Abstract
Dienogest (DNG), a progestin of 19-nortestosterone derivative, has good oral bioavailability and is highly selective for progesterone receptors. Owing to its antiovulatory, antiproliferative activities in endometrial cells, and its inhibitory effects on the secretion of cytokines, DNG is expected to be an effective treatment for endometriosis. Progesterone receptor-binding affinity is higher for DNG than for progesterone. Several pilot studies demonstrated that after 24 weeks of DNG treatment, there was a significant decrease in terms of dysmenorrhea, premenstrual pain, dyspareunia and diffuse pelvic pain. Most of the cases of genital bleeding occurring in the DNG treatment were spotting or breakthrough bleeding, which decreased with continued treatment and resolved either during treatment or after the end of treatment. The therapeutic effects of DNG 2 mg/day and norethisterone acetate 10 mg/day for endometriotic symptoms during a period of 24 weeks were almost similar. The only disadvantage of DNG seems to be the irregular bleeding. Good efficacy and tolerability of DNG in patients with endometriosis have been demonstrated in an open, randomized European clinical trial as compared with norethisterone acetate. In Japan, a Phase III, randomized, double-blind, multicenter, controlled trial was conducted to compare the efficacy and safety of DNG with intranasal buserelin acetate in patients with endometriosis. The study demonstrated that DNG is as effective as intranasal buserelin acetate in alleviating endometriosis symptoms, and causes less bone mineral density loss, resulting in the use on a commercial basis for endometriosis patients in Japan from 2008. This paper provides summarized data on this new promising drug for endometriosis.
Keywords
Endometriosis is characterized by the presence and growth of endometrial tissue in locations other than the uterine cavity. It is estimated to affect 10–15% of all women of reproductive age, and is frequently associated with painful symptoms such as dysmenorrhea, dyspareunia, chronic pelvic pain and infertility [1–3]. Endometriosis is a chronic and recurrent disease that adversely affects young patients' quality of life (QOL), requiring constant control of the symptoms. It has also been reported that 25–40% of women with infertility have endometriosis [1]. It is well known that the treated lesions would result in the recurrence at the high rate and no medication is curative, even if any treatments were provided for patients of endometriosis.
Surgical treatment of endometriosis is thought to be a most powerful and efficacious treatment modality for alleviating both pain and infertility problems. On the other hand, disease recurrence after operation is quite frequently observed and is a most troublesome issue both for patients and gynecologists [1–3]. Hormonal therapies are aimed to suppress ovarian function and reduce the serum estra-diol concentration and thus shrink the endo-metriotic lesions [4]. Gonadotropin-releasing hormone (GnRH) agonists are the principal therapeutic medications. Danazol, progestin and estrogen–progestin combinations are also used [4]. Although GnRH agonists exhibit considerable efficacy by reducing the serum estradiol concentration to postmenopausal levels, these agents are accompanied by a high incidence of hypoestrogenic symptoms; and their long-term use is associated with a substantial decrease in bone mineral density (BMD), limiting the length of time they can be used [5]. Thus, drugs that are highly effective and that can be used over an extended period of time are urgently needed.
Recently, a new device, levonorgestrel-releasing intrauterine system (LNG-IUS) could become the treatment of choice for chronic pelvic pain-associated endometriosis. This device releases LNG directly into the uterine cavity for 5 years. Both the LNG-IUS and the GnRH agonists were equally effective in the treatment of endometriosis [6]. The use of LNG-IUS after laparoscopic surgery for symptomatic endometriosis reduced the mediumterm risk of recurrence of moderate or severe dysmenorrhea [7].
Dienogest (DNG), a progestin derived from 19- nortestosterone, has good oral bioavailability and is highly selective for progesterone receptors [8]. Owing to its antiovulatory effects in humans, antiproliferative activities in isolated human endometrial cells and its inhibitory effects on the secretion of cytokines in endometriotic stromal cells [9–11], DNG is expected to be an effective treatment for endometriosis. Good efficacy and tolerability of DNG in patients with endometriosis have been demonstrated in an open, randomized European clinical trial as compared with norethisterone acetate [12]. In a questionnaire survey, approximately 55% of gynecologists in Japan have used DNG for patients with endometriosis since 2008.
