Abstract
Addiction research has historically neglected research on women, and most studies have been conducted on men only, with the concluding results generalized to the female population. The role of sex differences in vulnerability to drug abuse, their repercussions on prevention and treatment strategies all require detailed studies, as does the progression from recreational drug use to dependence. This review synthesizes evidence of gender differences in drug addiction, with particular emphasis on women's health and implications. We first reviewed behavioral studies showing sex differences in the preference for and self-administration of licit (i.e., alcohol and nicotine) and illicit (i.e., cocaine, amphetamine, heroin and cannabis) substances as revealed by animal models of addiction. Clinical studies demonstrating differences between men and women in craving, drug use, abstinence and relapse will then be examined. For both animal and human studies, the effects of hormones and estrous/menstrual cycle will be reviewed. Finally, neurobiological factors underlying gender differences in vulnerability to drug addiction (i.e., brain morphology and neurotransmission) and need for gender-specific detoxification treatments will be discussed.
Keywords
Drug addiction is widely recognized as a chronic relapsing illness that affects both women and men. However, studies on drug abuse and dependence typically involve men as predominant, if not exclusive, research subjects. Despite the huge progress made during the last decade in the addiction field, especially in the individuation of the brain pathways and mechanisms engaged, relatively little is known regarding the origins, treatments and prevention of substance-abuse disorders among women. The paucity of clinical studies specifically focused on the female population represents a serious concern, as initiation to drug use is triggered by factors that may differ between men and women, and dependence may develop differently in women than in men and likely require different treatment approaches.
Animal studies have revealed important sex-dependent differences in addiction, as females and males often differ for motivation to obtain a specific drug, levels of drug intake, or the propensity to reinitiate drug-seeking behavior following a period of abstinence. In humans, scarce information exists regarding social, cultural and environmental risk factors specific for men and women. This is probably owing to the fact that scientists began to be aware of the exclusion of women from clinical trials and of the insufficient attention paid to possible gender differences by international research only from 1990 onwards.
Retrospective reports suggest that women dependent on opioids, cannabis and alcohol may have an accelerated progression from initiation of use to treatment entry [1], suggesting an escalating effect that may be greater in women than in men. However, it has not yet been determined if women may react differently from men to stress agents (i.e., divorce or lack of work), nor whether domestic violence, experiences of sexual abuse, low self-confidence, or anxiety arising from having multiple roles (i.e., working mother) may predispose women to use drugs. Among the numerous studies carried out on abstinent subjects at risk of relapse, only few paid attention to possible gender differences, and even fewer were those considering the influence of the menstrual cycle in female patients. Sex-related differences are likely to exist with regards to the widespread habit of using two or more drugs concomitantly (i.e., polydrug abuse) as well as in the comorbidity between drug addiction and other diseases (i.e., chronic pain and psychiatric diseases) or in other forms of addiction, such as binge eating [2] or gambling [3,4].
Brain mechanisms underlying drug craving and abuse can be analyzed by means of animal models, which proved to be fundamental in the understanding of the abuse liability of drugs (intravenous drug self-administration, conditioned place preference procedures), the subjective responses to administered drugs (drug-discrimination tasks), the behavioral disturbances that are associated with the withdrawal states (abrupt termination of chronic drug exposure, administration of selective antagonists after chronic treatment), and relapse phenomena (reinstatement models of extinguished drug-seeking behavior). Most of these studies have used rodents (rats and mice) as subjects, but data are also available from studies using other animal species (i.e., monkeys) and human volunteers as subjects for abstinence-related studies.
Sex differences in drug addiction: preclinical evidence
Among animal models, oral and intravenous self-administration paradigms are those more closely resembling the different phases of the addiction cycle in humans, thereby greatly improving our understanding of the brain mechanisms implicated. In self-administration experiments, animals are given the opportunity to perform an action, usually to press a lever or to put their nose in a hole, in order to receive a contingent presentation of the drug. If animals reach a stable level of drug intake, brain lesions or pharmacological manipulations can be performed to determine whether it is possible to affect the reinforcing properties of the drug. Self-administration procedures have been especially useful for investigating the ‘acquisition phase’ of drug addiction, owing to the ethical implications of conducting experiments on the initiation of illicit drug use in drug-naive humans.
Whatever the specific drug available in self-administration studies, it appears that female rats are, in general, more responsive during all phases of the addiction cycle, and that sex differences are more consistently apparent in the transition phases of drug addiction, such as acquisition or relapse, than in the steady state, that is, maintenance phase (see Table 1). More specifically, female rats' operant behavior is typically more robust than males' during acquisition of drug self-administration [5], escalation of drug intake [6] and reinstatement of extinguished drug-seeking behavior [7]. Interestingly, sex differences also exist in drug-induced behavioral effects other than self-administration, with females displaying an enhanced behavioral responsiveness to a variety of these drugs. For example, female rats show enhanced cocaine- [8], amphetamine- [9] and methamphetamine- [10] induced locomotive activity relative to male rats.
Sex differences in female and male laboratory animals in drug self-administration, and influence of the estrous cycle on drug intake.

F: Female; M: Male.
