Abstract
Since the 1960s, antiphospholipid antibodies have been known to be associated with repeated miscarriages and fetal losses. Other complications of pregnancy, such as preterm birth, with pre-eclampsia or severe placental insufficiency were also frequently reported and are included in the current classification criteria of the antiphospholipid syndrome. The titer, isotype or antigen specificity of the antibodies may be important in risk determination. The pathogenesis of pregnancy failures is not only linked to the thrombophilic effect of antiphospholipid antibodies but also to a direct effect of antibodies on trophoblast differentiation and invasion. The study of experimental animal models provided sound evidence of the pathogenic role of antiphospholipid antibodies both in lupus-prone and -naive mice. The classification of pregnant antiphospholipid syndrome patients as being at a ‘high risk’ has completely changed their prognosis due to obstetric monitoring and the application of effective therapy. In fact, despite the high rates of complications and preterm delivery, a successful outcome can now be achieved in a large majority of cases.
Keywords
Antiphospholipid syndrome (APS) was first identified in the 1960s as the association of thrombosis, repeated miscarriages or fetal losses with antiphospholipid antibodies (aPL) [1,2]. The two aPLs that were first characterized are lupus anticoagulant (LA) and anticardiolipin antibodies (aCL). Although many women with LA also have aCL, the correlation is only partial [3,4]: 80% of patients with LA have aCL and 20% of patients positive for aCL have LA. β2 glycoprotein I (β2GPI) has been recognized as the target for the majority of aCL antibodies and some antibodies with LA activity [5,6]. Therefore, despite a number of methodological problems, the assay for anti-β2GPI antibodies raised a great deal of interest and is now included in the laboratory criteria to define APS. Laboratories often report antibodies to other phospholipids or phospholipid-binding proteins (i.e., antiphosphatidylserine, antiphos-phatidylinositol, antiphoshatidylethanolamine, antiphosphatidylcholine, antiphosphatidylglycerol, antiprothrombin, antiannexin V) that are not specific or frequent enough to be regarded as criteria.
APS can occur as a primary syndrome when present in otherwise healthy subjects, or, alternatively, within other autoimmune diseases such as systemic lupus erythematosus (SLE).
Since pregnant patients with this condition can be appropriately managed, APS has been defined as “one of the few tractable causes of pregnancy loss” [7]. The current management of pregnancy in women with aPL has significantly improved the rate of live births, which now reach 70% in most of the published reports [7,8]. However, the rate of preterm delivery remains high, although probably with fewer severe consequences owing to close obstetric monitoring, allowing a careful delivery timing, and due to the progress recently achieved by neonatal intensive care units.
As demonstrated in the literature, the presence of aPL, determined by different methods, is associated with obstetric pathology but, up to now, it is not clear if the titer, antibody isotype or specificity are important factors in the determination of risk level. However, the presence of aCL has proved to be a significant risk factor for unsuccessful pregnancy outcome in women affected by SLE and otherwise healthy women (primary APS) [9].
Update of the classification criteria
In 1999 during the International Consensus Conference of Sapporo, the preliminary classification criteria of APS were defined as thrombosis episodes or pregnancy losses associated with aPL, defined as aCL or LA [10]. This classification was revised in Sydney in 2004 when some features of obstetric complications were better defined and anti-β2GPI assay was added as an independent laboratory classification criteria (
The revised classification criteria of antiphospholipid syndrome.
Vascular thrombosis:
One or more clinical episodes of arterial, venous or small-vessel thrombosis, in any tissue or organ. Thrombosis must be confirmed by imaging or Doppler studies or histopathology, with the exception of superficial venous thrombosis. For histopathological confirmation, thrombosis should be present without significant evidence of inflammation in the vessel wall.*
Pregnancy morbidity:
One or more unexplained deaths of a morphologically normal fetus at or beyond the 10th week of gestation, with normal fetal morphology documented by ultrasound or by direct examination of the fetus; or
One or more premature births of a morphologically normal neonate before the 34th week of gestation due to: (a) eclampsia or severe preeclampsia defined according to standard definition or (b) recognized features of placental insufficiency; or
Three or more unexplained consecutive spontaneous abortions before the 10th week of gestation with maternal anatomic or hormonal abnormalities and paternal causes excluded.
