Abstract
This review summarizes the epidemiology and consequences of maternal smoking in pregnancy, with emphasis on the adverse effects on birth outcomes. In developed countries, approximately 15%, and in developing countries, approximately 8% of women smoke cigarettes, and adolescents and women from lower socioeconomic groups are more likely than other women to smoke while pregnant. Maternal smoking during pregnancy is the largest modifiable risk factor for intrauterine growth restriction. A meta-analysis of recent studies showed that the pooled estimate for reduction of mean birthweight was 174 g (95% confidence limits 132–220 g). Other studies confirm a weaker association between maternal smoking and preterm birth. The population attributable risk of low birthweight due to maternal smoking in the UK is estimated to be 29–39%. Tobacco smoke toxins damage the placenta and may lead to placental abruption, abortion or placenta praevia. Infants of mothers who smoke in pregnancy are at an increased risk of respiratory complications including asthma, obesity and, possibly, behavioral disorders. These effects may be dose-related, as there is good evidence that mean birthweight decrements are greater with increased numbers of cigarettes smoked during pregnancy. Cotinine is a useful indicator of tobacco smoke exposure in pregnant women and higher levels in body fluids have been related to lower birthweights. Maternal genetic polymorphisms of the cytochrome P (CYP)450 and glutathione-S-transferase (GST) subfamilies of metabolic genes influence the magnitude of the effect of nicotine exposure on birth outcomes through their influence on nicotine metabolism. Greatly increased risk of cigarette smoke-induced diseases, including low birthweight, has been found in individuals with susceptible genotypes. Interventions to control maternal smoking are also considered.
Smoking during pregnancy is considered the largest preventable cause of fetal and infant ill health and death [1]. Tobacco exposure during pregnancy remains a major public health problem, as there are more than 1 billion individuals globally using tobacco products [2]. In the UK in 2004, a fifth of women smoked during pregnancy [201] compared with 27% in 1988 [3], although smoking prevalence remains high in adolescent pregnancies [4].
Maternal smoking is well known to be associated with breast [5], cervical [6] and lung cancer [7]. Nicotine and tobacco-specific carcinogens have also been detected in the cervical mucus of smokers [8], and a significant reduction in the immune response of the cervix in smoking mothers has been demonstrated [9]. Women who smoke may have reduced fertility [10], and both active and passive smoking have been associated with delayed conception [11]. The fertility problems observed in smokers may result at least in part from changes in sex-hormone metabolism. An adverse effect of nicotine and cigarette smoke on the interaction of egg and sperm and implantation of the fertilized egg has been described in animal studies [12].
Maternal smoking is associated with an increased risk of spontaneous abortion, prenatal placental abnormality, birth complications, preterm birth and low birthweight [13]. A meta-analysis of data from studies on pregnancy-related smoking risk in the USA suggested that smoking during pregnancy caused an estimated 19,000–141,000 tobacco-induced miscarriages, 32,000–61,000 low-birthweight babies, 14,000–26,000 infant admissions to neonatal intensive care units and 1200–2200 sudden infant deaths every year [14]. Pregnancy smoking is also associated with an increased risk of ectopic pregnancy and this association remains after adjustment for other predisposing factors, such as sexually transmitted infections and pelvic inflammatory disease [15]. Risk of miscarriage (loss of the pregnancy before the 24th week) may be increased by 25% with smoking during pregnancy [16]. Pregnancy smoking has even been considered a risk factor for subsequent nicotine dependence, as offspring of mothers who smoked during pregnancy have been shown to be at elevated risk of developing nicotine dependence as adults [17].
This review will focus on birth outcomes in smoking mothers related to low birthweight, fetal growth and preterm birth. Mechanisms related to these effects will be considered along with how population risk is influenced by smoking prevalence, maternal age, passive or active cigarette smoke exposure and maternal genetic polymorphisms.
Socioeconomic factors affecting pregnancy outcomes
Maternal socioeconomic status & smoking risk
A social class-dimension model has been suggested related to the tobacco epidemic. Typically, the more advantaged groups are the first to commence smoking and the first to give it up. As the better off quit smoking, it becomes normative behavior only among those with less education and income. Data from the UK have indicated that among the most deprived, smoking prevalence did not decline during the 1980s, and among single mothers, prevalence increased [3]. In many northern European countries, smoking has been established as a marker of deprivation, and tobacco-related damage weighs most heavily on the most deprived women, who often already suffer the poorest health. Smoking prevalence in these countries is typically two- or three-times higher among the disadvantaged. This has been identified as an important reason for the gap in life expectancy in developed countries between rich and poor [18]. It is estimated that the middle-aged mortality gap between socioeconomic groups would be reduced by a half to two-thirds if smoking could be eliminated in developed countries [19].
In developed countries, approximately 15%, and in developing countries, approximately 8% of women smoke cigarettes. The proportion of maternal smokers in the EU varies from 7% in Portugal to 31% in Ireland. In the Asian and Pacific regions, approximately 12% of all women smoke, but prevalence is rising [20]. In England in 2001, people from manual socioeconomic groups were more likely to smoke than those in other groups (32 vs 21%). The widening of this gap over the past 20 years reflects a steeper decline in smoking prevalence among nonmanual, compared with manual classes [202]. Smoking women were reported as more likely to be younger, single and of lower educational achievement [21]. In the USA, it has been estimated that one in five pregnant women who smoked were alone [22], and better education was associated with reduced smoking prevalence during pregnancy [23].