Here, we describe the pharmacodynamics, pharmacokinetics, therapeutic efficacy and tolerability of DNG, as well as Japanese experience of using this drug on the market for the first time in the world.
Development of a new progestin: dienogest
Progestins are largely derived from two parental structures, the 19-nortestosterone derivatives (e.g., norethisterone, levonorgestrel, desogestrel, gestodene and norgestimate) and the progesterone derivatives. Although 19-nortestosterone derivatives have strong oral activity and are widely used as a component of oral contraceptives, these compounds tend to have strong androgenic effects. As the androgenic activity causes various metabolic side effects, the use of androgenic progestins for endometriosis in effective doses for long periods of time is limited. DNG is structurally related to 19-nortestos-terone derivatives but acts as an antiandrogen. Similar to the others in this family, DNG has the same 18-carbon nucleus as the estrane structure; however, it differs in structure from norethisterone by having a cyanomethyl group instead of an ethinyl group at C-17 and by the addition of a double bond between C-9 and C-10 (Figure 1) [13]. With these modifications, dienogest has strong endometrial activity with antiandrogenic profile by oral administration.

Dienogest.
Pharmacodynamic profile
Steroid hormone receptor binding & activation
In vitro, progesterone receptor (PR)-binding affinity is higher for DNG than for progesterone. DNG exhibits low binding affinity to the androgen receptor (AR). Affinity of DNG for estrogen receptors (ERs), glucocorticoid (GR) and miner-alocorticoid receptors (MRs) are negligible [14].
Using transactivation assays, agonistic/antagonistic activity of DNG on human PR, AR, GR, MR, ER-α or ER-β was measured. As shown in Table 1, DNG activates PRs (EC50 = 3.4 or 10.5 nmol/l). DNG has no agonistic activity on ARs but has antagonistic activity (EC50 = 420.6 or 775.0 nmol/l). It does not have any agonistic nor antagonistic action on GR and MR (3000 nmol/l). DNG does not activate either ER-α or ER-β (3000 nmol/l) (Table 1) [15].
Steroid hormone-receptor activation by dienogest.
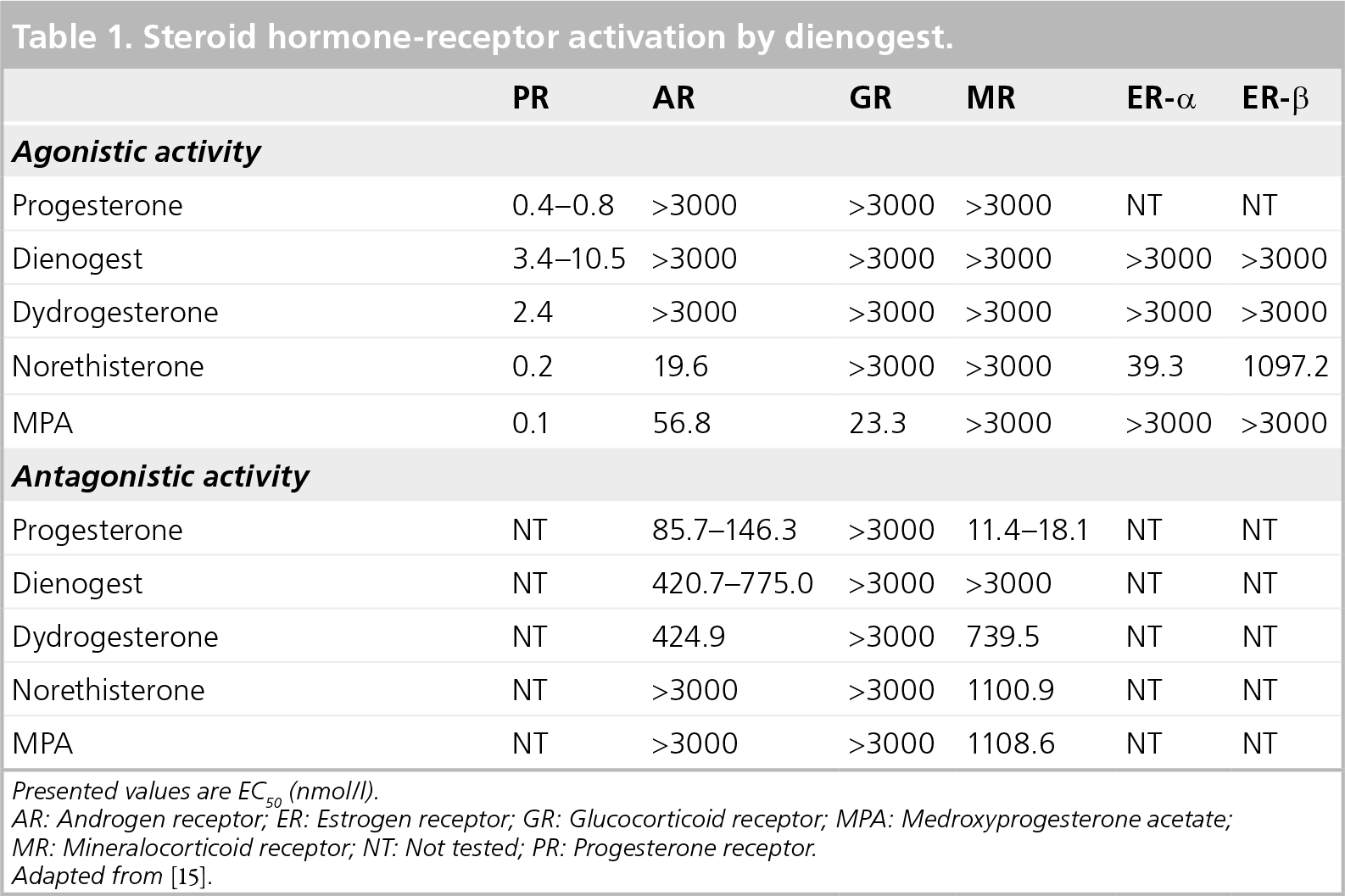
Presented values are EC50 (nmol/l).
AR: Androgen receptor; ER: Estrogen receptor; GR: Glucocorticoid receptor; MPA: Medroxyprogesterone acetate
MR: Mineralocorticoid receptor; NT: Not tested; PR: Progesterone receptor.
Adapted from [15].
Hormonal bioassay
