Abstract
Technological advances in fiberoptics and endoscopy have resulted in the development of minimally invasive transcervical tubal catheterization procedures with the potential of improved diagnostic accuracy of tubal disease and transcervical treatment of proximal tubal obstruction (PTO) with reduced risks, costs and morbidity compared with surgical procedures. Fallopian tube recanalization can be performed with catheters, flexible atraumatic guidewires or balloon systems under endoscopic (falloposcopy/hysteroscopy/laparoscopy), sonographic, fluoroscopic or tactile guidance. Falloposcopy provides a unique possibility to accurately visualize and grade endotubal disease, characterize and document endotubal lesions, identify the segmental location of tubal pathology without complications, objectively classify the cause of PTO and guide future patient management. This is in contrast to the surgical and radiological gold standards, laparoscopy and hysterosalpingography, respectively, that are often associated with poor or misdiagnosis of PTO. Nonhysteroscopic transuterine falloposcopy using the linear eversion catheter is a successful, well-tolerated, outpatient technique with a good predictive value for future fertility. Hysteroscopic–falloposcopic–laparoscopic tubal aquadissection, guidewire cannulation, guidewire dilatation and direct balloon tubuloplasty may be used therapeutically to breakdown intraluminal adhesions or dilate a stenosis in normal or minimally diseased tubes with high patency and pregnancy rates. However, guidewire cannulation of proximally obstructed tubes yields much lower pregnancy rates compared with other catheter techniques, despite the high tubal patency rates. Laparo-hysteroscopic selective tubal catheterization with insufflation of oil-soluble radiopaque dye has been reported to be an effective treatment for infertility associated with endometriosis. The various disadvantages associated with fluoroscopic and sonographic techniques limit their application, despite the reportedly high patency and intrauterine pregnancy rates. Recanalization is contraindicated in florid infections and genital tuberculosis, obliterative fibrosis and long tubal obliterations that are difficult to bypass with the catheter, severe tubal damage, male subfertilitY and previously performed tubal surgery. Distal tubal obstruction is not amenable to catheter recanalization techniques. Tuberculosis, salpingitis isthmica nodosa, isthmic occlusion with club-changed terminal, ampullar or fimbrial occlusion, and tubal fibrosis have been cited as reasons for recanalization failure. In lieu of the poor pregnancy outcomes in patients with severe tubal disease and poor mucosal health following tubal recanalization, as well as poor available technical skills and results with microsurgery, in vitro fertilization and embryo transfer is a valid option in such women. Despite the high diagnostic and therapeutic power of falloposcopic interventions, technical shortcomings with falloposcopy must be overcome before the procedure gains widespread acceptance.
Keywords
 Medscape: Continuing Medical Education Online
Medscape: Continuing Medical Education Online
This activity has been planned and implemented in accordance with the Essential Areas and policies of the Accreditation Council for Continuing Medical Education through the joint sponsorship of Medscape, LLC and Future Medicine Ltd. Medscape, LLC is accredited by the ACCME to provide continuing medical education for physicians.
Medscape, LLC designates this educational activity for a maximum of
Learning objectives
Upon completion of this activity, participants should be able to:
Describe the prevalence of false-positive and false-negative diagnoses of proximal tube obstruction (PTO) with hysterosalpingography
Describe the frequency of PTO as a cause of subfertility
Identify indications for and contraindications to transluminal salpingoplasty
List different uses of falloposcopy in the management of PTO
Identify advantages and disadvantages of falloposcopy over hysterosalpingography and laparoscopy in PTO
Rationale
Although proximal tubal obstruction (PTO), is a frequent finding on hysterosalpingography (HSG), approximately two-thirds of the Fallopian tubes resected for PTO reveal an absence of luminal occlusion [1]. False-positive diagnosis of PTO ranges from 16 to 40% [2], and can be as high as 50% [3]. The specific indications and limitations of Fallopian tube recanalization (FTR) makes a careful evaluation of the Fallopian tube prior to therapy an absolute requirement, underscoring the significance of endoscopy. The distinction between true pathologic occlusion, spasm or plugging and abnormalities of the mucosa is crucial in determining therapy [1], and the diagnostic test used has an important bearing on the selection of patients and, consequently, the results, further signifying the value of endoscopy [4]. While outlining the various cannulation techniques available to diagnose and treat tubal pathology, this article will highlight the applications, efficiency and success of endoscopic FTR, and the place of tubal cannulation against the challenge of IVF.
Introduction
Pathophysiology of PTO
Tubal disease is the cause of subfertility in approximately 30% of women and 10–25% of these are due to PTO. PTO has been a diagnostic and therapeutic dilemma since its recognition more than 100 years ago [5]. It can occur in either the intramural segment or the uterotubal junction, and is the result of tubal spasm or transient occlusion by mucus plugs in up to 40% of women [6]. Figure 1 depicts a HSG plate showing a bilateral cornual block. Proximal, distal and peritubal damage can be caused by a number of pathologic processes, such as inflammation, endometriosis and surgical trauma [3]. PTO has been associated with the presence of pathologic microflora in the oviducts of 36.6% women, as confirmed by bacteriological examination of tubal fluid [7]. Inflammatory etiology seems to be important in isthmic tubal occlusion, and in many cases, chlamydial infection may be the chronic irritant, which also causes muscular hypertrophy leading to salpingitis isthmica nodosa [8]. Induced abortion, uterine curettage, pelvic inflammatory disease and intrauterine devices may all influence PTO infertility [9].

Hysterosalpingogram plate showing a bilateral cornual block.

The Wallace ET catheter that we used for hysteroscopic Fallopian tube recanalization.
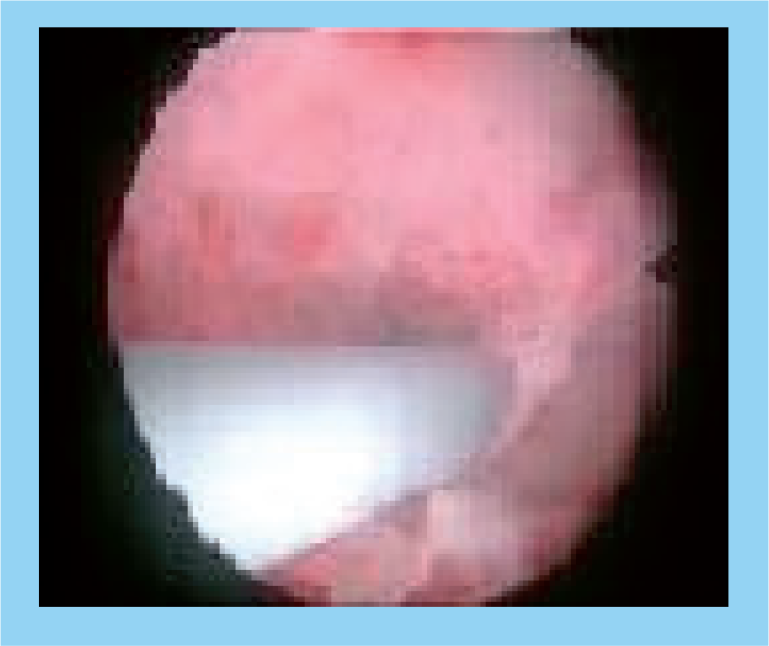
The Wallace ET sheath in the left ostium.

The guidewire seen entering the left tubal ostium.
