Abstract
Pregnancy outcome among women with Type 1 or Type 2 diabetes is still significantly poorer than in the background population [1]. This review will focus on pregnant women with Type 1 diabetes. Optimal glycemic control is crucial in order to reduce the risk of congenital malformations, stillbirth, macrosomia, pre-eclampsia and preterm delivery [2–5]. The results of population-based cohort studies conducted in Europe, mainly using human insulin, demonstrate unequivocally that women with Type 1 diabetes have an increased risk of late fetal loss, with an approximate four- to five-fold increase in perinatal death and a four- to sixfold increase in stillbirth relative to the national background population [1–6].
Reports have attributed the increased risk of congenital abnormalities in diabetic mothers to poor metabolic control during the critical period of organogenesis in the first trimester of pregnancy [7,8]. Population-based cohort studies have reported incidences of congenital malformations ranging from two- to ten-times higher than the background population in the UK [6]. The risk of major congenital abnormalities among offspring of women with diabetes is predominantly accounted for by congenital heart disease, bone malformations and neural tube anomalies [1].
The rate of premature deliveries (defined as delivery before 37 weeks' gestation) are four- to five-fold higher among pregnant women with diabetes compared with the general maternity population [7]. The rate of preterm delivery in the UK diabetic population was 36%, compared with 7% for the general maternity population. Preterm infants of women with pregestational diabetes are at significantly greater risk of complications, including hyperbilirubinemia, respiratory disorders, hypertrophic cardiomyopathy and asphyxia, than those born to nondiabetic mothers [7].
Macrosomia is probably mediated by fetal hyperinsulinemia in response to maternal hyperglycemia. Recent studies report that rates of macrosomia (>90th percentile weight for gestational age) among infants of women with pregestational diabetes are 3.5–4.5-times greater than among infants of nondiabetic mothers [7]. In the UK, 21% of singleton babies of mothers with diabetes weighed over 4 kg, with 6% weighing more than 4.5 kg in the background population. The corresponding figures for singleton babies of nondiabetic mothers in England and Wales are 11% over 4 kg and 2% over 4.5 kg [7].
In the general population, macrosomia has been associated with a variety of obstetric complications, including caesarean birth, chorioamnionitis, shoulder dystocia, fourth-degree perineal lacerations and postpartum hemorrhage [8]. Recent studies of pregnant women with pregestational diabetes have reported associations between macrosomia and the incidence of neonatal hypoglycemia [7], shoulder dystocia and Erb's palsy [7].
Rates of maternal morbidity and mortality are also higher among women with diabetes. The death rate of pregnant women with Type 1 diabetes is 109-times greater than women in the general population and 3.4-times higher than nonpregnant Type 1 diabetic women [9]. Risk of hypoglycemia remains a barrier to achieving tight glycemic control with insulin therapy [9]. Hypoglycemic coma [10–12], traffic accidents [12] and death [9] due to severe hypoglycemia in pregnancy constitute significant problems. Severe hypoglycemia has been described to be four-times as frequent in early pregnancy compared with the period prior to pregnancy [11] and highest in gestational week 8–16 [10,12].
Several authors have attempted to link percentage reduction in hemoglobin (Hb)A1c with relative risk of adverse pregnancy outcomes. These studies most commonly focused on congenital malformations, reporting relative risks of 1.63–2.34 per 1% increase in HbA1c [13,14]. Conversely, the risk of adverse outcome is halved with each percentage of HbA1c reduction prior to pregnancy [15].
Hiilesmaa et al. followed up 638 pregnancies in women with Type 1 diabetes and found that the risk of pre-eclampsia was associated with glycemic control in early pregnancy and with an insufficient improvement in metabolic control during pregnancy [16]. In a study of 213 pregnant women with Type 1 diabetes HbA1c at 28 weeks of gestation (p < 0.001) was the best predictor of preterm delivery [17]. The adjusted odds ratio per 1% increment in hemoglobin A1c at 28 weeks of gestation was 2.8 (95% CI: 1.7–4.4).
Many of the increased maternal and fetal complications correlate with poor maternal glycemic control during pregnancy, and improvement of metabolic control using insulin treatment is important to prevent complications [1–6]. However, tightening glycemic control is associated with an increased risk of severe hypoglycemia [10–12], with a potential adverse maternal outcome including coma, seizures and maternal death [9–12]. This emphasises the need for new tools in the treatment of diabetes during pregnancy.
Insulin treatment
Diet and exercise are a cornerstone in the treatment of diabetes during pregnancy. In addition, women with Type 1 diabetes also require careful adjustments of insulin dose during pregnancy. The aims are normal pre- and post-prandial glucose levels and avoidance of severe hypoglycemia. HbA1c should be within normal range for pregnancy (<5.6%) [18,19]. Multiple insulin injections with fast-acting insulin at mealtimes and long-acting insulin one- to three-times daily is commonly used in these women. Even with the use of frequent home glucose monitoring, these goals for metabolic control during pregnancy are not reached [20].