In view of these associations, socioeconomic factors and maternal age could well be confounding influences on the association between smoking and pregnancy outcome. These epidemiological arguments could relate to any of the birth outcomes associated with smoking in pregnancy. Other potential confounding factors to consider include maternal nutritional status and pregnancy infections. In many studies, confounding factors are not included in adjusted analyses. In developing countries, in particular, where low birthweight is common due to poor maternal nutritional status and malaria in pregnancy, the harmful effects of maternal smoking could be additive, leading to severely growth-restricted babies.
Adolescent pregnancy & smoking
Adolescent pregnancy is an important public health problem as it often occurs in the context of poor social support and maternal wellbeing [24]. Adolescents are more likely than older women to smoke while pregnant. In the USA, approximately one in five teenage girls were smokers. Of more than 140,000 women who died prematurely from tobacco-related illnesses each year, 80% began smoking while adolescent [25].
Young teenage mothers have a significantly higher risk of delivering a low-birthweight baby than other age groups, and lower birthweight at all gestational ages in babies of teenage mothers has been reported [4,26]. Children born to teenage smokers have lower birthweight, an increased risk of infant mortality and an increased risk of some congenital abnormalities [27].
In some countries where adolescent pregnancy smoking remains high, specific programs are required to target this high-risk group of girls, with a focus on prevention as well as cessation. Although the economic costs of such programs are high, their introduction is required in view of the high burden of disease in this group of mothers and the long-term consequences for their at-risk infants.
Birth outcomes associated with maternal smoking
Fetal growth restriction & low birthweight
Maternal smoking during pregnancy is the largest modifiable risk factor for intrauterine growth restriction (IUGR), which in turn influences infant mortality and the long-term health outcomes of surviving infants [28–30]. Figure 1 illustrates the lower birthweight percentile curves in smoking mothers at different gestational ages, compared with mothers who did not smoke during pregnancy [4].
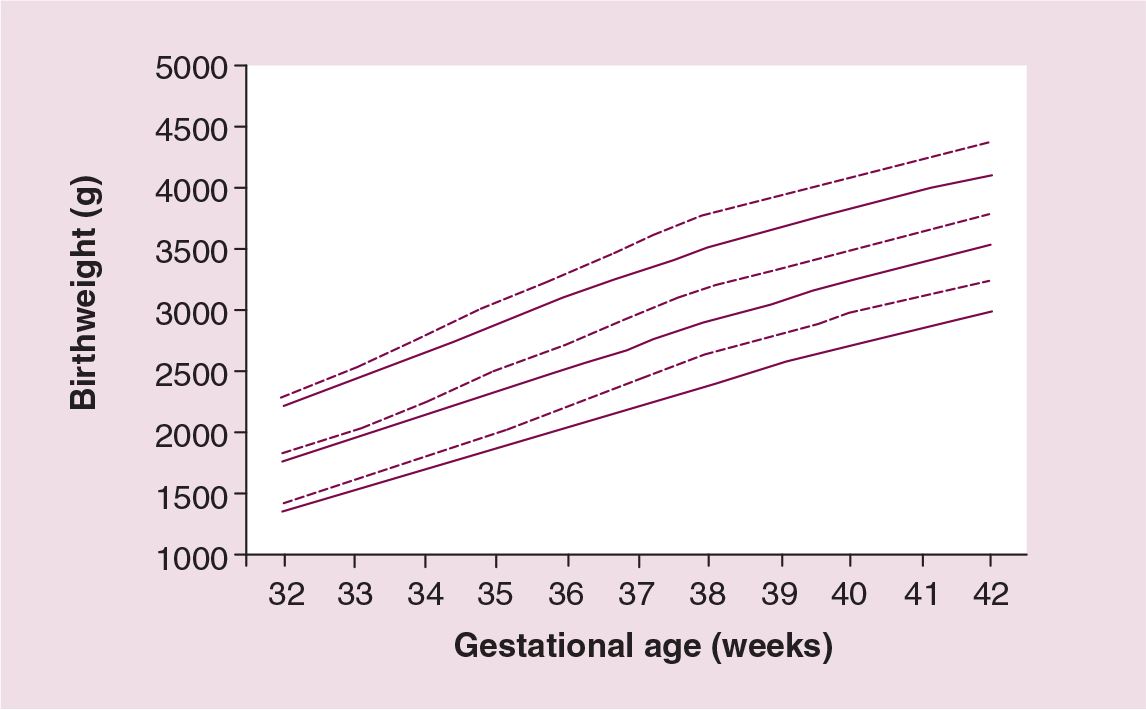
Birthweight for gestational age 10th, 50th and 90th percentiles for babies born to smoking (black lines) and nonsmoking (dotted lines) mothers.
A longitudinal study in the USA, which compared patterns of tobacco use during pregnancy over time and across population subgroups, has reported a higher incidence of low birthweight among singletons born to smoking compared with nonsmoking mothers [31]. Another longitudinal study in Bristol (UK) demonstrated that infants of smoking mothers were symmetrically small at birth (p < 0.0005), compared with those of nonsmokers [32]. A Swedish study comprising 15,185 births reported that babies of women who stopped smoking before the 32nd week of pregnancy were statistically indistinguishable from those of nondaily smokers in terms of birthweight and other pregnancy indices [33], suggesting that fetal effects primarily occurred in the last trimester of pregnancy (Figure 2).

Body size difference between a full-term growth-restricted and normal baby.