Hypothesis for the genesis of PTO
The small caliber, thick muscular wall and reduced proportion of ciliated cells in the epithelium of the proximal tube predispose this tubal segment to blockage. Increased muscle tone, reduced ciliary activity and increased tubal secretions at the uterotubal junction (UTJ) and the isthmus during the estrogen-dominant phase of the cycle can result in stasis of the tubal luminal contents and functional obstruction of the proximal tube. This event may biologically serve to delay the zygote in the ampulla for nutritional and developmental benefits. With relaxation of the UTJ musculature, increased ciliary activity and a reduction in tubal secretions, this functional proximal tube obstruction should normally be completely reversed during the progesterone-dominant phase of the menstrual cycle. Failure to do so may result in prolonged stasis of uterine material and, therefore, initially partial (tubal spasm) and then, in a given time, complete anatomical obstruction of the narrow intramural tubal lumen [10]. Calcification of this obstructing material can follow [11]. Fibrosis may represent a nonspecific final response to chronic injury of the transmural and isthmic segments of the oviduct [12], following which, tubal damage has become irreversible.
Classification of PTO
Proximal tubal obstruction has been classified into nodular (salpingitis isthmica nodosa or endometriosis), non-nodular (true fibrotic occlusion) and so-called ‘pseudo’ occlusion (detritus, polyps or hypoplastic tubes) [13]. Falloposcopic observations of endotubal isthmic plugs reveal a cast of debris-containing aggregates of histiocytic-like cells of endometrial stromal or mesothelial origin, or white to yellow mucus-like fragments of unknown physiological or pathophysiological significance [11]. Histological examinations of excised tubal segments reveal tubal abnormalities, such as obliterative fibrosis, salpingitis isthmica nodosa, endometriosis, chronic salpingitis, chronic tubal inflammation and tuberculosis in varying frequencies (Table 1). Obliterative fibrosis has been observed as the most common histologic tubal abnormality by both Wiedemann et al. [14] and Fortier and Haney [12] followed by salpingitis isthmica nodosa. The highest incidence of salpingitis isthmica nodosa was reported by Punnonen et al. (60%) [8], while the highest incidence of chronic tubal inflammation was reported by Zhang et al. (70.59%) [9]. On the basis of their observations on the pathologic spectrum of UTJ, Fortier and Haney demonstrated that there are multiple distinct histologic patterns and intraabdominal findings that do not predict the histology of the UTJ pathology [12].
Etiology of proximal tubal obstruction.
Proximal tube obstruction is the most treatable since it often occurs because of the accumulation of mucus or debris, which forms an impacted plug in the interstitial or proximal isthmic portion of the tube [11]. Until recently a domain of microsurgery [15], the long-standing surgical corrective approach to treat tubal occlusion has been replaced by noninvasive tubal recanalization methods in selected patients with endoluminal damage [16]. FTR is a minimally invasive procedure used to open blocked Fallopian tubes in patients with a history of infertility and confirmed PTO. There are several techniques to recanalize proximally obstructed tubes (Box 1). However, the following discussion will focus on the endoscopic techniques for FTR.
Clinical discussion
Evolution of tubal cannulation
Rapid progress has been made regarding minimally invasive access to the human Fallopian tube. Although the diagnosis of tubal occlusion relies primarily on HSG, hysteroscopy and laparoscopy, advances in ultrasound technology and fiberoptics have enhanced our ability to accurately and noninvasively diagnose and treat tubal pathology with innovative diagnostic procedures (e.g., sonosalpingography, falloposcopy and selective salpingography) and have enabled the visualization of the tubal endosalpinx, a portion of the reproductive tract that has evaded endoscopic evaluation. Endosalpingeal changes can be quantitated in the presence of hydrosalpinges, and possibly with endometriosis, and these changes measured with a scoring system [17]. Tubal cannulation has emerged as an excellent alternative to microsurgical tubal anastomosis to treat patients with cornual obstruction, avoiding a laparotomy and extended disability [3].
Instrumentation
Initial attempts in the treatment of proximal obstruction involved the use of a whalebone bougie positioned in the uterine cornua to dilate the proximal tube by Smith as early as 1849 [18]. Although it was considered impossible to atraumatically pass probes along the intramural segment of the human oviduct due to its tortuosity and small caliber until recently, these difficulties have been largely overcome by the miniaturization of cannulation devices and the development of coaxial catheter systems with flexible atraumatic guidewires initially used for coronary angioplasty and vascular embolectomy. Selective salpingography, transcervical cannulation and the injection of contrast medium directly into the Fallopian tube was first performed in 1966, using a curved metal cannula [19]. Technological advances have led to major improvements in the design and application of Fallopian tube cannulation devices using the transcervical approach [20]. Since the first description of fluoroscopic cannulation in 1985 and transcervical balloon tuboplasty a year later [21], there have been numerous reports of successful cannulation using ureteral catheters, ureteral stents, 19-gauge epidural catheters, guidewires and, more recently (and commonly), the coaxial systems [5]. Presently, such transcervical cannulation systems, incorporating a very fine endoscopic fiber, are being used to visualize the lumen of the Fallopian tube, displace debris that may block the tube, eliminate proximal endoluminal plaques, breakdown intraluminal adhesions, perform intratubal insemination or embryo deposition to facilitate conception and thus overcome infertility, or conversely, to facilitate the option of sterilization reversal [20]. After proven mild or moderate tubal pathology, blasting of proximal incomplete obstructive disease by the use of transcervical balloon catheter dilatation or tuboscopy-guided transcervically everting balloon catheter system is possible [22].
Fallopian tube recanalization techniques.
Hysteroscopic tubal catherization and hydrotubation
Laparo–hysteroscopic selective tubal catheterization with insufflation of oil-soluble radiopaque dye
Hysteroscopic–falloposcopic–laparoscopic
– Tubal aquadissection
– Guidewire cannulation
– Guidewire dilatation
– Coaxial tubal cannulation
– Direct balloon tubuloplasty
Nonhysteroscopic falloposcopy
– Linear everting catheter
Transcervical balloon tuboplasty with coaxial balloon catheter
Selective salpingography and tubal catheterization (Figure 8)

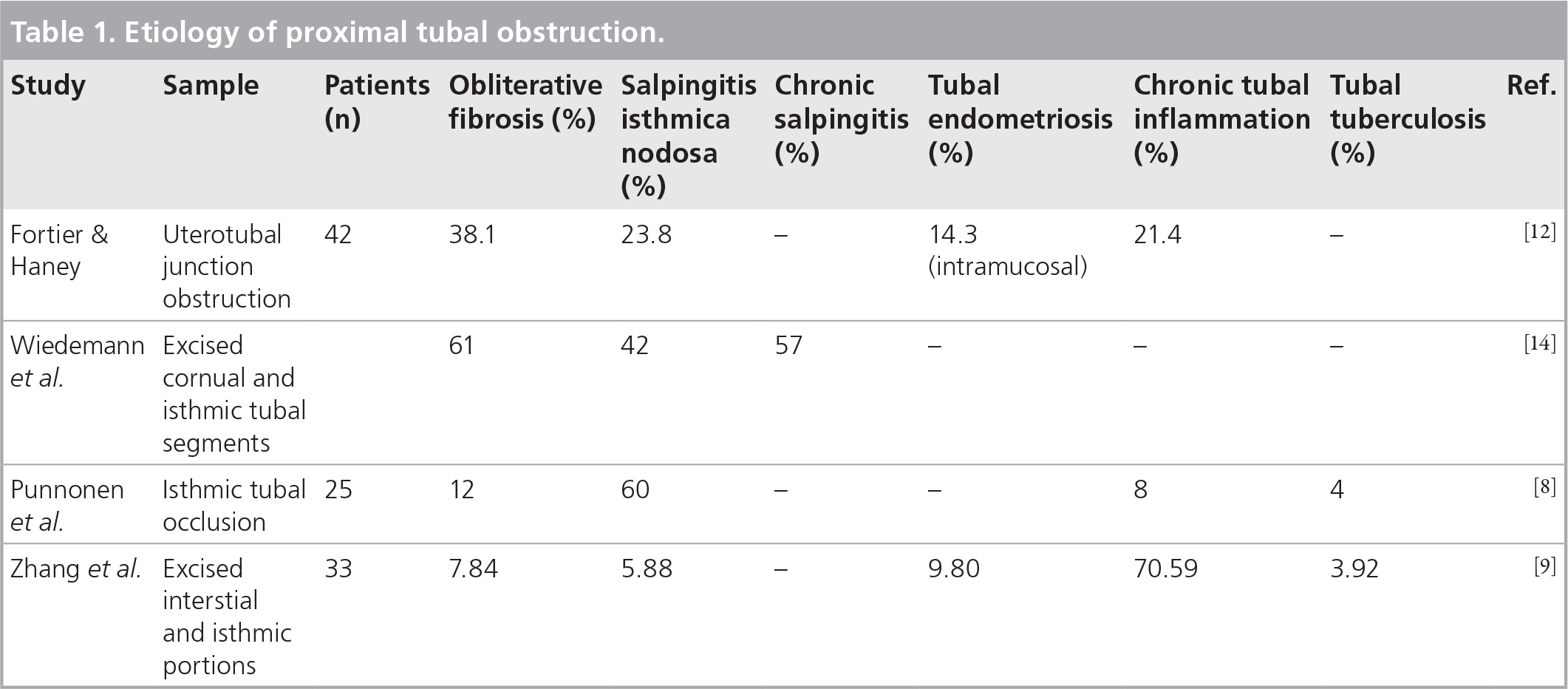
Laparoscopic view of the guidewire traversing the blocked left ostium.
