Abstract
The importance of mind–body health relationships has been recognized for decades, but only recently has the wider medical and cardiovascular community become engaged in understanding and addressing the complex, bidirectional risk relationship between cardiovascular disease (CVD) and depression. Furthermore, it has become increasingly clear that there are incompletely understood sex differences in incidence and outcomes for both conditions that should guide treatment and future research efforts. This review will explore the role of depression in women as a risk factor for incident CVD, its impact on women already suffering from CVD, proposed psychobiologic mechanisms and links, and the implications of sex differences on diagnosis and treatment.
Keywords
Cardiovascular disease (CVD) and depression are two of the most prevalent health problems afflicting women, not only in the USA, but worldwide. Stroke and heart disease kill more than half a million women each year, exceeding CVD deaths in men. Depression is second only to CVD as a cause of morbidity and premature mortality in industrialized countries [1], and almost twice as common in women, with 9.5% of women versus 5.3% of men in the USA suffering at least one major depressive episode each year [2]. Although CVD and depression commonly present as comorbid conditions, numerous epidemiological and observational studies have shown depression to be an independent risk factor for the development and progression of CVD and an important prognostic factor in women with heart disease. Depressed women are more likely than those without depression to develop CVD, die of or have recurrent cadiac events [3–5]. Conversely, CVD is a risk factor for both new and recurrent depression. Women suffering from both CVD and depression have more physical and social dysfunction, are less likely to attend cardiac rehabilitation, and have lower health-related quality of life, than women with CVD who do not have depression [6–8].
A major barrier to better understanding of the role of depression in women with heart disease is the lack of recognition of the condition. While most studies suggest that depression or depressive symptoms are present in at least 20–25% of hospitalized cardiac patients [9–14], and in one of three patients with heart failure [15], fewer than 15% of depressed patients hospitalized with myocardial infarction (MI) are identified [16,17], and even fewer are treated.
Depression & cardiovascular outcomes
Depression not only predicts development of overt CVD but is also associated with more rapid development of CVD risk factors. Women with major depression compared to those without experience faster progression across both the early and later stages of CVD [18]. In a study of over 700,000 members of a large health plan (53% women), a diagnosis of depression in the previous 2 years in both women and men was associated with a 1.5-2.6-times increased risk of developing hypertension or dyslipidemia compared to those without depression. Individuals who already had one or both of these risk factors had similar increased risk for development of additional CVD risks factors and progression to overt coronary artery disease and heart failure [18].
The presence of major depressive disorder (MDD) has been demonstrated to be a stronger predictor of adverse CVD outcomes than many more commonly acknowledged risk factors such as prior MI, heart failure, diabetes and left ventricular dysfunction [19,20]. In 93,676 women participating in the Women's Health Initiative Observational Study, of whom 15.8% met criteria for depression, depression was associated with elevated adjusted risks of CVD death (RR: 1.50; 95% CI: 1.10–12.03) and all-cause mortality (RR: 1.32; 95% CI: 1.16–11.52) that was unaffected by depression treatment [21]. The risk of recurrent CVD events, death and readmission are increased two- to five-times in patients with depression, depending upon the study population and how depression is defined [14,21–29]. Frasure-Smith and colleagues were among the first to report this association and found depression to be a significant predictor of mortality (p < 0.001) in 222 patients (22% women) 6 months after MI [14]. Depression remained a significant predictor of mortality (p = 0.01) even after multivariate risk adjustment in this predominately male study population. The risk associated with early depression that persisted was significantly related to 18-month cardiac mortality, even after controlling for other predictors (previous MI, Killip class and frequency of ventricular ectopy; p = 0.003) [23].
Even minor depressive symptoms are associated with a 50 to 100% increase in CVD risk in a ‘dose-dependent’ manner as measured by standard depression assessment tools such as the Beck Depression Inventory (BDI) (Figure 1) [30–32]. The highest risks associated with depression occur in patients with heart failure [33] and after coronary artery bypass (CAB) surgery [34,35]. Depressive symptoms are present in up to 50% of patients recently hospitalized for CAB surgery or heart failure, with women being disproportionately affected [36–38]. The presence of depression after CAB is associated with more pain, failure to return to activities, longer hospitalization, rehospitalization, repeat procedures, disease progression, and higher short- and long-term mortality [35,39].

Severity of depression and risk for mortality after myocardial infarction.
Impact on women
While there is little evidence that depression imparts greater absolute CVD risk to women than men, women are more often affected and possibly more disadvantaged when it comes to depression. Women are at twice the risk of developing depression compared to men [40–42]. In addition, in individuals with suspected coronary heart disease (CHD), the combination of depression and being female was associated with the lowest level of health-related quality of life in a study of 185 patients hospitalized with suspected CHD [43]. In a recent Canadian study of the association of heart disease and depression, depression was associated with incident heart disease in women (HR: 1.7; 95% CI: 1.1–2.5), but not men [44]. Similarly, while elevated BDI scores in a Finnish national risk survey (FINRISK Study) predicted all-cause mortality in both men and women without CVD at baseline, incident CVD events were associated with depression only in women (RR: 1.84; 95% CI: 1.06–03.20) [45]. Women with CVD are also among several patient populations whose psychological conditions are more likely to be missed by healthcare providers [46].
Compounding these risks, for virtually every CVD condition, women have higher mortality rates and poorer outcomes compared with men. While mortality from CVD has significantly declined over the past three decades, women have not experienced the same reductions in death and disability as have men [47]. There remains a significant sex-related mortality gap that is generally thought to be due to a combination of sex-based physiologic differences and disparities in care. The higher prevalence of depression in women may well be an additional factor accounting for this excess mortality. For instance, younger women with MI have up to twice the death rate compared with same-aged men [48] and also have strikingly higher depression rates. In a population of 2498 MI patients (33% women), Mallik and colleagues found that depression occurred in twice as many women (40%) than in men (22%) under 60 years of age, whereas older women and men had lower and more similar rates (22 and 15%, respectively) (Figure 2) [49]. Further analysis of 2411 individuals (807 women) from the same study population found that depressive symptoms were more prevalent in women overall (29 vs 18.8%, p < 0.001), predicted worse CVD outcomes to a similar degree in both men and women, and in multivariate analysis accounted for a measurable, but modest, proportion of women's poorer outcomes compared with men [50].