A case–control study, which included plasma cotinine and placental cadmium measurement, found that mean birthweight (± standard deviation [SD]) in smokers (cotinine-validated) was significantly reduced (3294 ± 101 g vs 3562 ± 92 g, respectively; p < 0.01). Placental cadmium concentration was positively correlated with the number of cigarettes smoked (p < 0.05) [34]. An inverse dose–response association between smoking metabolic adducts or maternal smoking during pregnancy and birthweight of the offspring (p < 0.04) has been reported [35]. A case–control study of the effects of smoking on placental morphology in 40 smoking and nonsmoking mothers showed extensive aging and placental degenerative changes [36]. Low birthweight, fetal growth restriction and preterm birth in environmental tobacco smoke (ETS) exposure are considered in the section on dose-related response. Figure 3 shows the results of ten recent studies demonstrating differences in mean birthweight due to maternal smoking during pregnancy. The pooled estimation for reduction in mean birthweight in these ten studies was 174 g (95% confidence interval [CI]: 132–220 g).

Differences in mean birthweights (g) between babies of smoking and nonsmoking mothers.
Transgenerational effects of parental smoking on birthweight
A previous study to investigate possible multi-generational influences on birthweight reported an association between birthweight and height of the maternal grandmother and social class of the maternal grandfather, after adjustment for strong predictors of birthweight such as maternal smoking during pregnancy. This suggested a multi-generational influence of smoking on birthweight, which passed through the maternal line [37]. To investigate the influence of maternal smoking during pregnancy on birthweight across generations, a cohort study undertaken in the UK using a random-effects model showed a negative effect of grandmothers' smoking during pregnancy on the grandchild's birthweight (β regression coefficient −24 g, from −50 to 3 g) [38]. Although the authors found that intergenerational birthweight associations depended on particular factors influencing birthweight, they concluded that the reduction in birthweight of offspring attributable to maternal smoking was compensated for in the subsequent generation. Surprisingly, a recent Swedish study on sex-specific, male-line, transgenerational responses in humans found that early paternal smoking was associated with a greater body mass index (BMI) at 9 years in sons, but not daughters [39].
Preterm birth
A number of studies confirm the association between preterm birth and maternal smoking during pregnancy [40]. A regression analysis, which estimated the effect of maternal smoking on birthweight after adjustment for nonmodifiable factors, accounted for 1.5–3.1% of the variance in gestational age at delivery. Nevertheless, it was concluded that most of the gain in birthweight on quitting smoking was due to an effect of smoking on fetal growth restriction [41]. A retrospective study on 1194 Japanese infants demonstrated that maternal smoking during any trimester of pregnancy significantly increased the risk of preterm birth. The birthweight of full-term babies whose mothers smoked only during the third trimester was lower than that of nonsmokers [42].
Smoking is thought to explain 15% of premature births globally [13], and smoking mothers had a two- to threefold increased risk of premature rupture of the membranes before 37 weeks gestation [15], although the overall birthweight reduction in smoking mothers remains primarily due to growth restriction [43,44]. This effect is reported in women of different ethnic origins [45]. A significant effect of passive smoking on preterm birth risk has been reported (odds ratio [OR]: 1.3; 95 % CI: 1.3–28.7) [46], and high maternal ETS exposure (more than 7 h/day) substantially increased the risk of very preterm birth (OR: 2.4; CI: 1.05–5.3) [47].
Other studies have found no significant effects on risk of preterm birth. In one study, an effect was only observed in twin pregnancies [44,48], and among women with daily caffeine intakes of less than 400 mg. Overall, no difference in risk of preterm birth was reported between smokers and nonsmokers [44]. The mechanisms explaining the preterm association with cigarette smoke exposure are not well understood. Maternal vasoconstriction effects may be important and could lead to premature rupture of the membranes through mechanical stress disrupting membrane integrity [49].
Estimation of population attributable risks for adverse birth outcomes
A population attributable risk (PAR) of 29–39% has been estimated for low birthweight due to maternal smoking during pregnancy across the UK [201]. Results from a large study in Merseyside (UK) demonstrated that almost a third of low-birthweight and small for gestational age cases and 15% of preterm births could be prevented if mothers stopped smoking during pregnancy [50]. Figure 4 illustrates PAR values for adverse birth outcomes due to maternal smoking during pregnancy by mothers' age for almost 13,000 deliveries in Merseyside [50].

Population attributable risk for adverse birth outcomes related to pregnancy smoking in adolescents (<20 years) and adults.
Placental damage
The human placenta is hemomonochorial, meaning that only one chorionic cell layer exist between the maternal and fetal blood vessels, favoring exchange of gases, nutrients and metabolic products [51]. Tobacco-smoke toxins readily cross the placental membrane [52]. Compounds in tobacco smoke such as nicotine, carbon monoxide and polycyclic aromatic hydrocarbons (PAHs) are known to cross the placenta, and are found in newborn blood [53]. Several placental complications occur as a result of this, including miscarriage [14] and placenta previa [13]. The increased risk of adverse outcomes is eliminated when women with fetal growth restriction, placental abortion or placental previa are excluded from data analysis [54], suggesting that smoking produces most of its effects through placental pathology. Analysis of 15 varied placental morphometric measurements demonstrated that only three of them were correlated with maternal cigarette-smoke exposure [55].