Insulin analogues
Recombinant DNA and protein-engineering technologies, allowing the amino acid sequence of insulin to be modified, enabled the development of a series of rapid-acting insulin analogues with a reduced tendency to self-aggregate, affording faster absorption and, therefore, faster onset of action when injected [21]. The first, in 1996, was insulin lispro (Humalog®, Eli Lilly and Co, IN, USA) [22]. A second insulin analogue, insulin aspart (NovoRapid®, Novo Nordisk, Denmark), was introduced in 1999 [23]. The most recent addition to the rapid-acting insulin analogues is insulin glulisine (Sanofi Aventis, France), which was made available in 2005 [24].
The rapid absorption of these insulin analogues from the injection site is associated with a faster peak insulin concentration and a shorter duration of action than that achievable with regular human insulin [22–24]. This represents a more accurate simulation of physiological insulin release in response to a meal and results in lower postprandial glucose concentrations. The rapid-acting insulin analogues may be injected immediately before a meal and therefore require less mealtime planning compared with regular human insulin.
Later efforts in the development of insulin analogues were driven by the recognition that once-daily injections of intermediate and long-acting human insulin preparations are associated with an unpredictable action profile, including high variability in absorption and insulin peaks, which confer a high risk of nocturnal hypoglycemia unless food intake can be timed to compensate [25]. Insulin glargine (Lantus®, Aventis Pharmaceuticals) and insulin detemir (Levemir®, Novo Nordisk, Denmark), are approved for clinical use, but not in pregnancy. The peakless action profile of insulin glargine and detemir confers a significant risk reduction for nocturnal hypoglycemia and a more stable daily plasma glucose profile [26–28]. For insulin glargine, some studies have demonstrated no adverse effects on diabetic pregnancies [29,30].
Insulin lispro has been widely used in pregnancy. Several small observational or randomized studies using insulin lispro during pregnancy have been published [31–39]. The offspring of 496 women treated with insulin lispro before and during pregnancy have been carefully investigated retrospectively, demonstrating a rate of major congenital malformations of 5.4%, comparable to other large-scale studies of pregnant women receiving regular human insulin [39]. US authorities have approved the use insulin lispro in pregnancy. Regarding the long-acting insulin analogues, experiences in pregnancy to date are too limited to make any recommendations at present. Insulin aspart has been investigated during pregnancy with a large, prospective, randomized study and this article will focus on the data on insulin aspart used in Type 1 diabetes.
Insulin aspart
In an open-label, parallel-group multicenter study, a total of 322 women who were pregnant or planning pregnancy were randomized to receive insulin aspart (n = 157) or regular human insulin (n = 165) plus neutral protamine Hagedorn insulin as basal-bolus therapy during the whole pregnancy [40,41]. The risk of major hypoglycemia or major nocturnal hypoglycemia was 28 and 52% lower with insulin aspart compared with regular human insulin, respectively; although clinically meaningful, these differences did not reach statistical significance. The risk of less severe hypoglycemic episodes was similar between treatment groups. Most patients achieved an HbA1c level of 6.5% or less during the second and third trimesters, with no difference between the treatment groups.
Insulin aspart showed better control of postprandial hyperglycemic excursions than regular human insulin (Figure 1). Mean plasma glucose levels after breakfast were lower among patients receiving insulin aspart than those receiving regular human insulin at the end of the first (p = 0.044) and third trimesters (p = 0.0007). There was no difference between the treatment regimens in preprandial glucose control.
