Abstract
Preterm birth is a major public-health problem in the USA, which has higher rates of preterm birth than most other developed countries. Attempts at the prevention of preterm birth have been largely unsuccessful. The recent publication of a large, multicenter, randomized trial of 17 α-hydroxyprogesterone caproate, which showed efficacy in preventing recurrent preterm delivery, has sparked renewed interest in progestational drugs in general and this compound in particular for use in preventing preterm delivery. Although this drug is not currently commercially available, an application has been made to the US FDA to produce 17 α-hydroxyprogesterone caproate, as Gestiva™ for the prevention of preterm delivery.
17 α-hydroxyprogesterone caproate (17HPC) is a drug that was first introduced in the 1950s and promoted for the treatment of a number of gynecological and obstetrical disorders such as infertility due to luteal-phase defect, threatened abortion or for women with a history of repeated early pregnancy loss or preterm delivery. Over time, the popularity of the drug declined, and it has been out of production in the USA since 2001. Recently, the drug has received renewed interest for its possible efficacy in preventing pre-term delivery in women at high risk. As a result, Adeza Corp. has submitted a New Drug Application to the US FDA to produce 17HPC as Gestiva™, for the prevention of preterm birth.
The problem of preterm birth
Preterm birth, that is, delivery before 37 completed weeks of gestation, is the major determinant of infant mortality in developed countries [1]. The rate of preterm birth is greater in the USA than in most developed countries, and is the factor most responsible for the relatively high rate of infant mortality in the USA [2]. In addition, the rate of preterm birth in the USA has increased progressively over the past two decades, from 9 to 12% of all births [2]. The rate of preterm delivery is particularly high for African–American women, who experience rates of low birthweight and preterm delivery twice as high as those for white women in the USA [1]. Preterm birth is hazardous to the health of the surviving children. Preterm birth increases the risk of developmental problems, and is the greatest single cause of cerebral palsy [1]. In addition, the financial costs of preterm birth are high. In 2001, the total national bill for premature babies was estimated at $13.6 billion [101]. For these reasons, preterm birth is a very significant public health problem in the USA.
Many attempts have been made to find ways to reduce the incidence of preterm birth. Beginning in the 1970s, drug therapy for the prevention of preterm delivery became widely used and focused mainly on the use of tocolytic drugs to halt preterm labor after it had begun. Although trials of various tocolytic drugs have shown effectiveness in delaying labor for up to several days, the use of these drugs has not reduced the incidence of preterm delivery and has not resulted in any improvement in perinatal outcome [3]. Cervical cerclage has long been employed for women thought to have a weakness of cervical integrity, but up to now randomized trials of cervical cerclage have generally not shown effectiveness [4]. Although vaginal infections are known to be associated with an increased risk of preterm delivery, the largest and best-controlled trials in low-risk women have found no improvement in rates of preterm birth as a result of screening for and treating vaginal infections [5,6]. Despite many trials of reduced physical activity, the use of tocolytic drugs to halt labor, antibiotic therapy, and other strategies for prevention, no effective and reproducible method of preventing preterm birth has been discovered [7]. Thus, the enigma of pre-term birth is widely considered to be the greatest problem in obstetrics in the developed world.
Actions of progesterone
Progesterone, produced by secretion from the corpus luteum and the placenta, is known to be essential for the maintenance of pregnancy early in gestation [8,9]. In addition, progesterone is known to have actions that maintain pregnancy later in gestation. Progesterone acts to relax smooth muscle in many organs, including the pregnant uterus. Progesterone has immunosuppressive activity against the activation of T lymphocytes and blocks the effects of oxytocin on the myometrium [9,10]. Perhaps most importantly, progesterone is a potent inhibitor of the formation of gap junctions between myometrial cells [11]. These intercellular communications are essential for the propagation of coordinated uterine muscle activity leading to labor.
In addition to the actions of progesterone in the general maintenance of pregnancy, evidence exists from data obtained from both animals and humans that changes in local or systemic concentrations of progesterone may play a role in the initiation of labor. The importance of progesterone in regulating the onset of labor is supported by the observation that, in sheep, goats, and many other mammalian species, a decrease in plasma progesterone and an increase in estrogen precedes the onset of labor [12]. These findings of changes in the progesterone/estrogen ratio are consistent with the concept of ‘progesterone block,’ which was advanced and championed by Csapo in the 1950s, based on his extensive and pioneering experiments in pregnant rabbits [13].