Tobacco smoke induces placental and fetal enzyme systems, which can endorse procarcinogen to mutagen forms and damage placental DNA [56]. Nicotine-induced vasoconstriction of blood vessels could restrict placental blood flow and reduce the supply of nutrients and oxygen to the fetus. Carbon monoxide in tobacco smoke displaces oxygen in the circulation, reducing the amount of oxygen available to the fetus and impairing fetal growth [57]. Carbon monoxide binds to maternal and fetal hemoglobin with an affinity that is more than 200-times that of oxygen [51]. Nicotine in cigarette smoke increases the maternal heart rate and induces vasoconstriction [58]. Elevated concentrations of toxic chemicals in cigarette smoke, such as benzo(α)pyrene, cyanide, thiocyanate and cadmium occur in the blood and urine of smoking mothers and interfere with fetal growth, contributing to low birthweight [59,60]. In smokers, elevated carboxyhemoglobin levels were reported to interfere with placental function, maternal–fetal oxygen transport and fetal circulation. The combination of intrauterine hypoxia and impaired placental blood flow is likely to result in fetal growth restriction [61]. Heavy smoking alters placental cadmium concentrations [62].
Effects of maternal pregnancy smoking on child health
Respiratory consequences
Infants of parents who smoke are at increased risk of respiratory complications compared with children of nonsmokers [63]. There are many studies linking maternal smoking during pregnancy to childhood predisposition to respiratory disorders. These include: deficient childhood lung function [64], increased childhood asthma [65], increased incidence of pneumonia and respiratory symptoms [66], wheeze [67] and lung complications during and after surgery [68].
A large Finnish study of 58,841 singleton births observed a positive relationship between maternal smoking during pregnancy, fetal development and the risk of asthma in childhood. The risk of asthma attacks during the first 7 years of life was significantly increased in children of smokers (OR: 1.3; 95% CI: 1.1–1.6), and children under 5 years were twice as likely to develop asthma compared with those free from exposure [65]. A meta-analysis of 17 studies reported that asthma occurred with a 45% greater frequency in children of smokers [69]. There is also substantial evidence confirming that maternal smoking during pregnancy causes sizeable adverse effects on neonatal lung function and increases the risk of infant respiratory disorders [65].
A dose–response relationship between maternal smoking during pregnancy and infant respiratory function is reported, with infants of mothers who smoked at least one pack of cigarettes/day having a 2.8-times greater risk of developing a lower respiratory infection [70]. A survey of 5300 children in New Zealand found an association between parental smoking and night cough and wheeze among 13–14-year-olds interviewed with a video questionnaire, but there was no association among 6–7-year-olds [71]. Conversely, a recent study reported that passive smoking was not associated with additional episodes, or increased prevalence or severity of childhood asthma [72]. Distinguishing between the residual effects of maternal smoking during pregnancy and the consequence of childhood ETS exposure to explain these deficits is methodologically complex.
Other disorders
The adverse effects on the child of maternal smoking during pregnancy are not limited to low birthweight. A population-based study including 482 Swedish women observed that children of mothers who smoked during pregnancy were at increased risk of overweight by 5 years of age using BMI measurements (relative risk [RR]: 2.5; 95% CI: 1.5–4.2) [73]. Data from the obligatory school entry health examination in Germany demonstrated that maternal smoking during pregnancy was also significantly associated with increased childhood risk of overweight (OR: 1.4; 95% CI: 1.1–1.9) and obesity (OR: 2.1; 95% CI: 1.3–3.2) [74]. Further confirmation of this comes from an analysis of 8765 German children (aged 5–7 years), which demonstrated that the risk of obesity (BMI >97th percentile) was increased with pregnancy smoking (OR: 2.3; 95% CI: 1.3–2.2). The effect was mainly before rather than during pregnancy (OR: 1.7; CI: 1.3–2.2), suggesting that early intrauterine exposure to cigarette smoke is of importance [75].
Maternal smoking during pregnancy also results in a reduction in organ size and function. A longitudinal study of 1335 normal infants investigated weight, length/height and head circumference measured at birth and on up to ten occasions to the age of 5 years, and found that infants of maternal smokers were symmetrically small at birth (p < 0.0005) compared with infants of nonsmokers. They showed complete catch-up growth over the first 12 months [32]. A prospective study of antenatal fetal growth assessed by ultrasound at 20 and 30 weeks gestation, in 1650 low-risk, singleton, white pregnancies in London (UK) also found a negative association between maternal smoking during pregnancy and fetal size at birth, including head circumference (p < 0.001) [76]. These effects on reduction in head circumference could have important implications for mental performance and ability during childhood.
Parental smoking is associated with approximately 24% of sudden infant death syndrome (SIDS) worldwide [77]. In Sweden, among more than 600,000 pregnancies/year, the risk of stillbirth increased by 40% in smoking compared with nonsmoking mothers [54]. There is some evidence that childhood behavioral difficulties including conduct disorders, attention deficit disorder [78,79] and idiopathic mental retardation occur more frequently in children of smoking mothers [82]. In a Danish study, iodine transferred to breast milk in smoking mothers correlated negatively with urinary cotinine concentration (p < 0.005), suggesting that maternal smoking could increase the risk of iodine deficiency, leading to possible brain damage in children [81].
Human as well as animal studies have recorded accelerated motor activity, learning and memory deficits in the offspring of mothers exposed to cigarette smoke during pregnancy. Indian scientists have exposed several groups of experimental rats to varying levels of nicotine, reaching levels of exposure experienced by heavy smokers. Animals were examined at different periods after birth. Observable effects included significantly reduced thickness of the cerebral cortex, smaller cerebral cortex neurons and reduced brain weight. The study has also demonstrated that the greater the dose of nicotine, the greater the biological effects upon the offspring, providing a good biological model to support the many other studies on the adverse effects of maternal cigarette smoking on maternal health and reproduction [82].
