Abstract
Osteoporosis-related fractures are a major public health problem worldwide. Antiresorptive drugs, which work principally by suppressing bone resorption, are the established therapeutic approach for the prevention of fragility fractures in patients with osteoporosis. Parathyroid hormone and its analogs represent a new class of agents with anabolic effects on the skeleton. The results of double-blind, randomized, placebo-controlled trials have shown that both the full length, 84 amino acid parathyroid hormone and teriparatide, the parathyroid hormone fragment (1–34) increase bone mineral density and reduce the risk of fracture when administered intermittently to postmenopausal osteoporotic women. Therefore, these drugs should be considered an alternative therapy in postmenopausal osteoporosis.
Keywords
Osteoporosis had previously been defined as a skeletal disease characterized by low bone mass and deterioration of trabecular microarchitecture, with a consequent increase in bone fragility and susceptibility to fracture [1]. More recently, the definition of osteoporosis has been updated by an NIH-sponsored Consensus Conference on Osteoporosis as a skeletal disorder characterized by compromised bone strength, predisposing sufferers to an increased risk of fractures [2]. According to this statement, bone strength may reflect the integration of bone density (a surrogate of bone mass) and bone quality (referring to architecture, damage accumulation [microfractures], mineralization and rate of bone turnover). Clinically, osteoporosis is recognized by the occurrence of characteristic low-trauma fractures, typically at the spine, hip and wrist.
Osteoporosis is the most prevalent metabolic bone disease in countries with an increasing proportion of elderly people. With subtle differences between diagnostic techniques and populations, different studies have demonstrated that the disease affects 30% of women after the menopause [3–5]. It is estimated that approximately 40% of white womenaged over 50 years of age in the USA will experience at least one clinically apparent fragility fracture in their life times [3]. Osteoporosis-related fractures increase with age and there is at least a twofold higher incidence among women compared with men. Moreover, since life expectancy is longer for women, there are proportionately more older women than older men. As a result, fracture prevalence among women is greater than would be predicted from the age-adjusted incidence ratio. There are substantial geographical and ethnic differences in the incidence of osteoporosis-related fractures. The highest age-adjusted rates of hip fracture have been reported in Sweden and the northern USA. Hip fracture rates are higher among whites than blacks regardless of geographical location [6]. In Oriental populations, rates overall are intermediate between those found in whites and blacks. Differences in bone density may count toward geographical and ethnic differences in fracture prevalence.
Osteoporosis-related fractures are a significant public health problem. In the USA, osteoporosis accounts for more than 400,000 hospitalizations, 44 million patient-days of nursing-home care and approximately US$14 billion in healthcare spending/year [7]. In a cohort of patients aged 50 years or older, the median incremental costs (case minus control) due to an incidental fragility fracture increased by 500% in the year following fracture [8].
Mortality associated with complications after hip fracture is high (12–20%) and increases progressively with advancing age. Up to 1 year after a hip fracture, 40% of patients are still unable to walk independently, 60% require assistance in essential activities of daily living and 80% are unable to perform at least one instrumental activity of daily living [9]. Approximately 7% of survivors of all types of fragility fractures have some degree of permanent disability, and 8% require long-term nursing-home care. While the greatest fracture-attributable morbidity and mortality arises from hip fracture, recent epidemiological studies have shown a continuous rise of vertebral fracture prevalence in elderly patients, which impacts on hospitalizations and mortality in a similar manner to hip fractures [10–12].
Until recently, the treatment of osteoporosis was limited to antiresorptive therapies. Hormone replacement therapy (HRT), calcitonin and several bisphosphonates are approved for the treatment of postmenopausal osteoporosis. Raloxifene, a selective estrogen-receptor modulator and the bisphosphonate alendronate are approved for both treatment and prevention of postmenopausal osteoporosis. These agents act primarily by slowing bone resorption and remodeling to prevent further bone loss and stabilize fracture rates. In contrast, parathyroid hormone (PTH) and its analogs, such as teriparatide, represent a new class of anabolic agents for the treatment of postmenopausal osteoporosis.
