Abstract
Until the last decade, oral hypoglycemic agents have not been recommended in pregnancy owing to fear of their potential adverse fetal effects, including teratogenicity and neonatal hypoglycemia. However, the evidence in support of these recommendations is weak and is principally based on case series involving the use of first-generation sulfonylureas. Studies using a single-cotyledon placental model have found glyburide to only minimally cross the placenta, an observation that paved the way for a landmark randomized clinical trial that found glyburide to be as safe and effective as insulin in the management of gestational diabetes mellitus. Still, contradicting results regarding its trans-placental transfer, lack of adequate data regarding its safety during the first trimester and reports of increased neonatal morbidity raise concerns regarding the universal application of glyburide as an alternative to insulin therapy in diabetic pregnant women. Thus, there is a need for large, randomized, controlled trials with adequate power to evaluate the possibility of increased neonatal metabolic complications as well as the long-term outcome of infants born to mothers treated with glyburide and insulin. Unless future studies refute current data regarding the efficacy and safety of glyburide, we believe that, owing to its ease of administration, convenience and low cost, glyburide will become the first line of medical treatment in patients with gestational diabetes mellitus within the next few years.
Gestational diabetes mellitus (GDM) is defined as glucose intolerance of variable severity with onset or first recognition during pregnancy, as determined by the 3 h 100 g oral glucose tolerance test (OGTT). The prevalence of GDM has increased during recent years in parallel with the obesity epidemic, affecting up to 8% of all pregnancies [1]. Moreover, recent studies have convincingly demonstrated that GDM is associated with adverse maternal and neonatal outcome [2,3] and that treatment of GDM, even in mild cases [4], helps to decrease the incidence of these complications.
Adherence to a strict diabetic diet is accepted as the initial treatment of GDM. Traditionally, in cases when dietary therapy in insufficient to maintain euglycemia, insulin was the only available medical therapy [5–7], considering the fact that oral hypoglycemic agents (OHAs) have not been recommended in pregnancy owing to fear of their potential adverse fetal effects, including teratogenicity and neonatal hypoglycemia [8–12]. However, the evidence in support of these recommendations is weak and is principally based on case series involving the use of first-generation sulfonylureas [8–10,13–17]. These recommendations persisted until the landmark randomized controlled trial (RCT) of Langer et al. in which glyburide, a second-generation sulfonylurea, was demonstrated to be as safe and effective as insulin in the management of GDM [18]. Indeed, the similarity in the pathogenesis of GDM and Type 2 diabetes [19], and the evidence regarding the efficacy of glyburide in the management of Type 2 diabetic patients [20], provides a strong rationale for the use of glyburide in the management of GDM. In view of these data, glyburide has gained acceptance as first-line therapy for GDM. Nevertheless, in spite of the increased use of glyburide in the management of GDM, a recent survey performed by the American College of Obstetricians and Gynecologists reported that only 13% of obstetricians and maternal–fetal medicine specialists are using glyburide as first-line medical therapy for GDM [21].
The purpose of this paper is to review the current evidence regarding the safety and efficacy of glyburide in the management of diabetes in pregnancy.
Safety of glyburide
Early studies
Historically, the use of OHAs in pregnancy was not recommended following reports of possible association with congenital malformations and prolonged neonatal hypoglycemia in animal models as well as in observational studies in humans [8,10,11,13,22]. In 1970, Kemball et al. reported on four infants of mothers treated with chlorpropamide and acetohexamide, first-generation sulfonylureas, who experienced prolonged neonatal hypoglycemia [10]. Subsequently, Piacquadio et al. studied 20 pregnant women with Type 2 diabetes who used OHA (mainly chlorpropamide) during the first-trimester and compared them to a control group of 40 Type 2 diabetics matched for age, race, weight, parity and glycemic control that were not exposed to OHA [13]. They found a greater than threefold increase in the rate of congenital anomalies in the group of women exposed to OHA (50 vs 15%; p = 0.002), with nine of the ten cases of anomalies occurring in patients treated with sulphonylureas. Importantly, most of the patients were poorly controlled having baseline glycosylated hemoglobin levels in the range of 7.2 to 14.2%. Furthermore, neonates in the OHA group had a significantly higher rate of metabolic complications, including prolonged hypoglycemia lasting up to 7 days, hyperbilirubinemia and polycythemia. Further support to a possible teratogenic effect of chlorpropamide comes from experiments in a model of mouse embryo culture, in which exposure to chlorpropamide at human therapeutic concentrations resulted in a higher rate of major congenital malformations and abnormal growth, independently of hypoglycemia or by-products of chlorpropamide metabolism [22].