When measured by secretory transformation of estrogen-primed endometrium in immature rabbits (Clauberg/McPhail assay), DNG has high oral progestational activity. The 50% effective dose for oral DNG was 0.11 mg/kg [16]. In postmenopausal women receiving oral ethinyl estradiol 50 μg/day, an oral DNG dose of 6.3 mg given over 14 days (0.45 mg/day) was required for complete secretory transformation of the endometrium according to the Kaufmann assay [16]. Thus, DNG has strong oral progestational activity in rabbit and human. This characteristic is attributable to its pharmacokinetic profiles and steroid hormonal specificity [15]. DNG has no androgenic activity but has antiandrogenic activity as demonstrated in an in vitro study. Ventral prostate weight in gonadectomized immature male rats (Hershberger assay) was not significantly increased by treatment with oral DNG doses less than 100 mg/kg/day or subcutaneous doses of 10 mg/day or 62.5 mg over 7 days. DNG had antiandrogenic activity that was approximately 40% of that of cyproterone acetate in the modified Hershberger assay in immature gonadectomized male rats treated with testosterone [17]. DNG had no mineralocorticoid activity or glucocorticoid activity showing no effect on urinary electrolyte excretion and glycogen deposition in adrenoectomized rats [18].
Effect on ovarian function
A dose-finding study indicated that a minimum oral DNG dose of 1 mg/day is required for inhibition of ovulation in cyclic women. During treatment with oral DNG 2 mg/day in women with regular menstrual cycle, serum progesterone levels were reduced to anovulatory levels; however, serum levels of LH and FSH were not greatly affected [19].
Studies by Schleussner demonstrated that the activity of DNG is likely to be associated with its strong peripheral effects on the ovary. DNG 2 mg/day was administered to women with a regular menstrual cycle daily over 21 days. The concentration of progesterone in the blood serum, the mean LH pulse frequency and the mean concentrations of FSH in the serum were determined, and in parallel, the sizes of ovarian follicles were measured by ultrasonography. Although DNG clearly induced anovulation as judged by serum progesterone levels, it did not inhibit the secretion of LH and its pulsatile secretion, or FSH [20]. Therefore, the effect on contraception is considered to be imperfect. In Japan, DNG is not permitted for contraception use. As shown by ultrasonography, the size of ovarian follicle-like structures on DNG was not greater than 10 mm. From these data, it is likely that DNG inhibits folliculogenesis by its peripheral effect [16,21].
