Abstract
Vaginal prolapse is a common health problem, and although severe morbidity is rare, it can have marked effects on quality of life. The treatment of vaginal vault prolapse can be a difficult and challenging problem. A detailed history and clinical evaluation is required in order to plan the appropriate choice of procedure. There are numerous surgical procedures that have been described using either abdominal or vaginal approaches. The choice of procedure is often dependent on the individual surgeon's choice and experience, and should be tailored to the individual patient. The ideal procedure should have a low risk of morbidity and mortality, but should also have long-term durability. There is a need for large, randomized trials to evaluate surgical techniques to correct vaginal prolapse and related urinary, bowel and sexual dysfunction.
Keywords
Vaginal pelvic organ prolapse (POP) is a common condition that has marked effects on quality of life. Prevalence estimates vary from 2% for symptomatic prolapse to 50% for asymptomatic prolapse [1]. Approximately 50% of parous women will have some degree of prolapse and only 10–20% of these seek medical help [2]. The lifetime risk of surgery for prolapse has been estimated to be approximately 11.1%, and 30% will undergo reoperation for recurrent prolapse [3]. Concomitant surgery at the time of POP repair is common, with approximately 50% of patients having more than one procedure during POP repair [4,5].
The maintenance of normal anatomy is dependent on pelvic floor support which is, in itself, dependent on the functional and structural integrity of the striated muscle of the pelvic floor and the surrounding connective tissue.
The etiology of prolapse is multifactorial. Advancing age, parity and collagen weakness are all quoted as significant predisposing factors [6]. Pathophysiological mechanisms that have been proposed include pelvic-floor denervation, direct trauma to the pelvic-floor musculature, abnormal synthesis and degradation of collagen, and defects in endopelvic fascia [7–10].
Assessment & management of prolapse
Prolapse is often asymptomatic and symptoms that are present may often not correlate with the site or severity of POP [11]. Symptoms from one compartment may overlap with another and include a sensation of pelvic pressure, vaginal ‘heaviness’ or ‘dragging’, recurrent irritative bladder symptoms, voiding difficulties, incontinence or defecatory difficulties. It is therefore important to evaluate the pelvic floor as an interrelated system. The ‘integral theory’ published by Petros and Ulmsten describes the pelvic floor as three anatomical zones – anterior, middle (apical) and posterior, and hypothesized that an intact musculovaginal unit plays a role in preserving continence (Figure 1) [12]. This theory relates specific symptoms, such as stress, urge incontinence and voiding difficulties to pelvic-floor laxity. The treatment of symptoms is thus often directed at restoring anatomical laxity in these specific compartments:

The pictorial diagnostic algorithm.
Anterior compartment – prolapse of the urethra or bladder (cystocele) or both into the vagina (cystourethrocele)
Apical compartment – uterine or vault descent and enterocele (herniation of the Pouch of Douglas)
Posterior compartment – prolapse of the rectum into the vagina (rectocele)
Traditionally, prolapse has been defined as first degree if it descends within the vagina, second degree if it descends to the introitus, or third degree if it descends outside the introitus. More recently, in an attempt to standardize examination and prolapse quantification, the International Continence Society has produced a POP quantification (POP-Q) system, which is highly reproducible [13]. This system assesses prolapse during a maximum Valsalva maneuver and describes the position of six vaginal sites relative to the hymen measured with a centimeter ruler. External measurements of the genital hiatus and perineal body are also taken (Figures 2 &3). This method allows the standardization of prolapse grading and can be used as a validated tool for surgical follow-up and research.
