Abstract
The use of oral contraceptives is associated with an increased risk of venous thrombosis. It is now generally accepted that women who use oral contraceptives that contain so-called third-generation progestins (desogestrel or gestodene) are exposed to a twofold higher risk of venous thrombosis than women who use oral contraceptives that contain the second-generation progestin levonorgestrel. Coagulation studies demonstrated that oral contraceptives increase the plasma level of prothrombin, decrease the level of protein S and induce acquired activated protein C resistance. The changes in hemostatic parameters can explain why women who use oral contraceptives are exposed to an increased risk of venous thrombosis and why the risk is further increased in third-generation oral contraceptive users.
The history of hormonal fertility control began in 1937, when AW Makepeace demonstrated that injections of progesterone could prevent ovulation in rabbits [1]. The antiovulatory effect of progesterone was later used as the basis for hormonal contraception, although natural progesterone, due to its low bioavailability following oral administration, was replaced by synthetic progestins. The first large-scale clinical trials of oral contraceptives (OCs) started in 1956 and 1957. The earliest preparations contained the progestin norethynodrel, which was accidentally contaminated with the estrogen compound mestranol. When the mestranol contamination was reduced, women experienced one of the most frequent side effects of progestin-only preparations, breakthrough bleeding. Therefore, the estrogen compound was retained for improved cycle control, establishing the principle of combined oral contraception [2].
In 1959 the first OCs were approved for birth control in the USA. Although OCs were widely welcomed, it did not take long before concerns were raised regarding their side effects. The first case reports describing venous and arterial thromboembolic events in women using OCs, including fatal cases, appeared shortly after the introduction of the pill [3–5]. Initially, the estrogen component of OCs was considered to be responsible for the prothrombotic effect of hormonal contraception [6]. Compared with nonusers, the risk of venous thrombosis (VT) was estimated to be four- to ten-times increased in users of the early contraceptive formulations (containing over 50 μg of ethinylestradiol) [7,8] and the death rate attributable to OC was considered to be approximately 3/100,000 users [9]. Subsequent reductions in the amount of estrogen in pills to the lowest dose necessary for effective cycle control (15–35 μg of ethinylestradiol) resulted in a decreased risk of thromboembolic events. However, the risk of VT is still reported to be increased for ‘low-dose’ OCs by three- to sixfold in healthy young women without other risk factors. Consequently, an incidence of approximately 1/10,000 women-years in nonusers is increased to 3–4/10,000 women-years during contraceptive use [10]. In spite of the low absolute risk, oral contraceptives are responsible for most of the VT cases in young women. The highest risk occurs during the first year of use and the increased risk persists until, but not beyond, discontinuation of the OC [11,12].
Further efforts to improve OC formulation were aimed at minimizing the androgenicity and adverse metabolic effects of the progestin compound. Since progestin prevents ovulation and, as a result, provides the contraceptive effect, the changes in this compound concerned the chemical composition rather than the dose. In the early 1980s, OCs containing so-called third-generation progestins (gestodene or desogestrel) were introduced on the market. Gestodene and desogestrel were expected to eventually replace the previous progestin generation (levonorgestrel) owing to their higher biological effectiveness, lower androgenicity and minimal adverse metabolic effects. However, in 1995, the thrombogenicity of OCs again became a topic of debate when a number of case–control studies indicated that both gestodene- and desogestrel-containing OCs (third-generation OCs) might further increase the risk of VT by 1.5- to 3-fold compared with levonorgestrel-containing OCs (second-generation OCs) [11,13–15]. These findings were questioned in later publications, in which the difference in the risk of VT was attributed to bias and confounding [16,17]. Potential sources of bias mentioned where:
Selective prescription of third-generation OCs to women with a higher risk of venous thrombosis;
The tendency that women with suspected VT who use newer formulations of OCs are more likely to be referred for diagnostic testing than women receiving other formulations;
So-called ‘attrition of susceptibles’, meaning that the group of users of older formulations of OCs might be relatively healthier and free of side effects since individuals susceptible to VT were gradually eliminated from the group due to thrombotic complications [18].