Dose-related associations
Nicotine dependence leading to smoking addiction is also strongly associated with the number of cigarettes smoked per day. Girls and women who smoke appear to be nicotine dependent in relation to the number of cigarettes smoked per day [202]. The importance of this is that the risk of adverse birth outcomes increases with an increasing number of cigarettes smoked daily [83], with heavy smokers delivering babies up to 458 g lighter than nonsmokers [84]. Overall, a significant dose–response relationship has been reported for the effects of maternal cigarette smoking during pregnancy on birthweight reduction [31]. A study of 60,000 singleton births in Finland reported that mean birthweight was 250 g lower among babies of mothers who were heavy smokers (> ten cigarettes/day), compared with nonsmokers [65]. Mothers who were light smokers (< ten cigarettes/day) delivered babies weighing on average of 96 g less than infants of nonsmokers [31]. Analysis of the birthweights of 7803 full-term singleton births in Vienna (Austria) also demonstrated a significant effect of any reduction in the numbers of maternal cigarettes smoked daily during pregnancy on birthweight restriction (p < 0.05) [85]. Other research has shown that even light maternal smoking during pregnancy (< five cigarettes/day), was significantly associated with elevated rates of low birthweight (p < 0.05) [86].
The amount of nicotine absorbed is clearly important. A study of 87 smoking and nonsmoking Croatian pregnant women reported a significant correlation (p < 0.01) between mean gestational age at delivery and heavy maternal smoking (> 20 cigarettes/day), confirming a dose-related effect [87]. A European longitudinal study also demonstrated that children of moderate/heavy-smoking mothers during pregnancy had, on average, a 245 g lower mean birthweight, 1.22 cm shorter body length and 0.66 cm smaller head circumference than babies of nonsmokers [88]. A retrospective cohort study of maternal smoking and delivery outcomes in Swedish infants born between 1983 and 1996 demonstrated a significant dose–response relationship between maternal smoking during pregnancy and preterm birth, IUGR and small head circumference (p < 0.05). These studies demonstrated that maternal smoking during pregnancy accounts for several negative delivery outcomes [89].
Comparison of birthweights for women who smoked low-, compared with high-yield cigarettes (low or high nicotine and carbon monoxide) has shown a dose-related effect of nicotine and carbon monoxide on birthweight reduction. The risks were primarily among those who smoked the equivalent of 13 or more cigarettes/day [90]. The dose–response effects of maternal cigarette smoking on pregnancy outcomes is illustrated in Figures 5 & 6 for several thousand women in the UK. There were no differences in birth outcomes between nonsmoking and light-smoking mothers. There was a trend towards shorter gestational ages and greater reductions in birthweight with increased number of cigarettes smoked daily [4].

Mean birthweight and number of maternal cigarettes smoked/day.
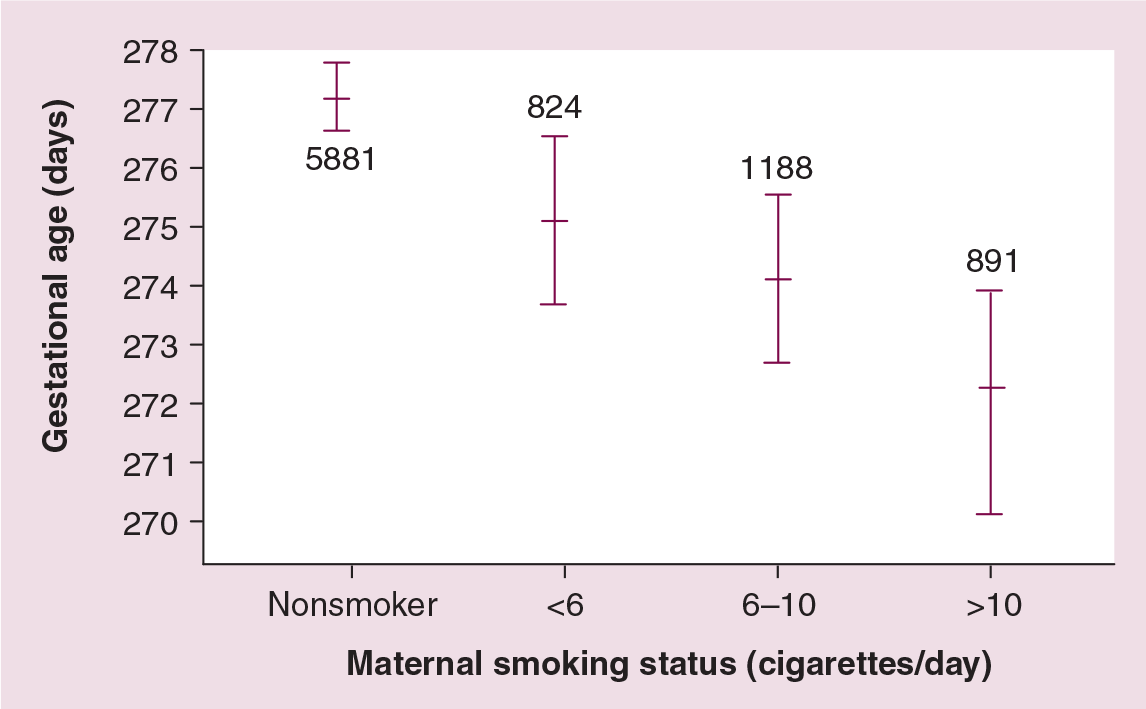
Mean gestational age, and number of maternal cigarettes smoked/day.