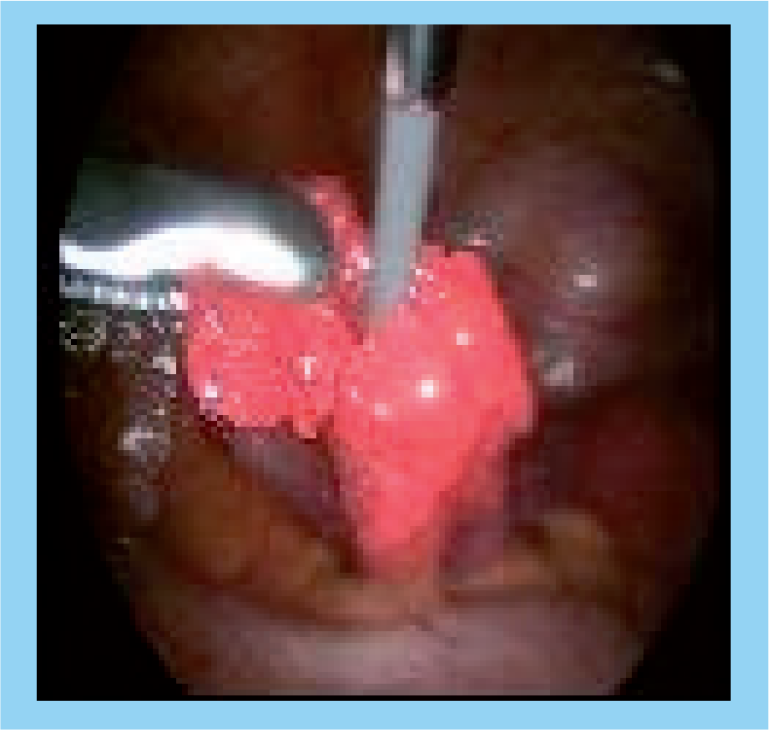
Laparoscopic view of the guidewire exiting the fimbrial end of the tube.
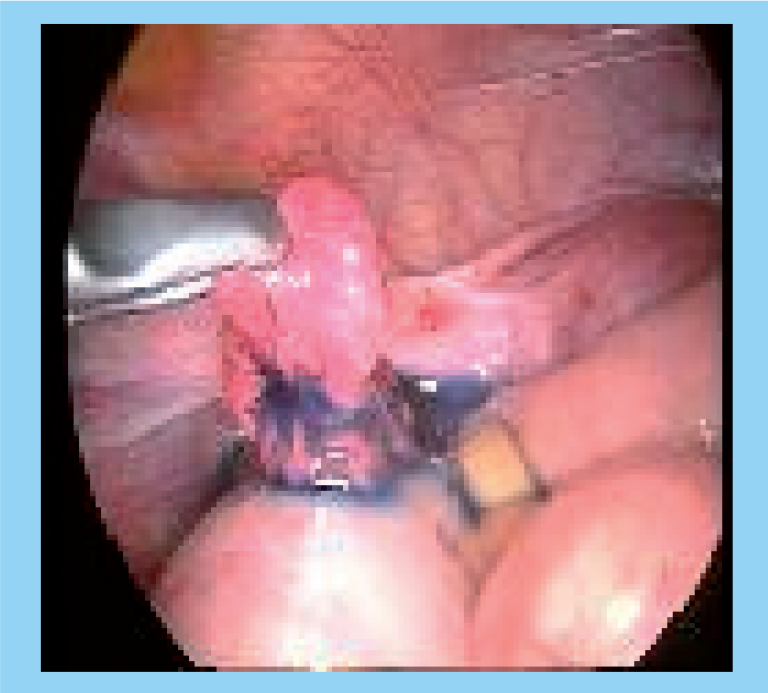
Laparoscopic view of the free spill of dye post-Fallopian tube recanalization.

The Cook Fallopian tube recanalization catheter in the right ostium.
Sonographically guided transcervical balloon tuboplasty
Sonographically guided Fallopian tube recanalization under laparoscopic control
Color Doppler ultrasound-guided coaxial cannulation and transcervical wire tuboplasty
Sonographically guided transcervical Fallopian tube catheterization using an ultrasound contrast agent

Tactile Fallopian tube recanalization with the Labotect cannula in progress.

The Labotect Fallopian tube recanalization cannula snugly in place in the right ostium.

The guidewire seen exiting the bulbous tip of the Labotect Fallopian tube recanalization cannula.
Hysteroscopic tubal cannulation with selective salpingography under fluoroscopic guidance.
Although Fallopian tube cannulation with coaxial catheters began under fluoroscopy and was adaptive to cornual cannulation, coaxial catheter systems are now being used with hysteroscopy, fluoroscopy, ultrasonography and tactile sensation with consistent success [23]. FTR can be performed with catheters, guidewires or balloon systems under sonographic, fluoroscopic or hysteroscopic guidance [6]. Figures 2–7 illustrate laparo–hysteroscopic guidewire cannulation. Figure 8 illustrates the Cook Fallopian tube recanalization catheter used for transcervical fluoroscopically guided catheter recanalization. Figures 9–11 illustrate tactile catheterization. Coaxial catheters and balloon systems have been used with similar success rates. However, the simplicity of coaxial catheters, particularly with the use of the hysteroscope and under laparoscopic control simplifies the technique, enables direct observation of the UTJs, tubal cannulation and evaluation of the entire pelvis, and avoids exposure to radiation [16]. Wenzl et al. introduced a specially developed linear everting catheter (LEC) in combination with a microendoscope that enables the visualization of the complete tubal mucosa from a vaginal approach [24]. With the development of the LEC system, it is now possible to evaluate the tubal lumen and to diagnose changes in the tubal wall or the tubal mucosa by direct visualization. Advantages of the new LEC system include both the ability to inspect the tubal ostium without cervical dilation or concomitant hysteroscopy, and virtually atraumatic access to the tube by means of an endoscope measuring 0.5 mm in diameter and with a magnification of 40 [24]. With adequate experience, this technique may also be performed in an outpatient setting [25]. Other potential applications of this new technology are the intratubal transfer of gametes and embryos, conservative treatment of tubal pregnancy, direct visualization of the tubal epithelium (falloposcopy) and contraception [23,25]. Collectively, these techniques offer the ability to define tubal pathology more precisely, facilitating proper directed therapy [23].