Anticardiolipin antibody of IgG and/or IgM isotype in serum or plasma, present in medium or high titer (i.e. >40 G phospholipid or M phospholipid, or > the 99th percentile), on two or more occasions, at least 12 weeks apart, measured by a standardized enzyme-linked immunosorbent assay.
Lupus anticoagulant present in plasma on two or more occasions at least 12 weeks apart, detected according to the guidelines of the International Society on Thrombosis and Haemostasis (Scientific Subcommittee on Lupus Anticoagulants/Phospholipid-dependent antibodies).
Anti-32 glycoprotein-I antibody of IgG and/or IgM isotype in serum or plasma, (in titer >99th percentile) present on two or more occasions, at least 12 weeks apart, measured by a standardized enzyme-linked immunosorbent assay, according to recommended procedures.
Superficial venous thrombosis is not included in the clinical criteria. From [9].
APS is present if at least one of the clinical criteria and one of the laboratory criteria are met, with the first measurement of the laboratory test performed at least 12 weeks from the clinical manifestation. Classification should be avoided if less than 12 weeks or more than 5 years separate the positive aPL test and the clinical manifestation.
It has to be noted that criteria are for the clinical research purpose of defining patients who all investigators agree to have the syndrome.
Coexisting inherited or acquired factors of thrombosis are not a valid reason for excluding patients from APS diagnosis. However, the two subgroups of APS patients should be recognized according to (a) the presence or (b) the absence of additional risk factors for thrombosis [11].
Investigators performing studies are advised to classify APS patients into one of the following categories:
I: More than one laboratory criteria present (any combination);
IIa: aCL antibody present alone;
IIb: LA present alone;
IIc: Anti-β2GPI antibody present alone.
There are many other clinical or laboratory features frequently associated with APS, but not included in the revised criteria because they are not specific or too rare. These include heart-valve disease, Livedo reticularis, thrombocytopenia, nephropathy, neurological manifestations, and, in the laboratory, aCL and anti-β2GPI of IgA isotype, antiphosphatidylserine antibodies, antiphosphatidylethanolamine antibodies, antibodies against prothrombin alone and antibodies to the phosphatidylserine–prothrombin complex [11].
Pregnancy losses associated with aPL are described as losses within the fetal period (≥10 weeks of gestation) and/or miscarriages in the pre-embryonic/embryonic period (<10 weeks of gestation) [13]. As suggested from clinical experience, fetal death is the most specific criterion, while recurrent early abortion may be the most sensitive [5]. In contrast to recurrent pregnancy losses, aPLs are not associated with sporadic pre-embryonic or embryonic pregnancy loss. The specificity of recurrent early abortion is uncertain because of the difficulty in excluding other known or suspected causes. The new classification criteria underline the conditions of early and severe pre-clampsia associated with APS. In order to improve its specificity, this criterion must include only cases requiring delivery before 34 weeks of gestation.
Antiphospholipid syndrome & pregnancy
The pathogenic role of aPL has been clearly demonstrated in experimental animals. In fact, pregnant mice infused with aPL develop fetal resorbtions (equivalent to miscarriages in women) or placental insufficiency [14].
Pregnancy in women who are positive for aPL can also be complicated by utero–placental insufficiency causing pre-eclampsia-HELLP (hemolysis, elevated liver enzyme levels and a low platelet count) syndrome and/or intrauterine growth restriction (IUGR) and, consequently, premature delivery [15].
These obstetric complications arise from the prothrombotic effects of maternal aPL on placental function. Thrombotic events at the placental level may be related to endothelial cell activation, inhibition of protein C/S system and fibrinolysis [16] as well as annexin V displacement [17]. However, it is thought that the thrombophilic state cannot explain all miscarriages, and a direct antibody-mediated damage to trophoblasts has been suggested [18]. During differentiation to syncytium, trophoblasts express cell-membrane anionic phospholipids [19] that can bind β2GPI, the main cationic phospholipid-binding protein, recognized by aPL. Adhered β2GPI might be recognized by the antibodies that, once bound, strongly interfere with in vitro trophoblast cell maturation, thus resulting in defective placentation and hormonal production. Notably, it was recently shown that complement activation is an absolute requirement in this pathogenic model, in fact pregnancy losses cannot be reproduced in complement-depleted or -deficient mice [20]. It has been suggested that these mechanisms play a role in early fetal loss, while thrombotic events would be responsible for losses occurring late in the pregnancy [21]. In addition, it may be useful to mention that pregnancy itself is a known risk factor for thromboembolic episodes; therefore, women with aPL bear two concomitant factors of enhanced vascular risk.