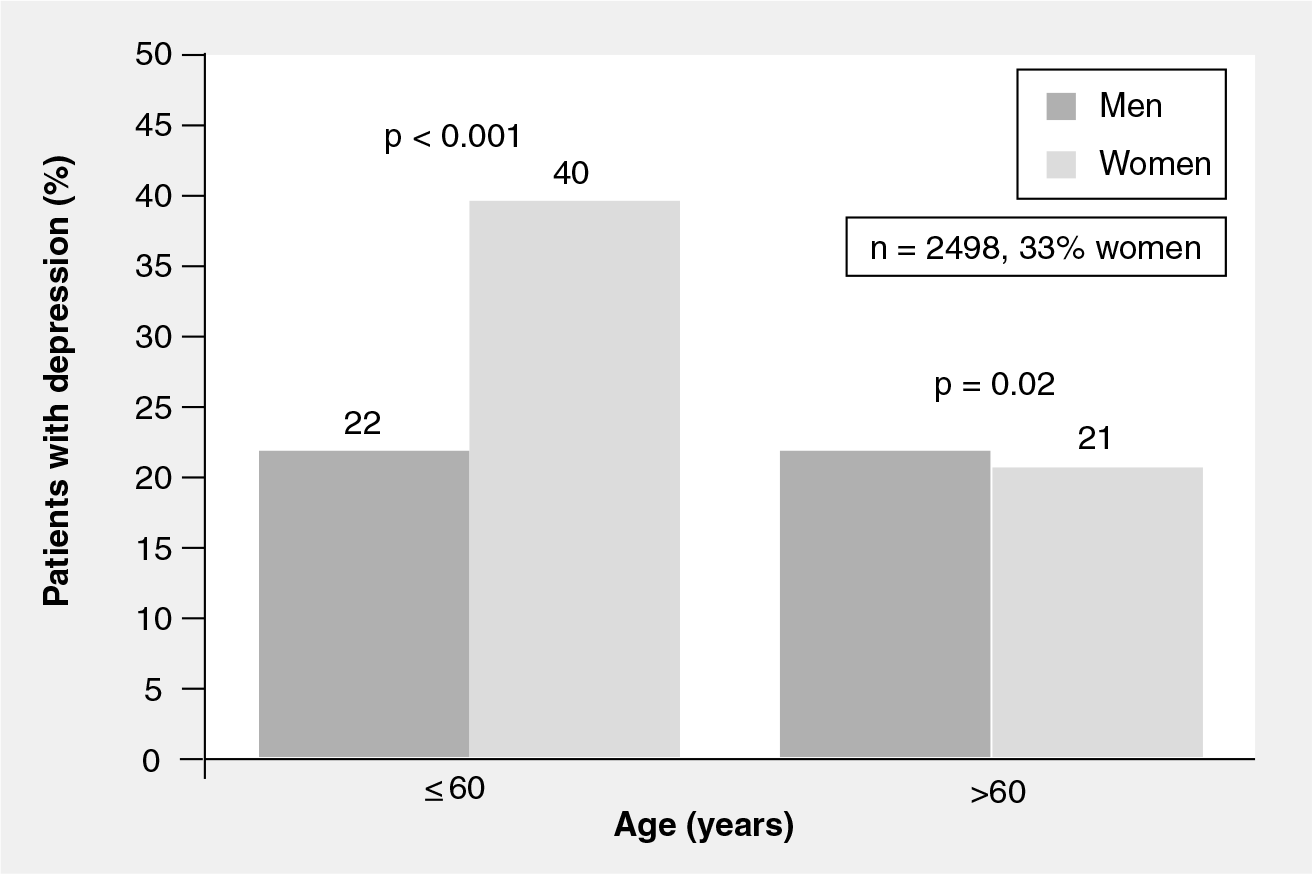
Effect of age and gender on depression rates after myocardial infarction.
After CAB surgery, depression is more common in women and younger patients and is associated with higher rates of graft disease progression [39]. Female outpatients with heart failure compared with males have significantly higher rates of depression (64 vs 44%; p = 0.048), somatic symptoms and functional impairment [38]. In women, but not men, depressive symptoms are associated with increased risk of metabolic syndrome, and the presence of metabolic syndrome parameters in adolescent girls is associated with adult depression [51]. Finally, depression in women with CVD is expensive. Investigators from the Women's Ischemic Symptom Evaluation (WISE) study found that depressed women had adjusted annual cardiovascular costs that were US $ 1550–3300 higher and 5-year cardiovascular costs 15–53% higher than nondepressed women (r = 0.08-0.12; p < 0.05), and concluded that improved management of depression in women with suspected myocardial ischemia could reduce medical costs [52].
Lack of data in women
There are significant challenges to fully understanding the CVD-depression link in women. First, the causal relationship between mood disorders and CVD events has not been fully determined for either men or women, and some investigators still maintain that depression is not a true risk factor but is simply an epiphenomenon or risk marker for adverse CVD events [53]. In addition, sex differences in epidemiology, pathophysiology, response to treatment and outcomes have been identified in many cardiovascular and mental health conditions, but women have been under-represented in cardiovascular clinical trials, including those investigating mental health conditions, and, therefore, these differences and their significance are incompletely understood. Even when included in adequate numbers, investigators frequently have not reported outcomes by sex, limiting application of findings to women [54]. The first sex-specific data regarding depression and CVD began to appear approximately a decade ago [19,24], but subsequent studies have only variably included women, typically making up less than 30% of study subjects. Only a minority have reported sex-specific findings.
Diagnostic challenges
Depression, particularly in women with CVD, is an often misdiagnosed illness [16,46]. The somatic symptoms that often accompany depression may be mistakenly attributed to the underlying medical condition or to treatment side effects, especially in women. This is compounded by the fact that cardiovascular healthcare providers typically have a low awareness of and little focus on mental health issues, and even when prompted to assess for depressed mood, do not do so with accuracy [55]. When an appropriate diagnosis is made, treatment is often not offered [46]. Ellis et al. found that over 40% of 490 patients (29% women) reported persistent depression after recent MI or acute coronary syndrome. However, only 10% were adequately treated with antidepressants and/or psychosocial support, and women were significantly more likely to be untreated or undertreated than men (p < 0.0001). Lack of treatment and undertreatment resulted in significantly lower mental health-related quality of life compared to adequately treated and nondepressed subjects [46].