Applications of FTR
Transcervical tubal catheterization procedures for the diagnosis of tubal disease, obliteration, recanalization and medication are minimally invasive procedures that allow transcervical treatment of PTO and can improve our understanding and diagnostic accuracy of tubal disease [26]. True occlusion by amorphous material, flimsy adhesions or a polyp can only be successfully treated by various uterotubal cannulation methods and guidewiring the Fallopian tubes can re-establish tubal patency and fertility [2]. By restoring tubal patency, tubal cannulation can be used effectively in the management of female subfertility secondary to isolated PTO, thus avoiding the need for expensive assisted reproductive techniques [3]. By identifying patients with proximal and distal occlusion (bipolar tubal occlusion) and differentiating between true and false diagnoses of PTO, tubal cannulation eliminates or postpone the need for a costly hysteroscopy or laparoscopy. In contrast to invasive laparotomic and laparoscopic microsurgical interventions, tuboplasty is advantageous because it is minimally invasive with lower peri- and post-operative morbidity, takes less time, anesthesia is rarely required and the risks (e.g., injury of the bowel or bleeding after vessel perforation) are reduced thus resulting in shortened convalescence [22].
Although radical changes have occurred in the treatment of PTO, the repair of distal and peritubal damage frequently yields disappointing results [27]. Fallopian tube catheterization is diagnostically useful and technically highly successful for treating occluded tubes, however, patients with distally blocked tubes are not good candidates for this procedure [28,29]. Distal isthmic, ampullary or fimbrial occlusions, commonly due to previous pelvic infection or endometriosis, are difficult to recanalize with poor pregnancy rates.
Contraindications
Contraindications to transluminal salpingoplasty include florid infections and genital tuberculosis, long tubal obliterations that are difficult to bypass with the catheter, severe tubal damage, male subfertility and previously performed tubal surgery. Because of the inflammation reaction in florid infections, the tubal wall is vulnerable, susceptible to rupture and, consequently, at risk for peritonitis, while long tubal obliterations that are difficult to bypass with the catheter may also result in a perforation of the tubal wall [22]. Tuberculosis, salpingitis isthmica nodosa, isthmic occlusion with club-changed terminal, ampullar or fimbrial occlusion, and tubal fibrosis have been cited as reasons for recanalization failure [30]. Cobblestone appearance of the distal tubes heralds significant mucosal damage, which is prone to progressive disease and, hence, there is a poor chance for conception [31]. Restoration of the tubal function (i.e., gamete or blastocyst transport) after tuboplasty is unlikely in cases with severe tubal damage. Cases with previous surgery must be well selected prior to tubal interventional surgery [22]. The rate of long-term post interventional re-occlusion seems to be high and must be evaluated in case of a failure to achieve a pregnancy after successful recanalization.
Fallopian tube recanalization techniques
Endoscopic techniques
Falloposcopic diagnosis of tubal disease
Falloposcopy provides a unique possibility to visualize and grade endotubal disease and may provide a valuable instrument for in vivo exploration of tubal physiology [32–35]. It has been successfully used to characterize normal and abnormal epithelial changes, document endotubal lesions ranging from accumulated debris, nonobstructive intraluminal adhesions, stenosis, polyps, to total fibrotic obstruction, as well as the identification of the segmental location of tubal pathology without complications [36].
A useful falloposcopic classification and scoring system of tubal lumen lesions is utilized to grade tubal damage [11]. Using this scoring system, Kerin et al. classified endotubal lumens as falloposcopically normal (46%), to contain mild-to-moderate disease in 29%, or severe-to-obstructive disease in 25% cases [35]. They reported endotubal lesions in 57% examinations in 55 women without complications and observed that the majority (70%) of lesions were confined to the medial third of the tube, between the UTJ and ampullary isthmic junction [36]. Using a microendoscopic transvaginal technique incorporating guidewire cannulation and direct balloon tuboplasty, Kerin et al. characterized endotubal lesions into intramural stenosis (five cases); isthmic stenosis (ten cases); isthmic obstruction (five cases); salpingitis isthmica nodosa (two cases); nonobstructive endotubal disease from intraluminal adhesions, associated devascularization and epithelial atrophy in the intramural, isthmic and ampullary segments (ten cases); hydrosalpinx (two cases); and intratubal polyp (one case) in 35 of 43 fallopo-scopies performed, with normal falloposcopic appearance of the fimbrial, ampullary, isthmic and intramural tubal epithelium being observed in 18.6% cases [37].
Falloposcopic tubal cannulation
Falloposcopic cannulation has been performed hysteroscopically under laparoscopic guidance by dilation [15] and coaxial catheters [38–41], and nonhysteroscopically with the LEC [34,42]. The results of the various endoscopic Fallopian tube recanalization procedures are presented in Table 2. Higher recanalization success rates have been reported with falloposcopic cannulation using the LEC compared with those achieved with coaxial cannulation. Kerin et al. reported technical difficulties related to the failure to negotiate the entire tubal lumen in the absence of obstructive disease, and minor difficulties due to ostial spasm secondary to attempted guidewire cannulation [36]. Technical and minor difficulties were experienced in 11 and 8%, respectively, of the 84 endoscopy cases, during coaxial falloposcopy [36]. They suggested that while technical difficulties with catheterization may be partly overcome by the incorporation of smaller directional guidewires, softer distortion-free Teflon catheters, improved microendoscopes and the acquisition of new surgical skills necessary for safe and successful endoscopy of the Fallopian tube, minor difficulties are generally overcome once spasm ceases [36].
Summary of results following various endoscopic Fallopian tube recanalization techniques.