The conflict in the interpretation of the data initiated a large number of comparative epidemiological studies. Most of these studies confirmed that women who use third-generation OC are exposed to a higher risk of VT than users of second-generation OCs [12,19,20], although some did not [21,22]. In a number of consecutive reports by Heinemann and colleagues [23–25], it was argued that study design, prescription and referral bias, as well as diagnostic suspicion, may have led to an overestimation of the increase in the risk of VT among users of newer OCs compared with those who use previous generation OCs.
However, careful reanalysis of the original studies with appropriate stratification for risk factors, such as smoking, obesity and duration of use, executed by independent experts, led to the conclusion that bias and confounding could not explain the consistent epidemiological findings of an increased risk of VT during use of third-generation OCs [10,26]. An effect of diagnostic suspicion and referral bias was excluded in the additional case–control study, in which patients and controls were subjected to the same referral and diagnostic procedures [27]. Thus, it is now generally accepted by major national and international drug regulatory authorities that third-generation OCs are more thrombogenic than second-generation OCs, although the absolute risk of VT among OC users remains very low. In this respect, the European Agency for the Evaluation of Medicinal Products (EMEA) issued position statements [101] and information for women using oral contraceptives [102], in which the Committee for the Proprietary of Medicinal Products (CPMP) concluded that “users of combined OCs containing desogestrel or gestodene with 30 μg of ethinylestradiol (mono-, bi- or tri-phasic formulation) have a small increased risk of venous thromboembolism compared to women using combined OC containing levonorgestrel with less than 50 μg of ethinylestradiol” and “while in users of levonorgestrel-containing products the frequency of venous thromboembolism is estimated to be approximately 20 cases per 100,000 women-years of use it is estimated to be approximately 30–40 cases per 100,000 women-years of use of desogestrel- or gestodene-containing products with 30 μg of ethinylestradiol”. Furthermore, a number of recent studies have indicated that the risk of VT in women receiving a cyproterone acetate-containing OC preparation was significantly higher (fourfold) than the risk of women using levonorgestrel-containing OCs [28].
Ever since VT was established as one of the most serious side effects of OC use, there have been investigations into the influence of estrogens and progestins on the hemostatic system in an attempt to provide a biological explanation for OC-induced thrombosis. It appeared that OCs affect the plasma levels of almost all proteins involved in coagulation and since, at the time of these studies, there was limited knowledge concerning the etiology of VT, these investigations did not provide a mechanistic basis for the thrombotic effect of OCs. However, the discovery of a large number of hereditary risk factors of VT in the 1990s enabled a better interpretation of the relation between the OC effects on hemostatic parameters and the risk of V T.
Risk factors for VT
Under normal conditions, the hemostatic system (Figure 1) is in a delicate balance, which is the result of the complex interaction between the procoagulant and anticoagulant pathways. However, increased levels of procoagulant proteins or defects in the anticoagulant system may disturb this balance and lead to a so-called prothrombotic state, which, under certain conditions, may result in excessive thrombin formation and the development of VT. The occurrence of thrombosis is not just a matter of chance, but can often be associated with the presence of one or more risk factors that can be either of acquired or genetic origin (Table 1).
Major risk factors of venous thrombosis.
OC: Oral contraception; TFPI: Tissure factor pathway inhibitor; vWF: von Willebrand factor.