The relationship between aPL and pre-eclampsia is debated: in several studies the occurrence in patients with primary or secondary APS is high [15,22,23] and in obstetric population with pre-eclampsia a high frequency of aPL-positive patients ranging from 11–17% has been observed [24–27]. The association is strongest in women with severe, early onset pre-eclampsia (<34 weeks of gestation) while, in general, term pre-clampsia is not associated with increased levels of aPL [28].
In 2001, Branch and colleagues evaluated 317 pregnant patients with an episode of pre-eclampsia in a previous pregnancy, thus considered at high relapse risk, and found that IgG antiphosphatidylserine antibodies were associated with severe pre-eclampsia and IgG aCLs were associated with IUGR. However, these tests showed a limited predictive value for the occurrence of such complications [29].
Our study, dealing with 132 pregnancies prospectively followed in 92 SLE patients, showed a significant association between LA and pre-eclampsia, with a high predictive value (RR: 9.2) [30]. In a different prospective study of 510 healthy pregnant women, we observed that positive anti-β2GPI antibodies detected at 15–18 weeks of gestation were significantly associated with the onset of pre-eclampsia and eclampsia [12].
IUGR is reported with a frequency ranging from 12–30% in different series of pregnant patients with APS; despite the use of different prophylactic treatments, a number of studies underline a significant increase in the occurrence of IUGR in patients with APS compared with the normal obstetrical population [15,23,31,32] but contrasting results have also been reported [33,34]. The inclusion of some women with low positive aPL titers probably accounts for these discrepancies.
Uteroplacental insufficiency, pre-eclampsia and IUGR further increase the risk of preterm delivery in women with APS. The risk is greater in women with high titers of aPL antibodies who meet the strict criteria for APS. In one report, approximately a third of pregnancies in women with APS treated with different combinations of medications (prednisone and/or low-dose aspirin and/or heparin and immunoglobulin) occurred before 34 weeks of gestation [15]. In order to investigate if aPLs are associated with fetal damage different from prematurity, we studied the neonatal outcome of 71 babies from mothers with APS receiving a prophylactic treatment during pregnancy, comparing it with that of 71 babies from healthy mothers [35]. The two groups were matched for gestational age, birthweight, mode of delivery and obstetrical complications. Neonatal outcome, in terms of neonatal intensive-care unit admission, prevalence of respiratory distress syndrome, chronic lung disease, intraventricular hemorrhage, periventricular leucomalacia and retinopathy, did not show a significant difference between cases and matched controls (
Outcome of neonates from mothers with antiphospholipid syndrome.
NICU: Neonatal intensive-care unit. From [35].
However, in children from mothers with APS, case reports of neonatal thrombosis were recorded, involving the brain or other areas. From 1987–2002, 13 neonatal thromboses related to aPL have been reported; among them, arterial vessels were involved ten-times, six-times in the cerebral territory, venous thrombosis occurred in only two cases, arterial and venous thrombosis in one case [36].
We also observed a fetal stroke associated with maternal aPL, which was found at 2 months of age in the cerebral artery territory by ultrasound and computed tomography (CT) scan, probably due to an intrauterine event. The child then developed West syndrome and left hemiparesis [37]. Therefore, infants born from mothers with aPL should be screened at birth and those found to be positive should be closely monitored, at least until the antibodies are completely cleared from their circulation. To improve knowledge in this area a European registry has been started that will join clinical and biological data in this group of children [36]. Certainly, thromboses in the offspring of mothers with aPL are extraordinarily rare when compared with the number of pregnancies reported in this group of patients. This observation can have two possible explanations. The first is that children are protected from thrombosis because they do not have a concomitant risk factor. In fact, according to the ‘two-hit hypothesis’, the presence of aPL is not enough to cause the thrombosis [38]. Alternatively, the placenta might be involved in the clearance of a consistent part of aPL because it carries their target antigen, impairing the amount of antibodies actually reaching the fetal circulation [39].