Biological activity of parathyroid hormone
PTH is a polypeptide consisting of 84 amino acids that is synthesized and secreted by the parathyroid glands. Extracellular calcium level signals an increase in PTH secretion in response to a decrease in calcium concentration via the calcium-sensing receptors on the parathyroid cellular membrane. The physiological role of PTH is to control calcium homeostasis directly, by regulating calcium reabsorption by the kidney, and indirectly, by regulating calcium absorption from the intestines via 1,25-dihydroxyvitamin D. In bone, PTH increases the number and activity of osteoclasts and osteoblasts and thus increases bone turnover. With sustained elevations in PTH levels, osteoclastic activity exceeds that of osteoblasts, resulting in a net release of calcium from bone and a decrease in bone mineral density (BMD). In contrast, single daily injections of the hormone, which raise PTH levels transiently, increase osteoblastic activity preferentially and result in a net increase in BMD [13]. This effect was first reported in humans in a landmark study by Reeve and colleagues in 1980 [14].
Although the secreted peptide in humans is 84 amino acids in length, early structure–function studies suggested that all of the in vivo biological effects of PTH reside in the N-terminal 34 amino acids (hPTH [1–34]), now named teriparatide. However, N-terminal residues capable of receptor activation do not exist in the circulation under normal physiological conditions. The major immunoreactive circulating PTH species are C-terminal fragments of the hormone. These fragments are secreted by the parathyroid cell [15] and also arise from cleavage of intact PTH by target tissues [16]. In vitro and in vivo studies suggest that C-terminal fragments of PTH may have significant effects in bone, and there is increasing evidence of the existence of a separate receptor for the C-terminus of PTH. Cell culture studies with osteoblasts showed that C-terminal fragments containing the last 30 or more amino acids of PTH can stimulate the production of alkaline phosphatase and other markers of osteoblast activity [17]. Bringhurst and colleagues demonstrated that C-terminal fragments enhance apoptosis of osteocytes [18]. It is therefore conceivable that, as compared with teriparatide, the full-length 1–84 amino acid PTH may have slightly different biological actions when used as a treatment for osteoporosis.
Parathyroid hormone as a bone anabolic agent
As discussed previously, intermittent administration of exogenous PTH stimulates bone formation in association with an increase in the bone remodeling rate. This increase in bone remodeling has to be distinguished from other high bone-turnover states, such as estrogen deficiency, which are deleterious for bone. PTH administration increases the amount of bone laid down in each remodeling unit. The combination of high remodeling rate and increased amount of bone laid down in each remodeling unit result in a net gain in the amount of bone tissue [19]. In addition, it has been suggested that teriparatide may initially uncouple formation from resorption, stimulating formation without the requirement of prior resorption [20,21]. The molecular mechanisms whereby PTH stimulates bone formation when intermittently applied are poorly understood. PTH appears to stimulate bone-forming osteoblasts at all levels of their life cycle. Recently, it has been shown that PTH suppresses expression of the specific expression of the sclerosteosis (SOST) gene in vivo and in vitro [22]. The SOST gene is strongly expressed in osteocytes and its protein product sclerostin inhibits proliferation and differentiation of osteoblasts. Therefore, SOST downregulation could play a critical role in PTH bone anabolic actions.