The role of progesterone or of changes in the progesterone/estrogen ratio in human beings and other primates is less well known. No consistent evidence exists of changes in plasma progesterone or the progesterone/estrogen ratio before the onset of labor at term or before term. Nonetheless, some evidence exists that local changes in progesterone or the progesterone/estrogen ratio in the placenta, decidua, or fetal membranes may be important in the initiation of labor in human beings [14,15]. Several investigators have reported the effects of administering progesterone antagonists to women at term. The results were an increased rate of spontaneous onset of labor and, in the women whose labor was induced with oxytocin, an increased sensitivity to oxytocin compared with placebo-treated controls [16,18]. Although these data support the concept that progesterone plays a role in maintaining gestation in human beings, the actual mechanisms by which progesterone therapy may avert preterm labor and delivery are not known.
Clinical pharmacology of 17HPC
17 α-hydroxyprogesterone (unesterified) is a naturally occurring progestin that was originally isolated from the adrenal glands and has minimal progestational activity. 17HPC is a synthetic progestin hormone, which is an esterified derivative of 17 α-hydroxyprogesterone. 17HPC has substantial progestational activity and a prolonged duration of action compared with progesterone and other progesterone derivatives. [19] The route of administration is by intramuscular injection. Like progesterone, 17HPC exerts no androgenic effect [20]. Pharmacokinetc studies have shown that once-weekly intramuscular administration of 17HPC would provide continuous systemic serum levels of 17HPC. The half-life of 17HPC was estimated to be approximately 7.8 days [21].
Early trials of 17HPC
Among the earliest prophylactic trials of 17HPC for the prevention of preterm delivery was conducted by Papiernik in 1970 [22]. This trial enrolled women identified as being at risk of pre-term delivery by using a risk-scoring system. A total of 99 women were randomly assigned to receive 17HPC or placebo injections. Preterm delivery occurred less frequently in the treated group (4%) than in the placebo group (18%).
In 1975, Johnson reported the results of a trial of 17HPC for women who had a history of two spontaneous abortions, two preterm deliveries, or a combination of these outcomes in the pregnancies immediately preceding the index pregnancy [23]. Of the 43 women in the trial, 18 were randomized to weekly injections of 17HPC 250 mg, and 22 were randomized to placebo injections. The treatment was started as soon as the patients registered for care and was continued to 37 weeks of gestation or delivery. The primary outcome was delivery at less than 36 weeks of gestation. The rate of preterm delivery in the placebo group was 41% (nine women), whereas none of the treatment group delivered before 36 weeks. The treatment group showed significant differences from the control group in mean duration of pregnancy, mean birthweight and perinatal mortality rate.
The results of these early reported trials of progesterone for the prevention of preterm labor and delivery were evaluated by two different meta-analyses. In 1989, Goldstein and colleagues published the results of a meta-analysis of randomized, controlled trials involving the use of progesterone or other progestational agents for the maintenance of pregnancy [24]. A total of 15 trials of variously defined high-risk subjects were felt to be suitable for analysis. The trials employed six different progestational drugs. The pooled odds ratios for these trials showed no statistically significant effect on rates of miscarriage, stillbirth, neonatal death or pre-term birth. The authors concluded that, “progestogens should not be used outside of randomized trials at present” [24].
In response to this publication, Keirse presented the results of an analysis of a more focused selection of trials in 1990 [25]. This meta-analysis was restricted to trials that employed 17HPC, the most fully studied progestational agent, and included all placebo-controlled trials that used this drug. Pooled odds ratios found no significant effect on rates of miscarriage, perinatal death, or neonatal complications. However, in contrast to Goldstein's review, the odds ratio for reduction of preterm birth was significant, 0.5 (95% confidence interval [CI]: 0.30–0.85), as was the odds ratio for birthweight less than 2500 g, 0.46 (95% CI: 0.27–0.80).
The National Institute of Child Health and Human Development Maternal Fetal Medicine Units Network Trial
The recent publication of a trial of 17HPC conducted by the Maternal Fetal Medicine Units (MFMU) network and sponsored by the National Institute of Child Health and Human Development (NICHD), has stimulated renewed interest in 17HPC and progesterone compounds in general for preventing preterm birth. Meis and colleagues reported the results of a large, multicenter trial of 17HPC conducted by the MFMU Network [26]. The study enrolled women with a documented history of a previous spontaneous preterm delivery, which occurred as a consequence of either spontaneous preterm labor or preterm premature rupture of the fetal membranes. After receiving an ultrasound examination to rule out major fetal anomalies and to determine gestational age, the subjects were offered the study and given a test dose of the placebo injection to assess compliance. If they chose to continue, they were randomly assigned, using a 2:1 ratio, to weekly injections of 17HPC 250 mg or a placebo. Treatment began between 16 and 21 weeks of gestation and continued until delivery, or 37 weeks of gestation, whichever came first. The study planned to enroll 500 subjects, a sample size estimated to be sufficient to detect a 37% reduction in the rate of preterm birth. However, enrollment was halted at 463 subjects, 310 in the treatment group and 153 in the placebo group, following a scheduled evaluation by the Data Safety and Monitoring Committee, which found that the evidence of efficacy for the primary outcome was such that further entry of patients would not be ethical. In this study, delivery at less than 37 weeks was reduced from 54.9% in the placebo group to 36.3% in the treatment group. Similar reductions were seen in delivery at less than 35 weeks, from 30.7 to 20.6%, and delivery at less than 32 weeks, from 19.6 to 11.4% (

Rates of births at less than 32 weeks.