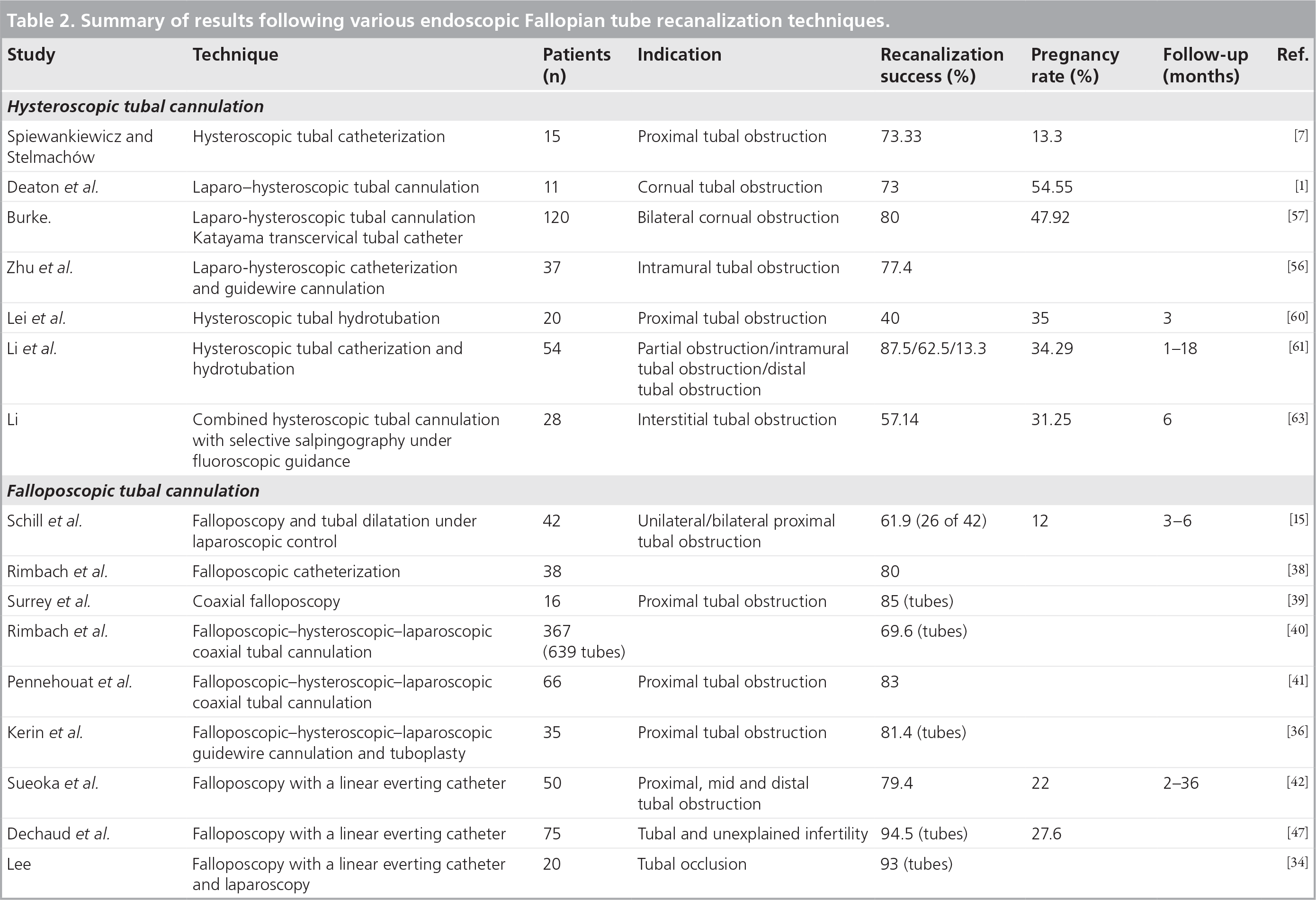
Falloposcopy may be used therapeutically for dislodging intraluminal debris and breaking down filmy adhesions in normal or minimally diseased tubes [32,35], and may additionally aid in differentiating between patients suitable for tubal surgery and those who should be referred for IVF [33,34]. Techniques of tubal aquadissection, guidewire cannulation, guidewire dilatation and direct balloon tubuloplasty under hysteroscopic–falloposcopic–laparoscopic control have been devised for attempting to breakdown intraluminal adhesions or dilate a stenosis. Following falloposcopic-directed, selected tubal cannulation and aquadissection techniques, isthmic plugs occluding the entire isthmic lumen have been successfully mobilized in 4% of the cases studies and tubal patency restored in all the cases [11]. Using one or more of these techniques, obstruction in 81.4% tubes containing a lesion could be opened up [36], while a combination of guidewire cannulation and direct balloon tubuloplasty procedures under hysteroscopic–falloposcopic–laparoscopic control could breakdown nonobstructive intraluminal adhesions in 60% of cases, dilate intramural or isthmic stenoses in 40% of cases and negotiate an isthmic stricture secondary to salpingitis isthmica nodosa in 50% of cases. The techniques were unsuccessful in bypassing fibrotic obstructions [37]. Using aquadissection, flexible wire cannulation or direct balloon tuboplasty, Kerin et al. reported conception rates of 21% in women whom at least one tube was normal and 9% in women with mild-to-moderate disease within 1 year of the procedure; no conception was obtained in women with severe endotubal disease [35]. They concluded that while falloposcopic tuboplasty may have a therapeutic role in normal or minimally diseased tubes, the presence of severe disease remains resistant to the use of current endotuboplasty treatments, as reflected by poor pregnancy outcome, and such women should be provided with the option of microsurgical tubal repair or IVF and embryo-transfer procedures [35].
Coaxial falloposcopy
The coaxial falloposcopy system consists of a hysteroscope, stabilizing device to maintain uterotubal alignment, a flexible coaxial catheter and guidewire and a 0.4-mm outer diameter (OD) falloposcope with enhanced fiberoptics [43]. Coaxial falloposcopy is a transvaginal endoscopic technique that utilizes a small flexible microendoscopic instrument, the falloposcope (OD: 0.5 mm), for effective visualization of the entire length of the human Fallopian tube from the uterotubal ostium to the fimbria. Using a transvaginal approach, upon visualization of the tubal ostium, tubal cannulation is performed via the working channel of a small flexible operating hysteroscope (OD: 3.3–4.5 mm; operating channel diameters: 1.5–1.8 mm). Fluid is constantly administered via the flexible cannula. A coaxial technique, incorporating guidewire cannulation and placement of an over-the-wire Teflon catheter may be used, following which, the guidewire is replaced with a falloposcope for video documentation of the endotubal surface anatomy [36]. Common problems associated with the technique include white-out due to the intense light in close proximity of the tissues and kinking leading to catheter damage and impediment of successful falloposcope insertion. Lastly, if the endometrium is in the advanced proliferative phase, it may be difficult to visualize the ostia [44].
Falloposcopy with the linear everting catheter
Although falloposcopy initially involved the hysteroscopic insertion of a falloposcope through a flexible cannula, a miniature tubular balloon system that is rolled out along the Fallopian tube lumen by the use of hydraulic pressure, without the aid of a hysteroscope, under sedation or with local anesthesia, and that concurrently carries the falloposcope forward (linear eversion system), has recently been employed [45]. The LEC has been developed to safely guide a falloposcope into the entire length of Fallopian tube in order to observe the tubal lumen, and may also be used therapeutically for the recanalization of occluded tubes [42] and for intratubal gamete transfer [46]. Falloposcopic tuboplasty has been established as a highly useful, less invasive and novel treatment for tubal infertility that may be useful in selected patients with proximal, mid and distal tubal occlusion [42] with a good predictive value for future fertility [47]. Sueoka et al. successfully accessed 85.3% of tubes with an overall patency rate of 79.4% on follow-up HSG with the LEC [42]. Using the LEC, Dechaud et al. reported a tubal catheterization rate of 94.5% and a spontaneous pregnancy rate of 27.6% in patients who had normal tubes as defined by falloposcopy, whereas this dropped to 11.5% for mild and 0% for severe endotubal lesions [47]. Transuterine falloposcopy, using the linear eversion, is a well-tolerated technique that can be performed in an outpatient clinic with high rates of luminal cannulation and visualization. Scudamore et al. reported successful identification of tubal damage in 66.67% of the tubes identified [48].
Atraumatic access to the tube and visualization of its lumen offer exciting opportunities for diagnosis and treatment of tubal conditions [48]. Among the 15 cases with hydrosalpinx or fimbrial obstruction following falloposcopy with a LEC and laparoscopy, Lee considered 67% of the cases with flattened mucosa in the endosalpinx and endotubal adhesions suitable for IVF, while 27% of cases with normal mucosa were suitable for tuboplasty [35]. Interstitial tubal obstruction was overcome with the LEC [34].
Falloposcopic gamete intra-Fallopian transfer, using the LEC under laparoscopic control, has been reported to be a safe and efficient procedure and a less invasive alternative than laparoscopic transfer with a 20% delivery rate [46].