Other studies, however, found sulfonylureas to be relatively safe, even during the first trimester. In a study of 50 women with GDM treated with chlorpropamide (100 mg/d), Sutherland et al., found the rates of neonatal hypoglycemia and macrosomia to be similar to those observed in a control group of untreated GDM patients [14]. In another study of 78 women with Type 2 diabetes who were treated with chlorpropamide during the first trimester, there were only two cases of major congenital anomalies [17]. Furthermore, Towner et al. prospectively studied 332 pregnant patients with Type 2 diabetes treated with diet, sulfonylureas or insulin during the first trimester [23]. The overall rate of congenital anomalies was 16.9%, and was similar in each of the three treatment groups. Multivariable logistic regression analysis revealed that maternal age and glycosylated hemoglobin levels at initial prenatal presentation, but not treatment modality, were associated with major malformations.
There are several possible explanations for the apparently contradicting results of these early studies. Most of the data are based on case series and retrospective studies of first-generation sulfonylureas. In addition, most studies included only a small number of subjects, and were thus underpowered to demonstrate clear relationships between the drugs and neonatal outcomes as well as to adjust for potential confounders.
Furthermore, most of the studies did not adjust the results for maternal glycemic control, which is currently believed to be the primary cause of congenital malformations [23–25]. Despite these limitations, most authorities based their recommendations on these studies [5,6], stating that the use of OHAs in pregnancy should be avoided.
Transplacental transfer of sulfonylureas
As second-generation sulfonylureas (glyburide, glipizide and glimepiride) became available, their safety in pregnancy was examined as well. Elliott et al., using a single-cotyledon placental model from human term placentas, evaluated the transport and metabolism of tritiated glyburide with liquid scintillation spectrometry and high-performance liquid chromatography [26]. The authors did not observe any transfer of glyburide, and no appreciable metabolism of the drug was detected, even when maternal glyburide concentrations were increased to 100-times the therapeutic concentration. In another study of the same group, the same model was used to compare the placental transport of different sulfonylureas (glyburide, glipizide, chlorpropamide and tolbutamide) [27]. The transport rate of the different agents varied significantly, and was found to be strongly correlated with molecular properties of the drugs. Specifically, tolbutamide diffused across the placenta most freely, followed by chlorpropamide, then glipizide, with glyburide demonstrating the lowest maternal–fetal transfer rate. Thus, glyburide levels on the fetal side were insignificant even with maternal levels up to five-times the therapeutic dose, reaching levels of up to 1 to 2% of maternal concentrations. The author's conclusion was that that fetal exposure to maternally administered glyburide is, thus, most likely to be insignificant.
The reason as to why glyburide does not cross the placenta despite the fact that its molecular weight is less than 500 g/mol is not entirely clear. Possible explanations are its high protein binding (99.8%) and short elimination half-life [28]. In addition, is has been shown that the human placenta actively effluxes glyburide from the fetal to the maternal circulation. This active process was suggested to be carried out by the breast cancer resistance protein (BCRP) pump, but not by P-glycoprotein or multidrug resistance proteins [29,30].
Clinical evidence
Based on these observations, Langer et al. conducted a RCT to compare the efficacy and safety of glyburide (n = 201) and insulin (n = 203) in the management of women with GDM [18]. There were no differences in the rate of maternal and neonatal adverse outcome between the glyburide and insulin groups. In concordance with the in vitro studies, glyburide was not detected in cord serum (high-performance liquid chromatography, limit of detection of 10 ng/ml) despite the mothers having therapeutic concentrations of glyburide in their blood. This landmark trial provided the first and, as for now, the most substantial clinical evidence regarding the safety and efficacy of glyburide in the management of GDM during the second and third trimesters. Subsequent clinical studies evaluating the outcome of infants born to mothers with GDM treated with glyburide during the second and third trimesters further confirmed these observations [31–34].