Early studies using synthetic PTH have shown either a maintenance or loss of cortical bone in comparison with improvements in trabecular bone [23,24]. These findings raised concerns that the gains in trabecular bone may be achieved at the expense of cortical bone [25]. Clinical studies with both teriparatide [26] and intact PTH [27] have demonstrated small decreases in BMD, as measured by dual x-ray absorptiometry (DXA), at cortical sites. However, evidence is accumulating that such effects in areal BMD are due to changes in bone structure induced by PTH. Zanchetta and colleagues assessed cortical architecture in the distal radius after 18 months of treatment with teriparatide or placebo using peripheral quantitative computed tomography (pQCT) [28]. Teriparatide administration induced new periosteal apposition, but cortical thickness did not change due to concurrent endocortical remodeling. However, the greater periosteal circumference resulted in an increase in bone strength, as assessed by axial and polar moment of inertia. The same effect of teriparatide treatment has been reported for other critical bone sites, such as the femoral neck [29]. These results suggest that caution must be exercised in interpreting projected BMD measurements by DXA in the assessment of the effects of PTH treatment. A decrease in areal BMD may not result in a loss of bone strength if it is accompanied by an improvement in bone geometry. Furthermore, areal DXA may underestimate improvements in bone mass induced by PTH due to the increased volume of relatively poorly mineralized osteoid that occurs when bone turnover is increased. An increase in cortical bone volume at the hip has been reported in postmenopausal women after 12- [30] and 18-month [31] treatment with PTH (1–84), as assessed by QCT. Therefore, the utility of new, noninvasive imaging modalities needs to be explored for the assessment of the effects of PTH on cortical and trabecular bone.
Introduction to recombinant human parathyroid hormone & teriparatide
Teriparatide
Teriparatide (rDNA origin) is the nonproprie-tary name for recombinant human PTH (1–34) (rhPTH [1–34]), which has an identical sequence to the 34 N-terminal amino acids of human PTH. Teriparatide has a molecular weight of 4117.8. It is manufactured by Eli Lilly and Company (IN, USA) using a strain of Escherichia coli modified by recombinant DNA technology and marketed under the name of Forsteo™ in the EU and Forteo™ in the USA and other countries.
Recombinant human parathyroid hormone (1–84)
rhPTH (1–84) is identical to the endogenous 84-amino acid human PTH and has a molecular weight of 9425. It is produced by and secreted from E. coli as a single-chain polypeptide and carries no post-translational modifications.
Pharmacokinetics
Teriparatide
After subcutaneous injection, the absolute bioavailability is approximately 95% based on combined data from 20, 40 and 80 μg doses [32]. Peak teriparatide serum concentrations are achieved within 30 min of subcutaneous injection and then decline with an apparent half-life of approximately 1 h (Figure 1). This half-life is influenced by the rate of absorption from the injection site, since the true half-life following intravenous administration is approximately 5 min. Serum teriparatide concentrations fall below the limit of quantitation within approximately 3 h [32]. Metabolism or excretion studies have not been performed with teriparatide.

Pharmacokinetic profile of teriparatide and changes in serum calcium.
Recombinant human parathyroid hormone (1–84)
The serum concentration profile of rhPTH (1–84) was assessed in 32 healthy, postmenopausal volunteers after single-dose subcutaneous administration in doses ranging from 0.02 to 5.0 μg/kg (1.2–300 μg for a 60 kg equivalent subject) [33]. Serum concentration–time curves of rhPTH (1–84) exhibited a double-peak profile, with an initial peak appearing 5–10 min after dosing and a second peak occurring approximately 1.5–2 h after administration. The serum terminal half-life of rhPTH (1–84) was approximately 2.5 h [33].
Pharmacodynamics
Teriparatide
The effects of teriparatide on mineral metabolism are consistent with the known actions of endogenous PTH (e.g., it increases serum calcium and decreases serum phosphate levels). In a Phase III clinical study of postmenopausal women with osteoporosis, serum calcium concentration was measured 4–6 h after doses of 20 or 40 μg of teriparatide [34]. After a subcutaneous injection of teriparatide, the serum calcium concentration increased, reaching a maximum concentration between 4 and 6 h, and began to decrease after approximately 6 h, returning to baseline 16–24 h after dosing (Figure 1).