Environmental tobacco smoke exposure & birth outcomes
It is less certain whether maternal exposure to environmental tobacco smoke has a similar effect on birthweight, although sizable birthweight reductions have been reported (20–120 g) [35,91]. The adjusted OR for small for gestational age births in ETS-exposed compared with unexposed mothers has been reported as 0.82 (95% CI: 0.51–1.33) [92]. Babies born in the UK to women with heavy passive cigarette smoke exposure were 70 g lighter than those in the least-exposed group, and were 20% more likely to be low-birthweight babies [92]. A recent review concluded that, on average, infants born to women exposed to passive smoke during pregnancy were 40–50 g lighter than those born to mothers who were not exposed [15]. There was some evidence that women with ETS exposure during pregnancy were at increased risk for preterm birth [47]. Other studies have demonstrated birthweight reductions of 19–53 g in smoking mothers exposed to ETS, primarily due to growth restriction [93].
Cotinine assessments & birth outcomes
Cotinine concentration is a useful indicator of both active smoking and ETS exposure. A cohort study of 2418 pregnant women in the USA who smoked during pregnancy reported that smoking was related to cotinine concentration at the time of delivery (OR: 2.2; 95% CI: 1.1–4.5) [94]. In a US study, infants of non-ETS exposed mothers had lower cotinine levels (<2 ng/ml) than children of exposed mothers (cotinine 2–10 ng/ml), whose babies were on average 45 g lighter, although this effect was not statistically significant (95% CI: −126 to 36 g) [95]. Pregnant women in the highest quintile of salivary cotinine concentrations (>1.7 ng/ml) had babies with a significantly lower mean birthweight than mothers in the lowest quintile range [96]. Assessing the accuracy of self-reported maternal smoking with plasma and urinary cotinine levels in pregnant women has demonstrated good levels of agreement, with 84.6% of women who reported smoking and 94.5% of women who denied smoking correctly identified [97].
In two Polish studies, a negative association (p = 0.06) between fetal biparietal diameter and pregnancy serum cotinine level at 20–24 weeks gestation and birthweight has been reported [98].
Similar findings were reported from the USA [99] and South Korea [100] using urinary cotinine measurement. In a New Zealand smoking-cessation program, analysis of 4178 cotinine samples of 3082 pregnant women demonstrated that adjusted cotinine validated measurements in the first, second or third trimester were significantly reduced by 4.7% (p = 0.02) [101]. A biochemical study on 1254 women booking for antenatal care using maternal plasma cotinine level further demonstrated this, as cotinine levels in smoking mothers were more closely related to birthweight than the number of maternal cigarettes smoked [91].
A population-based study in southeast Finland of 389 nonsmoking women who were exposed to ETS used the hair nicotine level as a biomarker. The authors found that the risk of preterm birth in the high exposure group (>4 μg/g) was six-times that of the lowest exposed group (OR: 6.1; 95% CI: 1.3–28.7). The low birthweight risk estimates were OR: 1.1 (95% CI: 0.96–1.2), and for fetal growth restriction OR: 1.0 (95% CI: 0.96–1.2) [46]. A significant correlation between paired newborn/maternal samples and levels of aromatic-DNA adducts (p < 0.001) and plasma cotinine (p < 0.001), but not levels of polycyclic aromatic hydrocarbon (PAH)-DNA adducts, has been previously reported [102].
Maternal genetic factors, smoking & birth outcome
DNA adducts
Many chemicals require some kind of biotransformation to activate derivates, which can be harmful to the body. Most human metabolizing enzymes are genetically polymorphic and these may affect enzyme activity or inducibility. The sensitivity to procarcinogens differs among individuals, and this may be of substantial importance in carcinogenesis [103]. Drug-metabolizing enzymes, which often display genetic polymorphisms, convert many tobacco carcinogens into DNA-binding metabolites in target cells, and thus regulate intermediate-effect markers such as DNA adducts [104]. Use of DNA adduct levels as a measure of exposure has established a dose–response relationship between intensity of smoking exposure and birthweight reduction. This could prove useful for understanding the relationship between maternal smoking, birthweight and, perhaps, other adverse pregnancy outcomes [35,104–106]. Chemicals entering the body from the environment are metabolized by cellular enzymes to reactive or inert metabolites.
Metabolism of nicotine & other toxins in tobacco smoke
In humans, a significant proportion of the metabolic genes involved are polymorphic. These genes are classified as phase I (including some activity enzymes in which the original nonpolar compound becomes polar and reactive) and phase II (detoxifying enzymes in which the transformed polar compound is conjugated with certain endogenous functional groups) [107]. The adverse health effects of cigarette smoke may depend on the combined effects of the phase I and II enzymes associated with nicotine metabolism. The magnitude of this effect for pregnant women and its impact on birthweight and preterm birth depends on the cytochrome P (CYP)450 and glutathione-S-transferase (GST) subfamily of genes involved in the metabolism of cigarette-smoke toxins [108]. These results could have significance for maternal genotype screening, although we do not know all the genes involved in nicotine metabolism, and different toxins from cigarette smoke need different enzyme-inducting genes. Genetic properties that are detrimental for one toxin may be accompanied with advantageous properties for other toxins within the same individual. Genotyping in modern epidemiological studies is useful for understanding the different outcomes after similar exposures. Further research in different population groups is required to identify other genes, which may also be responsible for adverse pregnancy outcomes.
The product of the GSTM1 gene catalyzes the detoxification of alkyl and polycyclic aromatic hydrocarbons that are intermediary forms of many carcinogens, such as tobacco smoke. Interest in GSTM1 has been stimulated by studies indicating that homozygosity for GSTM1 could increase the risk of various diseases, including adverse birth outcomes [109].