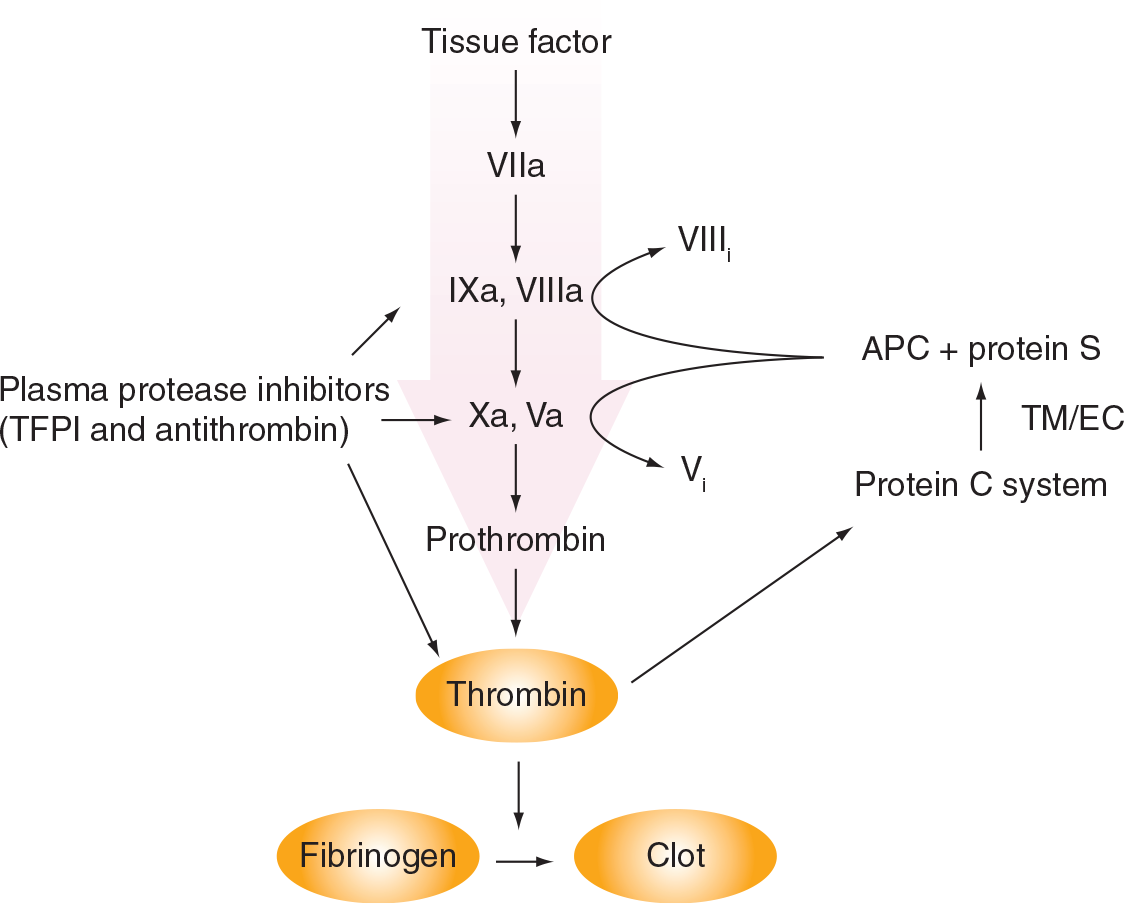
Regulation of the coagulation cascade.
The plasma levels and activity of proteins involved in blood coagulation and the anticoagulant pathways are determined by a genetic background and environmental influence. The G20210A prothrombin mutation (which is associated with elevated prothrombin levels), the factor V Leiden mutation (which results in a decreased sensitivity of factor Va for activated protein C [APC]), and deficiencies of antithrombin (AT), proteins C and S are the most well known hereditary risk factors of VT (Table 1). A combination of two or more genetic defects leads to an interaction and synergistically increases the thrombosis risk [29]. In addition, acquired risk factors, such as pregnancy, purperium, malignancy, surgery, OC use and hormone-replacement therapy, greatly enhance the risk of VT introduced by a genetic background. For instance, it has been reported that OC use further increases the risk of VT in women heterozygous for deficiencies of AT, protein C and protein S [30], in heterozygous and homozygous carriers of the G20210A prothrombin [31], and the factor V Leiden mutations [32]. Combinations of acquired risk factors, such as obesity and OC use, are also associated with a greater risk of VT than would be expected on the basis of their individual effects [33,34]. The synergistic effect on the risk of VT may be explained by additive effects of obesity [34–36] on the changes of coagulation parameters that occur during OC use (see later) [37–39].
A prothrombotic condition may not only be caused by changes in the plasma levels of clotting factors and anticoagulant proteins, but may also be a result of qualitative alterations. In 1993, a hereditary prothrombotic condition was described that appeared to be associated with resistance to the anticoagulant protein, APC [40]. APC, together with its cofactor protein S, inhibits coagulation by inactivating the activated forms of coagulation factors V and VIII (factors Va and VIIIa) by limited proteolysis (Figure 1). One year after the discovery of APC resistance, its molecular basis was identified as a single-point mutation in the factor V gene (factor V Leiden), which renders the factor Va molecule less susceptible to inactivation by APC [41]. Later, APC resistance was also shown to occur in the absence of the mutation. This condition, which, in the majority of cases, was associated with increased plasma levels of procoagulant proteins (prothrombin and factor VIII) or decreased levels of the anticoagulant, protein S [42,43], appeared to be an independent risk factor for VT [44].