Remarkably, studies investigating sex differences in reward and craving for drugs provided compelling evidence for a key role played by the estrous cycle in such differences, with estrogen administration and ovariectomy typically increasing and dampening, respectively, operant behavior in female animals [11–13].
Licit drugs: nicotine & alcohol
Under particular experimental conditions, sex differences can be observed in nicotine intake, as female rats acquire nicotine self-administration behavior more rapidly than males (although this difference only occurs at low nicotine doses), show a shorter latency to the first drug infusion and work significantly more than males [14]. Intriguingly, while few differences were found between male and female rats in the number of active responses, number of infusions, or total intake of nicotine under a fixed-ratio schedule of reinforcement, females reached higher break points on a progressive ratio schedule [14], evocative of a higher motivation to self-administer nicotine. Adolescent female mice also consume more nicotine than adolescent males, suggesting that female periadolescent animals may be more sensitive to the effects of nicotine than males [15]. The incentive salience of nicotine-paired cues (i.e., light or sound previously associated with drug delivery) may also differ as a function of sex, as comparisons between male and female rats revealed subtle but significant sex differences in operant responding not only for nicotine but also for nicotine-paired visual stimuli [16]. However, females do not change nicotine self-administration behavior as a function of the estrous cycle under either fixed- or progressive-ratio schedules [14], at least in food-restricted animals previously trained to press a lever for food.
Animal models for the study of alcoholism reveal that, in general, females drink more alcohol than males and have sex-related differences in drinking patterns and responses to alcohol. For example, female vervet monkeys display a higher frequency of alcohol intake than males, with juveniles showing higher frequencies of intake than adults [17], while female mice consume more ethanol than males and exhibit increased ethanol consumption across days of drinking experience [18]. Moreover, although male and female rats initiate alcohol drinking at similar rates, females drink more alcohol than males and maintain their preference for ethanol over a longer period of time [19]. However, other authors reported that male rats consume higher alcohol concentrations and reach higher blood alcohol levels than females [20].
With regards to the influence of hormones on alcohol intake, it was found that either ovariectomy or progesterone replacement in ovariectomized rats resulted in ethanol-drinking patterns that were not different from those observed in intact females [21]. Similarly, no major differences were found between gonadectomized and sham-operated animals during long-term ethanol intake [22]. The fact that neither ovariectomy nor castration affects ethanol self-administration, along with the inconsistent results observed after replacement steroid treatments, indicates that sex-steroid milieu has little influence on ethanol-drinking behavior. Notably, in humans the difference between the sexes is the opposite of rodents, with males drinking more than females (see Table 2), indicating that sex differences and differences between humans and rodents in drinking behavior may rely on a complex interaction of social, genetic, hormonal, neurobiological and environmental factors.
Gender differences in women and men in drug addiction and dependence, and influence of the menstrual cycle on drug intake.
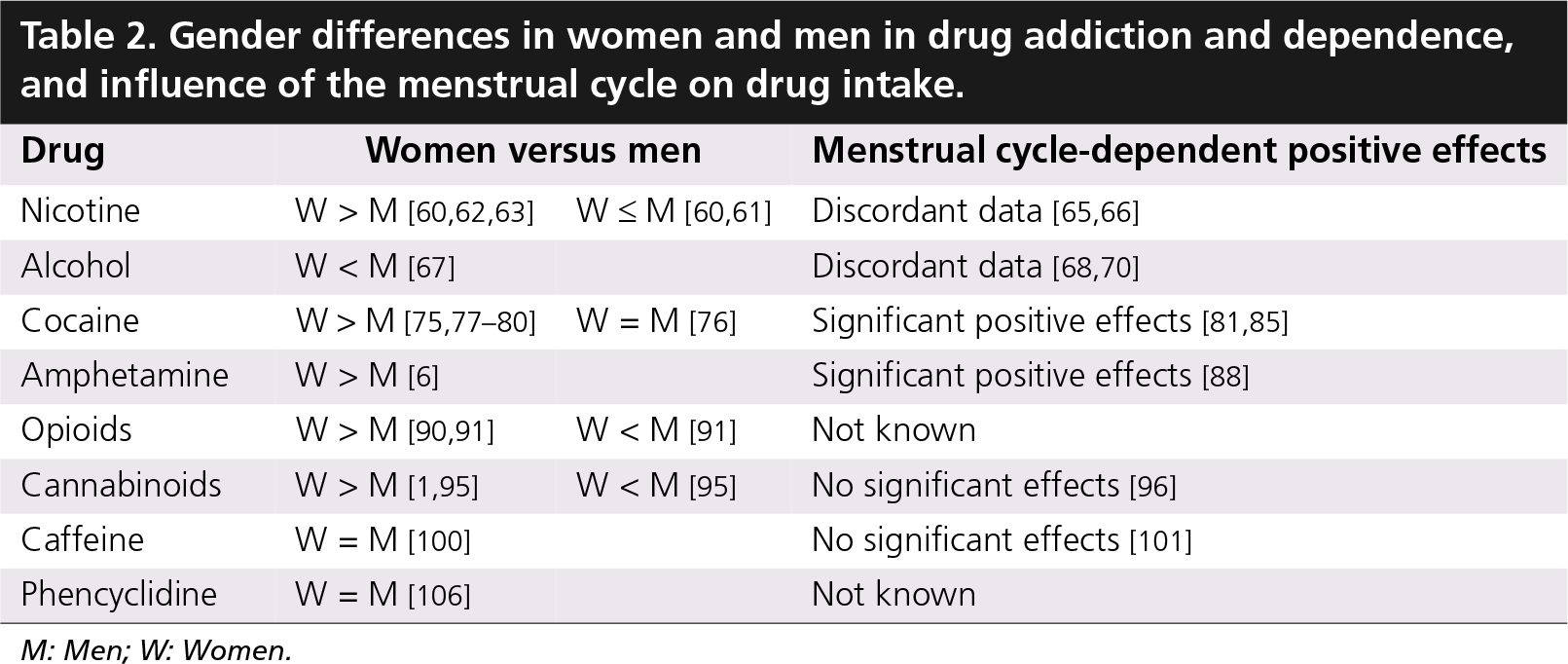
M: Men; W: Women.