The sample size of the study was not powered to examine effects on neonatal morbidity and mortality, and a statistical significance was not demonstrated for rates of perinatal death. However, a strong trend was found for reduction in rates of neonatal death, transient tachypnea, respiratory distress syndrome, bronchopulmonary dysplasia, need for ventilatory support, supplementary oxygen, intraventricular hemorrhage, necrotizing enterocolitis, patent ductus arteriosis and retinopathy. These differences were statistically significant for rates of intraventricular hemorrhage (but not for grades III–IV), necrotizing enterocolitis and need for supplemental oxygen and ventilatory support (
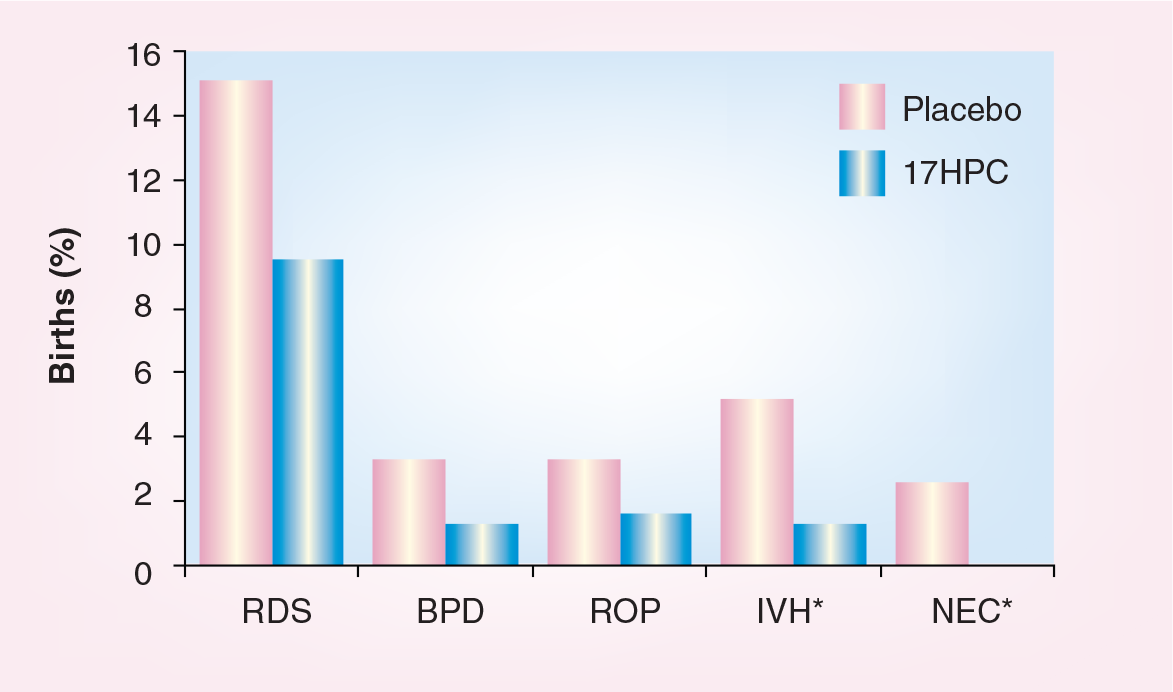
Rates of neonatal morbidity.
Further analysis of the results of the trial by Spong and colleagues found that treatment with 17HPC was especially efficacious in preventing recurrent preterm delivery in women whose prior preterm delivery occurred very early in gestation at 20–27 weeks [27].
Recently, two new systematic reviews have evaluated the results of progesterone trials to prevent preterm birth [28,29]. The meta-analyses of these reviews included the MFMU Network trial and a trial of progesterone suppositories by da Fonseca, as well as some earlier trials. Although the reviews differed as to which of the earlier trials were included, the results of the meta-analyses were similar in showing efficacy for progesterone treatment to prevent preterm delivery at less than 37 weeks of gestation, with odds ratios around 0.5.
Safety of 17HPC
The evidence for the safety of the use of 17HPC in pregnancy consists of theoretical considerations, animal studies and clinical studies. 17 hydroxyprogesterone is a naturally occurring metabolite of progesterone. Progesterone and 17 hydroxyprogesterone are produced in large amounts in human pregnancy [30]. The quantities produced in pregnancy, mainly by the placenta, exceed pharmacological doses employed in clinical use. 17 hydroxyprogesterone has no androgenic activity [20]. Logically, it is not reasonable to expect ill effects of a nonandrogenic progestin, naturally found in large quantities, upon human gestation.