Knowledge regarding hereditary risk factors for VT and their influence on the coagulation system sheds new light on the relationship between the effects of OCs on hemostatic parameters and the increased risk of VT during OC use.
Effects of OCs on the procoagulant system
The effects of OCs on parameters of the procoagulant, anticoagulant and fibrinolytic systems and differences in effects between second- and third-generation OCs are summarized in Table 2. These data were obtained in a well-designed randomized cycle-controlled crossover study [37–39,45] and were later confirmed in a large study initiated by the EMEA [45,46].
Effects of oral contracetives on haemostatic parameters.
Not significant (p > 0.05). aPTT: Activated partial thromboplastin time; ETP: Endogenous thrombin potential; nAPC-sr: Normalized APC sensitivity ratios; OC: Oral contraceptive; PAI: Plasminogen activator-inhibitor; SD: Standard deviation; TAFI: Thrombin-activatable fibrinolysis inhibitor; tPA: Tissue-type plasminogen activator.
These studies demonstrated that the plasma levels of the coagulation factors VII, VIII and X, fibrinogen and prothrombin are increased and that the factor V level is decreased during OC use (Table 2). Moreover, the increase in prothrombin and factor VII (both antigen and activity) [45] and the decrease in factor V levels were significantly more pronounced during the use of desogestrel-containing OCs than during the use of levonorgestrel-containing OCs [37]. The changes in the concentrations of coagulation factors V, VII and X may not have clinical implications since they are not considered to be risk factors for VT; however, the elevated plasma levels of fibrinogen, prothrombin and factor VIII are more likely to contribute to the increased risk of VT associated with OC use. However, since the difference between the plasma prothrombin levels during the use of third- and second-generation OCs are small, it is unlikely that changes in prothrombin level alone can fully explain the difference in the risk of VT between these OCs.
Effects of OCs on the anticoagulant system
OC use considerably affects the anticoagulant system (Table 2), decreasing the plasma level of the major inhibitor of several activated coagulation factors, AT and the concentration and activity of tissue factor pathway inhibitor (TFPI) [38,47]. However, the most pronounced changes occur in the protein C pathway. OCs induce a small increase in concentration and activity of protein C, which is presumably counterbalanced by elevated levels of the major protein C inhibitors, protein C inhibitor (PCI), α1-antitrypsin and α2-macroglobulin [38,48]. By contrast, the total and free protein S concentration, as well as the APC-independent anticoagulant activity of protein S are considerably reduced during OC use [38,48,49]. Furthermore, the concentration of total and free protein S and its APC-independent anticoagulant activity are significantly lower in women receiving third-generation OCs than in users of second-generation OCs [38,48,49].
An interesting phenomenon related to the protein C system was observed in OC users: they were shown to be more resistant to the anticoagulant effect of APC than nonusers. Acquired APC resistance during OC use was demonstrated by measuring the effect of APC on the activated partial thromboplastin time (aPTT) [50] and thrombin generation [51]. These tests have been validated clinically as predictors of risk of VT development both in the presence and absence of the factor V Leiden mutation [44,52,53]. However, the difference in the sensitivity to APC between non-OC users and users of different OC formulations, determined with the aPTT-based assay, is subtle and does not always reach the level of statistical significance. By contrast, the thrombin generation-based APC resistance test demonstrates substantially higher APC resistance in all OC users compared with non-OC users and in users of third-generation compared with users of second-generation OCs (Figure 2) [54]. Furthermore, an amplification of genetically determined APC resistance in carriers of the factor V Leiden mutation by OC use was observed with the thrombin generation-based APC resistance test (Figure 2) but not with the aPTT-based assay [54].

Effect of oral contraceptive use and factor V Leiden mutation on activated protein C resistance.