The studies described above, however, did not address the safety of glyburide in the first trimester, when a teratogenic effect is possible. In a meta-analysis evaluating the safety of OHAs in the first trimester, ten studies (471 women) were included [35,36]. There were no significant differences in the rates of major malformations between women with first trimester exposure to glyburide compared with nonexposed women. However, the authors emphasized that the studies were heterogeneous, of an overall small sample size, and did not always adjust the results for glycemic control. Therefore, their conclusion was that the routine use of OHAs in early pregnancy warrants further safety evaluation.
Limitations
There are still some caveats with regard to placental transport of glyburide. Previous studies in pregnant rats demonstrated a high maternal-to-fetal transfer rate [37]. Although this observation may be related to interspecies variation, the authors concluded that more studies are needed to evaluate the trans-placental transfer of glyburide. In addition, Hebert et al. prospectively studied 40 women with GDM treated with glyburide [38]. At delivery, glyburide levels in maternal plasma and cord venous blood were measured using a sensitive high-performance liquid chromatography–mass spectrometry assay. Maternal plasma glyburide levels ranged from less than the limit of detection to 32.7 ng/ml. In contrast to the findings of Langer et al., glyburide was detected in the umbilical cord blood samples, reaching a mean level of 70% of maternal plasma glyburide levels (ranging from less than the limit of detection to 12.5 ng/ml). Furthermore, in approximately 20% of umbilical cord samples, glyburide concentrations were greater than or equal to the mean maternal steady-state trough concentrations. This apparent discrepancy may be in part related to the use of a more sensitive assay in the latter study compared with the assay employed in the study of Langer et al. [18] (limit of quantification 0.25 ng/ml vs 10 ng/ml).
Summary
Although it is not clear as to what degree glyburide crosses the placenta, clinical data suggest that glyburide is safe to the mother and the neonate during the second and third trimesters. Nevertheless, patients should be informed regarding the current official recommendations of the American Diabetes Association (ADA) and the American College of Obstetricians and Gynecologists (ACOG) regarding the use of glyburide in pregnancy, as well as that glyburide is not approved by the US FDA for the treatment of GDM [39,40]. Furthermore, there are currently no data regarding long-term neonatal outcome following in utero exposure to glyburide. Further studies are needed to evaluate the safety of glyburide, especially during the first trimester, before the universal application of glyburide as an alternative to insulin therapy in diabetic pregnant women.
Efficacy of glyburide
Glyburide has been demonstrated to be effective in the management of patients with Type 2 diabetes [20]. Since GDM patients have a less severe hyperglycemia compared to Type 2 diabetic patients, it is reasonable to assume that glyburide for the management of women with GDM would be at least as effective as in the case of Type 2 diabetics.
Randomized controlled trials
In the study of Langer et al. described above (Table 1) [18], 404 women with GDM at 11–33 weeks of gestation and with fasting plasma glucose concentrations between 95 and 140 mg/dl were randomized to glyburide or insulin treatment. The primary end point was achievement of the desired level of glycemic control, and secondary end points included maternal and neonatal complications. The two groups were similar with regard to baseline characteristics. The authors found no significant differences in degree of glycemic control and neonatal outcome, including the rate of large for gestational age (LGA) infants, macrosomia, neonatal intensive care unit (NICU) admission, neonatal hypoglycemia and fetal anomalies. With regard to maternal outcome, there was a significantly higher rate of maternal hypoglycemia in the insulin group (20 vs 2%; p = 0.03), although none of the women reported severe hypoglycemia-related symptoms. Although the glyburide failure rate was reported to be as low as 4% (defined as the initiation of insulin treatment because of failure to meet glycemic goals on the maximal glyburide dose for a 2-week period), only 82% of the women in the glyburide group and 88% of the women in the insulin group achieved the target level of glycemic control, thus representing a glyburide actual failure rate of 18%.
Studies assessing efficacy of glyburide in the management of gestational deabetes.