To examine the underlying mechanism of the antifolliculogenic effect of DNG, female cynomolgus monkeys were treated with a single oral dose of 0.1 mg/kg DNG on day 7 of the menstrual cycle. Plasma FSH, estradiol and progesterone levels were measured up to 15 days after dosing. In an additional experiment, ovaries were excised 24 h after dosing for histological examinations. As a result, DNG induced a decrease in plasma E2 levels by 24 h after dosing, while the drug did not decrease FSH levels prior to E2 decline. After decline of E2 levels, the low level of E2 was sustained for more than 10 days after dosing and it is thought that a single oral dose of DNG induced atresia of the dominant follicle. In the histological examination, two out of three animals demonstrated a decline in E2 level. The ovarian dominant follicles from these animals showed apoptotic changes in granulosa cells with scattered aromatase expression by 24 h after dosing. These results indicate that the induction of atresia of the ovarian dominant follicle exclusively by direct action would be a possible mechanism of DNG in inhibition of plasma E2 levels [21].
Direct action on endometriotic cells in vitro
Direct actions of DNG on endometriotic cells are reported in recent studies. Using endomet-riotic stromal cells from ovarian ‘chocolate’ cyst, Horie et al. demonstrated that DNG (10−7 mol/l) attenuates the expression of IL-8 by reducing TNF-α-induced NF-κB activation in endo-metriotic stromal cells, suggesting a possible molecular mechanism of hormone therapy for controlling the growth of endometriosis and inflammation [11]. Fu et al. examined the effects of DNG on the proliferation of the cultured endometriotic stromal cells. DNG at concentrations of 10−7 mol/l and 10−6 mol/l significantly inhibited 5-bromo-2′-deoxyuridine (BrdU) incorporation into DNA at 24 and 48 h. DNG significantly increased the cells in G0/G1 phase and reduced the cells in S phase and G2/M phase in 24 and 48 h. These results indicate that DNG can inhibit the proliferation of the endometriotic stromal cells with G(0)/G(1) arrest, suggesting a possible direct effect of DNG in the treatment of endometriosis [22].
It is suggested that ectopic endometrial stromal cells have an enhanced contractile profile that may contribute to the pathological condition of endometriosis. In vitro decidualization inhibited the contractility of eutopic and ectopic endometrial stromal cells. By decidualization, DNG can attenuate the contractility of these cells. Reduced contractility may be one of the action mechanisms by which oral contraceptives and progestins ameliorate endometriosis [23].
Other actions on nonreproductive organs
Dienogest at oral doses of 0.01–0.1 mg/kg/day, significantly suppressed the 17 β-estradiol benzoate (E2)-dependent tumor growth of HEC-88neu cells, which were unresponsive to known progestins such as medroxyprogesterone acetate (MPA; 100 mg/kg/day, administered orally) and norethisterone (NES; 100 mg/kg/day, administered orally). In addition, DNG suppressed the E2-dependent tumor growth of both Ishikawa and MCF-7 cells [24].
The inhibitory effect on angiogenesis by DNG is reported by Nakamura et al. DNG has antag-onistic activity for angiogenesis. Topical DNG treatment in chicken egg dose-dependently inhibited embryonic angiogenesis, the 50% inhibitory dose (ID[50]) value being 6.4 nmol/egg. Oral administration of DNG (1 mg/kg/day) for 5 consecutive days significantly suppressed angio-genesis induced by S-180 mouse tumor cells in the mouse dorsal air sac assay [25].
Dienogest has less effect on the expression of adhesion molecules by endothelial cells in comparison with other progestins [26]. Human vascular endothelial cells were stimulated by IL-1 β for 24 h with or without various steroids, and then the cell-surface expression of intercellular adhesion molecule-1 and vascular cell adhesion molecule-1 was semiquantified. The addition of progesterone (10−10-10−8 mol/l) or DNG (10−10-10−8 mol/l) did not affect IL-1β-stimulated ICAM-1 or VCAM-1 expression, while MPA, norethisterone acetate and levonorgestrel (10−10-10−8 mol/l) dose-dependently increased cell adhesion molecules. These results suggest that DNG, unlike other synthetic progestins, lacks stimulation of cell-adhesion molecules.