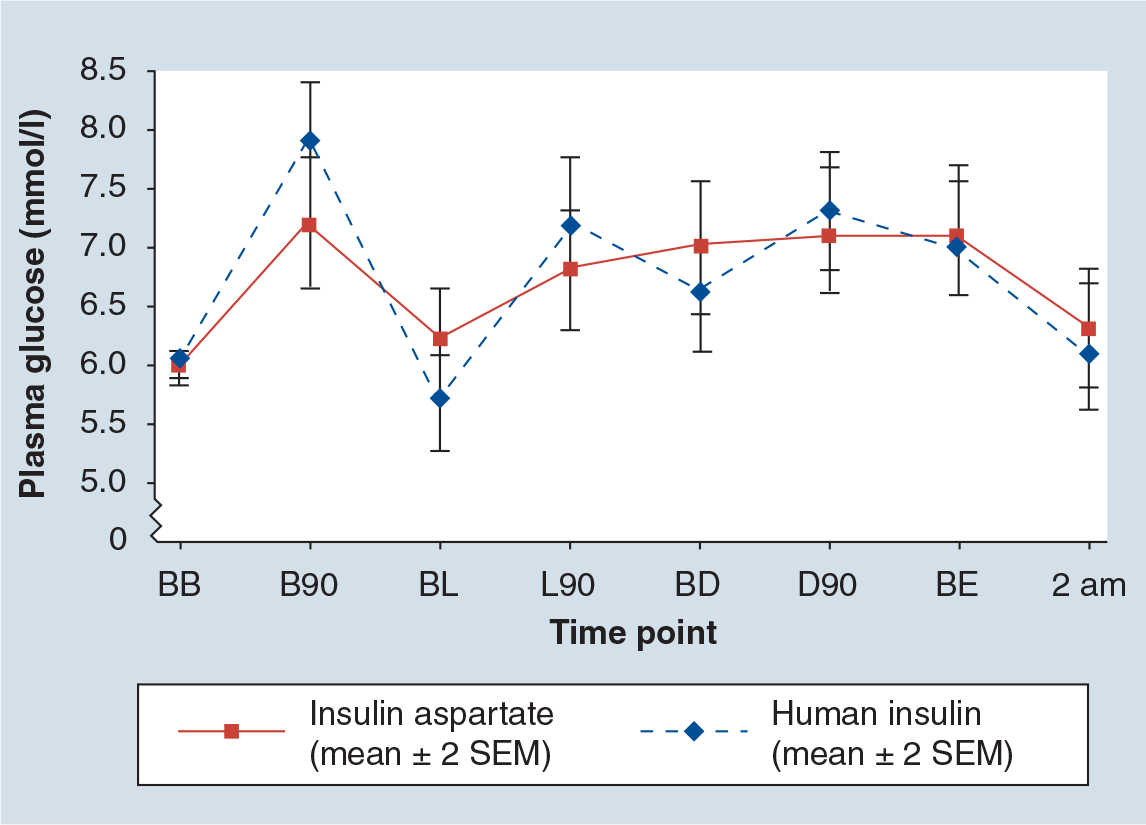
Mean eight-point plasma glucose profile.
In terms of maternal outcomes, both insulins were generally well tolerated, with a similar frequency of adverse events between the treatment groups. Patient satisfaction, assessed using the Diabetes Treatment Satisfaction Questionnaire, was significantly greater among those receiving insulin aspart than in the group assigned to regular human insulin (p = 0.031). The between-treatment difference was largely attributable to differences on two subitems of the Diabetes Treatment Satisfaction Questionnaire; namely, patients receiving insulin aspart were more satisfied with the flexibility of their treatment regimen and were more willing to continue with their assigned treatment [40].
In terms of fetal outcome, there were 137 and 131 live births and 14 and 21 fetal losses in the group administered insulin aspart and human insulin, respectively [41]. Perinatal mortality was 14 and 22 per 1000 births; the number of congenital malformations were six and nine; standard error of mean (SEM) birth weight corrected for gestational age was 3438 g [72] and 3555 g [73], (p = 0.09). Preterm delivery (before week 37) occurred in 20.3% (insulin aspart) and 30.6% (human insulin) of pregnancies (p = 0.05).
In addition, initiation of treatment with insulin aspart in pregnancy did not induce a rise in insulin antibodies and a transfer of measurable amounts of insulin aspart to the fetal circulation was not detected [42].
Focusing on four parameters of clinical outcomes: severe hypoglycemia, malformations, fetal losses and preterm deliveries, it is worth noting that even though none of these outcomes reached a significant difference, all four clinically important outcomes had 30% lower incidence in the insulin aspart group compared with the human insulin group (Figure 2).

Outcome in women with Type 1 diabetes randomized to insulin aspart (n = 151) or human insulin (n = 152).
Conclusion
The data presented suggest that insulin aspart is safe in pregnancy for mother and fetus and offers benefits in controlling postprandial glucose levels and quality of life during pregnancy. Insulin aspart is now approved for use in pregnancy by the US and European health authorities.
Future perspective
The fast-acting insulin analogues insulin aspart and insulin lispro are now regarded as safe and are widely used during pregnancy. The safety and efficacy of long-acting insulin analogues such as insulin detemir and insulin glargine have to be tested further in pregnancy. A large, randomized study of insulin detemir in pregnancy complicated by Type 1 diabetes was initiated in 2007 and the results will be presented in 2010. Continous glucose monitoring might be a useful tool in obtaining near-normal glucose levels with reduced levels of severe hypoglycemia, and clinical trials are warranted in this field.
Executive Summary
Both insulin aspart and human insulin are regarded as safe in pregnancy.
Insulin aspart improves postprandial glucose control.
Insulin aspart might induce fewer episodes of severe hypoglycemia.
Insulin aspart might be associated with fewer preterm deliveries.
Initiating insulin aspart in pregnancy did not induce insulin anitbodies.
Footnotes
The author has no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending, or royalties.
No writing assistance was utilized in the production of this manuscript.