GDM: Gestational diabetes mellitus; lUGR: Intrauterine growth restriction; LGA: Large for gestational age; NICU: Neonatal intensive care unit; NR: Not reported; Obser: Observation; OR: Odds ratio; Pro: Prospective; RCT: Randomized controlled trial; Retro: Retrograde; SGA: Small for gestational age.
Retrospective & observational studies
This study was followed by several case-control studies and one small RCT (Table 1). Except for one study that found glyburide to be effective in only appoximately half of the women with GDM [41], most subsequent studies have found glyburide to be an effective alternative for insulin in the management of GDM, with a failure rate ranging from 7.3% [34] to 20.8% (Table 1) [42]. Overall, glycemic control in women treated with glyburide appears to be comparable to that achieved with insulin, and in one study [32] the proportion of women that achieved target levels of glycemic control was even higher in the glyburide group (86 vs 63%; p < 0.001).
With regard to maternal and neonatal outcome, most studies reported overall similar results for women treated with glyburide compared with insulin. Still, in contrast to the finding of Langer et al., few studies reported a higher rate of neonatal complications in patients treated with glyburide, including neonatal hypoglycemia [33,42], LGA infants and macrosomia [31,33,34,42], small for gestational age (SGA) infants [34], need for phototherapy [32] and longer NICU stay [32]. With regard to maternal outcome, Jacobson et al., in a large retrospective study, found the rate of pre-eclampsia to be two-times higher in the glyburide group (12 vs 6%; p = 0.02), despite the fact that women treated with glyburide achieved a better glycemic control compared with those treated with insulin [32]. These findings, however, were not observed by others.
In a recent systematic review, Nicholson et al. reviewed four RCTs and five observational studies of maternal and neonatal outcome in women with GDM treated with OHA compared with insulin [43]. They conducted a meta-analysis of infant birth weight from the three RCTs comparing glyburide and insulin and found no statistically significant difference in infant birth weight (−93g, 95% CI: −191 to 5 g). The author's conclusion was that there is no consistent evidence for an increase in adverse maternal or neonatal outcomes with the use of glyburide compared with insulin. However, the authors note that the small number of studies available and the small samples sizes in the study of Langer et al. contributed most to the overall results and, thus, limited their ability to draw substantial conclusions. In another meta-analysis, Moretti et al. have reported similar results [44].
In a recent study, Lain et al. suggested that neonatal body composition may more precisely reflect the effects of the maternal environment and exposures on fetal growth and may better estimate fetal effects of various methods of maternal glycemic control [34]. They randomized 82 women with GDM requiring medical therapy to insulin or glyburide (Table 1). The primary outcome was percent neonatal fat mass. Secondary outcomes included anthropometrics (ponderal index, BMI, skinfold sum and arm fat area), glycemic control, and maternal plasma and cord blood biomarkers (C-peptide, insulin, glucose and total adiponectin). Although there was a higher rate of macrosomia and LGA infants in the glyburide group, the glyburide and insulin groups were similar with regard to neonatal body composition, measures of glycemic control and cord metabolic biomarkers. Nevertheless, the study was stopped early secondary to technical reasons, so that the power of the study to detect differences between the two groups was lower than originally planned.
In a secondary analysis of the data from his RCT, Langer et al. stratified the data by severity of GDM using the fasting glucose levels from the OGTT [45]. The authors found that degree of glycemic control and perinatal outcome were similar for patients in the glyburide and insulin groups at all severity levels of GDM. They also found, using logistic regression analysis, that the risk of LGA was related to severity of GDM, glycemic control, weight gain in pregnancy and a previous delivery of an infant with macrosomia, but not to the treatment modality.
Limitations of current literature
There are notable limitations to the studies presented above. In his recent critical review, Moore raised concerns regarding the possibility of a type 2 error owing to an inadequate sample size [46], as power analysis for the different outcome variables was not available in the study of Langer et al. [18]. He noted the higher rate (although not statistically significant) of neonatal hypoglycemia and hyperbilirubinemia in the glyburide group in this study (9 vs 6% and 6 vs 4%, respectively), and calculated that a larger sample size (approximately 1300 women per group) may have determined these differences to be statistically significant. He also suggested that the study of Jacobson et al. [32] was too small to convincingly demonstrate equivalency in neonatal outcome between the glyburide and insulin groups as it was only powered to detect at least a twofold increase in the rate of macrosomia/LGA and a 44% increase in rate of neonatal hypoglycemia.