Infants born from mothers with aPL-positive autoimmune disease and prospectively evaluated for aCL and anti-β2GPI antibodies were all found aCL-negative at 12 months [39]. This observation suggests that aCL is the best assay to evaluate the disappearance of maternal aPL and estimate the potential risk of thrombosis associated with these antibodies. On the other hand, the high rate of anti-β2GPI antibodies detected in 12-month-old children may indicate a de novo synthesis of these antibodies, possibly stimulated by aspecific factors [39]. Experimental work in progress will clarify these interesting aspects.
There are few data regarding the long-term outcomes of children born to patients with aPLs [40].
A study conducted in our clinic on children born to SLE patients reported that the occurrence of learning disabilities seems increased in subjects whose mothers were aPL-positive [41]. However, these children have a normal intelligence level. In support of this finding, a prolonged exposure to aPL was demonstrated to cause hyperactivity and anxiety in animal models and in vitro data showed that aPL can bind to brain tissue and brain endothelial cells [42].
Thromboembolic events are the most serious complications of pregnant patients with APS. Their occurrence is related to the potential synergy between the thrombophilic risk typical of pregnancy and/or puerperium and the risk represented by aPL antibodies.
Management
Owing to the above-mentioned increased thromboembolic risk and because APS pregnancies are classified as high risk for obstetrical complications, patients should undergo a close follow-up by an ad hoc team including rheumatologists, obstetricians and other specialists needed for the care of the mother, working in collaboration with a neonatal intensive-care unit.
Patients are seen periodically throughout the pregnancy. In our institution, patients undergo monthly visits until 20 weeks of gestation, and then more frequently (every 2 or 3 weeks) up to delivery. At each visit we recommend a physical examination and the control of blood parameters and urine analysis, while the immunological profile should be checked every 3 months. As uterine artery Doppler performed at 24 weeks is recognized as a useful screening test for pre-eclampsia and small-for-gestational-age infants, all APS patients attending our institution are examined at 24 weeks [43].
According to this protocol, maternal and fetal wellbeing are strictly monitored, allowing the clinical team to modulate maternal treatment or decide the delivery time, as needed. Careful management, together with appropriate treatment, have considerably improved obstetrical outcomes (
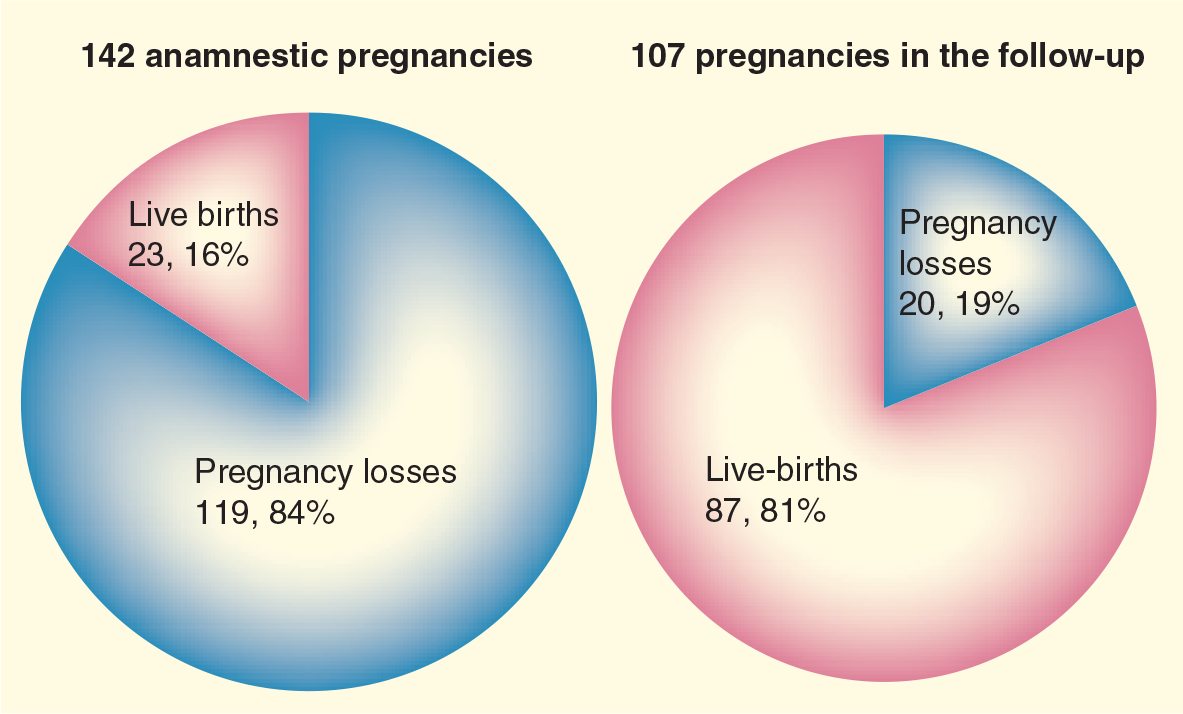
Outcome of pregnancy in 75 primary antiphospholipid syndrome patients followed in Brescia Hospital, Italy, between 1984 and 2004.