Illicit drugs: cocaine, amphetamines & opioids
Several studies demonstrate that female rats acquire cocaine self-administration faster and self-administer significantly more cocaine than male rats under fixed-ratio schedules of reinforcement [5], display marked increases in motivation to obtain cocaine when given extended access to cocaine [23], and show a reduction in levels of responding for cocaine under progressive-ratio schedules of reinforcement [24]. Female rats also show greater reinstatement of extinguished cocaine-reinforced responding than males [4]. Similarly, female mice self-administer slightly greater levels of cocaine than male mice [25].
However, different results are also reported by an interesting study showing no significant differences between females and males in the shape or position of the cocaine dose-effect curve when various doses of cocaine are examined under a fixed schedule of reinforcement [26], thus indicating that the unit dose of cocaine and the schedule of reinforcement are important variables in studies of sex-dependent effects on cocaine self-administration.
Importantly, there are differences in the effects of circulating ovarian hormones on acquisition of cocaine self-administration, with estradiol administration enhancing acquisition of cocaine self-administration in ovariectomized female rats, and concurrent administration of progesterone with estradiol inhibiting this effect [27–29]. However, once they acquire stable cocaine self-administration behavior, the amount of drug consumed by female rats during estrous is significantly less than that consumed by males, while that consumed during proestrous and metestrous-diestrus is not significantly different [30]. Moreover, with respect to nonestrous female rats, females in estrus show greater responding during self-administration, extinction and cocaine-primed reinstatement [31,32]. Thus, variability in hormone levels during estrous may be an important factor giving rise to the observed sex differences in cocaine intake. However, when female monkeys were given access to cocaine at higher schedules of reinforcement (i.e., FR-30), estradiol administration did not alter the abuse-related effects of cocaine, as measured by self-administration and drug-discrimination procedures [33].
With regards to amphetamine and its derivatives, a greater percentage of female versus male rats meet the methamphetamine acquisition criterion [34], and they do so in significantly fewer days compared with males. Female rats also display increased responding for a range of doses of methamphetamine, and maintain greater levels of drug intake compared with male rats [34].
Notably, when estrogen is high and progesterone is low (i.e., during estrous), rats show increased locomotor activity, rotational behavior, and stereotypes in response to amphetamine, while ovariectomy attenuates amphetamine-induced behavioral responses [35].
Gender differences were observed in numerous aspects of the pharmacology of opioids, including a higher sensitivity in male rats to their antinociceptive activity [36], discriminative stimulus effects [37] and their ability to generate physical dependence [38], as well as an enhanced responsiveness in females to their positive reinforcing properties, as assessed by conditioned place preference [39] and intravenous self-administration [40] procedures. Females acquire self-administration of opioids at a much faster rate than males, and self-administer more heroin and morphine both in terms of the absolute amount of the self-infused drug and the amount of work they expend to obtain it [5].
Intriguingly, although estradiol injection to ovariectomized female rats facilitates acquisition of heroin self-administration and enhances levels of drug intake with respect to nontreated ovariectomized females, no differences in the self-administration of heroin or morphine are engendered by the estrous cycle [41].
Other drugs: cannabis, caffeine & phencyclidine
Although difficult to demonstrate in animal models of addiction, the addictive properties of cannabis derivatives have been assessed in self-administration [42–45] and relapse [46] studies. That is, both rodents and squirrel monkeys displayed voluntary cannabinoid intake, and reinitiated cannabinoid-seeking behavior following a period of drug withdrawal when primed with cannabinoid agonists. A very recent study demonstrates that female rats are more sensitive to the rewarding effects of cannabinoids than males, and that the ovarian cycle is a determining factor underlying these sex-related differences [47]. Indeed, when compared across all phases of cannabinoid self-administration, intact females show faster acquisition and higher levels of drug intake with respect to both males and ovariectomized females [47], thus highlighting the importance of estrogens in the perception of the motivational and rewarding value of cannabinoids. Accordingly, an endocannabinoid link between steroids and neuronal signaling transmission has been detected in both the periphery [48] and the brain [49].