rhPTH (1–84)
In a Phase I clinical study, healthy, postmenopausal women received subcutaneous injections of rhPTH (1–84) in a dose range from 0.02–5 μg/kg [33]. Increases in serum ionized and total calcium concentrations were first observed at a dose of 0.5 μg/kg (30 μg for a 60 kg equivalent subject) and remained within normal ranges through a dose of 1.5 μg/kg (90 μg for a 60 kg equivalent subject). Urinary cyclic adenosine monophosphate (cAMP), an indirect measure of PTH action on the kidneys, and the urinary phosphate:creatinine ratio increased in samples collected during the 12 h after administration for doses of 1.5 μg/kg and higher. The urinary calcium:creatinine ratio increased in 12–24 h and 24–36 h urine collections for doses higher than 2.5 μg/kg. No specific studies have been conducted to investigate the distribution, metabolism and excretion of rhPTH (1–84).
Clinical efficacy in postmenopausal osteoporosis
Teriparatide
The efficacy of teriparatide as a treatment for postmenopausal osteoporosis was assessed in the Fracture Prevention Trial (FPT) [34]. This was a Phase III, double-blind, randomized, placebo-controlled study of 1637 postmenopausal women randomized to receive daily self-administered subcutaneous injections of either teriparatide at doses of 20 or 40 μg or placebo, plus calcium and vitamin supplements, for a median of 19 months of treatment. Women included in this trial were at least 5 years postmenopausal and had a minimum of either one moderate or two mild nontraumatic vertebral fractures. Patients with less than seven evaluable nonfractured vertebrae were excluded.
Effects on bone mineral density
Teriparatide significantly increased BMD at all skeletal sites except the distal radius [34]. Mean percentage changes in patients treated with teriparatide 20 μg/day and placebo at the end point of the study are presented in Table 1. Teriparatide consistently increased BMD at predominantly trabecular bone sites and, to a lesser degree, at mixed cortical/trabecular sites, such as the femoral neck. In the group receiving 20 μg/day, the mean increment in lumbar spine BMD was 9.7%, compared with 2.8% at the femoral neck. Changes in femoral neck BMD were statistically significant compared with the placebo group. Decreases in distal radius BMD were significantly different to placebo only in the 40 μg dose at the midshaft site, and were not associated with an increase in the number of wrist fractures. As previously discussed, the changes in BMD at the radius might reflect the positive effects of teriparatide in the cross-sectional geometry of the distal radius [28], rather than an adverse effect on cortical bone.
Changes in bone mineral density and total body bone mineral content after a median 19 months of treatment in the Fracture Prevention Trial.
p < 0.001 compared with placebo (pairwise comparison).
TPTD20: Teriparatide 20 μg/day; SD: Standard deviation.
Reduction in fracture risk
In the FPT, teriparatide was shown to decrease both vertebral and nonvertebral fractures at doses of 20 and 40 μg/day [34]. The 20 μg dose chosen for marketing reduced the risk of new radiographic vertebral fractures by 65% compared with placebo (Figure 2). The treatment reduced not only the number but also the severity of vertebral fractures. When the analysis was restricted to moderate or severe deformities, in patients treated with teriparatide 20 μg/day the risk reduction was 90%.
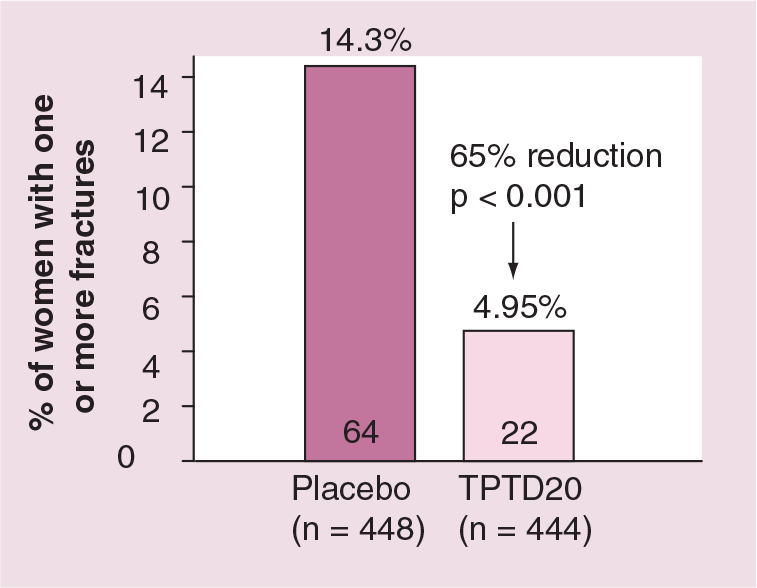
Reduction in the risk of new vertebral fractures after a median 19 months of treatment with teriparatide 20 μg/day in postmenopausal women with osteoporosis.