Coaxial versus balloon catheter set
A pilot study by Shinmoto et al. reported successful selective catheterization of the uterine cornu through a balloon catheter wedged at the internal uterine os in 87.5% of 16 occlusive Fallopian tubes (11 cases), a recanalization success rate of 75.0% of the affected tubes and a subsequent pregnancy rate of 27.27% of the cases [49]. They concluded that the technique is convenient, safe and effective and it will be accepted as the first choice in the diagnosis and treatment of Fallopian tube obstruction [49]. However, Rösch et al. concluded that the new hysterograph with the coaxial catheter set is more suitable for recanalization of the obstructed Fallopian tubes than the previously used balloon catheter set [50]. Using this instrumentation, they accomplished a recanalization success in 96% of 28 PTOs in 25 women and in 33% of six midisthmic obstructions unrelated to surgery, following selective transcervical Fallopian tube catheterization of the uterine cornua. However, repeat recanalization attempts were met with tubal perforations without apparent clinical effects in four tubes, one with proximal and three with midisthmic postsurgical obstructions [50].
Guidewire cannulation
According to Gleicher et al., although guidewire cannulation of proximally obstructed tubes achieves tubal patency in a large percentage of cases, comparable to other catheter techniques, it yields much lower pregnancy rates [51]. They suggested that guidewire cannulation alone does not represent adequate treatment for patients with proximally occluded Fallopian tubes. Guidewires, used with coaxial and balloon catheter systems, are not responsible for the pregnancy success reported for these procedures [51].
Free-hand cannulation technique
In a feasibility study on 66 patients with proximal and/or distal suspected tubal damage, Pennehouat et al. performed transcervical falloposcopy under laparoscopic control, catheterization being performed with either a transhysteroscopic or a free-hand tubal cannulation technique [41]. They demonstrated that the transcervical free-hand cannulation technique with the tubal embryo transfer catheter was as effective as the transhysteroscopic approach and this technique is a simple and effective aternative to the transhysteroscopic approach [41].
Falloposcopy versus laparoscopy/hysterosalpingography
Current diagnostic techniques, such as laparoscopy and HSG, can detect only a fraction of the causes of tubal infertility and in many cases, misdiagnosis results [33]. Although interstitial Fallopian tube obstruction has been reported in 15% of HSG performed for infertility, conventional HSG or laparoscopy may not differentiate cornual spasm or other temporary causes from true obstruction [52]. Falloposcopy offers the advantage of objectively classify the cause of PTO and demonstrating that isthmic plugs can cause reversible proximal PTO [11]. Comparing falloposcopy employing a new coaxial system with traditional laparoscopic chromotubation and HSG in a prospective, multicenter clinical trial at five tertiary infertility centers, Surrey et al. concluded that falloposcopy with the new coaxial system allows improved visualization with less bulky and less traumatic instruments [39]. The system provides valuable information regarding the Fallopian tube lumen that correlates poorly with that obtained with more traditional techniques. Management was changed in 52.4% of women as a result of falloposcopic findings [39].
Despite increasing evidence of its potential clinical value, falloposcopy has not yet found widespread use [40]. Lundberg et al. were able to obtain endosalpinx images in 60.5% of the women; however, none of the images were of sufficient quality to describe the entire tubal mucosa in detail [32]. They concluded that although falloposcopy represents a unique tool for visualization of endotubal disease and a valuable instrument for in vivo exploration of tubal physiology, certain technical problems limit the usefulness of this method in routine clinical practice, and these problems must be solved before falloposcopy can achieve a central position in the investigation and treatment of tubal disease [32]. In a large prospective international multicener study, Rimbach et al. reported 6.1% failures during hysteroscopy (10.6% during the cannulation step and 16.4% during visualization) following falloposcopic coaxial tubal cannulation using hysteroscopic ostium access and retrograde visualization under laparoscopic control [40]. Predominantly intracavitary pathology or thick endometrium were found to interfere with hysteroscopic ostium access, while technical insufficiencies, resulting in catheter damage or vision disturbing light reflections, were identified to be responsible for most cannulation and visualization failures. The number of patients who received a complete falloposcopic evaluation did not exceed 57%. The authors concluded that owing to the technically limited results, the method currently qualifies for selected indications rather than for routine clinical application [40]. Hence, although falloposcopy provides information regarding the condition of the tubal mucosa that is unavailable by any other technique, adding precision to surgical techniques when they are deemed necessary while directing other patients to assisted reproductive technologies, it is still in its infancy and data from larger studies are needed [53].
Hysteroscopic tubal cannulation
Advances in hysteroscopy in the past decade, including the introduction of small-caliber endoscopes, microhysteroscopy, the flexible steerable hysteroscope and the use of video systems in monitoring hysteroscopic evaluations, have extended the application of hysteroscopy to tubal cannulation both for treating cornual obstruction and in conjunction with new reproductive technologies [54]. While cannulation with coaxial catheters began under fluoroscopy, the use of the hysteroscope simplifies the technique. Hysteroscopic tubal catheterization in patients with PTO can be used both as a diagnostic and considerably effective therapeutic method [7]. Initial attempts with hysteroscopic proximal tube catheterization and balloon dilatation for recanalization proved intraoperatively successful in more than 80% of the cases [55]. With laparoscopy, the hysteroscopic approach enables tubal cannulation and evaluation of the entire pelvis. Treatment of additional problems affecting the Fallopian tubes, particularly adhesions and endometriosis, is possible. While laparoscopy helps monitor the procedure and visual assessment of tubal patency, the ability to observe the UTJs directly by hysteroscopy provides an excellent approach for tubal cannulation [5] (Figures 4–7).
The results of the various hysteroscopic cannulation techniques employed to diagnose and treat PTO are presented in Table 2. Laparo-hysteroscopic tubal cannulation with or without guidewire cannulation has yielded an average recanalization success rate of 76%% with an average intrauterine pregnancy rate as high as 39% [1,7,56,57]. Using combined laparoscopy and hysteroscopic tubal cannulation, Deaton et al. concluded that hysteroscopic cannulation of the Fallopian tube is a safe diagnostic procedure that can be used to identify those patients with true proximal occlusion, and may also serve as a therapeutic procedure in some of these patients [1]. However, conception in their study was achieved after tubal cannulation and adjunctive distal tubal surgery confounding the results [1]. Zhu et al. performed laparo-hysteroscopic cannulation of the proximal oviduct with a flexible guidewire to evaluate and treat intramural Fallopian tube obstruction, and also concluded that this procedure is an effective method for the evaluation and treatment of cornual obstruction [56].
Lin et al. described a soft and rigid operating fiberoptic hysteroscope (OD: 4.8 mm) that can be used clinically for transcervical tubocornual recanalization for the management of cornual occlusion [58]. The functional part of the telescope consists of three sections: a soft, flexible front section; a rigid rotating middle section; and a semi-rigid, self-retaining rear section offering advantages of an easy, close and direct approach to the intrauterine target, usually with no cervical dilation or anesthesia, with the operator in a comfortable position and without reported complications. This new hysteroscope has proved to be a very useful tool for the treatment of intrauterine lesions in the theater or an office setting [58]. Clinical results in 1503 women who underwent panoramic, televised fiberoptic hysteroscopy without cervical dilation suggest that the soft and rigid structure of the Fujinon diagnostic fiberoptic hysteroscope offers advantages over rigid scopes or conventional fiberscopes with full-length soft, malleable parts [59].