Other limitations are related to the nonrandomized retrospective design of most of the studies. One example is a selection bias that may significantly confound the results. In the study of Kitzmiller et al., the women included in the glyburide group were those who chose (rather than were allocated) to use glyburide [41]. In the retrospective study of Jacobson et al., 25% of the women who failed diet therapy and were intended to be included in the glyburide group were instead treated with insulin [32]. Further analysis done by the authors revealed that the patients who were allocated to the glyburide group had lower BMIs, lower mean fasting glucose, and were more likely to be Asian. Similarly, Ramos et al. have also found that older women and those with lower BMIs were more likely to receive glyburide than insulin [33]. The fact the neonatologists are aware of the treatment modality may also affect the results, so that neonates born to mothers treated with a certain modality may be more likely to be transferred to the NICU or to be diagnosed with certain conditions [32,33]. Finally, none of the studies provided information as to what proportion of patients who failed glyburide therapy achieved adequate control with insulin, thus preventing appropriate interpretation of the glyburide failure rates. Other important limitations are that most studies lack information regarding glycemic control and patient compliance with regard to glucose monitoring and treatment.
Summary
Overall, glyburide appears to an effective alternative to insulin in most women (∼80%) with GDM, and results in glycemic control and perinatal outcome similar to that achieved with insulin. The limitations presented above, along with differences between the studies with regard to study population (ethnicity and obesity), inclusion criteria (gestational age and fasting glucose levels), definitions of glyburide failure and outcome measures, and differences in the dosage and titration of glyburide, may all contribute to the variation in results from the different studies.
Patient selection
One of the major drawbacks of using glyburide as the first-line of medical treatment in GDM is the concern that women who will eventually fail glyburide therapy may be suboptimally controlled for several weeks until treatment is switched to insulin [47,48]. This delay in achieving glycemic control, which is the result of the need to gradually titrate glyburide dose before glyburide treatment is determined as failure, and may theoretically result in an increased risk of adverse pregnancy outcome. The best way to avoid this delay is through the identification of risk factors for glyburide failure, and thus to define the group of patients in which insulin should be used as first-line for medical treatment following failure of treatment by diet alone.
Risk factors for glyburide failure
Several studies have tried to identify risk factors for glyburide failure (Table 2). Following the RCT of Langer et al., Conway et al. retrospectively studied 75 women with GDM treated with glyburide in San Antonio, TX, USA [49]. They found that women who failed glyburide had higher glucose levels on their OGTT at all time points. However, these differences were not found to be clinically useful as receiver operating characteristic analysis revealed no clear cut-off points for prediction of glyburide failure. Similarly, Rochon et al. reported that the result of the 50 g glucose challenge test (GCT) was the only factor predictive of glyburide failure both as a continuous variable (OR:1.01; 95% CI: 1.01-1.03) and as a dichotomous (>200 mg/dl) variable (OR: 2.84; 95% CI: 1.01-07.98) [48].
Studies assessing risk factors for glyburide failure.