The different results obtained with the two assays can be explained by the differences in the determinants of the assays [42]. The clotting of plasma, which is used as an end point in the aPTT-based assay, occurs when only a small amount of thrombin is generated. Thus, the outcome of the test mainly probes the initiation phase of thrombin generation. By contrast, the thrombin generation-based assay mainly depends on the propagation and termination phases of thrombin generation, which results in a higher sensitivity to defects of anticoagulant components, such as AT, TFPI and the proteins of the protein C pathway. For instance, total and free protein S levels appear to be the major determinants of the thrombin generation-based APC resistance test, whereas they have only a small influence on the aPTT-based assay [42].
In the thrombin generation-based APC resistance test, the response to APC is expressed as an APC sensitivity ratio (APC-sr), which is obtained by dividing the result of the measurement in the presence of APC, the endogenous thrombin potential (ETP+APC), by the result of the measurement in the absence of APC (ETP-APC). This mathematical operation, which provides a correction for non-APC-related changes of the ETP, raised a question regarding the influence of the components of the equation (ETP-APC and ETP+APC) on the final result and on the clinical relevance of an increased APC-sr [55,56]. It appears that ETP-APC and ETP+APC are elevated in users of OCs, compared with nonusers. It was noticed by Gris and colleagues [55] that, in one of the original studies [51], the absolute differences between the ETP-APC and the ETP+APC in pooled plasma from users of second- and third-generation OCs were identical. This led to a debate as to whether the elevation of APC-sr in OC users is caused by changes in basal ETP-APC, rather than by resistance to APC [56]. However, recent observations from our laboratory in a large population of second- and third-generation OC users and users of cyproterone acetate- and drospirenone-containing OCs indicated that all OCs significantly increase the ETP-APC, but that there is no significant difference between the effects of the various OCs on the ETP-APC (manuscript in preparation). Multiple regression analysis showed that the main determinant of the APC-sr is the ETP+APC and that there is no significant influence by the ETP-APC on the APC-sr value. Thus, these observations support the hypothesis that elevated APC-sr in OC users reflect an impaired response to APC and that the higher APC-sr values of users of third-generation OCs are indicative of a more APC-resistant phenotype compared with users of second-generation OCs.
At present, acquired resistance to APC is believed to explain the increased risk of VT during OC use, as well as the differential risk in users of second- and third- generation OC. Yet, the molecular basis of acquired APC resistance in OC users remains uncertain. The differences in plasma protein S and prothrombin levels between nonusers and users of second- or third-generation OCs cannot fully explain the differences in sensitivity to APC, which implies that other hemostatic parameters, that were not included in the regression analysis, contribute to acquired APC resistance during OC use [42].
Effects of OC on the fibrinolytic system
For a long time, it was believed that the procoagulant changes in plasma during OC use were at least partially compensated by an enhanced fibrinolytic activity. Particularly, the decreased concentration and activity of plasminogen activator inhibitor (PAI)-1 and the increased plasma levels of tissue plasminogen activator (tPA) and plasminogen during OC use were considered to counteract the prothrombotic effects of the pill [39]. However, it is not clear whether these findings have clinical implications, since changes in the fibrinolytic system have not been demonstrated to affect the risk of V T. Moreover, the increased activity of the fibrinolytic system during OC use is counteracted by elevated thrombin-activatable fibrinolysis inhibitor (TAFI), the plasma level of which has been shown to be higher in women receiving desogestrel-containing OCs than in users of levonorgestrel-containing OCs [39].
Differentiation between the effects of progestins & estrogens on hemostatic parameters
At present, it is not clear whether the more pronounced effect of third-generation OCs on coagulation is determined by a difference in the modulating effect of the progestin components on the ethinylestradiol-promoted changes of the hemostatic system or an independent effect of the progestins. It is hypothesized that overall effects of OCs are dependent on their so-called ‘estrogenicity’, which rises with increasing dose of estrogen, but decreases with increasing antiestrogenic capacity of progestin [57]. Sex hormone binding globulin (SHBG), which is upregulated by estrogens [58] and downregulated by progestins [59], is considered to be one of the most sensitive indicators of the ‘estrogenicity’ of hormone preparations [57]. The SHBG level during OC use correlates with the risk of VT and, furthermore, there appears to be an inverse correlation between the SHBG level and the concentrations of free protein S and the APC sensitivity ratios in OC users [60]. Considerably higher plasma concentrations of SHBG are observed in users of more thrombogenic formulations of OC [57]. Thus, OCs containing the less androgenic progestins cyproterone acetate (CPA), gestodene or desogestrel are considered to be more estrogenic, which is explained by a weaker antiestrogenic effect of these progestin compounds.