Interestingly, gender differences were found in the effects of perinatal exposure to δ-9-tetrahydrocannabinol (THC), primarily responsible for the psychotropic effects of Cannabis sativa, on intravenous morphine self-administration in adulthood. Indeed, adult female rats born from mothers treated with THC during gestation and lactation exhibit a significant increase in the acquisition rate of morphine self-administration behavior compared with females born from vehicle-exposed mothers and to THC-exposed male offspring [50]. Such a significant influence of cannabinoid prenatal exposure on the rewarding value of morphine in adulthood is not surprising, as the endocannabinoid system is well known to strictly interact with the endogenous opioid system in the reciprocal modulation of the reinforcing, and hence addictive, effects [51–54].
Similarly to cannabis derivatives, drug-experienced female rodents also self-administer significantly more caffeine [55] compared with males. Finally, female monkeys consume significantly greater amounts of phencyclidine (PCP) than water, while males do not show significant differences between PCP and water intake [56].
Gender differences in drug addiction: clinical evidence
In the human population, the first drug experienced by a drug-naive individual is usually alcohol or nicotine, that is, a licit drug. However, a well-developed sequence from licit to illicit drug abuse was determined, although with significant differences between the sexes [57]. Comparing women and men on intake patterns of alcohol, tobacco, cocaine, amphetamine, opioids, cannabis, caffeine and phencyclidine use, it is proven that women take less time to progress to dependence than men [1]. Long- and short-term effects of drugs may impose differential consequences on women and men's health, and in many cases women appear to be more sensitive to the adverse effects of drugs than men. Importantly, although basic research indicates that in female animals the estrous cycle is able to increase motivation to self-administer most drugs of abuse (see Table 1), it is still not completely clarified whether the menstrual cycle has a similar effect on women's drug-taking behavior (see Table 2).
Gender differences have also been demonstrated in behaviors other than addiction, with anxiety, panic disorders and post-traumatic stress disorders being more prevalent among women than men [58,59]. Phobia, depression and other psychiatric conditions can all encourage abuse of mood-altering drugs and lead to ingestion of alcohol or cigarette smoke in an attempt to self-medicate. The high degree of comorbidity between drug abuse and psychiatric or mood disorders is therefore not surprising.
Licit drugs: nicotine & alcohol
While alcohol seems to be crucial in males' progression to illicit drug abuse, nicotine smoking represents the female's first step of this progression. Compared with men, female smokers show less sensitivity to the discriminative stimulus effects of nicotine and to the change in nicotine dose via nasal spray [60], while subjective and reinforcing effects of smoking are less influenced in women by the nicotine dose present in the cigarette [61]. By contrast, female smokers consistently show shorter or less frequent abstinence periods compared with men, along with greater sensitivity to nonpharmacological stimuli that are associated with cigarette use [60]. For example, in a study on gender differences in the contribution of visual and olfactory cigarette-associated stimuli to smoking, blockade of olfactory stimuli significantly reduces subjective hedonic ratings in women, but not in men [62].
Women are more vulnerable than men to develop dependence on nicotine, and over 30% of women who have experienced cigarette smoking meet the criteria for dependence [63]. Gender differences in vulnerability to nicotine addiction and success in quitting smoking could be related to factors of physiological (i.e., differential sensitivity and tolerance to nicotine, greater withdrawal symptoms, timing of quit attempts in relation to the menstrual cycle) and/or psychological (i.e., fear of weight gain, need for social support, depression and negative states) nature [64].
Importantly, withdrawal and craving in female smokers may vary as a function of the menstrual phase, while oral contraceptive use and estrogen-replacement therapy may affect intake and effects of nicotine [65]. Nicotine and cotinine metabolism is faster in women than in men, and it is faster in women receiving oral contraceptives compared with those who do not take pills, thus suggesting that estrogens may accelerate nicotine metabolism and therefore affect smoking behaviors in women [66].
Discrepancies between humans and rodents in alcohol-drinking behavior and responses to alcohol were reported, as alcoholism and a heavy drinking habit are generally higher among men than women. Numerous differences between men and women in the rate of alcohol absorption and metabolism are likely to be genetic in origin and may influence the development of alcoholism. Women consume lower levels of alcohol and are less likely than men to drink daily or to engage in binge patterns of use. However, women are more sensitive than men to the physiological effects of alcohol, achieve higher blood alcohol concentrations, report feeling more intoxicated, develop brain damage more readily than men and show higher vulnerability to alcohol dependence [67].
The issue of whether the physiological responsiveness of women to alcohol varies during the menstrual cycle, owing to changes in levels of sex-steroid hormones, is still contradictory (see Table 2). Some women develop alcohol dependence as a result of drinking to alleviate premenstrual dysphoria [68]. Other subjects declare that they have more negative effects during menstruation (not premenstruation) and that they drink alone more frequently during this phase to relieve tension or negative moods [69]. Women who drink more tend to be significantly more extroverted, spontaneous, carefree and open to change. By contrast, women who report greater overall menstrual distress tend to be less capable and secure, and report a greater number of emotional and psychological problems. Therefore, it could be possible that a relationship between hormone levels and ethanol effects (and, hence, alcohol drinking) exists in women with strong mood variations across their cycle, such as those with premenstrual syndrome. In line with this, women with significant premenstrual dysphoria might consume more ethanol during the premenstrual (late-luteal) phase [69]. However, the alcoholism-menstrual cycle hypothesis is challenged by few studies reporting no correlation between alcohol consumption and menstrual distress, with women not drinking more during any menstrual phase and drinking not being correlated with negative mood states [70]. This latter study also reports that the menstrual cycle phase does not significantly modify the direct subjective, behavioral and physiological effects of ethanol across the menstrual cycle, nor does it alter choice for additional ethanol drinks.