This lack of recognition and undertreatment is likely multifactorial, and several potential reasons have been identified. Clearly, there continues to be a social stigma associated with depression by both women and healthcare providers that prevents some referrals for mental health care. Physicians may not address depression since they have not been adequately trained to recognize depressive symptoms and frequently cite time constraints as a barrier, particularly in the inpatient setting. Often the cardiac care team lacks experience in depression treatment modalities and follow-up or feels that mental health issues are either not in their ‘job description’ or scope of practice or assume the issues will be dealt with by other members of the healthcare team. Some physicians do not address depression because they feel that treatment is not warranted owing to the lack of data to prove that treatment of depression effectively reduces CVD risk, while ignoring the fact that treating depression relieves suffering and improves health-related quality of life. In order to optimally address the mental health needs of women who present with increasingly complex cardiovascular disease states, better integration and coordination of care by primary care providers, mental health specialists and cardiac rehabilitation staff will be necessary. This is especially important in light of the frequent coexistence of social isolation and other mental health disorders such as anxiety, adjustment disorder and exacerbation of preexisting psychiatric illness that may require additional or longer term management.
Causality, mechanisms & pathobiology
In the past, depression occurring around the time of MI or a diagnosis of heart failure was deemed a ‘normal’ response to a patient's underlying condition with no specific impact on outcomes [56]. We now know that depression is not a natural consequence of cardiac illness but instead is a complex psychobiologic condition that independently increases CVD risk. The more we discover about the complex interplay of depression and CVD risk, the more potential interactions, similarities and inter-relatedness are found, frequently leading investigators and clinicians to ask the ‘chicken and egg’ question as to which condition precedes the other. Direct, indirect and common causal pathways between depression and CVD have been proposed (Figure 3). The ‘direct’ hypothesis is prefaced upon the fact that there are many adverse physiologic changes associated with depression, such as elevated levels of inflammatory markers, enhanced platelet aggregation and susceptibility to cardiac arrhythmias that may lead to development of atherosclerosis and to CVD events. Indirectly, depression is associated with a number of maladaptive health behaviors, particularly tobacco use, sedentary lifestyle and several coronary risk factors that in turn can promote development of CHD. The ‘common pathway’ hypothesis is built upon observations that underlying risk factors and pathophysiology simultaneously influence the risk for both depression and CHD. At present, there is evidence to suggest that all of these relationships likely contribute to some extent (Figure 4), but the relative contributions of each pathway are less certain. It should be noted that the vast majority of the mechanistic studies do not report sex-specific results, and the summary that follows therefore reflects combined gender results unless otherwise noted.

Potential causal relationships between depression and CVD.
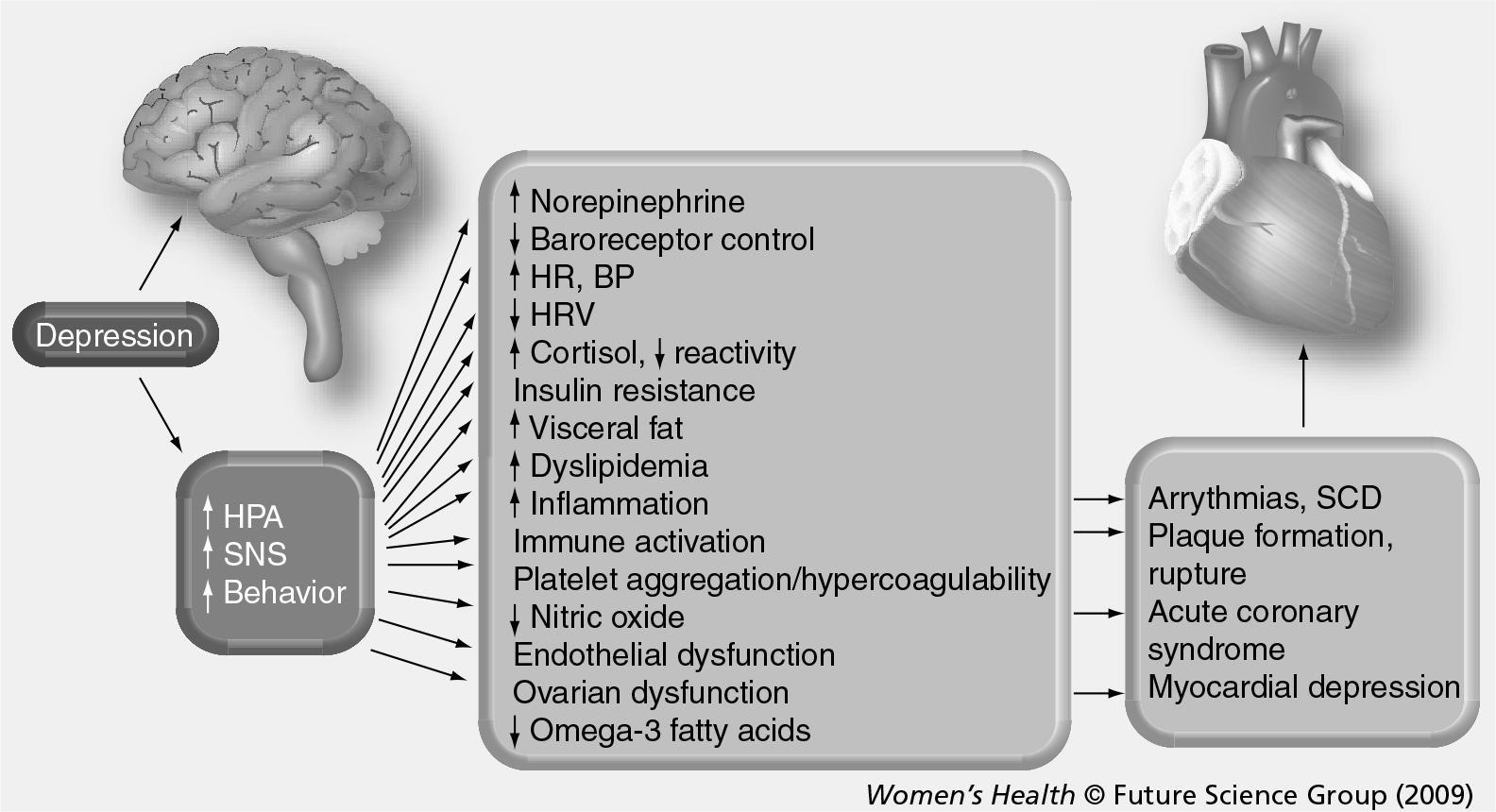
Depression and cardiovascular disease: potential psychobiologic interactions.