There are important differences between males and females in terms of genetic susceptibility. Female susceptibility to smoking-related diseases may be higher than males. A Polish study that investigated the combined genotypes of GSTM1 and GSTT1 found that 17.7% of women had an absence of the GSTT1 genotype, compared with 15.2% of men. Although this difference was not statistically significant, it raises the possibility that sex differences in susceptibility to disease may also be caused by genotypic differences in detoxifying enzymes [110].
It has been suggested that alterations in the patterns of growth at different stages in gestation will lead to different anthropometric phenotypes at birth. A British study of 1650 mothers found that shape at birth was influenced by the sex of the child, with males having greater birthweight, length and head circumference, and smaller skin-folds than females [76]. The authors have already reported a gender-specific effect of maternal age and pregnancy smoking in which male babies born to smoking adolescents had lower mean birthweight than females. The difference was largely due to shorter gestational age and increased preterm birth among the male babies (p < 0.01) [4].
Gene–environment interactions & pregnancy outcomes
Pregnant women with certain metabolic genotypes are at high risk, and are more likely to experience adverse outcomes if they smoke [100]. This may relate to the fact that not all women who smoke cigarettes during pregnancy have low birthweight babies. The reason for this variability is unclear, but could relate to maternal genetic polymorphisms [108]. Both genetic and environmental influences play an important role in interactions related to maternal cigarette smoking [111]. Mothers who smoke heavily may pass on a genetic predisposition for cigarette smoking or other smoking-associated conditions. A significant gene–environment association among female smokers compared with nonsmokers has recently been reported [112]. A strong familial aggregation of low birthweight has been determined by fetal genotype (10%) as well as maternal genotype (24%).
In a molecular epidemiological study of metabolic gene polymorphisms and low birthweight risk in smoking and nonsmoking mothers, a significant positive association was found between smoking and low birthweight based on maternal genetic polymorphisms [108]. For the CYP1A1 genotypes, the OR for low birthweight was 1.3 (95% CI: 0.6–2.6) for the AA genotype versus 3.2 (1.6–6.4) for the Aa, or aa genotypes. The OR for low birthweight associated with the GSTT1 genotype was 1.7 (0.9–3.2) if present versus 3.5 (1.5–8.3) if absent. For nonsmokers, genotype alone did not confer a significant adverse effect. The greatest reduction in birthweight and gestational age was found when both the CYP1A1 (Aa/aa genotypes) and GSTT1 (absent genotypes) polymorphisms were present (−1285 g and −5.2 weeks). There was a statistically significant difference in the percentage of CYP1A1 genotypes in blacks versus whites (p = 0.005) but not in the percentage of the GSTT1-absent genotype (p = 0.39). Unless maternal genotype was considered, there was no significant difference between blacks and whites with respect to the association of maternal smoking and infant birthweight.
A case–control study of 304 Chinese singleton live-born mother–infant pairs showed that both genotypes of the CYP1A1 (MspI and Hinc II) polymorphism were significantly associated with reduced birthweight by −95 and −132 g, respectively, after adjusting for passive smoking exposure. There is evidence that if both mother and baby had the null allele of GSTT1, the joint effect of smoking and presence of the genotype was higher (OR: 4.9; 95% CI: 0.7–36.9) [113]. An American study of 266 pregnant women described an association between smoking and birthweight in subjects with the null allele for the GSTT polymorphism (p < 0.01) [100]. In the same study, there was a significant negative association between maternal cotinine levels and birthweight (p < 0.05).
The CYP17 enzyme mediates both steroid 17-hydroxylase and 17, 20-lyase activities and functions at key steps in the genesis of human sex steroid hormones. Pregnant women homozygous for the A1 allele of CYP17 are reported to have higher frequencies of fetal growth restriction; for example, women carrying the A2 allele of CYP17 have a lower risk of fetal growth restriction [114]. This study was limited by not considering pregnancy smoking; therefore, future research is required to determine the role of the CYP17 polymorphisms in the metabolism of nicotine and other toxins in relation to pregnancy outcomes.
Interventions to control maternal smoking
Only nonsmoking is the right way to protect the fetus and child. Advantages of smoking-cessation programs include the benefits of population health gains through reductions in smoking prevalence [115], reduction in the risk of dying from smoking-related diseases [116], as well as reductions in hospital admissions [117]. Giving up smoking during pregnancy also reduces the risk of infant illness, with 8.8% of infants whose mothers stopped smoking requiring hospital care in the first month of life compared with 11.4% of infants whose mothers continued to smoke [118]. Women may be more likely to stop smoking during pregnancy than at other times due to their awareness of the additional hazards to their baby [119]. A meta-analysis of randomized, clinical trials in pregnant smokers demonstrated that the active delivery of smoking-cessation methods in prenatal care increased the quitting rate by 50%, although these cessation rates were still modest [120].
Many women who succeed in stopping smoking during pregnancy later return to smoking after the birth of their baby. Of mothers who gave up smoking in the year before or during pregnancy, 22% relapsed 6 weeks postpartum [121]. An American study reported that 56% of women who quit smoking during pregnancy started again within 1 month of delivery [41]. Similar relapse rates are reported by others, with up to two-thirds of women who stopped smoking during pregnancy returning to smoking within 6 months of delivery [122]. Within a year of childbirth, most mothers may have recommenced smoking, thus also exposing their child to the harmful effects of secondhand smoke [123]. In view of the high relapse rates after delivery among women who have quit during pregnancy, specific intervention programs provided through pediatric consultations during child follow-up visits should be strongly recommended.