Hysteroscopic hydrotubation with a hydrotubation solution consisting of hydrocortisone (20 mg), gentamicin (160,000 IU) and procaine (80 mg) in 20 ml distilled water may be an alternative treatment for tubal blockage [60]. Although the addition of hydrotubation to hysteroscopic tubal catheterization resulted in low recanalization rates compared with laparo-hysteroscopic tubal cannulation (Table 2) [60,61], studies have reported effective recanalization of partially obstructed tubes, intramural tubal obstruction and distal tubal obstruction with an intrauterine pregnancy rate of 34.29% over a 12-month follow-up after treatment [61]. Laparo–hysteroscopic selective tubal catheterization with insufflation of oil-soluble radiopaque dye has been reported to be an effective treatment for infertility associated with endometriosis with higher conception rates compared with women without endometriosis (60 vs 36.5%) [62]. Combined hysteroscopic tubal cannulation with selective salpingography under fluoroscopic guidance has been reported as a safe and simple diagnostic method that has also been used to identify and successfully treat interstitial Fallopian tube obstruction [52,63]. However, compared with the other hysteroscopic cannulation techniques, the addition of selective salpingography under fluoroscopic guidance to hysteroscopic tubal cannulation appears to have yielded the lowest patency and pregnancy rates (Table 2). A systematic review of observational studies [64] showed that hysteroscopic tubal cannulation was associated with a higher pregnancy rate (49%) than salpingography and tubal catherization (21%) in women with PTO [65].
Similar intrauterine and cumulative pregnancy rates and obstetric outcomes have been reported with both hysteroscopic cannulation as well as tubal microsurgery in patients with PTO [66,67]. However, in lieu of the lower ectopic pregnancy rates in the cannulation group (none of 21 [0%] vs seven of 24 [29.1%]), hysteroscopic cannulation has been recommended as the first choice in the management of PTOs in selected patients. It may be a treatment option for delayed occlusion after successful cannulation or resection anastomosis [67]. Hysteroscopic gamete intra-Fallopian transfer has been reported as an alternative, safe, effective and noninvasive technique for fertility problems with a satisfactory pregnancy rate [68] and may be carried out in some cases where general anesthesia was not advisable or possible (i.e., difficulties in tubal catheterization due to pelvic adhesions, extended distal tubal damage, patients’ intolerance or lack of available operating theatre [69]).
Fluoroscopic Fallopian tube recanalization
Selective salpingography & transcervical FTR
Selective salpingography and transcervical FTR is a fluoroscopically guided procedure that employs catheterization of the tubal ostium and visualization of the transcervical instillation of contrast media under fluoroscopic imaging. In the event of a PTO, this may be followed by tubal catheterization and guidewire cannulation, passing a soft 2 or 3 French catheter, loaded over a 0.015-inch guidewire, through the obstruction via the cornual catheter to clear the obstruction [47]. The use of a 4F glide catheter with a 0.89 mm guidewire advanced transcervically streamlines the procedure. Fluoroscopic guidance may be used to negotiate the guidewire beyond the intramural portion of the tube and selective salpingography to document the outcome of the recanalization [70]. A recanalization success rate ranging from 71 to 92% with an average pregnancy rate of 30% has been reported in the literature [71]. Selective salpingography and tubal catheterization offer patient-friendly, less-invasive and cost-effective alternatives to tubal microsurgery and IVF in patients with tubal occlusion [70,65] with high success rates and improved overall management of infertility caused by tubal obstruction [71]. However, major disadvantages of the use of fluoroscopy include the difficulty in ruling out tubal spasm, inability to evaluate distal tubal disease and other pelvic abnormalities [16], and the risk of radiation exposure. Allergy to the contrast medium may be a contraindication to the use of the technique [6]. Results with selective salpingography and tubal catheterization/guidewire cannulation suggest that while guidewire cannulation is the most effective method used to achieve tubal patency, the prognosis with regard to pregnancy is poor, and alternative therapy such as microsurgery or IVF should be considered early [72].
Fluoroscopically guided transcervical balloon tuboplasty
Fluoroscopically guided transcervical balloon tuboplasty, which utilizes a coaxial balloon catheter, has been reported as a safe, noninvasive, outpatient technique to treat PTO that may represent an alternative to IVF or microsurgical reanastomosis of Fallopian tubes [73,74]. Balloon tuboplasty has reported to be more effective in restoring patency in cases with failed selective salpingography. However, the choice of balloon tuboplasty catheter system may affect success rates [75].
Sonographic FTR
Sonographically guided transcervical tubal catheterization and transcervical balloon tuboplasty may be successfully performed to diagnose and treat patients with PTO. It may be aided by fluoroscopy [76], color Doppler ultrasound-guided coaxial cannulation and transcervical wire tuboplasty [77], hysteroscopic/laparoscopic insertion of small intraluminal ultrasound transducers into catheters of diameters of 3.5 F and 5 F during transcervical Fallopian tube catheterization [78], and transvaginal sonography-guided trans-uterine cannulation of the tubes with the Jansen–Anderson catheter and injection of sterile fluid [79]. However, the requirement of fluoroscopy in sonographically guided transcervical tubal catheterization and transcervical balloon tuboplasty for identification of the catheter tip and successful cannulation of the internal tubal ostia, entailing a risk of radiation exposure [76] and the inability to differentiate between tubal epithelium and muscularis with the intraluminal ultrasound transducers despite successful catheterization [78], limit the application of these techniques. On the other hand, Lisse and Sydow reported high recanalization success rates (91.2%), patency rates (84.2%) and intrauterine pregnancy rates (31.6%) at a 6-month follow-up interval following laparoscopically controlled sonographic transvaginal catheterization in patients with bilateral PTO [80]. They suggested that the technique may be used early in the diagnostic schedule of the infertile patient and call into question the application of microsurgical treatment of a selected group of patients [80]. Transcervical wire tuboplasty with a coaxial catheter and guidewire cannulation under color Doppler mapping ultrasound guidance has been reported to be an effective technique with high 1-year patency (96%) and pregnancy rates (38%), while avoiding the risk of radiation and allergic reaction [77]. Trans-uterine cannulation of the tubes with the Jansen–Anderson catheter and injection of sterile fluid under transvaginal sonography has been reported to be an easy and safe method to evaluate the tubal status, prove tubal patency in the early diagnostic stage and thus prevent loss of time and futile treatment cycles [79].
Tubal cannulation or IVF?
In patients with documented tubal disease, options for management would essentially include expectant management, tubal surgery or IVF. Advances in minimally invasive endoscopic cannulation techniques and the soaring acceptance and applications of IVF question the surgical management of patients presenting with tubal damage. Until the widespread use of IVF in the beginning of the 1980s, tubal surgery was the only available option for restoration of fertility in patients with PTO. Although tubal microsurgery and IVF may be complementary options in the management of patients with tubal obstruction following failed FTR, and although microsurgery to correct localized damage has the advantage of long-standing restoration of fertility, poor pregnancy rates with tubal microsurgery in patients with severe tubal damage and the lack of technical skill required to perform these procedures has resulted in a liberal referral to IVF. Selection of patients with tubal disease for future therapeutic management is based on tubal lesions, including the aspect of tubal mucosa and tuboperitoneal environment [4], and the severity of the tubal damage and the health of the mucosa is key in determining the outcome. Where the mucosa is unhealthy, surgery is not justified; early referral for IVF is indicated [81].