Treatment
Corticosteroids plus LDA was the first therapy used for pregnant patients with APS [44]. However, in 1992, a controlled trial showed that heparin (20,000 U/d) with LDA (81 mg) was equally effective in treating severe gestational complications with fewer maternal side effects [45].
The use of LDA was introduced due to the previous experience of treating pregnancy complications. In obstetric patients LDA has been extensively used in order to improve pregnancy outcome by preventing hypertension, pre-clampsia, preterm birth, intrauterine growth restriction and perinatal death, particularly in women with historical risk factors [46]. In fact, a recent Cochrane review, including data from 35,500 patients treated with antiplatelet drugs during pregnancy, shows a 19% reduction of the risk of developing pre-eclampsia, 7% reduction of preterm delivery and 16% reduction of perinatal death [47]. Despite the known effect of therapeutic doses of nonsteroidal anti-inflammatory drugs, the use of LDA in pregnancy is safe. Exposure in the first trimester does not increase the risk of congenital malformations [48]; in the second and third trimester it does not increase fetal side effects such as impaired renal function, pulmonary hypertension and hemorrage in the newborn, and it has no effect on the ductus arteriosus [49].
The administration of LDA results in selective inhibition of cycloxygenase 1 that regulates prostanoid-dependent functions in tissues and cells. The possible consequences of LDA administration are:
Thromboxane reduction, not only at platelet level but also at placental level. In vitro, aspirin suppresses the thomboxane release of normal placenta in the presence of LA-positive IgG [50];
Increased production of leukotrienes, which stimulate interleukin (IL)-3 production. IL-3 enhances placental and fetal development (it is a trophoblast growth factor) and was found to be reduced in APS patients. LDA restores IL-3 levels in animal models of APS and completely eliminates aPL-related obstetric complications [51];
Suppression of nitric oxide synthetase activity, preventing oxidative stress that induces trophoblast apoptosis. In fact, the increased apoptosis of a throphoblastic cell line, induced by the serum of patients with early miscarriages, is significantly reduced [52].
The rationale for heparin use (unfractioned and then low molecular weight) involves the pathogenic role of thrombotic phenomena in aPL-associated pregnancy loss. Heparin is the standard drug to prevent thromboembolic complications during pregnancy in patients with known risk factors.
In patients with APS and pregnancy morbidity only, the use of heparin has been related to the prevention of placental ischemia. Moreover, heparin can be helpful in preventing early miscarriages, probably because it impairs the binding of anti-(32GPIs to trophoblast cells [18,53]. Alternatively, since some complement components were shown necessary to the aPL pathogenic effect at placental level, heparin can exert its role by inhibition of the complement system. In fact, treatment with heparin prevented complement activation in vivo and in vitro and protected mice from pregnancy complications induced by aPLs. Different anticoagulant molecules (fondaparinux and hirudin), not binding complement, do not protect the mice from pregnancy losses [54].
The optimum time to initiate heparin has been debated, especially in patient who are receiving an oral anticoagulant due to previous thrombosis. Some authors advise stopping warfarin before conception in order to avoid its theratogenic effects, relevant between the 7th and 12th weeks of gestation. However it is difficult to know how long the period before conception will be and the potential side effect of heparin could be significant. The prevalent opinion is to advise patients to conduct a pregnancy test at the missed period and promptly switch from warfarin to heparin [8].