In a post hoc analysis, Marcus and colleagues demonstrated that the reduction in vertebral fracture risk was largely independent of age, baseline BMD and prevalent fractures [35].
Administration of teriparatide 20 μg/day was also associated with a statistically significant 53% reduction in the risk of nonvertebral fractures [34]. The absolute numbers of nonvertebral fractures were fewer in teriparatide-treated patients at each skeletal site. However, the number of new nonvertebral fractures was too small to estimate the effect of treatment on fracture incidence at specific skeletal sites, such as the hip.
Approximately 77% of the patients who completed the FPT participated in an 18-month follow-up study to assess the long-term durability of the effects of teriparatide treatment. During this follow-up study, patients were permitted to receive any approved treatment for osteoporosis, at the discretion of the investigator. The antifracture efficacy of teriparatide persisted after treatment discontinuation. During the 18-month follow-up, patients previously given teriparatide 20 μg/day showed a 40% reduction in the incidence of new vertebral fractures compared with the original placebo group [36]. In general, patients assigned to receive teriparatide during the FPT maintained a lower incidence of new or worsened, mild, moderate or severe vertebral fractures for 18 months after treatment ended [35]. Similarly, there was a sustained non-vertebral fragility fracture risk reduction after withdrawal of teriparatide treatment [37].
Recombinant human parathyroid hormone (1–84)
The results of the pivotal Phase III trial to assess the safety and efficacy of rhPTH (1–84) in the treatment of postmenopausal osteoporosis have recently become available [38]. The Treatment of Osteoporosis with PTH (TOP) study was a randomized, double-blind, placebo-controlled trial that included 2532 postmenopausal women, randomly assigned to receive daily self-administered subcutaneous injections of rhPTH 100 μg (1–84) or placebo, plus calcium and vitamin D supplements, for 18 months. Only 19% of the women enrolled in this trial had one or more prevalent vertebral fractures at baseline. Patients with hypercalcemia (>10.7 mg/dl), hypercalciuria (urine calcium:creatinine ratio ≥1 mmol/mmol) and two or more lumbar or five or more thoracic or lumbar deformities were excluded.
Effects on bone mineral density
The pattern of changes in BMD induced by rhPTH (1–84) administration was similar to that observed with teriparatide [38]. Compared with baseline, lumbar spine BMD increased by 5.6 and 6.5% after 12 and 18 months of treatment, respectively. Changes in BMD were smaller at nonvertebral sites. Nevertheless, there was a statistically significant increase in BMD at the total hip and femoral neck at 12 and 18 months in the rhPTH (1–84) group compared with placebo-treated patients [38]. A significant decrease, as compared with placebo, was observed in forearm BMD. However, QCT scans performed in a subset of patients showed an increase in cortical bone volume at the femoral neck without a significant decrease in volumetric cortical BMD after 18 months of treatment [31]. These findings suggest that rhPTH (1–84) stimulates the growth of cortical bone. Therefore, the smaller changes in BMD at predominantly cortical sites might result from limitations of BMD measurements by DXA as a surrogate for bone mass, rather than a lesser effect of PTH treatment on cortical bone.