Fallopian tube catheterization is diagnostically useful and technically highly successful for treating occluded tubes; however, patients with distally blocked tubes are not good candidates for this procedure [28,29] and distal tubal obstruction, caused by fibrosis and peritubal disease, is often not amenable to catheter recanalization techniques. While a functional obstruction may be amenable to conservative management, true occlusion may require management by microsurgical techniques or IVF [14]. Cases of failed fluoroscopically guided tubal canalization have been attributed to severe intrinsic tubal disease and tubal occlusion rather than to the technique [14]. Significantly higher pregnancy rates have been reported in patients without distal disease than those with bipolar tubal disease (49 vs 12%, life table-adjusted rate; p = 0.0002) independent of underlying etiology for tubal disease [73]. Dechaud et al. reported no pregnancies in patients with severe endotubal lesions despite a high tubal catheterization rate following falloposcopically guided cannulation [47]. Following nonhysteroscopic falloposcopy under laparoscopic control, Lee reported that while interstitial tubal obstruction was overcome with the LEC, among the 15 cases with hydrosalpinx or fimbrial obstruction, 67% of the cases with flattened mucosa in the endosalpinx and endotubal adhesions were suitable for IVF and 27% cases with normal mucosa were suitable for tuboplasty [34]. Letterie and Luetkehans reported no pregnancies following Fallopian tube canalization and microsurgery in patients with bipolar tubal occlusion after 12 and 18 months follow-up [82]. They suggested that in lieu of the lower patency rates and higher recurrence rates of PTO when compared with data of prior studies, IVF, although more costly, may ultimately represent the most expedient and effective method of management of coexistent proximal and distal tubal disease (bipolar disease) [82]. Microsurgical repair of bipolar tubal damage yields poor pregnancy rates and the expertise required to perform these techniques is not always available. Because distally obstructed tubes cannot be successfully catheterized, the potential impact of FTR depends on the percentage of cases in which the occlusion is proximal. Tubal surgery or IVF treatment is not influenced adversely by prior transcervical tubal recanalization and remains an option for patients who failed to attain pregnancy [83].
Future challenges
Despite the diagnostic superiority of falloposcopy in the visualization of endotubal disease and in vivo exploration of tubal physiology and its therapeutic advantages, technical shortcomings associated with falloposcopy (e.g., white-out due to the intense light in close proximity of tissues, kinking leading to catheter damage and impediment of successful falloposcope insertion) as well as personal expertise required with the technique, limit the usefulness of this method in routine clinical practice. Hence, we envisage a robotically assisted hysteroscopic-falloposcopic FTR technique under ultrasonic vision using the atraumatic Cook echotip embryo-transfer catheters to facilitate visualization and cannulation. While overcoming the necessary technical shortcomings associated with the technique, a robotically assisted manipulation would facilitate the catheter maneuver along the tortuous trajectory of the proximal Fallopian tube. Ultrasound-guided cannulation would help map the catheter path, avoiding tubal perforations during guidewire cannulation. The micromanipulation could alternatively be simulated on a monitor akin to that used for intracytoplasmic sperm injection. The use of thermally controlled catheters/guidewires may present the possibility of effectively clearing fibrotic occlusions that have so far evaded recanalization.
Conclusion
The diverse applications, safety, efficacy, credibility, noninvasiveness, reduced risks, costs and morbidity and encouraging results with endoscopic FTR in the evaluation and treatment of tubal obstructions make this procedure an excellent alternative to surgical invasive procedures (e.g., microsurgical tubal anastomosis and assisted reproduction), and should be offered as the initial method to treat proximally obstructed Fallopian tubes in well-selected patients. Falloposcopy is a diagnostically accurate procedure for evaluating and grading tubal disease with a good predictive value for future fertility, and falloposcopically-guided interventions under hystero–laparoscopic control have been therapeutically used to breakdown intraluminal adhesions or dilate a stenosis in normal or minimally diseased tubes with high patency and pregnancy rates. However, severe tubal disease with poor mucosal health, obliterative fibrosis, distal tubal obstruction and bipolar tubal damage are not amenable to recanalization techniques, and following an accurate diagnosis of the tubal mucosa and tubo-peritoneal environment, such cases must directly be referred for IVF. Moreover, technical shortcomings associated with falloposcopy limit the routine application of this procedure. The latest improvements in sonographic equipment and catheter technology may help eliminate radiation and replace fluoroscopy during the performance of transcervical balloon tuboplasty, making sonographic transcervical tubal catheterization a simple and cost-effective procedure for the diagnosis and treatment of patients with PTO [76].
Executive summary
Minimally invasive transcervical tubal catheterization procedures provide an excellent alternative to invasive and expensive surgical procedures and assisted reproductive technologies for the diagnosis of tubal disease and treatment of minimally diseased proximal Fallopian tubes.
Fallopian tube recanalization (FTR) can be performed with catheters, flexible atraumatic guide wires or balloon systems under endoscopic (falloposcopy/hysteroscopy/laparoscopy), sonographic, fluoroscopic or tactile guidance.
Falloposcopy provides a unique possibility to accurately visualize, characterize and grade endotubal disease, identify the segmental location of tubal pathology without complications, objectively classify the cause of proximal tubal obstruction and guide future patient management in contrast to laparoscopy and hysterosalpingography that are often associated with poor or misdiagnosis of proximal tubal obstruction.
Nonhysteroscopic transuterine falloposcopy, using the linear eversion catheter, is a well-tolerated technique that can be performed in an outpatient clinic with high rates of luminal cannulation and visualization and a good predictive value for future fertility.
Techniques of tubal aquadissection, guide wire cannulation, wire guide dilatation and direct balloon tubuloplasty, under hysteroscopic-falloposcopic-laparoscopic control, have been therapeutically used to breakdown intraluminal adhesions, and dilate a stenosis in normal or minimally diseased tubes with high patency and pregnancy rates.
The difficulty in ruling out tubal spasm, inability to evaluate distal tubal disease and other pelvic abnormalities and radiation exposure with fluoroscopy, and requirement of fluoroscopy for successful cannulation of the internal tubal ostia and failure to effectively evaluate the tubal mucosa with sonographic techniques limit the application of these techniques despite the reportedly high patency and intrauterine pregnancy rates.
Guidewire cannulation of proximally obstructed tubes yields much lower pregnancy rates compared with other catheter techniques despite the high tubal patency rates.
Recanalization is contraindicated in florid infections, genital tuberculosis, obliterative fibrosis, long tubal obliterations that are difficult to bypass with the catheter, severe tubal damage, male subfertility and previously performed tubal surgery.
Distal tubal obstruction is not amenable to catheter recanalization techniques, and tuberculosis, salpingitis isthmica nodosa, isthmic occlusion with club-changed terminal, ampullar or fimbrial occlusion and tubal fibrosis have been cited as reasons for recanalization failure.
In lieu of the poor pregnancy outcomes in patients with severe tubal disease, poor mucosal health following tubal recanalization and poor available technical skills and results with microsurgery, such women should be provided with the option of IVF and embryo transfer.
Despite the high diagnostic and therapeutic power of falloposcopic interventions, technical shortcomings with falloposcopy must be overcome before the procedure gains widespread acceptance.

Fallopian tube recanalization
To obtain credit, you should first read the journal article. After reading the article, you should be able to answer the following, related, multiplechoice questions. To complete the questions and earn continuing medical education (CME) credit, please go to www.medscapecme.com/journal/wh. Credit cannot be obtained for tests completed on paper, although you may use the worksheet below to keep a record of your answers. You must be a registered user on Medscape.com. If you are not registered on Medscape.com, please click on the New Users: Free Registration link on the left hand side of the website to register. Only one answer is correct for each question. Once you successfully answer all post-test questions you will be able to view and/or print your certificate. For questions regarding the content of this activity, contact the accredited provider,






Footnotes