Autonomic imbalance
Depression is associated with autonomic imbalance and dysregulation and activation of the hypothalamic–pituitary–adrenocortical (HPA) axis [57]. Excessive sympathetic stimulation or diminished vagal stimulation or both are associated with higher rates of morbidity and death, and depressed patients have more activation of the HPA and sympathetic nervous system (SNS). These features consisting of high neurohormonal activation, with increased levels of norepinephrine, renin–angiotensin–aldosterone, arginine vasopressin and endothelin-1 are found in both depressed patients and those with decompensated heart failure [58]. High endogenous cortisol levels may accelerate the development of hypertension and atherosclerosis and result in endothelial vascular injury. Activation of the SNS leads to higher catecholamine levels, predisposing to vasoconstriction, elevated heart rate, platelet activation and reduced heart rate variability (HRV).
Heart rate variability describes the beat-to-beat variations in R–R intervals over time and is influenced by the balance and interplay between sympathetic and parasympathetic influences on the sinus node. Diminished HRV is one proposed common mechanism to explain the ventricular arrhythmias and excess cardiovascular mortality found in women with CVD and comorbid MDD [57,59,60]. A high degree of HRV is observed in normal hearts with good cardiac function, and HRV is significantly reduced in women with heart failure and severe CHD as well as in depression. The lowest HRV occurs in women who have both a cardiac condition and MDD [59–61]. A few small trials suggest that HRV may improve with selective serotonin-reuptake inhibitor (SSRI) therapy [62,63]. Prospective studies aimed at understanding the relationship between depression, CVD and these neurohormonal alterations, particularly in women, are virtually nonexistent and, therefore, the extent and impact of HPA/SNS dysregulation on these conditions is unknown.
Hypercoagulability
A prothrombotic state owing to enhanced platelet aggregation, elevated fibrinogen levels and coagulation abnormalities is frequently observed in depressive disorders and may play an etiologic role in the development or the progression of atherosclerosis via thrombus formation and subsequent myocardial ischemia. Patients with both depression and coronary artery disease have higher plasma levels of platelet factor IV, β-thromboglobulin and other markers of platelet activation than those with only one or neither condition [64–66]. Alterations in platelet serotonin-mediated function that occur in depressed patients and those with CVD have also been observed [67–69] and have generated considerable interest owing to the potential for the antidepressant class SSRIs to directly reduce thrombotic potential [70].
Endothelial & vascular effects
Endothelial dysfunction and increased arterial stiffness are hallmarks of atherosclerosis and have also been found in depressed women without CVD [71–73]. In otherwise healthy adolescent girls, endothelial function as assessed by digital tonometry was inversely related to higher BDI scores [74]. Similarly, in a population of 143 patients with CHD (44 women), women were more likely to be depressed (BDI > 10; p = 0.013), and higher depression scores in both men and women were associated with impaired flow mediated dilatation (FMD) while antidepressant use was associated with better FMD [75]. Nitric oxide, a vasodilator and marker of endothelial health, is reduced in depressed patients [76] and increased with citalopram treatment [77], suggesting that treatment of depression may play a role in improving vascular health.
Inflammation
The hypothesis that inflammation causes or is at least strongly associated with CVD has been studied and debated extensively [78,79]. Depression has been associated with higher levels of inflammatory markers including C-reactive protein (CRP), IL-1, IL-6 and TNF-α [79–82]. There have been a few small studies demonstrating a decline in inflammatory marker levels with successful treatment of depression [77,83,84]. If this is found to be a consistent effect, inflammation might become an appealing therapeutic target and marker of treatment success, especially for women who have higher baseline levels of several inflammatory markers, including CRP, compared with men. However, inflammation may only be a minor contributor to the risk imparted by depression. In the WISE study of approximately 900 women being evaluated for chest pain syndromes, investigators found that depressive symptoms and a history of depression were strongly predictive of the presence of CHD and CVD events. In this population, ‘definite’ depression (BDI > 10 and a history of depression) and ‘possible’ depression (either elevated BDI or depression history) was directly related to elevated levels of CRP and IL-6 compared with nondepressed women [80]. Depressed women had 70% higher CRP (p = 0.0008) and 25% higher IL-6 (p = 0.4) levels, and possible depression was associated with elevations of CRP and IL-6 of 30% (p = 0.02) and 28% (p = 0.01), respectively. Although both markers were independent risk factors for CVD outcomes, when added to a multivariate model these inflammatory markers were only minor contributors to the elevated CVD risk associated with depression.
Indirect causation, behavior & adherence
Recognition and treatment of major depression is crucial, especially for women, since depression is linked to maladaptive coping behaviors such as persistent smoking, poor self-care, social isolation, chronic life stress, poor adherence with medical therapy, inability to make desirable lifestyle changes, and low attendance at cardiac rehabilitation [59,85–94]. Ziegelstein et al. evaluated 204 post-MI patients (43% women) and found that those with mild-to-moderate depressive symptoms had poorer adherence to diet, exercise, smoking cessation, stress reduction and had less social interaction [92]. More severe depression was also associated with medication nonadherence. The Heart and Soul Study investigators explored this risk factor in their study population (18% women) and have found that depression was also associated with medication nonadherence (Figure 5) [90]. Furthermore, self-reported medication nonadherence was more common in women (p = 0.005) and associated with a greater than twofold increased risk of CHD death, MI or stroke during 3.9 years of follow-up. Nonadherence was as powerful a predictor of adverse cardiovascular events as diabetes and current smoking [95]. In the same study population, investigators found evidence to support treatment nonadherence, especially as it relates to participation in cardiac rehabilitation programs, and physical activity recommendations may prove to be the most powerful indirect influence on adverse CVD outcomes in women with depression. Depressive symptoms were associated with a 31% increased rate of adverse CVD events [96]. However, after controlling for behavioral factors, especially physical inactivity, the association between depression and adverse CVD events was almost completely attenuated, suggesting that most of the elevated risk of depression in CVD patients is mediated through behavioral factors that are theoretically modifiable with counseling, effective depression treatment, education and enhanced participation in cardiac rehabilitation. Effective treatment and remission of depression may result in improved medication adherence and conceivably could improve adherence to other medical recommendations.