Reduction in fracture risk
In the TOP study, rhPTH (1–84) significantly reduced the overall incidence of new vertebral fractures by 61% compared with placebo (Figure 3). In patients without a prevalent fracture, rhPTH (1–84) administration resulted in a significant 68% reduction in the relative risk of a first vertebral fracture [38]. The TOP study failed to demonstrate antifracture efficacy for nonvertebral fractures.

Reduction in the risk of new vertebral fractures after 18 months of treatment with rhPTH (1–84) 100 μg/day in postmenopausal women with osteoporosis.
Safety & tolerability
Teriparatide
The FPT was the only study large enough to assess adverse events in patients receiving teriparatide as compared with placebo [34]. The more frequent adverse events associated with teriparatide 20 μg/day are listed in Table 2. Only the frequency of dizziness (9%) and leg cramps (3%) was significantly greater in treated patients than in the placebo group [34].
Incidence of adverse events in patients receiving teriparatide 20 μg/day or placebo during the Fracture Prevention Trial.
Significantly different to placebo.
TPTD: Teriparatide.
Adapted from [34].
A total of 16 (3%) of the patients receiving teriparatide 20 μg/day developed circulating antibodies to the drug. However, there was no apparent difference in any clinical outcome between patients who did and did not develop antiteriparatide antibodies [34].
Postdose hypercalcemia occurred at least once in 11% of the patients in the 20 μg group [34]. Most increases in postdose (4–6 h) serum calcium returned to normal within 24 h. Sustained hypercalcemia was confirmed in only 3% of the patients and led to a 50% reduction in teriparatide dosing [34]. Teriparatide induced a mild increase in 24-h urinary calcium excretion by a median of 17% that was significantly different to placebo at 6 months only [34]. However, the trial excluded patients with hypercalciuria or a history of nephrolithiasis and, in patients who developed hypercalciuria, the dose of calcium supplements was reduced.
The FPT was prematurely terminated due to the findings of teriparatide-induced osteosarcoma in a carcinogenicity study in Fischer 344 rats [39]. In this study, rats were given teriparatide for near lifetime (from 8 weeks of age through 2 years). Osteosarcoma in response to teriparatide has not been reported in human studies, but the rarity of these tumors in humans makes the assessment of the relative risk impossible at this point. An independent oncology advisory board concluded that the rat carcinogenicity finding is very unlikely to have relevance to humans treated with teriparatide for a relatively small fraction of the normal life span.
Recombinant human parathyroid hormone (1–84)
The more frequent adverse events associated with rhPTH (1–84) (100 μg/day) during the TOP study are shown in Table 3 [38]. With the exception of vomiting, the side effects associated with rhPTH (1–84) administration were very similar to those reported for teriparatide. However, there were some differences in the frequency of events between the two drugs.
Incidence of adverse events in patients receiving recombinant human parathyroid hormone (1–84) 100 μg/day or placebo during the TOP trial.
Significantly different to placebo.
TOP: Treatment of Osteoporosis with Parathyroid hormone.
Adapted from [38].
Hypercalciuria occurred in approximately twice the percentage of treated patients (46%) than in the placebo group (22.3%) during the TOP study [38]. Hypercalcemia was reported in 27.8% of the patients receiving rhPTH (1–84) in the TOP study [38]. However, the frequency of hypercalcemia in clinical studies of rhPTH (1–84) has been variable. The PTH and alendronate (PaTH) trial was a study on the effects of rhPTH (1–84) and alendronate, alone or in combination, in postmenopausal osteoporosis [30]. The incidence of hypercalcemia in the PTH and PTH plus alendronate groups in the PaTH study was approximately half (13.5 and 13.6%, respectively) that observed in TOP [30]. The differences may relate to the selection criteria employed in these studies. Notably, vitamin D sufficiency was not an eligibility criterion in TOP, as it was in PaTH. However, it should be noted that an assessment of safety based on the PaTH study might not be appropriate given the small size of the study.