Pharmacokinetic profile
A Phase I study using healthy male volunteers demonstrated that oral bioavailability of DNG was approximately 90% [27]. In female volunteers who received single oral doses of DNG, the mean maximum serum concentrations were 27.5 ng/ml with 1 mg, 53.9 ng/ml with 2 mg, 101.1 ng/ml with 4 mg, and 212 ng/ml with 8 mg. Mean values for AUC306, 577, 1153 and 2292 ng/ml/h, for 1, 2, 4 and 8 mg of doses, respectively. Thus, the pharmacokinetics of oral DNG are linear [28]. Maximum serum DNG concentrations were reached within approximately 2 h.
The free nonprotein-bound concentration of DNG in plasma after oral administration is almost 10% of the total concentration of DNG. This biologically active fraction is relatively high compared with that of other progestins (0.5–4%). The remaining 90% of compound is bound to albumin (
Comparison of pharmacokinetic parameters of progestins.
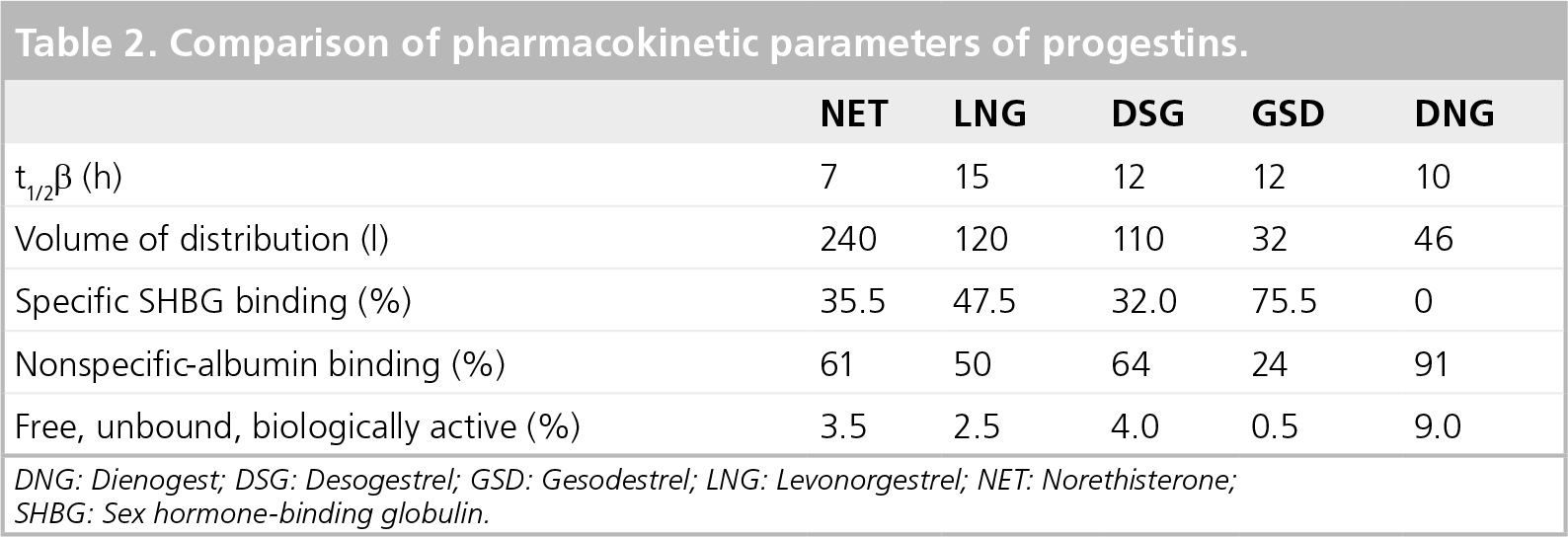
DNG: Dienogest; DSG: Desogestrel; GSD: Gesodestrel; LNG: Levonorgestrel; NET: Norethisterone; SHBG: Sex hormone-binding globulin.