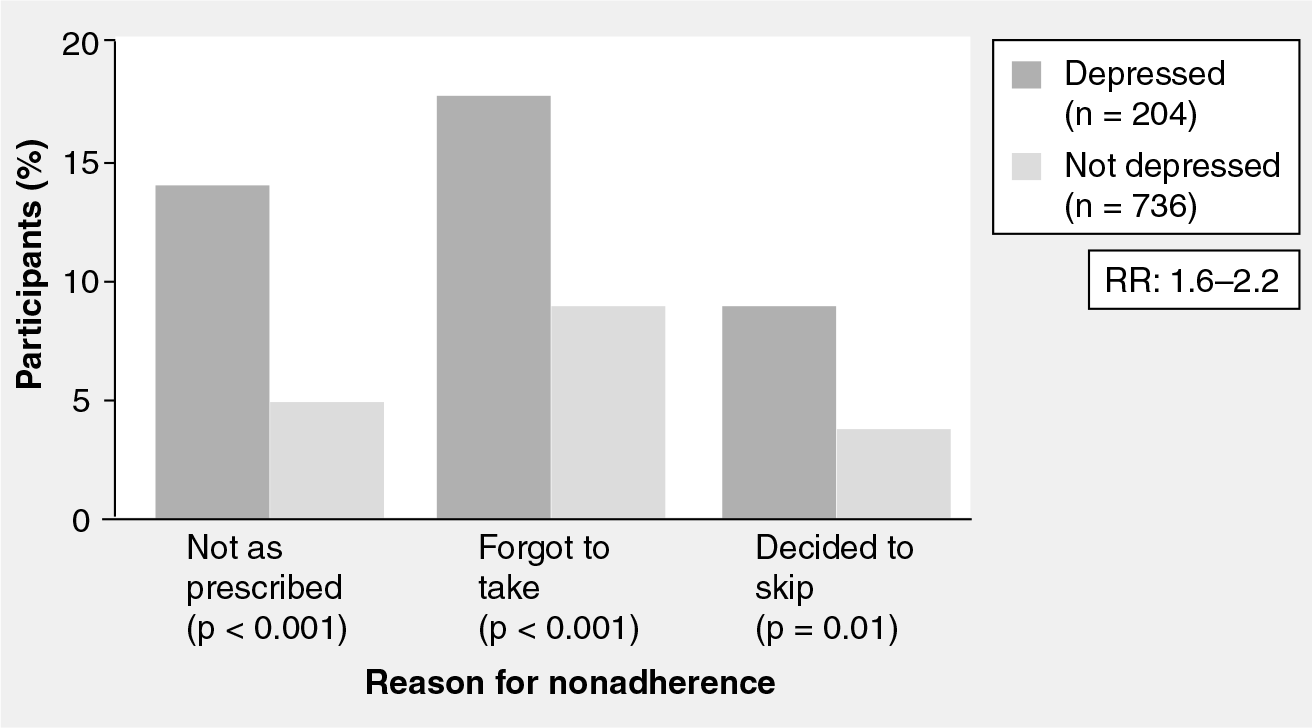
Medication nonadherence in women and men with coronary heart disease with and without depression.
Challenges in diagnosis & screening
Diagnosing depression in women with CVD can be challenging, despite clear diagnostic criteria (Box 1). Frequent overlap exists between the symptoms of acute or chronic CVD and those of depression: fatigue, poor appetite and sleep disturbances are common to both conditions. Although a full discussion of the impact of anxiety on women with CVD is beyond the scope of this review, anxiety is a frequent comorbid condition that can challenge both the diagnosis and treatment of depression, and independently increase CVD risk [97]. Clinicians must have a high index of suspicion for depression to be accurately identified in these high-risk women (Box 2). The American Heart Association recommends routine depression screening for women with CVD followed by appropriate treatment and referral [98,99], but the perfect screening tool for depression in women with CVD has not yet been identified. Several reasonable options are available that are easy to administer in a busy clinical practice, including the BDI and Patient Health Questionnaire (PHQ)-9 and −2 (Table 1) [100–102]. While diagnostic accuracy is imperfect with any tools, especially in medically ill populations, and there are few sex-specific data, unless formal screening is performed most depressive symptoms in women with CVD will be missed [55].
Depression screening with the Patient Health Questionnaire-9*.

Patient Health Questionnaire (PHQ)-2 utilizes the first two items of the PHQ-9 (anhedonia and depressed mood) with a total score range of 0 to 6. Some investigators have modified the PHQ-2 to prompt a yes or no response by asking “Over the last 2 weeks, have you been bothered by either of the following problems?” A positive response to either question requires follow-up.
Total score: 1–4 minimal depression; 5–9 mild depression; 10–14 moderate depression; 15–19 moderately severe depression; 20–27 severe depression.
Adapted from the Primary Care Evaluation of Mental Disorders Patient Health Questionnaire (PRIME-MD PHQ). PHQ-9 Copyright © Pfizer Inc. All rights reserved. Reproduced with permission from [100].