The newest OCs containing drospirenone have an estrogenic potential comparable with that of third-generation OCs, which suggests that they may increase the risk of VT to the same extent as gestodene- and desogestrel-containing OCs. In this respect, it is interesting to note that the increase in SHBG plasma level and resistance to APC in users of drospirenone-containing OCs has been shown to be similar to the increase observed for desogestrel-, gestodene- and CPA-containing OCs [61].
Comparison of progestin-only preparations with combined OCs may provide an answer to the question of why OCs with third-generation progestins are more thrombotic than second-generation OCs and why they have more pronounced effects on hemostatic parameters. It has been reported that progestin-only preparations used to treat menstrual disorders increase the risk of VT [62]. However, there is no evidence that progestin-only OCs, when used for contraception in the general population, are associated with an increased VT risk [63,64]. Although there is no information on the risk of VT in thrombophilic women using progestin-only preparations, many clinicians prescribe progestin-only preparations (pills, implants, injections or progestin-bearing intra-uterine devices) for women with a personal or family history of VT.
Unfortunately, the effects of progestin-only OCs on the coagulation system have been studied to a much lesser extent. It has been shown that they exert an influence on the anticoagulant system opposite to combined OCs. Progestin-only pills decreased the plasma levels of protein C, elevated the levels of total and free protein S and increased the sensitivity of plasma towards APC with more pronounced effects in women who used levonorgestrel-only preparations compared with users of desogestrel-only preparations [48]. Thus, it was hypothesized that the progestin component in the combined OC has an anti-thrombotic effect that counteracts the estrogen-induced prothrombotic changes. The data on the effects of progestin-only preparations on hemostatic parameters further indicate that levonorgestrel has a more potent antithrombotic effect than desogestrel. These observations can explain the more pronounced changes of hemostatic parameters in women using third-generation OCs and the higher risk of VT in third-generation pill users.
Overall hemostatic effects of OCs
Although the OC-induced thrombophilic alterations are rather small and the levels of coagulation factors mostly stay within the normal range, OCs appear to have a net prothrombotic effect that can explain the increased risk of VT in OC users, demonstrated in numerous epidemiological studies. For several years, it was argued that changes of coagulation parameters during pill use are too small to explain the thrombotic effect of OCs. However, it is now known that a small increase in the plasma levels of coagulation factors or relatively small decreases in the levels of anticoagulant proteins increase the risk of VT [65–75]. For instance, the prothrombin G20210A mutation, which, in the heterozygous state, is associated with an approximately 20% increase in the plasma prothrombin level, increases the risk of VT two- to threefold [65]. Therefore, the mean prothrombin level of heterozygous carriers of this mutation (approximately 120%) is well within the normal range, although is still associated with a two- to threefold increase in thrombotic risk.
The homeostasis of the coagulation system is maintained by complex mechanisms that allow compensation for the effects of unfavorable environmental factors. Recently, it has been shown that, in the healthy population, the plasma level of prothrombin correlates with that of protein S [76]. Although the mechanism of the prothrombotic effect of elevated prothrombin levels is not yet fully understood, evidence has been provided that at least part of this effect may be explained by inhibition of APC-mediated factor Va inactivation by prothrombin [77] and APC resistance [78]. Protein S was demonstrated to reduce prothrombin-induced APC resistance [76]. Thus, the effect of elevated prothrombin, which is an independent risk factor VT, is modulated by an opposite effect of protein S which, in normal individuals, simultaneously increases with prothrombin. Interestingly, in OC users, the correlation between these two proteins disappears since during OC use the level of prothrombin is increased, whereas the plasma level of protein S is decreased. As a result of the shift in the prothrombin/protein S ratio, the elevated level of prothrombin in OC users is not counterbalanced by an increase in protein S. Thus, women using OC are exposed to a higher risk of VT than nonusers, with an equally elevated concentration of prothrombin owing to the presence of the prothrombin G20210A mutation [76].