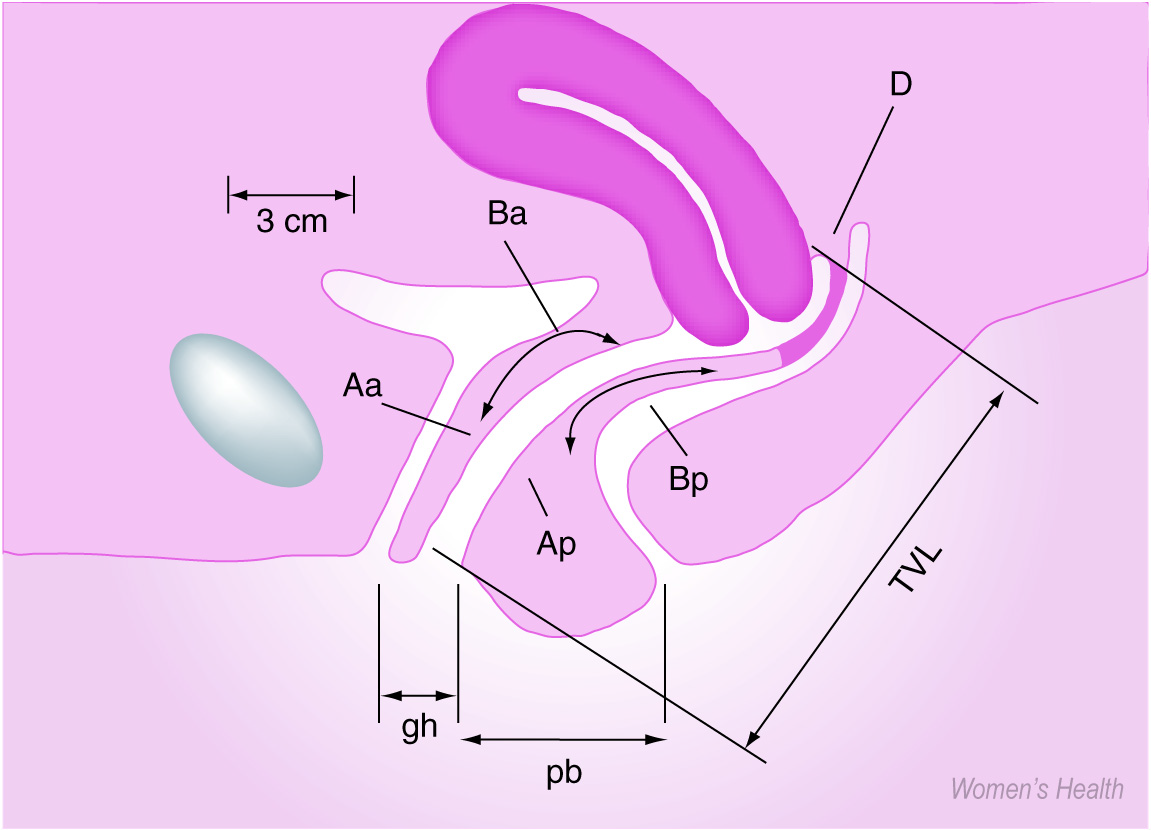
Representation of points used to quantify prolapse.

Contrasting measurement between normal support and posthysterectomy vaginal eversion.
Treatment
The choice of treatment method depends on the severity of symptoms, degree of prolapse and the suitability of the patient for surgical intervention. In cases where prolapse is present, but the patient is asymptomatic and the overlying vaginal skin is healthy, the prolapse can be observed and the patient reassured that treatment is not necessary.
Nonsurgical treatment
Conservative measures are generally recommended for those who have not completed childbearing or are unwilling or unfit for surgery.
Pelvic-floor exercises are useful in the treatment of urinary stress incontinence and mixed incontinence; however, evidence of their efficacy in the prevention or treatment of POP, especially if severe, is limited [14,15].
There are many pessaries available made from a wide variety of materials including rubber, clear plastic, soft plastic with metal reinforcement and silicone. These provide support to the pelvic organs and relieve pressure on the bladder and bowel. Ring pessaries are widely used, but they rely on adequate perineal support to retain them, which may not be present. The Gelhorn, cube and donut pessaries are thought to be most effective for both uterine or vault defects [16]. Disadvantages of pessary use are that they require regular replacement, usually at 6-monthly intervals, to avoid vaginal erosion and ulceration, and they may interfere with coital function.
Surgical treatment
The goal of surgical treatment is to restore normal anatomy and the vaginal axis, preserve vaginal length and function, and reduce recurrence. There are numerous surgical procedures that have been described using either abdominal or vaginal approaches. No one technique is completely satisfactory, reflecting how poorly the pathophysiology, and therefore its correction, is understood. To address the high recurrence rates, the use of a wide variety of synthetic and biological graft materials have been proposed for reconstructive surgery.
The choice of procedure is often dependent on the individual surgeon's choice and experience. The procedure should be tailored to the individual patient, but should also take into account how best to anatomically correct the defect and address the underlying pathophysiology.
Obliterative procedures: colpectomy & Le fort colpocleisis
Most procedures to correct prolapse aim to restore normal anatomy; however, in the frail and those who do not wish to retain sexual function, obliterative procedures may be considered. The primary advantage of these procedures is that they are relatively quick to perform and can be carried out under local or regional analgesia, so reducing morbidity further. Partial colpocleisis involves excision of a triangle of tissue from the dorsal and ventral surfaces of the prolapse, and the vagina is then inverted with scarring of the raw surfaces. In the colpectomy, all the vaginal skin is removed. The fact that obliterative procedures preclude further assessment of the cervix and uterus if this is left in situ needs to be borne in mind. In addition, if the anterior vaginal wall is involved in the obliteration, there is a risk of developing urinary incontinence.