Diagnostic challenges are even greater in hospitalized patients where the combination of sedatives, analgesics, interrupted sleep, pain and disruption of routine may result in cognitive slowing that can be mistaken for depressed affect and result in elevated scores on BDI and other depression screening tools. For example, women with low energy, disturbed sleep, fatigue and problems with cognition related to their acute illness may have a high score on BDI or PHQ-9 owing to somatic symptomatology but yet not meet criteria for MDD. However, these individuals still represent a group with elevated CVD morbidity and mortality [17,103]. An analysis of two acute MI registries (46% women) found that somatic symptoms (e.g., fatigue and sleep disturbances) of depression compared to cognitive symptoms (e.g., sadness and anhedonia) were more strongly associated with poor outcomes, while cognitive symptoms were associated with better depression detection rates [17]. Findings were similar for women and men and overall; only 29% of depression cases were recognized. Several studies have shown that after adjustment for the severity of the medical condition, women with depression compared to depressed men [104], or women without depression report a higher number of somatic symptoms, including anginal frequency, emphasizing the impact depression can have on health-related quality of life [20,38,105]. Women's Ischemia Syndrome Evaluation (WISE) investigators examined the relationship between depression (BDI score > 17) and cardiac symptoms in 505 women being evaluated for suspected MI [106] and found that women with depressive symptoms had more cardiac and somatic symptoms compared to those without depression [106]. Depressed women also tend to suffer more and slightly different symptoms compared with depressed men, and differentiating between these cognitive and somatic symptoms of depression may have important implications for management [17,103]. Among the WISE population, the 21 BDI items were divided into somatic and cognitive/affective groupings and compared with subsequent CVD event rates over 5.8 years [103]. Even after controlling for disease severity, somatic (HR: 1.63; 95% CI: 1.28-22.08) but not cognitive/affective depressive symptoms (HR: 0.87, 95% CI: 0.68-61.11) were associated with CVD events. The results of these studies suggest that a more sophisticated approach to diagnostic and prognostic assessment of women with depressive symptoms may be needed and that the presence of certain symptoms may have different therapeutic implications.
Criteria for major depressive disorder*
Depressed mood
Loss of interest in most activities
Significant change in appetite or weight
Insomnia or hypersomnia
Psychomotor agitation or retardation
Fatigue or loss of energy
Feelings of worthlessness or inappropriate guilt
Diminished ability to think or concentrate
Recurrent thoughts of death or suicide
Must have five symptoms present over at least 2 weeks.
Minor depression = two to four symptoms.
Data taken from [138].
Features of depressive illness in women with cardiovascular disease.

Past history of major depressive illness
More symptoms than men
More frequent family history of depression
Hormonal milieu or shifts may impact symptoms
Seasonal mood changes more common
Profound and chronic fatigue or lack of energy
Hostility and irritability
High levels of stress and/or anxiety
Difficulty coping with change or loss
Rumination
Listlessness or apathy
Social withdrawal from family and friends
Helplessness
Poor hygiene
Nonadherence to recommended lifestyle modifications, medications and cardiac rehabilitation
Frequent emergency department or provider visits for complaints that have no physical cause after appropriate evaluation
Persistent anxiety and pessimism, unresponsive to reassurance and education
Failure to return to usual activities after an appropriate recovery period
Requests for extension of sick leave, disability
Treatment issues
Determining the optimal treatment for women with CVD and depression remains a challenge. With the strong association and potential causal relationship of depression on elevated CVD risk, it previously seemed reasonable to assume that treat-L ing depression would lead to improved outcomes in women with or at risk for CVD. Treatment I and/or remission of depression in women with: CVD favorably affects mood, quality of life and physiologic parameters that are dysregulated in depression [84,107,108]. However, many therapeutic interventions have had disappointing or ‘negative’ results, at least in terms of having an impact on CVD outcomes [109,110]. One potential explanation may be that most tested treatments only partially alleviated depressive symptoms, so we cannot entirely dismiss the possibility that more effective treatment might improve CVD prognosis. Another challenge is that women appear to respond differently than men to psychosocial interventions, at least as they have been applied in several of the largest intervention trials [110,111]. Finally, many trials suffer from small sample sizes, post hoc and subgroup analyses, inappropriate control groups, and a lack of sex-specific result reporting, which reduces the significance of the findings for women.
One of the first trials to recognize sex differences in outcomes of depression treatment in CVD patients was the Montreal Heart Attack Readjustment Trial (MHART) [111]. The intervention, based on results from a successful pilot study performed in a small group of men, used cognitive-behavioral therapy (CBT), telephone calls and home nursing visits in the first year after MI in 1376 patients (473 women) with psychological distress and/or depressive symptomatology. The overall trial did not find that supportive care had any impact on survival compared with usual care. Disturbingly, women in the intervention group had an unexpected trend toward higher rates of cardiac (p = 0.064) and all-cause mortality (p = 0.051), even after 5 years [112,113]. Further subgroup analysis found that ‘highly anxious’ men benefited from the supportive care provided (HR: 0.48; 95% CI: 0.26-20.89), while ‘highly anxious’ women were ‘harmed’ (HR: 1.76; 95% CI: 1.01-03.05; HR: 4.04 vs men), and that both men and women who tended to ‘repress’ their emotions and problems were adversely affected by treatment.
The Enhancing Recovery in Coronary Heart Disease (ENRICHD) study was the first randomized, controlled clinical trial to test whether intervening on depression with CBT soon after acute MI, supplemented with an SSRI antidepressant when indicated, reduced the risk of recurrent nonfatal infarction and death [110]. ENRICHD enrolled 2481 patients (1084 women) who had depression and/or low perceived social support. The intervention group received an individually tailored CBT-based intervention, initiated 2–3 weeks after MI and continued for a median of 11 sessions over 6 months. Patients with more severe depression or who were unresponsive to CBT after 5 weeks were eligible to receive an SSRI on a nonrandomized basis. There were modest improvements in psychological outcomes at six but not 30 months and no differences in CVD outcomes in the overall study group. It has been postulated that this may have been owing to the high level of unexpected SSRI use by control patients (20.6% control vs 28% intervention), and lower than expected levels of depression, morbidity and mortality in both groups. Sex and racial differences were again observed, with benefit observed in white men and a trend toward harm in women (Figure 6). A recently published reanalysis of ENRICHD found that after multiple adjustments there was a nonsignificant reduction in all-cause mortality and recurrent MI in women in the subgroup who participated in both individual and subsequent group training (31% of the treatment arm) that was not different from results in men [114].