No detectable titers of PTH antibodies were found in any patients in a Phase II study of the effects of rhPTH (1–84) 100 g/day in postmenopausal osteoporotic women [40]. No information regarding development of circulating antibodies for rhPTH (1–84) in the TOP study is yet available.
A carcinogenicity study in Fischer 344 rats receiving rhPTH in doses up to 150 μg/kg/day for 2 years demonstrated that rhPTH (1–84) did not increase the incidence of osteosarcoma at a dose equivalent to a rat-to-human exposure ratio of 4.6-fold [41]. However, higher doses induced a dose-related increase in the incidence of osteosarcoma in rats [41].
Regulatory affairs
Teriparatide is approved for the treatment of postmenopausal osteoporosis in women who are at a high risk of fracture. It is currently approved and marketed in the USA, the EU and 21 other countries. Teriparatide is also approved for the treatment of osteoporosis in men, a topic that is beyond the scope of this review. Due to the rat carcinogenicity findings of osteosarcoma, the use of teriparatide is limited to no more than 2 years. In addition, teriparatide is not indicated in patients considered to have increased risk of osteosarcoma, including patients who have Paget's bone disease, unexplained high serum levels of alkaline phosphatase, those who have had radiation therapy involving bone sites, have ever been diagnosed with bone cancer, have any metabolic bone disease other than osteoporosis, or are children or growing adults. The use of teriparatide is contraindicated in patients with hypercalcemia.
Currently, rhPTH (1–84) is under US FDA and European Medicines Agency review and is not yet marketed in any country.
Conclusion
Teriparatide and rhPTH (1–84) are the first in a new class of bone anabolic agents for the treatment of postmenopausal osteoporosis. Both have demonstrated antifracture efficacy in large, placebo-controlled clinical trials in postmenopausal women with osteoporosis. However, different inclusion/exclusion criteria resulted in quite different study populations in the pivotal Phase III trials. This and the lack of head-to-head studies prevent comparisons on safety and efficacy between the two drugs at present. Teriparatide should represent an alternative treatment to the existing antiresorptive agents for postmenopausal women with severe osteoporosis. rhPTH (1–84) has yet to receive regulatory approval.
Future perspective
While the development of anabolic agents represents an important advance in osteoporosis treatment, several key questions on the use of these drugs in daily practice must be addressed. A consensus on which patients should be considered candidates for anabolic therapy remains to be achieved. Patients with a pre-existing vertebral fracture seem logical candidates for treatment with teriparatide. Patients with very low BMD are considered to be at a high risk for fracture, an approved indication for teriparatide. However, the antifracture efficacy of teriparatide has not been tested in postmenopausal women without prevalent vertebral fractures. In contrast, rhPTH (1–84) has demonstrated a reduction in the risk for a first vertebral fracture in postmenopausal osteoporotic women. However, no evidence is available that rhPTH (1–84) or teriparatide are superior to bisphosphonates in reducing the risk for vertebral fractures. The higher cost of anabolic drugs may not justify their use as a first-line treatment in these patients. Patients who have contraindications for, or present with an unsatisfactory response to, antiresorptive therapy (i.e., occurrence of fracture in the face of significant reduction in BMD) may be considered for anabolic treatment. However, previous use of potent bisphosphonates, such as alendronate, has been shown to delay the anabolic response to teriparatide [42]. This effect has not been demonstrated for less potent antiresorptive agents such as raloxifene [42]. The efficacy of PTH therapy in patients who are already receiving antiresorptive treatment remains to be determined.
It has been hypothesized that the anabolic effects of PTH could be enhanced by the concurrent administration of an antiresorptive agent. This hypothesis is not supported by the current evidence. Clinical studies on the effects of treatment with rhPTH (1–84) [30] or PTH (1–34) [43] in combination with alendronate in postmenopausal osteoporotic women showed no evidence of synergy between PTH and alendronate. In fact, the results from both studies suggested that the concurrent use of alendronate might reduce the bone anabolic effects of PTH in terms of changes in BMD.