Whatever the relationship is between alcohol use and the menstrual cycle, many of the medical consequences of excessive alcohol consumption develop more rapidly among women than among men [71]. For example, alcoholic women develop cirrhosis [72], alcohol-induced weakening of the heart muscle (i.e., cardiomyopathy) [73], and nerve damage in the body's extremities (i.e., peripheral neuropathy) [74] after fewer years of heavy drinking than alcoholic men.
Illicit drugs: cocaine, amphetamines & opioids
With regard to psychostimulants, controversial data have been obtained when comparing cocaine use between women and men. Occasional cocaine female users typically report greater cocaine use than men, although they take longer to detect the subjective effects of the drug and report less euphoria and dysphoria [75]. Males are more likely than females to have opportunities to try cocaine, yet both sexes progress to actual use at an equal rate once exposure to cocaine has occurred [76]. Although no sex differences in rates of cocaine use in adulthood are found, cocaine use is higher in adolescent females than in adolescent males, with adolescent females being more likely to use cocaine at an earlier age and with a greater frequency [77]. Interestingly, women report shorter abstinence periods between cocaine use than men [78], experience more nervousness after intranasal administration of cocaine [78], and have more severe drug use at intake [79] and stronger craving in response to cocaine cues compared with men [80].
A woman's response to cocaine may vary with the menstrual cycle. Females have greater subjective responses to cocaine in the follicular phase of the menstrual cycle, that is, when estradiol is elevated and progesterone is minimal, than they do during the luteal phase, that is, when both estradiol and progesterone are elevated [81,82]. In conjunction with the rodent literature, these findings indicate that estradiol enhances the effects of cocaine. Interestingly, administration of progesterone in women during the follicular phase attenuates the positive subjective effects of cocaine relative to their normal luteal phase [83], whereas only minimal changes are observed in men [84]. These results indicate that progesterone modulates the response to cocaine in women and suggests that fluctuations in endogenous progesterone levels may account for some of the sex differences observed in humans [85]. However, although it is known that in female animals the estrous cycle typically increases motivation to self-administer cocaine, human experiments are needed to determine whether the menstrual cycle has a similar, motivating effect on women's cocaine intake.
Relative to cocaine use, the use of amphetamines appears to induce a faster progression towards regular use and subsequent need for treatment as, compared with cocaine users, methamphetamine users exhibit a shorter period of time from first use to regular use, and from first use to treatment entry. Human response to repeated low doses of amphetamine shows that the subjective ratings of vigor and euphoria induced by this drug are generally greater in women than men, although one study reported that following an amphetamine challenge, men display higher ratings of the positive effects and greater striatal dopamine release than women [86]. Women are typically more susceptible to the initiation of methamphetamine use, and may be more motivated to obtain methamphetamine once they have been introduced to the drug's effects [6]. However, when examining treatment outcomes among methamphetamine-abusing patients, women demonstrate greater improvement from baseline to follow-up, even though they are unemployed, with childcare responsibilities or physically abused [87].
Amphetamine-induced subjective stimulation varies across the menstrual cycle and is enhanced when estrogen levels are high and progesterone levels are low, that is, during the follicular phase. In the presence of both estrogen and progesterone, that is, during the luteal phase, estrogen levels are not related to the effects of amphetamine [88]. Thus, estrogen may enhance the subjective responses to amphetamine in women, but this effect may be masked in the presence of progesterone. If so, one could argue that women who initially use amphetamines for recreational purposes during the follicular phase may be more likely to repeat use of the drug because of its stronger effects, while women who are trying to abstain from drug use may be more likely to succeed if the initial abstinence is scheduled to occur during the luteal phase, when the drug effects are less potent.
Compared with men, women addicted to opioids have an earlier onset of heroin use as well as an overall greater level of psychiatric distress [89]. However, once dependence develops, addicted women report significantly lower levels of conduct problems and higher levels of internalizing problems during childhood and, once adult, are no ‘sicker’ or ‘more impaired’ than males [89]. Notably, unlike men, the initial use of heroin by women is highly influenced by a spouse or partner who is often a daily heroin user [90]. In both sexes, opioids use is strictly correlated with stress: as increase in stress levels enhances the likelihood of opioid use and relapse. That is, the major problem in treating opiate addiction is the occurrence of relapsing episodes within a few months after withdrawal. Intriguingly, men and women differ in complex ways in responsiveness to drug-associated cues that are likely to trigger relapse to heroin. For example, presentation of script-guided imagery of heroin-related cue situations elicits more heroin craving, sadness and cardiovascular changes in women than in men, while presentation of heroin-associated paraphernalia (i.e., needles, syringes, spoons, aluminum foil) does not produce gender differences in craving, although it produces more anger in men than in women [91].