Anterior compartment
There are a number of procedures advocated for the repair of the anterior compartment including anterior coloporrhaphy, paravaginal repair and colposuspension.
Anterior colporrhaphy
This procedure entails plication of the vesicopelvic fascia and bladder neck. It aims to correct the distension defect of the cystocele and stabilize the periurethral fascia. The technique can be modified by the extent of lateral dissection and whether an interposing mesh is placed in the anterior vagina for additional support.
Randomized, controlled trials report anterior colporrhaphy to be successful in only 47–57% of cases [17,18]. Mesh placement has been used to improve this success rate, although good-quality evidence for its use is limited. These meshes can be natural materials, such as resected parts of the vaginal wall, rectus or cadaveric fascia. Permanent or absorbable meshes can also be used, although there is a high rate of erosion and infection with the permanent meshes, often necessitating their removal. Julian reported a 66% cure rate for recurrent anterior prolapse using a standard anterior colporrhaphy, but 100% when a Marlex© mesh was used [19]. However, mesh-related complications occurred in 25% of patients.
Anterior colporrhaphy should not be used for the treatment of stress incontinence, even though stress incontinence often co-exists with anterior vaginal-wall prolapse. In these cases, the insertion of a midurethral tape may be considered. Burch colposuspension can also be used; in these cases, the paravaginal fascia on either side of the bladder neck and the base of the bladder are approximated to the pelvic side wall by sutures placed through the ipsilateral ileopectineal ligament.
Paravaginal repair
This procedure aims to correct lateral defects by re-attaching the detached lateral vagina to the level of the arcus tendineus fascia pelvis. The failure rate has been reported at 3–14% [20]. Paravaginal repair can be performed vaginally or abdominally. Speight and colleagues reported a laparoscopic paravaginal repair technique performed on 18 patients, with a short operating and fast recovery time [21]. Importantly, there were no ureteric injuries, bladder injuries or excessive blood loss.
Apical compartment
Adequate support of the vaginal apex is the key to surgical success. Without this, the anterior and posterior vaginal walls are exposed to increased intra-abdominal pressure and their correction is more likely to fail. Surgical correction of apical defects can be divided by the presence or absence of the uterus, either posthysterectomy vaginal-vault prolapse or uterine prolapse. For posthysterectomy vault prolapse, procedures can be supportive or obliterative. Supportive procedures aim to replace the damaged or absent upper supports while fixing the upper third of the vagina over the levator plate.
Vault support at the time of hysterectomy has been advocated using either culdoplasty techniques or sacrospinous fixation.
Enterocele repair
Vaginal enterocele repair is often performed in conjunction with vaginal hysterectomy, apical support procedures and colporrhaphy. The vaginal approach is advantageous as it is associated with a quicker recovery time and decreased morbidity. The enterocele is identified and dissected from the vaginal epithelium, and the neck of the sac ligated. Additional support can be given by plicating the uterosacral–cardinal ligament complex. Potential complications include rectal or bowel injury and ureteric kinking. The vaginal skin is then closed and plicated over. Long-term follow-up results have generally been poor [22].
Abdominal repair of enterocele
Although this approach is associated with increased morbidity, it can be considered if there is a concomitant abdominal procedure. These include the Moschowitz and Halban procedures, which aim to obliterate the pouch of Douglas [23,24]. Care needs to be taken with both of these techniques not to kink the ureter or involve the bowel.
Site-specific repairs have been advocated for enterocele repairs, which aim to identify fascial defects and re-approximate the detached edges. There are limited data on this technique. Miklos and colleagues reported a combined laparoscopic and vaginal site-specific approach to enterocele repair with resuspension of the vaginal vault [25]. In 17 patients who underwent this procedure, there were only two cases of vault descent, but no recurrent enterocele. There were no significant complications.