The effects of 17HPC upon pregnancy in experimental animals has been studied in rats, rabbits, mice and monkeys (Macaca mulatta) [31–34]. These studies found no evidence of androgenic or glucocorticoid activity, no virilizing effects upon female fetuses, and no teratogenic effects.
A number of authors have reported the results of well-controlled clinical studies that examine the safety of 17HPC in human pregnancy. None of these studies found any evidence of adverse effects on the fetus or the outcome of the pregnancy.
Several extensive reviews of the literature have been published regarding the safety of the use of progestins in pregnancy. Schardein, in an extensive review, found “no justification (exists) for undue concern over the induction of nongenital malformations through hormone use in pregnancy” [35]. This review observed that, although some androgenic compounds have the potential for masculinization of the female fetus, progesterone and 17HPC have no such potential. Raman-Wilms and colleagues performed a meta-analysis of the literature after reviewing 186 published articles [36]. The meta-analysis showed no association between first-trimester exposure to sex hormones and external genital malformations. In the current REPROTOX17 computer database, supported by Micomedex, the review of hydroxyprogesterone concludes that, “There is no available evidence that the administration of this agent (17HPC) during pregnancy is harmful.” [37]. In summary, the safety of 17HPC administration in pregnancy is well documented by animal and clinical studies. Reviews of this topic by knowledgeable authors have uniformly concluded that no evidence exists to support the idea that administration of 17HPC in pregnancy represents a significant risk to mother, fetus or newborn. While evidence of possible teratogenic effects of 17HPC is slight, it would be prudent to withhold the start of 17HPC treatment for the prevention of preterm delivery until 16 weeks of gestation.
Indications for treatment
Currently, the group of patients who have been found to benefit from prophylactic treatment with progesterone to prevent preterm delivery are women at high risk for preterm delivery who have a history of prior spontaneous pre-term delivery caused by spontaneous preterm labor or preterm premature rupture of the fetal membranes. Treatment with 17HPC for other high-risk conditions, such as multiple gestation or short uterine cervix, should not be encouraged outside of randomized trials. Several such trials are in progress or in development at the time of this writing. The only published trial of 17HPC therapy for twin pregnancy failed to show efficacy, although this failure may have been related to the fact that therapy was begun at 29 weeks of gestation or later [38].
Although 17HPC treatment is effective in preventing recurrent preterm delivery in women at risk, treatment solely for this group of pregnant women can have only a modest impact on the rate of preterm birth in the general population [39]. More trials are urgently needed to determine whether 17HPC treatment is effective for women with other high-risk pregnancy conditions. In addition, improved methods of screening for risk and subsequent prophylactic treatment are needed for women in their first pregnancy.
The timing of initiation of progesterone therapy is likely to be important for its effectiveness. Successful trials have started treatment at no later than 24 weeks of gestation. It is reasonable to wait until 16 weeks of gestation to start treatment, and we recommend beginning as soon after this time as possible.
Conclusions
In summary, evidence is sufficient for clinicians to choose to use progesterone therapy to prevent recurrent preterm delivery in women at risk. The greater body of evidence of efficacy at this time is for treatment of 17HPC at 250 mg/week beginning as soon as possible after 16 weeks of gestation and continuing to 36 weeks or delivery. The safety of the use of this drug has been clearly documented. Some evidence exists to support the use of daily progesterone suppositories [40]. Further research will be necessary to support the use of progesterone for other high-risk conditions. In addition, further research is needed to elucidate the mechanism of action of 17HPC and progesterone in preventing preterm labor and delivery.
Future perspective
The successful use of 17HPC to prevent recurrent preterm delivery in women at risk would have only a modest impact on the overall rate of pre-term birth, If trials of this drug are successful for women with other high-risk conditions, such as multiple gestation, or women whose preterm labor has been temporarily halted with tocolytic drugs, the impact of the use of this drug could potentially have a much larger effect in reducing the rate of preterm birth. It is possible that other progesterone compounds and other routes of administration may show similar efficacy to 17HPC. Trials of progesterone vaginal suppositories are currently underway in several countries.
Executive summary
Preterm birth is a major public health problem in the USA.
Preterm birth is a leading cause of morbidity and death of newborn infants.
Preterm birth is responsible for very high financial costs.
Until now, no successful treatment for prevention of preterm delivery has been available.
Trials of 17 α-hydroxyprogesterone caproate (17HPC) have shown consistent efficacy in reducing the risk of recurrent preterm delivery in women with a history of a previous preterm delivery.
The usual dose is 250 mg injected intramuscularly.
Treatment should begin at 16–20weeks of gestation and continue to 36 weeks.
At present, no evidence exists for efficacy of 17HPC for other groups of high-risk pregnant women.
The results of future trials may broaden the indications for the use of this drug.