It is known that heroin-dependent women have substantial amounts of menstrual-cycle dysfunctions under either no treatment or early in methadone maintenance [92]. Opioids probably affect menstruation via disruption of the hypothalamic-pituitary-ovarian axis with changes in gonadotropin level, which can lead to amenorrhea and oligomenorrhea. Despite its depressive effects on pituitary hormones, no clear effects of ovarian hormones were reported on women's total intake of heroin. Subjects addicted to heroin present a significant reduction of testosterone and dihydrotestosterone concomitant with higher plasma concentrations of the drug, which returns to the initial levels after the decrease of heroin concentration [93].
Other drugs: cannabis, caffeine & phencyclidine
For both sexes, the first illicit drug most frequently tried is marijuana, but men are more likely to use cannabis than women, suggesting a lower lifetime risk for cannabis dependence among women. However, although the severity of cannabis dependence in women is comparable with that of men, gender-dependent effects are found in the progression of cannabis dependence [1]. Indeed, women experience fewer years of regular use of cannabis before entering treatment, thus showing an accelerated progression to treatment entry. Gender differences are more evident when looking at factors associated with the onset of cannabis use among adolescents. Indeed, although adolescent females are currently overtaking adolescent males in their rate of initiation of cannabis use [94], a recent study demonstrates that the onset of cannabis use is associated with a prior history of tobacco smoking (male > female), alcohol consumption (male > female), antisocial behavior (male ≥ female), intention to use drugs (female ≥ male), drug use among friends (female > male) and spending leisure time in bars or discos (female > male). Moreover, attending state schools, low academic performance and living in a single-parent family also independently correlate with cannabis use among girls [95].
With regards to the influence of menstrual-cycle phases on cannabis dependence, no effect of cycle phase seems to exist on marijuana use [96], and no significant differences in heart rate or changes in mood are observed across the cycle after smoking marijuana [97]. However, marijuana smoking suppresses plasma luteinizing hormone levels in women during the luteal phase of the menstrual cycle [98], while women who increase marijuana use during the premenstruum report significantly greater premenstrual dysphoria than women whose marijuana use decreases or remains the same [99].
Caffeine acts as a positive reinforcer in both men and women, although women show a higher tendency to develop dependence on caffeine. There are no sex differences in the hemodynamic responses to caffeine consumption as well as in the pattern of stress responses following caffeine ingestion [100]. Similarly, when the role of the menstrual-cycle phase in response to caffeine is examined, it turns out that self-reported caffeine intake does not vary systematically across the menstrual cycle in healthy women [101]. In turn, the menstrual-cycle phase does not affect plasma levels peak concentration, half-life, clearance or distribution of orally administered caffeine [102]. Accordingly, gender and physical exercise, with or without an additional thermal stress, has no effect on the pharmacokinetic measurements or urine caffeine in men and women, and only a trend for higher plasma caffeine and lower plasma paraxanthine concentrations is found in women [103]. However, oral contraceptive steroids impair caffeine disposition and elimination [104]. Some authors have hypothesized that caffeine elimination may be slowed in the late luteal phase, prior to the onset of menstruation, leading to increased accumulation of caffeine with repeated coffees during the day [105]. However, consequent effects may be too small to be of clinical significance in most women.
Finally, a study comparing use of PCP with cocaine in jail detainees reveals that female PCP users reported more dysphoria and aggressiveness when not taking PCP, while men were more likely to report aggressive behavior and dysphoria under the influence of the drug [106], suggesting that men who prefer PCP to cocaine may be self-stimulating and women who prefer PCP may be attempting to self-medicate.
Putative factors underlying gender differences in drug addiction
Many individual biological, environmental, genetic and psychosocial features normally affect health and disease in men and women. There is likely a biological basis of sex differences in vulnerability to drug abuse as well as in all phases of the addiction cycle [107,108]. Females appear more vulnerable than males during the transition periods of drug use that are characteristic of drug addiction and relapse, and in many aspects seem to be more sensitive to the rewarding effects of drugs than males. Research has just begun to focus the major factors that could provide a neurobiological basis for gender-based differences in drug addiction.
Ovarian hormones
Estrogens and progesterone are not just sex hormones that influence ovulation and reproduction; they also affect a large number of cognitive and affective functions. Presumably, sex differences in the effects of addictive drugs would reflect hormonal differences between women and men and, in turn, addictive drugs may affect hormone release and metabolism. In line with this assumption, alcohol and cocaine use are associated with disruption of the normal menstrual cycle and compromising of female reproduction, possibly through interference with the release of luteinizing and adrenocorticotropic hormones [109]. On the other hand, hormones represent a key factor underlying nicotine [110], opioids [111] and cannabinoid [47] intake, while increases in the levels of sex steroids due to pregnancy or oral contraceptive steroid use significantly decrease the rate at which caffeine is eliminated from the body [105].
Brain morphology & neurotransmission
Among humans and laboratory animals there are manifest sex-dependent differences in brain morphology and neurotransmitter/neuromodulator systems. The adult human brain is characterized by sexual dimorphisms, although very little is known regarding how these dimorphisms develop.
