OA by decreasing pain and improving function, quality of life and mental health, as well as delaying the onset of disability with activities of daily living. Longer duration exercise programs (>2 years) have not been evaluated, so conclusions regarding effectiveness of longer intervention are not currently available.
Future perspective
This review highlighted the potential contributions of estrogen, obesity and physical activity on the incidence and progression of OA, with emphasis on sex-specificity when possible. Despite the higher incidence of OA in women than men, few studies have adequately examined sex-specific differences. Why women are more susceptible to OA remains unclear and necessitates further research. Although many studies have explored the relationships of estrogen and reproductive history with the risk of OA or joint replacement, there is no clear consensus on the role of these factors. Obesity increases the risk for developing OA, but whether obesity affects women and men differently has not been thoroughly investigated. Finally, moderate levels of physical activity do not appear to increase the risk of developing OA, even in overweight or obese adults, and may even have a weak protective effect. Once OA has developed, moderate levels of physical activity may mitigate some symptoms associated with OA progression in both men and women. Future investigations should focus on sex-specific mechanisms for the development of OA and determine whether there are modifiable factors that can be targeted through prevention and treatment strategies to mitigate the increased prevalence of OA in women.
Executive summary
Arthritis is the leading cause of physical disability in the USA.
The most common form of arthritis is osteoarthritis (OA), which affects nearly 27 million people in the USA.
Women have a higher prevalence of OA than men, especially in the knee.
Clinical signs of symptomatic OA include slowly developing joint pain, stiffness and enlargement with limitation of motion.
Radiographic signs of OA include osteophytes, joint space narrowing and sclerosis.
Risk factors for the development of OA include: obesity, female sex, older age, previous knee trauma and occupational load.
Incidence and progression of symptomatic and radiographic knee OA may involve different processes.
The incidence of OA rises more steeply in women than in men after 50 years of age, suggesting an association with the menopausal withdrawal of ovarian hormones. Evidence to support this is mixed.
Some studies, but not all, have found a lower incidence or prevalence of knee and hip OA in hormone therapy (HT) users, suggesting that estrogens may have a protective effect on joints.
Few RCTs have been performed to evaluate the effects of HT on OA, and none has evaluated HT on OA risk as a primary outcome. The available evidence suggests that estrogen, but not estrogen plus progestin, therapy reduces OA risk.
There appears to be no significant association of the use or duration of use of hormonal contraceptives with OA.
The increased incidence of OA around the time of menopause suggests that the development of OA is linked with the menopausal withdrawal of ovarian hormones.
Attempts to establish a temporal relationship between the onset of OA and menopause have been inconclusive.
The bulk of evidence suggests that age at menopause and menopausal status (pre/peri versus post) are not related to incidence of OA.
Only a few studies have investigated the relationship between age of menarche and OA risk; some have reported that a younger age at menarche may be related to increased incidence of OA, while others have not found such an association.
Few studies have found an association between parity and risk for OA.
Obesity is a primary risk factor for developing OA.
Mechanical loading of joints and/or systemic and hormonal factors responsible for bone and cartilage metabolism may explain why people with higher BMIs are at greater risk for developing OA.
Weight loss reduces symptoms of knee OA, possibly because of reduced compressive knee-joint loads.
Physical activity and OA have a potentially complex association because a certain level of mechanical joint stress is essential for good joint health, but excessive joint stress may promote the development of OA.
There is no evidence that regular moderate to vigorous physical activity, in amounts that are commonly recommended for general health benefits, increases the risk of developing OA.
Moderate physical activity may have a weak protective benefit for developing OA.
Whether exceeding some threshold of physical activity promotes the development of OA is unclear.
Physical activity does not appear to increase the incidence of OA in individuals who are overweight.
There is strong evidence that endurance, resistance and combined endurance and resistance exercise programs reduce symptomatic OA by 1) decreasing pain, 2) improving function, quality of life and mental health, and 3) delaying the onset of disability with activities of daily living.
The Physical Activity Guidelines Advisory Committee Report concluded that people with OA can experience improvements in pain and physical function by engaging in moderate-to-vigorous intensity, low-impact physical activities for approximately 150 min per week. Walking is recommended as a particularly appropriate exercise.
Despite the higher incidence of OA in women than men, few studies have adequately examined sex-specific differences. The reasons why women are more susceptible to OA remain unclear and necessitate further research. Therefore, future investigation should focus on sex-specific mechanisms for the development of OA and determine whether there are modifiable factors (e.g., estrogen, obesity, physical activity) that can be targeted through prevention and treatment strategies to mitigate the increased incidence and prevalence of OA in women.
Prospective RCTs are needed to investigate the association of menopausal estrogen-based HT with OA. To date, few RCTs have evaluated the effects of HT on OA from an intervention perspective, and none have evaluated OA risk as a primary outcome.
Further investigation of how obesity impacts OA via either systemic hormonal influences or the biomechanical effects of altered joint loading is also an area for further investigation, especially with respect to how these factors may affect women more than men.
Prospective investigations evaluating how physical activity levels contribute to OA incidence and progression in men versus women are warranted. More specifically, a more comprehensive understanding of how different physical activity levels contribute to OA incidence would provide valuable information for clinical guidelines to prevent OA. Similarly, investigations of how physical activity intervention (especially longer than 2 years) affects OA progression would provide alternatives to pharmacological options for treating OA.
Finally, a better understanding of how the complex interactions between obesity, weight loss and energy expenditure through physical activity in men and women contribute to OA would enhance current treatment guidelines.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