Osteoporosis incidence associated with heparin treatment in pregnancy is between 0.2% (low molecular weight) and 2% (unfractioned); this side effect is most relevant in patients treated with corticosteroid, and a calcium supplementation is suggested in heparin-treated patients [55].
Some authors demonstrated that in patients without previous thrombosis and only obstetric complications, heparin plus aspirin (75–100 mg/d) were more effective than aspirin alone [31,56]. By contrast, Farquharson and colleagues observed a successful outcome in approximately 75% of pregnancies observed in both arms [57].
Branch and colleagues demonstrated that intravenous immunoglobulin added to conventional treatment with heparin/aspirin was not able to improve reproductive prognosis in APS patients, therefore such treatment should be reserved for cases in which conventional therapy has failed or for the treatment of additional problems occurring in pregnancy, such as severe thrombocytopenia [58].
The intensity of treatment is related to the patient's history. Patients with a previous thromboembolic event require a therapeutic, weight-adjusted dose of unfractioned or low molecular weight heparin; patients with obstetrical complications only need a prophylactic dose throughout pregnancy. It is still debated if patients with aPL but without clinical events need to be treated. However, it is widely accepted to administer LDA to pregnant patients as primary prophylaxis of pregnancy loss if they show repeated positive values of aPL, detected by LA, aCL or anti-β2GPI antibodies [8].
Executive summary
Antiphospholipid syndrome (APS) was first identified in the 1980s as the association of thrombosis, repeated miscarriages and fetal losses with antiphospholipid antibodies (aPL).
APS was defined as “one of the few tractable causes of pregnancy loss”: in fact, the rate of live births now reach 80% in most of the published reports.
Laboratory criteria to define APS require the persistent positivity of anticardiolipin antibodies and/or anti-β2 glycoprotein-1 and/or lupus anticoagulant.
Pregnancy losses due to aPL may be within the fetal period (>10 weeks gestation) and/or in the pre-embryonic/ embryonic period.
The pathogenesis of pregnancy failures is linked to the thrombophilic effect of aPL but also to different mechanisms, including a direct effect of antibodies on the throphoblast differentiation and invasion; a direct antibody-mediated damage on the trophoblast has been suggested to play a role in early fetal loss, while thrombotic events may be responsible for losses occurring later.
The study of experimental animal models confirmed the pathogenic role of aPL, which when infused during pregnancy caused fetal resorbtions (equivalent to miscarriages in women) or placental insufficiency.
Pregnancy in women who are positive for aPL can also be complicated by utero–placental insufficiency, causing pre-eclampsia–hemolysis, elevated liver enzyme levels and low platelet count (HELLP) syndrome and/or intrauterine growth restriction and consequently premature delivery, the major problem of aPL-complicated pregnancy. For this reason the referral hospital for pregnant patients with APS should have a neonatal intensive-care unit.
A recent case-control study on infants from APS mothers and healthy mothers did not show any difference in the occurrence of neonatal complications.
In children from mothers with APS, case reports (luckily rare) of neonatal thrombosis were recorded, involving the brain or other areas.
In children from mothers with systemic lupus erythematosus and aPL, an increased occurrence of learning disabilities but a normal intelligence level was described.
Maternal thromboses can occur in pregnancy and are due to the potential synergy between the thrombophilic risk typical of pregnancy and/or puerperium and the risk represented by aPL antibodies.
The management of pregnant patients with APS is nowadays mainly based on the use of antiaggregant/anticoagulant agents (with aspirin and heparin) to prevent thromboses in the uteroplacental circulation. Interventions with these drug therapies and close monitoring of pregnancy have increased fetal survival.
Conclusion
According to our and others' experience, the new and more precise classification of APS, standardization of laboratory tests for diagnosis, careful obstetric surveillance conducted by a multidisciplinary team (obstetricians, rheumatologists and neonatologists), and progress in neonatal intensive care have greatly improved the survival rate of newborns from APS mothers and reduced the incidence of long-term handicap [7,35].
Future perspective
In the future, the correct throphoblastic invasion might be tested in the first trimester by uterine artery Doppler evaluation, if the sensitivity and specificity of the method are confirmed [59].
The identification of specific antibody profiles and their correlation with particular aspects of the syndrome might enable us to perform tailored monitoring and treatment, hopefully improving pregnancy outcomes even if standard care sometimes fails.