Whether an antiresoptive agent should be administered after cessation of PTH treatment in order to maintain gains in BMD is still controversial. In the follow-up study after discontinuation of teriparatide, a subset of patients receiving only calcium and vitamin D supplements were followed for 18 months [44]. This study demonstrated BMD decreases at the lumbar spine, femoral neck and total body 18 months after teriparatide was terminated, but BMD at each site remained higher than baseline values. Moreover, BMD at the distal radius and the positive changes in bone geometry induced by teriparatide administration did not change during the 18 months of follow-up [45]. These results are in agreement with the sustained antifracture efficacy of teriparatide observed in a larger pool of patients [36]. Despite the fact that patients were permitted to receive additional osteoporosis treatment during follow-up, the sustained reduction in fracture risk was largely independent of treatment assignment [36]. On the other hand, Black and colleagues reported that in postmenopausal women treated for 12 months with rhPTH (1–84), the gains in BMD were maintained or increased by alendronate but lost when rhPTH (1–84) was not folowed by alendronate administration [46].
The efficacy of shorter cycles of anabolic treatment in combination with alendronate has been investigated. Cosman and colleagues compared the effects of alendronate plus daily injections of synthetic PTH (1–34) with those of alendronate plus synthetic PTH (1–34) in 3-month cycles alternating with 3-month periods without PTH on spinal BMD in postmenopausal osteoporotic women over 15 months [47]. The results suggest that daily and cyclic PTH plus alendronate are equally efficient in increasing spinal BMD [47]. The efficacy of combination, sequential and cyclic schemes on the risk of fractures in postmenopausal osteoporosis should be evaluated in larger trials.
Animal and human studies suggest that a small analog of PTH, Ostabolin-C™ ([Leu27]cyclo[Glu22-Lys26]hPTH [1–31] NH2) is a potent stimulator of bone formation, but a poor stimulator of bone resorption [48]. If confirmed, this property would result in a lower incidence of hypercalcemia, compared with the larger PTH molecules, allowing tolerance for higher doses and faster bone building.
New agents featuring a combination of anabolic and antiresorptive actions are at different stages of clinical development. An orally active vitamin D analog, 2MD, has been demonstrated to restore trabecular and cortical bone mass and strength by stimulating periosteal bone formation and decreasing trabecular bone resorption in ovariectomized rats with low bone mass [49], and a Phase II trial on its effects in postmenopausal women is ongoing. It is anticipated that many other approaches to increasing bone formation will be developed. Despite the enormous progress in the treatment of osteoporosis in the last 10 years, no available treatment reduces the risk of fractures to zero. Could the next decade witness the introduction of a ‘cure’ for osteoporosis?
Executive summary
Unlike bisphosphonates and other approved antiresorptive agents, teriparatide and parathyroid hormone (PTH) (1-84) exhibit bone anabolic effects when administered intermittently by subcutaneous injection.
Phase III clinical trials have demonstrated that teriparatide and PTH (1-84) increase bone mineral density and reduce the risk of vertebral fractures in postmenopausal osteoporotic women.
Teriparatide has demonstrated a reduction in the relative risk of nonvertebral fractures in postmenopausal women with osteoporosis.
PTH (1-84) has been shown to reduce the risk for a first vertebral fracture in postmenopausal women with osteoporosis.
Teriparatide and PTH (1-84) have generally been shown to be well tolerated.
The development of osteosarcoma in rats given teriparatide and PTH (1-84) in carcinogenicity studies is considered unlikely to have relevance in humans, but remains a concern. The use of teriparatide is limited to 2 years.
Teriparatide should be considered an alternative therapy in severe osteoporosis. PTH (1-84) has not yet received approval for marketing.
The benefits of teriparatide and PTH (1-84) in combination or sequential therapy with antiresorptive agents remain to be established.