Therapeutic efficacy
Pilot study in Germany
The effect of several doses of DNG on endometriosis was investigated in an open, controlled, randomized, clinical trial conducted in nine centers in Germany [12]. A total of 68 patients (mean age: 30 years) with histologically confirmed endometriosis (stage I–III according to revised American Fertility Society [r-AFS] score) were enrolled in the study. The patients were randomly allocated to be treated with DNG 1, 2 or 4 mg once-daily for 24 weeks. The primary outcome was changes in r-AFS scores evaluated by laparoscopy before and after the treatment. Secondary end points included typical pain symptoms in endometriosis, toler-ability, cycle control and laboratory parameters. The 2-mg group consisted of 29 patients and the 4-mg group of 35 patients. Only four patients were included in the 1-mg group and recruitment was terminated owing to bleeding in this group. Irregular bleeding occurred in 55.2% of the patients treated with DNG 2 mg. Bleeding occurred as spotting progressively with duration of treatment, with the rate of amenorrhea reaching as high as approximately 35% at the end of treatment.
After 24 weeks of DNG treatment with either 2 or 4 mg, there was a significant decrease in the mean r-AFS scores. According to this change in r-AFS points there was an improvement of 68% in the DNG 2-mg group, and of 57% in the DNG 4-mg group. Dysmenorrhea, premenstrual pain, dyspareunia and diffuse pelvic pain almost disappeared in both dose groups. In 90.9% of patients treated with DNG, signs and symptoms of endometriosis had improved or disappeared. Treatment-induced reduction in r-AFS scores by DNG appears equally or even more effective than other medical agents, such as danazol and GnRH agonists (reported to be 33.8–59.1%) [12]. There was no significant difference in improvement of r-AFS scores and pain symptoms between the DNG 2- and 4- mg groups.
High-dose pilot study in Europe
In an open, uncontrolled study, 21 women aged 18–52 years with laparoscopically and histologically proven endometriosis (r-AFS stage I– IV) were treated with DNG 20 mg/day for 24 weeks [29]. Treatment efficacy was analyzed objectively by second-look laparoscopy, and serum hormone measurements and evaluation of endometriosis-related symptom was performed and side effects were recorded. The baseline values of the total score determined by laparoscopy were between two and 106 (stage I–IV). By the end of the study, the mean total score had fallen by 59% from its baseline value. Both total score and AFS stage improved in 19 patients, with marked reduction (by at least 50%) in 15 and cure in the four. The proportion of subjects who reported key symptoms of endometriosis, such as dyspareunia, painful defecation, pelvic pain and dysmenorrhea premenstrual pain were significantly decreased after treatment with DNG.
Treatment with DNG terminated regular men-strual bleeding, but DNG increased the duration of bleeding. During treatment, 15 women reported spotting, which lasted between 2 and 72 days and ceased spontaneously. No subject reported unacceptable bleeding and five experienced no vaginal bleeding throughout the study.
Comparison between DNG & triptorelin (GnRH agonist)
This is a Phase III, multicenter, open randomized trial of patients who have stage II–IV endometriosis according to r-AFS classification [30]. Patients were given 16 weeks of treatment with DNG, 1 mg tablet twice-daily; or with triptorelin 3.75 mg intramuscular injection every 4 weeks. The main outcome of the study is a change in the patient's r-AFS score at the post-treatment laparoscopy. After exclusion owing to protocol deviations, 59 patients were allocated to the DNG group and 61 to the triptorelin group. The difference between the changes in the r-AFS score observed at second laparoscopy between the two groups after 4 months of treatment is not statistically significant. The authors concluded that DNG is as effective as triptorelin for consolidation therapy after surgery for the treatment of endometriosis. The safety profile of DNG differed from triptorelin. With regard to clinical safety, the symptoms in GnRH agonist-treated patients mainly involved the autonomic nervous system. The climacteric syndrome is usual in GnRH agonist treatment. Hot flashes were reported in 61% of women treated with GnRH agonist, compared with 11% of women treated with progestogen. Vaginal bleeding was the most common complication observed with DNG (61.6% of cases in comparison to 25.4% of those treated with the GnRH agonist).