Gender-specific differences have been found in brain weight, brain tissue volume and composition (proportions of grey and white matter), and measures of cortical surface anatomy. Adult males have more axosomatic synapses and fewer dendritic spine synapses than adult females, and these sex differences are a result of gonadal steroid exposure during the sensitive period of sexual differentiation [112]. Significant morphological differences may be detected between men and women in brain areas critically involved in craving, addiction and relapse, such as the cerebral cortex, with females showing a larger extent of cortical neuropil and lower neuronal numbers [113], the medial amygdala, whose volume is approximately 20% smaller in females [114], and the caudate putamen and hippocampus, whose volumes are larger in females than in males [115]. Evidence was also provided for gender differences in the expression and functionality of brain receptors normally activated by addictive drugs. For example, density and regional distribution of μ-opioid receptors vary between the sexes and, in females, across the ovarian cycle [116], while women have a higher number of D2-like receptors in the frontal cortex than men [117].
The brain is one of the primary sites of hormonal interaction, and it is conceivable that steroids and ovarian hormones directly alter neurotransmission. Estrogen exerts profound effects on mood, mental state and memory by acting on monoamine (i.e., dopamine, serotonin) and neuropeptide transmitter mechanisms in the brain [118], while steroids exert profound influences on aminoacid transmitters, GABA and glutamate [112]. Given that ovarian hormones exert a physiological influence on dopaminergic systems, it is expected that hormone levels can modulate behavioral responses to dopaminergic drugs. Estrogen and progesterone appear to have direct and opposing actions on brain monoamine systems, which suggests that monoaminergic drugs, such as amphetamine, may produce differential effects in men and women, and in women across the menstrual cycle. Both hormones interact with γ-aminobutyric acid and dopamine systems, each of which is crucially involved in the behavioral and physiological effects of ethanol. Therefore, it is possible that estrogen and progesterone alter the subjective, behavioral or physiological effects of ethanol in women at different phases of the menstrual cycle.
Others
Besides hormones and brain dimorphism, other factors are likely to contribute to gender differences in drug addiction, among which drug pharmacodynamics and pharmacokinetics deserve special mention.
Most psychoactive drugs have a lipophilic nature and are therefore fat-soluble, with absorption strictly depending on the amount of time that the drugs are in the GI tract. Thus, sex differences in the effect of drugs of abuse might be due, at least in part, to differences in muscle mass and fat tissue distribution between women and men, as well as in the gastric emptying time, which undergoes significant changes during the menstrual cycle.
Intriguingly, basic research shows that female mice may metabolize nicotine more quickly or eliminate cotinine more slowly than males, which would result in increased nicotine consumption by females to maintain steady plasma nicotine levels [119]. Sex-dependency in the pharmacokinetics and pharmacodynamics of amphetamines is also known in rats [120]. Body composition, volume of distribution, level of gastric and hepatic alcohol dehydrogenase, gastric absorption, capacity of the microsomal ethanol-oxidizing system, and putative hormonal effects on achieved blood alcohol levels are all key features underlying gender differences in alcohol addiction and dependence [121].
Finally, it should be kept in mind that nicotine smoking represents a serious risk factor of coronary heart disease for women using oral contraceptives [122], while hormone-replacement therapy may compromise bone quality and increase osteoporosis risk in alcohol-dependent women [123].
Gender-specific prevention & treatment strategies
Women and men have different physiological responses to drugs and medications, and may develop different manifestations of disease as a product of drug abuse. Clinical practice demonstrates that women may respond to treatment strategies that are different from those that men respond to, and may respond less to treatments that are successful with men. Women typically begin using substances later than men do, are strongly influenced by partners to use, report different reasons for maintaining the use of the substances (i.e., self-medication) and enter treatment earlier in the course of their illness than do men. While men tend to medicate themselves with alcohol and other drugs, women tend to seek medical help for emotional problems, thus explaining why more women than men seek treatment for mood disorders or other medical problems. However, only a smaller proportion of women seek treatment for drug abuse and, in general, women are less likely than men to accept enrolment in research studies. Accordingly, women involved with the criminal-justice system are typically no more likely than their male counterparts to report a need for treatment [124], and are less likely to participate in treatment [125]. However, once entering treatment programs, addicted women are more likely than men to complete them, and appear more responsive to psychosocial and behavioral treatments. Programs aimed at promoting positive life options and increasing socialization and self-esteem are particularly effective in treating drug misuse in women. Unfortunately, several factors may interfere with women's ability to attend detoxification programs, that is, childcare responsibilities, while others may undermine the positive outcomes of the programs, that is, coexisting mood alterations or concurrent use of tranquilizers or antidepressants.
A viable strategy for minimizing drug-induced injuries when abstinence from drugs is not feasible is the ‘harm-reduction’ policy, a variety of pragmatic strategies aimed to counteract harm brought about drug use rather than insisting on abstinence. Many women who abuse drugs practice personal harm-reduction strategies, ranging from reducing the amount of the drug to improving diet, from taking vitamins to changing lifestyle, to getting more sleep. Another frequent harm-reduction strategy is to substitute the abused drug with another one that is perceived as less harmful, that is, replacing crack with marijuana.
A drug abuse prevention program for women should be alert of specific research-based risk factors, such as low-income, poor maternal-child relationship, child neglect, violent experiences, sexual abuse, post-traumatic stress disorder, excessive stress pressure, and social isolation (often experienced by single parents), by addressing, at the same time, numerous and complex variables that could have important implications for a positive treatment outcome (i.e., comorbidity, emotional distress, poor self-esteem, coping skills and psychiatric conditions).