Effect of the Enhancing Recovery in Coronary Heart Disease Patients (ENRICHD) intervention on risk of death or nonfatal myocardial infarction.
The results of MHART and ENRICHD challenge the appeal of using CBT and supportive care for post-MI depression, particularly when associated with anxiety, at least in the manner used in these studies. It was previously believed that psychological support approaches had inherent advantages over pharmacotherapy by focusing on both the depressive symptoms and those features of depression that may contribute to increased mortality (i.e., treatment nonadherence) and could do so without adverse side effects. In both of these studies, not only did women experience no benefit from CBT, but they did less well than those in control groups. While we should not entirely discount the efficacy of CBT in treating women with depression and CVD, further studies should explore the nuances of various treatment effects and be designed to address sex differences in response to different therapies.
One such variation was used in a prospective randomized trial aimed at improving outcomes in high-risk women after MI in which depression was not an inclusion criterion. The Stockholm Women's Intervention Trial for Coronary Heart Disease (SWITCHD) investigators [115] found that a psychosocial intervention program based on CBT principles, and emphasizing stress reduction and coping in 237 women (mean age 62 years), resulted in significantly improved survival. Women-only support groups met over a 1-year period and were compared to usual care. Over a mean of 7 years after entering the study, women in usual care had a mortality rate of 20%, whereas those in the psychosocial intervention had a mortality rate of 7% that remained significant after adjustment for baseline factors (p = 0.009). While not designed to treat depression, the intervention was specifically devised to meet the unique needs of women and may be a model upon which to confirm whether this type of intervention might also benefit depressed women with CVD.
Pharmacologic therapy
Despite the pleiotropic beneficial effects of SSRIs on platelet function, immune and neurohormonal activation, and cardiac rhythm disturbances in patients with CVD [116,117], the use of SSRIs in depressed cardiac patients has not shown a consistent beneficial effect other than a modest improvement in depressive symptoms [107,110]. While no reduction of CVD end points has been achieved with pharmacologic therapy of depression, post hoc and subgroup analysis of several trials has suggested a potential beneficial effect [118]. In an analysis and comparison of SSR Is use in the ENRICHD population, treatment with antidepressants was associated with a significant 43% lower risk of death or recurrent MI (HR: 0.57; 95% CI: 0.38-30.84), adjusted for disease severity and other factors [119], but the findings of this observational post hoc analysis need to be investigated further and confirmed in a randomized controlled trial. The Sertraline Antidepressant Heart Attack Randomized Trial (SADHART) [107] was primarily a drug-safety trial for sertraline in depressed post-MI patients. This doubleblind, placebo-controlled trial of 369 subjects (36% women) found sertraline to be safe and well tolerated from a cardiovascular standpoint. Other findings of note were a high spontaneous depression remission rate (53%) and a nonsignificant reduction in the incidence of death, and recurrent nonfatal MI in the sertraline-treated group compared with the placebo group (14.5 vs 22.4%). While suggesting the potential for a beneficial effect of SSRI use, this was a post hoc analysis with insufficient power to be definitive.
Tricyclic antidepressants should generally be avoided in patients with CVD owing to side effects and elevated risk, but SSRIs have generally been found to be safe in women with CVD. While some studies have found an association with SSRI use and adverse outcome in univariate analysis, in multivariate analysis most show that SSRI use is simply associated with more severe depression and does not significantly modify depression-associated CVD risks positively or negatively [117,120,121]. However, cautious use of SSRIs is appropriate since a few trials have suggested potential adverse effects [5,121]. Analysis of the Nurse's Health Study population, a prospective study of over 63,000 women without known CVD at baseline [5], found that depressive symptoms were associated with sudden cardiac death (SCD), fatal CHD and nonfatal MI. Antidepressant use was also associated with increased SCD risk, suggesting that pharmacologic therapy could either be proarrhythmic or alternatively, a marker of more severe depression or treatment nonadherence. There have been a few reports of increased risk of minor bleeding associated with SSRIs in cardiac patients [118], but other investigators have not observed an increased risk of bleeding despite a high prevalence of concomitant antiplatelet drug use [122]. Kim et al. provide sex-specific results on 1076 SSRI users (449 women) and 304 non-SSRI user controls (127 women) undergoing CAB surgery and found no excess bleeding risk or differences in other adverse events in either women or men.
Further study on the effect of pharmacologic therapy of depression in women with CVD is clearly warranted, but based on data from interventional trials coupled with the relatively safe drug-interaction profile of sertraline and citalopram, these two SSRIs are currently the preferred agents when drug therapy of depression is necessary in women with CVD [107,123].
Other treatment options
Most studies have shown that symptoms of depression are reduced in women with symptomatic CHD who enroll in cardiac rehabilitation [124,125]. Participation in cardiac rehabilitation by women with recent CVD events or heart failure, particularly when physical activity levels increase, is also associated with improved behavioral parameters (anxiety, somatization and hostility), health-related quality of life, reduction in levels of classic CVD risk factors, and improved CVD outcomes [124,126,127]. Exercise training alone has also been shown to be comparable to antidepressant medication (sertraline) and better than placebo in alleviating depressive symptoms in patients with MDD (202 patients, 153 women) [128]. Since women attending cardiac rehabilitation have a higher prevalence of depressive symptoms and MDD [42], they stand to benefit even more than men in this regard. Unfortunately, in virtually every population studied, women are less likely to participate in cardiac rehabilitation programs than men [129–131]. Numerous barriers to cardiac rehabilitation participation by women have been identified and include older age [129–131], lack of physician referral [132,133], not feeling that rehabilitation is necessary [134,135], social isolation, and inability to drive [134–137]. Greater effort is needed to ensure that after major cardiac events all women, especially those with depressive symptoms, are referred to and attend formal cardiac rehabilitation programs, or at least a supervised exercise program.