Phase II study in Japan
A randomized, double-blind, multicenter, parallel study was designed to confirm the dose– response relationship in efficacy and safety of DNG in patients with endometriosis in Japan. In this study, 187 patients diagnosed with endometriosis were enrolled and randomized to three groups, and were administered oral DNG 1, 2 or 4 mg/day for 24 weeks. The primary end point for efficacy was global efficacy, which was an overall integrated assessment of improvements in seven endometriosis symptoms and signs (lower abdominal pain, lumbar pain, defecation pain, dyspareunia, pain during internal examination, induration in the pouch of Douglas and limited uterine mobility) at the end of treatment. The primary end point for safety was global safety, which was assessed with the incidence, severity, outcome of adverse events, laboratory parameters and genital bleeding episodes. In addition, serum estradiol levels were monitored.
The proportion of patients assessed who improved in global efficacy was 63.8, 66.7 or 73.2% with 1, 2 or 4 mg/day treatment, respectively; no statistically significant dose– response relationship was found. The proportion of patients assessed as tolerable in global safety was 85.2, 95.0 or 82.3%, respectively; furthermore, no significance was found. The most common adverse event was genital bleeding, which was observed at almost the same frequency and severity by the three groups; however, the bleeding was well tolerated in all the groups. Serum estradiol levels were not changed in the 1 mg/day group, but decreased significantly in the 2 and 4 mg/day groups. In addition, the mean estradiol level in the 2 mg/day group (37.4 pg/ml) was within a recommended level (30–50 pg/ml) of the ‘therapeutic window’ theory for efficacy and safety in the medical treatment of endometriosis, whereas it was below the window in the 4 mg/day group (26.2 pg/ml). All DNG doses (1, 2 or 4 mg/day) showed high efficacy and tolerability. DNG at a dose of 2 mg/day would be suitable for the treatment of endometriosis because the global efficacy, tolerability and safety of DNG were assessed.
Phase III study in Japan
A randomized, double-blind, multicenter, controlled trial was performed to compare the efficacy and safety of DNG with intranasal buserelin acetate (BA) in patients with endometriosis as a Phase III study in 24 study centers in Japan [31]. A total of 271 patients with endometriosis were included in the study and subjected to either DNG (2 mg/day, orally) or BA (900 μg/day, intranasally) for 24 weeks of treatment. The pre-to post-treatment changes in the scores of five subjective symptoms during nonmenstruation (lower abdominal pain, lumbago, defecation pain, dyspareunia and pain on internal examination) and two objective findings (induration in the pouch of Douglas and limited uterine mobility) were main outcome measures. DNG reduced the scores of all symptoms and findings at the end of treatment, and the mean changes in the scores of all symptoms and findings except induration in the pouch of Douglas were comparable with those obtained with BA. The two groups were similar in terms of the changes in the VAS for lower abdominal pain and lumbago from baseline to the end of treatment. The mean reduction ± standard deviation (mm) from baseline to the end of treatment in the DNG group was −30.2 ± 31.8 for lower abdominal pain and −15.7 ± 28.7 for lumbago, whereas the corresponding values for the BA group were −27.3 ± 33.8 and −17.3 ± 24.8. Compared with BA, DNG was associated with more frequent irregular genital bleeding (DNG: 95% vs BA: 67%) and with fewer hot flushes. The reduction in BMD during DNG treatment was significantly lower than that during BA treatment. The authors concluded that DNG is as effective as intranasal BA in alleviating endometriosis, and causes less BMD loss (DNG: −1.0 ± 2.3% vs BA: −2.6 ± 2.3%; the percent change from baseline to the end of treatment).
Tolerability
In a Japanese Phase III study, the incidences of adverse events and adverse drug reactions (ADRs) were 100% (129/129) and 100% (129/129), respectively, in the DNG group and 96% (121/126) and 93% (117/126), respectively, in the BA group. Serious adverse events occurred in three (2.3%) of the patients in the DNG group and in one (0.8%) of the patients in the BA group. Of these cases, one in each group was considered as treatment related (DNG: peritonitis; BA: bleeding in ovarian cyst). The peritonitis in the DNG group resolved post-treatment, whereas the bleeding in ovarian cysts resolved during the treatment period with BA.
The most frequent ADRs in the DNG group and the BA group were genital bleeding (122/129 [95%] vs 85/126 [67%], respectively), hot flushes (64/129 [50%] vs 85/126 [67%]) and headache (32/129 [25%] vs 43/126 [34%]). More patients reported genital bleeding, and fewer patients reported hot flushes, in the DNG group. None of the patients in either group discontinued treatment owing to genital bleeding or headache, and one patient in the DNG group and two patients in the BA group discontinued treatment owing to hot flushes. Most of the cases of genital bleeding occurring in the DNG group were spotting or breakthrough bleeding, which decreased with continued treatment and resolved either during treatment or after the end of treatment.