Conclusion
Traditionally, drug abuse was considered to be primarily a problem specific to men, but the recent focus on drug addiction in women has brought attention to numerous sex differences in the epidemiology of abuse-related disorders, neurobiology of substances of abuse, etiologic considerations and psychiatric comorbidity [126]. For example, animal studies show male-female differences in the speed of the acquisition of drug self-administration behavior, the amount of drug self-administered, the percentage of subjects that acquire self-administration, motivation to self-administer the drug, and the tendency to relapse following drug cessation. Despite the fact that most animal studies indicate that females are more susceptible to develop drug addiction than males, the rate of substance abuse and dependence in humans is higher among men than it is among women, although women have been found to progress through the various stages of drug addiction at an accelerated rate and to enter treatment programs earlier than men [127,128]. This could be explained, at least in part, by considering the societal and cultural factors governing the roles of women in our societies, such as the higher social disapproval of drug use in women or the female apprehension to consume drugs during pregnancy.
Long-term effects of and biological responses to addictive drugs may differ between women and men, so that different prevention and treatment strategies may be required. Epidemiologic studies revealed that, in humans, the prevalence of taking drugs differs markedly depending on the specific drug, with highest prevalence for licit drugs, such as tobacco in women and alcohol in men. Among illicit drugs, cannabis is used most frequently, followed by cocaine. Differences between sexes are slightly increased with age, with level of drug use also differing between women and men. Women and men have different physiological responses to drugs and may develop different manifestations as consequences of drug consumption. Women initiate cocaine use sooner [129], although they are more likely than men to remain abstinent [130], have more emergency-room visits following crack use [131], become more intoxicated after similar levels of alcohol intake [132], and take less time than men to become addicted to opioids and alcohol after initial use [132]. Women are also likely to develop more side effects and more fatal drug reactions to psychotropic substances than men. Moreover, the incidence of mixing medications for other than the intended purpose (i.e., polypharmacy) is particularly high among elderly women.
Future perspective
The challenges of future clinical trials will be the better understanding of the gender differences in vulnerability to drug abuse as well as the optimization of sex-specific prevention and treatment approaches. Indeed, although important progress has been made in the knowledge of gender differences in drug addiction and dependence, noteworthy gaps still remain, among which is the identification of the precise role of hormones. Drug-abuse prevention and medication should be tailored to sex, with a deeper comprehension of the differential effects and health consequences that abused drugs may induce in women and men.
In light of the promising findings of animal studies, clinical trials should take into great consideration the currently recognized risk factors for the transition from casual and recreational drug use to compulsive and uncontrolled drug-seeking and drug-taking behaviors. Accordingly, gender-specific factors triggering drug use should be considered while treating abstinent patients at high risk of relapse after detoxification.
At the same time, we feel that more attention should be paid to the medical problem of drug use in pregnant women, as most drugs of abuse readily cross the placenta, thus impinging on both the structural and functional development of the fetus. Substance use during pregnancy may have physical and mental health implications that go beyond childhood, which are likely to impact on the future adolescent's behavior in society.
Randomized animal and large-scale, well-designed human studies are urgently needed to further investigate:
Neurobiological basis for gender-related differences in drug addiction
Role played by hormones in vulnerability to develop drug dependence
How abused substances may affect women of childbearing age and pregnant women
Which environmental and sociocultural risk factors may contribute to drug abuse and relapse in men and women
Executive summary
Background
In the last several decades, appreciation has increased for the importance of the role of gender in the prediction of vulnerability to substance abuse and risk to develop psychological and/or physical dependence. Prior to that time, most preclinical studies and treatment strategies focused on male patients, resulting in an under-representation of outcome literature relating to women.
Animal studies
Self-administration studies in animals have clearly demonstrated that nicotine, alcohol, cocaine, amphetamines, opioids, marijuana and, possibly, caffeine and phencyclidine exert sex-dependent effects, and that the estrous cycle is a critical factor in determining such differences. Thus, female and male animals may differ for either preference for a specific drug, motivation to work for it, levels of drug intake or the propensity to reinstate drug-seeking behavior following a period of abstinence.
Human studies
Clinical practice has detected important gender differences in drug addiction and dependence, including differences in epidemiology, physiological factors, genetic, psychological and sociocultural features, clinical characteristics, psychiatric comorbidity, and treatment considerations. In general, women begin using substances later than men do, are strongly influenced by partners to use, report different reasons for maintaining the use of the substances (i.e., self-medication), and enter treatment earlier in the course of their illness than men.
Possible factors responsible for gender differences in drug addiction
Although a number of genetic, social and environmental factors have been proposed to explain the gender differences observed in addictive behavior, influence of hormones, brain sexual dimorphisms (i.e., different brain morphology and neurotransmission), drug pharmacokinetics and pharmacodynamics represent the major factors underlying gender differences in drug addiction and dependence.
Footnotes
The authors have no relevant affiliations or financial involvement with any organization or entity with a financial interest in or financial conflict with the subject matter or materials discussed in the manuscript. This includes employment, consultancies, honoraria, stock ownership or options, expert testimony, grants or patents received or pending or royalties.
No writing assistance was utilized in the production of this manuscript.