Conclusion
Depression in women, even in mild forms, is associated with a significantly increased risk of developing CVD and dying from cardiac causes. As such, depression should be added to the conventional risk factors for CVD when assessing risk levels. Women who already have CVD who then develop depression have more adverse outcomes than those who are not depressed. While treatment of depression has not been shown to reduce CVD risk, treatment does improve quality of life and adherence to recommended treatment regimens. Despite the fact that women are more vulnerable to depression and that CVD is the leading cause of death among adult women in the USA, relatively little research has focused on the etiology and pathogenic mechanisms of major depression among women with CVD. Future studies should focus on gender-specific psychosocial and physiologic measures as they relate to CVD. Better identification of depression and appropriate management, especially via participation in cardiac rehabilitation, is both humane and medically appropriate in women suffering from CVD.
Future perspective
The pathophysiology associated with, and relationship between CVD and depression, particularly in women, is complex and future research must focus upon clarifying mechanistic pathways and causal relationships (direct, indirect and common risk factors), in order to optimally treat depressive symptoms and reduce the associated elevated CVD risk. The results of this research will be critical in guiding management.
If depression is found to increase CVD risk directly via specific pathobiologic mechanisms, such as inflammation or thrombosis, then antidepressants or therapies that target those mechanisms may reduce the risk of CVD more effectively than those that do not. Alternatively, if depression exerts its adverse effects indirectly through nonspecific mechanisms, such as by deterring adoption of healthy behaviors, then a focus on remission of depression, regardless of how remission is achieved, may be an effective strategy, assuming treatment has an acceptable safety profile. Finally, if research determines that depression is simply another risk factor for CVD, but relief of depression does not reduce risk, then more aggressive management of other CVD risks (e.g., lower lipid or blood pressure target levels) in depressed women could be effective in attenuating the risk imparted by depression.
In light of previously observed sex differences in treatment outcomes and large gaps in data specific to women, future research must place a priority upon designing and reporting results from clinical trials so that sex-specific data is available to guide pharmacologic and behavioral treatment of depression in women with CVD. In particular, gender-specific therapy aimed at improving coping skills and anxiety warrant additional exploration.
Executive summary
Depression is an independent risk factor for incident cardiovascular disease (CVD) and adverse outcomes in women with prevalent CVD.
CVD is also a risk factor for new and recurrent depression.
Depression is associated with two-to five-times increased mortality and morbidity in women with CVD, with the highest risks in women with heart failure and after cardiac surgery.
Even mild forms of depression or depressive symptoms increase CVD risk, and do so in a dose-dependent manner.
Compared with men, women have double the risk of depression and experience poorer outcomes in virtually all CVD conditions.
Higher depression rates in women may be a significant factor in explaining the sex-based excess morbidity and mortality, particularly among younger women with CVD.
Sex-specific research is lacking, and only recently have investigators begun to report results by gender and explore the CVD-depression relationship among women-only study cohorts (i.e., the Women's Health Initiative, the Women's Ischemic Syndrome Evaluation and the Nurse's Health Study).
When analyzed, important sex differences in symptoms, effects of treatment, and outcomes have been found.
Depression is difficult to diagnose in women with CVD and other medical illnesses owing to atypical symptoms and the overlay of somatic complaints.
This challenge is compounded by the fact that cardiovascular healthcare providers typically have a low awareness of and little focus on mental health issues and neither accurately diagnose nor treat depression in the vast majority of CVD patients they care for.
The exact relationship between CVD and depression is not clear, but it is no longer believed that the development of depressed mood after a CVD event is simply a ‘normal’ response to illness.
Depression causes numerous adverse physiologic and behavioral changes that may directly contribute to heightened CVD risk.
Several factors, such as increased thrombotic potential and inflammation, are common to both depression and CVD leading some investigators to propose a common pathway for both conditions.
Specific causal or common risks include autonomic dysfunction, hypothalamic–pituitary–adrenocortical axis activation, hypercoagulability, endothelial dysfunction and inflammation.
Indirect causal factors associated with depression that may impact CVD risk include maladaptive coping behaviors, such as persistent smoking, poor self-care, social isolation, chronic life stress, poor adherence with medical therapy, inability to make desirable lifestyle changes, and low attendance at cardiac rehabilitation.
Studies performed to date have not been able to demonstrate that treatment of depression results in a reduction in CVD morbidity or mortality, but that should not dissuade healthcare providers from identifying women with depression since the depressive symptoms may warrant treatment independent of other conditions.
Several depression screening tools are widely available and easy to use (e.g., the Beck Depression Inventory, the Patient Health Questionnaire-2 and the Patient Health Questionnaire-9), and despite their limitations they should be used routinely to assess women with CVD in order to improve diagnosis and treatment rates.
Women appear to respond differently compared with men to psychosocial interventions such as counseling and cognitive behavioral therapy for depression and receive low social support after myocardial infarction.
In two large trials (the Montreal Heart Attack Readjustment Trial and the Enhancing Recovery in Coronary Heart Disease), women, especially those with anxiety, receiving interventions aimed at improving mood and levels of social support had unexpectedly worse outcomes than treated men, who benefited from the interventions, or women receiving usual care.
Programs designed specifically for women, such as the support and education provided in women-only groups in the Stockholm Women's Intervention Trial for Coronary Heart Disease (SWITCHD), have demonstrated promising reductions in CVD risk factors and mortality and suggest that further exploration of what types of behavioral interventions might benefit women is warranted.
Comprehensive cardiac rehabilitation programs and regular physical activity are strongly associated with both lower depression levels and CVD morbidity and mortality and should be strongly recommended to women with CVD, especially those with depression.
Nonrandomized trials and subgroup analyses suggest a potential treatment benefit from selective serotonin-reuptake inhibitors (SSRIs), but there are little sex-specific data.
SSRIs may favorably impact CVD risk by modifying adverse physiologic conditions (e.g., thrombotic potential and abnormal heart rate variability) and by improving mood enough to adopt healthy lifestyle behaviors and adhere to treatment recommendations.
Sertraline and citalopram are the preferred first-line antidepressants for women with coronary heart disease (CHD) based on the SADHART and the Canadian Cardiac Randomized Evaluation of Antidepressant and Psychotherapy Efficacy Trial studies that documented acceptable safety, tolerability and limited interaction with commonly used cardiac medications in patients with CHD.
Tricyclic antidepressants should generally be avoided owing to increased CVD risk.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