High-quality interventions to help pregnant women quit smoking can produce an absolute improvement of 8.1% in the validated late-pregnancy quit rate [124]. Suggested methods to do this include various forms of smoking-cessation programs, nicotine-replacement therapy and educational self-help programs, all of which may be targeted. It is difficult to make recommendations regarding which specific components should be included in an intervention, due to the diversity of methods that have been tested [125]. Self-help materials appear to be useful, especially when used in the populations for whom they were developed [126]. It is ineffectual to merely provide risk information to pregnant women. It is necessary to enroll them specifically in smoking-cessation programs [124].
Counseling should be aimed at developing behavioral aids to enable quitting, address specific concerns about quitting, such as fear of weight gain or withdrawal symptoms, and be responsive to individual needs [127]. Cognitive behavioral counseling programs show the highest success rates. The two main factors associated with success appeared to be involvement in an intense counseling program administered by medical, nursing or other trained clinicians and exposure to self-help materials that are specific to the characteristics of the individual [124]. A strong relationship between person-to-person session length in intervention programs and success in quitting has been reported [128].
Nicotine-replacement therapy has been considered as an alternative method for reducing the body's nicotine and can aid removal of other harmful constituents. It can assist highly nicotine-dependent mothers who are already motivated to quit. The intention is to reduce nicotine withdrawal symptoms while quitting, and to facilitate breaking the habit. Nicotine-replacement therapy can include the use of chewing gum [129], nicotine patches [130], nicotine inhalers [131] and prescription drugs such as bupropion (Zyban®) [132].
Conclusions
Smoking during pregnancy is considered the largest preventable cause of adverse birth outcomes in developed countries. Young teenage mothers have a significantly higher risk of delivering a low-birthweight baby than other age groups. Tobacco smoke toxins readily cross the placental membrane, and nonsmoking mothers who are exposed to ETS, either from their partner or in the workplace, are still at risk for similar effects to those associated with active maternal smoking during pregnancy. Babies born to mothers who smoked during pregnancy were 175 ± 50 g lighter than babies born to nonsmoking mothers, with a dose–response pattern. Maternal metabolic gene polymorphisms are associated with adverse birth outcomes in smoking mothers, confirming a gene–environmental interaction. Fundamental public health interventions such as school- and clinic-based health education, nicotine-replacement therapy, maternal genetic screening and maternal counseling are required. Nicotine replacement therapy during pregnancy may be harmful to the fetus and, if used, assessment of pregnancy outcomes will be important. In some countries where adolescent pregnancy smoking remains high, specific programs will be required to target these high-risk young women and girls with a focus on prevention as well as cessation. Pediatricians and pediatric nurses could be more actively involved in preventing postnatal maternal smoking relapses during infant follow-up visits for their babies.
Future perspective
Surveillance of adolescent maternal smoking in pregnancy is critical to facilitate targeted strategies for smoking interventions.
Mother–infant–child cohort studies are required to identify more clearly the long-term consequences of smoking in pregnancy on child health. These studies should incorporate characterization of the fetoplacental unit in terms of placental histology, endocrinology and function.
The combined effects of maternal smoking and alcohol consumption on the fetus should be assessed and the influence of specific GST and CYP gene polymorphisms on susceptibility to adverse outcomes in mothers with these exposures should be determined.
Intergenerational effects of maternal and paternal smoking should be better characterized with detailed family studies.
Innovative strategies are required to greatly increase public awareness of the major health problems for the child arising from maternal pregnancy smoking.
Executive summary
A total of 15% of women in developed countries and 8% in developing countries smoke during pregnancy.
Women who smoke during pregnancy are more likely to be young and of lower socioeconomic status.
Many studies link maternal smoking to respiratory disorders such as asthma, which is 45% more frequent in children born to smokers. In older children, it is difficult to isolate effects of passive smoking from residual effects of maternal smoking.
Other potential interactions include sudden infant death syndrome, childhood obesity, behavioral difficulties and brain damage.
Tobacco smoke toxins cross the placenta and restrict placental blood flow, reducing available oxygen and impairing fetal growth.
Smoking is associated with numerous adverse reproductive events including infertility, pregnancy complications and nicotine dependence in the offspring.
Maternal smoking is associated with a lower mean birthweight (132–220 g), intrauterine growth restriction and preterm birth. The population attributable risk for low birthweight due to maternal smoking is estimated to be 29–39%.
On average, babies born to women exposed to passive smoking during pregnancy are 40–50 g lighter than those born to nonexposed women.
Effects on intrauterine growth are dose-dependent and greatest during the third trimester. Cotinine concentration is a useful indicator of active and passive smoking.
Drug-metabolizing enzymes convert tobacco carcinogens into DNA-binding metabolites that regulate DNA adducts. Adduct levels provide a marker of intensity of smoking exposure.
Mothers who smoke heavily may pass on a genetic predisposition for cigarette smoking.
Glutathione S-transferase (GST) and cytochrome P (CYP) gene subfamilies are involved in detoxification of polycyclic aromatic hydrocarbons, and polymorphisms of these genes may influence susceptibility to disease.
Presence of both the CYP1A1 (Aa/aa) and GSTT1 (absent) genotypes is associated with a synergic reduction in birthweight and gestational age in smoking mothers.
Adverse birth outcomes would be reduced if women stopped smoking during pregnancy.
Women are more motivated to quit smoking during pregnancy but often restart following delivery.
Interventions include nicotine replacement therapy, educational self-help programs and counseling based on cognitive therapy.
Specific programmes should focus on pregnant adolescents who have the highest risk of adverse birth outcomes.