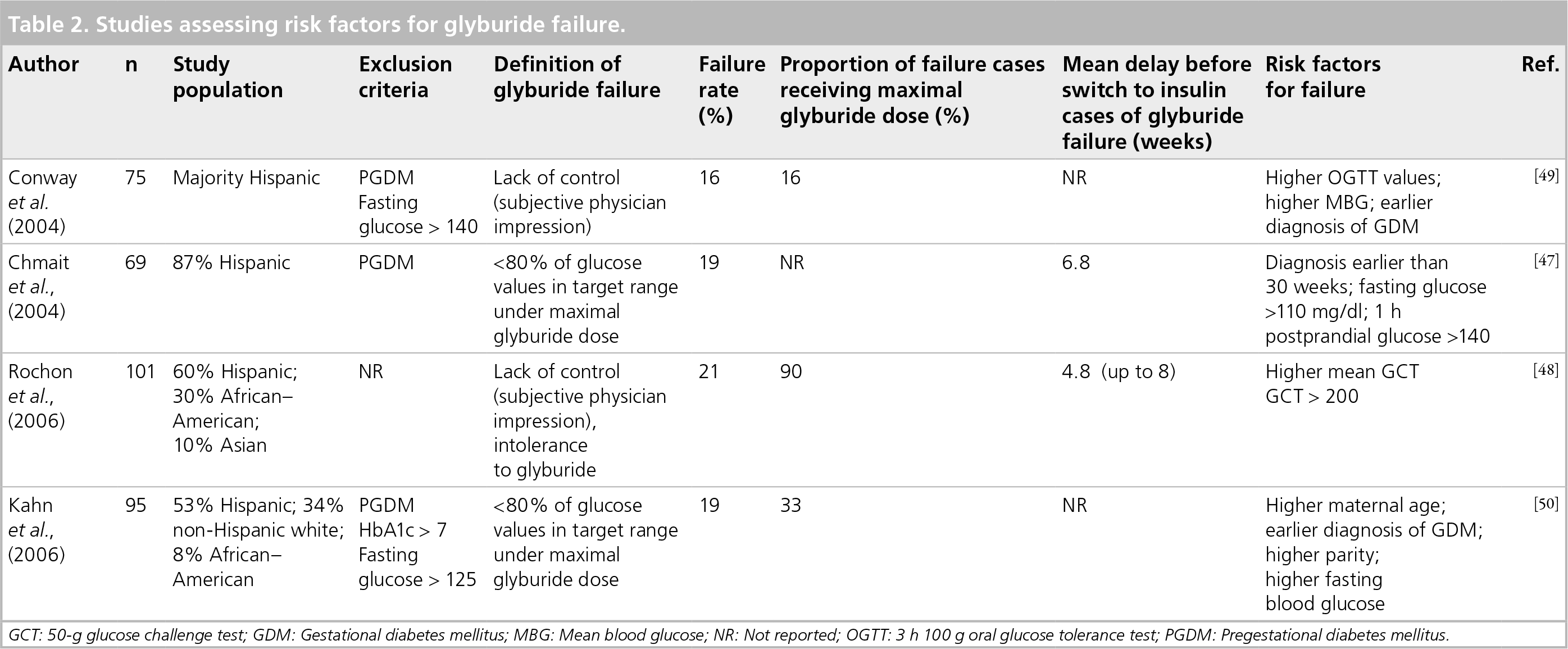
GCT: 50-g glucose challenge test; GDM: Gestational diabetes mellitus; MBG: Mean blood glucose; NR: Not reported; OGTT: 3 h 100 g oral glucose tolerance test; PGDM: Pregestational diabetes mellitus.
Three studies have found that earlier diagnosis of GDM, and thus initiation of glyburide earlier in pregnancy, is associated with increased risk of failure (Table 2) [47,49,50]. Kahn et al., using multivariable logistic regression to adjust for confounders, reported that women diagnosed with GDM at a gestational age of less than 25 weeks were 8.3-times more likely to fail glyburide compared with those diagnosed after 25 weeks [50]. Bertini et al. calculated that the combined criteria of initiation of treatment with glyburide after 30 weeks or low fasting and 1 h postprandial glucose levels (<110 mg/dl and <140 mg/dl, respectively) prior to the initiation of treatment with glyburide predicted glyburide success with a sensitivity of 98%, specificity of 65%, positive predictive value of 90% and a negative predictive value of 92% [47]. However, the clinical value of these factors should be further validated since these calculations were performed on the same cohort from which these criteria were derived. In addition, earlier diagnosis of GDM may actually be a confounder reflecting an unrecognized pregestational diabetes mellitus (PGDM) rather than an independent risk factor for glyburide failure. Although women with the diagnosis of PGDM were excluded from all studies, only in one study [50] was there an attempt to identify unrecognized PGDM by measuring glycosylated hemoglobin levels.