Of 87 patients, 41 in the DNG group and 46 in the BA group except one case with a concomitant user of calcium/vitamin D supplement, were eligible for analysis of BMD. BMD (mean ± SD) at baseline was 1.04 ± 0.086 g/cm2 in the DNG group and 1.03 ± 0.093 g/cm2 in the BA group. The percent change from baseline to the end of treatment in the BMD was −1.0 ± 2.3% in the DNG group and −2.6 ± 2.3% in the BA group, and there was a statistically significant difference (p = 0.0030) between the two treatment groups.
The mean serum concentrations of estradiol at baseline and at treatment week 16 were 86 ± 60 pg/ml and 38 ± 56 pg/ml, respectively, in the DNG group; whereas the corresponding values in the BA group were 87 ± 61 pg/ml and 21 ± 38 pg/ml. The mean serum concentrations of CA125 at baseline and at the end of the treatment were 65.5 IU/ml and 41.5 IU/ml, respectively, in the DNG group; whereas the corresponding values were 58.3 IU/ml and 28.6 IU/ml in the BA group [31]. Regidor and Regidor reported data regarding other gestagens. GnRH agonists was more effective than gestagens (lynestrenol) in the suppression of serum estradiol level after 6 months of treatment (mean: 27.7 ± 9.3 pg/ml vs 42.6 ± 59.3 pg/ml) [Regidor & Regidor, Unpublished Data]. Owing to the limited treatment of 6 months' duration of GnRH agonist, gestagens might be used as second-line drugs for long-term and continuous treatment in the management of endometriosis to maintain the primary beneficial effect of GnRH agonist treatment.
1-year experience in Japan
Dienogest 1 mg, twice-daily was standard pre-scription in Japan. DNG treatment was favorably accepted by Japanese patients and gynecologists. Almost 20,000 patients were administered DNG after 14 months since its appearance on the Japanese market. It is worth noting that several severe bleeding cases were reported. Eight endometriosis patients associated with adenomyosis developed severe anemia owing to continued genital bleeding and were either given blood transfusions or underwent hysterectomy. Although, these cases were rare, gynecologists should be aware of this problem and carefully check bleeding status and severity of anemia.
Executive summary
Dienogest (DNG) is a 19-nortestosterone derivative that has strong endometrial activity with antiandrogenic profile by oral administration.
DNG has moderate affinity to the progesterone receptor. DNG shows low binding to the androgen receptor and almost negligible to the estrogen receptor, glucocorticoid receptor and mineralcorticoid receptor.
DNG has strong oral progestational activity but antiandrogenic activity.
An oral DNG dose of 1 mg/day is required for inhibition of ovulation in cyclic women.
DNG has an inhibitory effect on growth and cytokine production of endometriotic cells.
Maximum serum DNG concentrations were reached within approximately 2 h.
After 24 weeks of DNG treatment with 2 or 4 mg, there was a significant decrease in the mean revised American Fertility Society (r-AFS) scores.
A high-dose pilot study (DNG 20 mg/day) demonstrated that 24 weeks of treatment reduced total r-AFS score to 59% from baseline.
DNG is as effective as triptorelin (gonadotropin-releasing hormone agonist) for consolidation therapy after surgery for the treatment of endometriosis.
DNG is as effective as intranasal buserelin acetate for the relief of pain symptoms associated with endometriosis.
The most frequent adverse events were genital bleeding but none of the patients in the DNG group discontinued treatment.
Careful observation for anemia should be provided for continued bleeding in patients with adenomyosis.
Future perspective
Dienogest has good oral bioavailability and is highly selective for progesterone receptors. Clinical studies and Japanese experience after going on the market suggest that DNG is a promising drug as a new oral progestin for the treatment of endometriosis.
Footnotes
Tasuku Harada has received consulting fees from Mochida Pharmaceutical Co. Ltd. The authors have no other relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript apart from those disclosed.
No writing assistance was utilized in the production of this manuscript.