Some investigators dispute whether changes in hemostatic parameters determined during OC use by in vitro tests provide reliable information regarding VT risks and differences in risks between various OC preparations [79,80]. However, it appears that elevated levels of, for example, prothrombin [65], factor VIII [69,70] and factor IX [71] and decreased levels of protein S [73] and AT [73], determined by in vitro assays as well as APC resistance [81], are well established as risk factors for VT, even in the absence of thrombotic polymorphisms/mutations [65,81]. This also applies to acquired APC resistance resulting from OC use, which, when determined via the ETP-based assay, predicts an increased risk of VT independent of factor V Leiden [53]. In summary, the multiple changes of the hemostatic parameters that occur during OC use, which amplify each other, can very well explain the increased risk of VT in OC users and the risk differences between second- and third-generation OCs.
Conclusions
Resistance to APC is likely to be a pathophysiological basis for OC-induced thrombosis. The decreased sensitivity to the natural anticoagulant APC is at least partially caused by the multiple effects of OC on the hemostatic system, for instance, decreased plasma levels of protein S and TFPI and increased plasma prothrombin concentrations [37,38,42]. In the absence of hereditary risk factors for VT, these alterations lead to an APC-resistant phenotype, the severity of which is comparable with that observed in heterozygous carriers of the factor V Leiden mutation [48,54]. Nevertheless, the overall risk/benefit ratio of OC use remains favorable. The rare side effects are at least partially counterbalanced by the advantages of OC use, including not only prevention of undesired pregnancies and negative health consequences of the induced terminations of such pregnancies, but also their therapeutic effects. Worldwide, more than 100 million women use OCs for birth control and/or treatment of premenstrual syndrome, dysmenorrhea, endometriosis, menorrhagia and iron-deficiency anemia, and only relatively few of them experience any serious side effects. Moreover, among those who develop VT, most have additional thrombotic risk factors, such as an impaired genetic background or acquired risk factors such as obesity, malignancy or trauma.
Future perspective
In the past 10 years, great advances have been made in the understanding of the underlying mechanisms of the prothrombotic effects of OCs. This progress will facilitate the design of safer OC formulations and aid prudent prescription of OCs. Physicians who prescribe OCs should consider the specific benefits and adverse effects of the formulation and other personal risk factors for each individual patient. OCs that cause a higher risk of VT should be administered with caution in women who already have a high baseline risk of VT due to inherited or acquired disorders.
Executive summary
The association between the use of oral contraceptives (OCs) and an increased risk of venous thrombosis (VT) was recognized soon after the introduction of OCs in the 1960s.
Despite a lowering of the estrogen component in OCs, an elevated thrombotic risk remained, which, compared with nonusers, was estimated to be three- to sixfold higher in women who used so-called second-generation OCs containing 30–50 μg ethinylestradiol and the progestin levonorgestrel.
Surprisingly, the risk of VT increased again 1.5- to twofold when the progestin component of OC was changed from levonorgestrel to the so-called third-generation progestins, desogestrel or gestodene.
The biological basis of OC-induced VT has remained obscure for a long time.
In the 1990s, several risk factors for venous thrombosis (VT), such as elevated plasma prothrombin levels, decreased plasma levels of the anticoagulant proteins antithrombin, protein C and protein S, and resistance to activated protein C (APC), were identified.
The discovery of these risk factors shed new light on the association between the effects of OCs on the hemostatic system and the increased risk of VT in OC users.
It is now generally accepted that OC use increases the plasma level of prothrombin, decreases the level of protein S and overall, induces acquired APC resistance.
Together, these changes can explain why women who use OCs are exposed to an increased risk of VT.
Compared with second-generation OCs, third-generation OCs caused more pronounced changes in the plasma levels of prothrombin and protein S and in APC resistance.
Progestin-only preparations change several hemostatic parameters in a direction that is opposite to that observed with combined OCs.
It is hypothesized that ethinylestradiol has a thrombotic effect and progestins have a counterbalancing antithrombotic effect, the magnitude of which depends on the type of progestin.
Differences in the antithrombotic effects between desogestrel/gestodene and levonorgestrel can explain the more pronounced effects of third-generation OCs on hemostatic parameters.
Insight into the biological basis of OC-induced thrombosis and the discovery of new risk factors for VT will facilitate the development of safer OC formulations with an improved risk/benefit ratio and guide the choice of a proper method of contraception.