Limitations of the studies
Although providing important information, the studies described above are limited for several reasons. The small sample size in these studies limits their power to identify risk factors for glyburide failure as well to control for potential confounding variables. Indeed, logistic regression analysis was feasible only in the study of Kahn et al. [50]. Another major problem is the definition of glyburide failure. While some studies provided a clear definition for glyburide failure [47,50], in other studies failure was determined by the subjective physician impression (Table 2). Consequently, in some of these studies, only a small proportion of the women defined as glyburide failure received the maximal glyburide dose [49,50]. Thus, in these cases it is possible that the ‘failure’ of glyburide is related to insufficient dose, and thus reflects physician failure to persist with glyburide to its maximum accepted dose rather than true glyburide failure.
Summary
Several risk factors have been suggested for the prediction of glyburide failure in GDM patients. Despite the limitations presented above, these risk factors likely represent a population with more advanced insulin resistance in which glyburide cannot stimulate a sufficient insulin response to sustain euglycemia, and thus may benefit from using insulin as first-line of medical treatment. Nevertheless, at the moment, the clinical implications of these risk factors are limited.
Conclusion
During the last decade, a significant amount of data have accumulated that demonstrate the efficacy and short-term safety of glyburide as an alternative for insulin in the management of GDM. Since glyburide is considerably more convenient and less expensive than insulin [51], it is clearly preferred by most patients [47,49], and can thus lead to increased patient compliance. For that reason, glyburide has gained increasing acceptance as first-line therapy for GDM [52–54].
Still, the use of glyburide in GDM patients has some drawbacks, including the conflicting evidence regarding its placental transfer, and possibly increased neonatal morbidity as reported by some of the studies presented above. Another important obstacle is the possible delay in achieving adequate glycemic control in women who will eventually fail glyburide therapy. Possible solutions for this latter problem include appropriate selection of patients for glyburide therapy according to the risk factors described above, as well as individualization of the starting dose according to patient's characteristics such as BMI and severity of glucose intolerance as reflected by fasting glucose levels [45,47,50].
The use of glyburide in pregnant women with Type 2 diabetes is also of concern considering the lack of adequate data regarding its safety during the first trimester. According to the current recommendation of the ADA [55], oral hypoglycemic agents should be stopped and insulin started before conception, and in cases of women who become pregnant while taking oral medications, insulin should be started as soon as possible. Nevertheless, the ADA states that limited first trimester data suggest that glyburide can be continued until insulin is started, in order to avoid severe hyperglycemia during early gestation.
Future perspective
Considering the limitations of the current literature, there is a need for large, randomized, controlled trials with adequate power to evaluate the possibility of increased neonatal metabolic complications as well as improved glycemic control with glyburide treatment. In parallel, studies should also compare the long-term outcome of infants born to mothers treated with glyburide and insulin.
Nevertheless, because of its ease of administration, convenience and low cost, we believe that during the next few years glyburide will become the first-line of medical treatment in patients with GDM, and possibly an alternative to insulin for patients with Type 2 diabetes.
Executive summary
Until the last decade, oral hypoglycemic agents have not been recommended in pregnancy owing to fear of their potential adverse fetal effects, including teratogenicity and neonatal hypoglycemia.
There is concern that women who eventually will fail glyburide therapy may be suboptimally controlled for several weeks.
Several studies found that higher plasma glucose levels, earlier diagnosis of gestational diabetes, higher maternal age and higher parity are associated with increased risk of glyburide failure.
At the moment, the clinical implications of these risk factors are limited.
It is currently accepted that glyburide crosses the human placenta in only minimal amounts.
Lack of glyburide placental transfer is probably related to its high protein binding as well as active transport of glyburide from the fetal to the maternal side.
In 2000, a landmark randomized controlled by Langer et al. found glyburide to be safe for the management of gestational diabetes during the second and third trimesters.
A meta analysis of patients with Type 2 diabetes suggests that glyburide may also be safe during the first trimester.
Most studies have found glyburide to be as effective as insulin with regard to glycemic control and maternal and neonatal outcome in patients with gestational diabetes.
Glyburide failure rates range from 7.3 to 20.8%.
Few studies reported higher rate of pre-eclampsia, neonatal hypoglycemia and excessive fetal growth in patients treated with glyburide compared with insulin.
Current data are limited by inadequate sample size, selection bias and lack of information regarding glycemic control.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