Uterine prolapse
A vaginal hysterectomy is the standard procedure to correct uterine prolapse and can be performed together with an anterior and posterior colporrhaphy. However, the role of hysterectomy in this situation has been challenged on the basis that hysterectomy does not address the underlying deficiencies that cause prolapse, does not increase the durability of the repair and may even increase morbidity [26]. In addition, it has been suggested that hysterectomy and its associated pelvic-floor dissection may increase pelvic-floor neuropathy and disrupt natural structures. It may also be associated with an increased risk of urinary incontinence, bladder dysfunction, vault prolapse and, possibly, sexual dysfunction [27,28]. The concept of uterine preservation in prolapse surgery was first suggested by Bonney, who described a passive role of the uterus in the development of prolapse [29]. Since then, various authors have described reconstructive procedures with uterine preservation. Options include the Manchester procedure, which shortens the uterosacral and cardinal ligaments, with amputation of the cervix. This procedure is rarely performed currently due to complications such as dyspareunia, dysmenorrhea, recurrent uterine prolapse and enterocele. There are also detrimental effects on fertility [30,31]. Other procedures involve transfixation of pelvic structures to the anterior abdominal wall, sacrohysteropexy and sacrospinous fixation. The sacrohysteropexy attaches the uterus to the anterior longitudinal ligament using a graft [32]. There are few data on the long-term results of these techniques, including the incidence and impact of mesh erosion.
Sacrospinous hysteropexy also has limited efficacy data, but seems to be less detrimental to fertility. Kovac and Cruishank reported on a series of 19 patients aged 17–37 years, of whom five attained normal pregnancies [33]. Maher compared this procedure in 34 women with 36 women who had vaginal hysterectomy and sacrospinous fixation [34]. Subjective and objective success rates and patient satisfaction were high and similar between groups.
There are limited controlled data on laparoscopic plication of the uterosacral ligaments to elevate the uterus, which involves plication of the uterosacral ligaments using nonabsorbable Ethibond© at two points: the origin of the ligaments and 2 cm posterior to the origin of the ligaments. This is a short procedure and one case report in a 36-year-old woman reported that at 2-year follow-up, the woman remained asymptomatic with the uterus well supported. Both this procedure and sacral hysteropexy can be performed laparoscopically or abdominally [35].
Vault prolapse
There are many procedures used to repair vault prolapse. The main techniques are sacrospinous fixation and transabdominal sacrocolpopexy.
Sacrospinous fixation
Sacrospinous fixation involves fixation of the vault to the sacrospinous ligament. This is not anatomical, as it results in a lateral and posterior deflection of the vagina and exposes the anterior vaginal wall to increased stress during rises in intra-abdominal pressure. Other complications include vaginal shortening, dyspareunia, pain and bleeding [36]. Injury to the pelvic organs has been reported in 0.8% and gluteal pain in 3% of patients undergoing this procedure [37]. Gluteal pain is most likely due to injury to the sacral nerve that runs through the coccygeal–sacrospinous complex, or posterior cutaneous or sciatic nerve trauma, and may necessitate reoperation to remove the sutures. Although there are a number of studies assessing this technique, there are very few objective data, and follow-up is often not specified or is too short to be valid. Sze and Karram performed an analysis of the literature and found many of these studies were of poor quality, with cure rates ranging from 8–97% [37]. Several authors have suggested that the marked vaginal retroversion after sacrospinous fixation predisposes patients to recurrent support defects, particularly in the anterior vagina. The development of cystoceles has been reported in 6–21% of patients followed-up over various lengths of time [38,39].
Ileococcygeus hitch
The ileococcygeus hitch involves fixation of the vault bilaterally to the ileococcygeus muscle, anterior to the ischial spine. This procedure has a lower incidence of complications than sacrospinous fixation, as it is less likely to traumatize the pudendal vessels and nerves. However, although more anatomical, it results in less elevation, as the ischial spines are inferior to the normal position of the apex [40].
Posterior intravaginal slingplasty
More recently, minimally invasive techniques have been used to support the apex and create an artificial neoligament. The posterior intravaginal slingplasty (IVS; infracoccygeal sacropexy) has been described, which uses an IVS Tunneller™ to insert an 8-mm polypropylene tape between the perineum and vaginal vault. The rectovaginal fascia and perineal body are then repaired to reinforce support. Preliminary reports suggest that this technique has a high symptomatic cure rate for prolapse, with no significant bleeding, and has the advantage that it can be performed as a day-case procedure [41,42]. The main complication described is tape erosion, noted in 5.3% of patients undergoing this treatment.
McCall culdoplasty
The McCall culdoplasty obliterates the pouch of Douglas using a series of continuous sutures suspended to the uterosacral ligaments, which are then plicated into the midline [43]. The procedure can be performed after a vaginal hysterectomy or in the posthysterectomy patient with an enterocele or vaginal-vault prolapse. This provides better support of the apex compared with simple peritoneal closure or vaginal Moscowitz procedures [44].
Uterosacral plication
A uterosacral plication can be performed to correct vault prolapse, and this technique can be performed laparoscopically, although this route requires further evaluation. There are limited data on the efficacy of uterosacral plication and suspension. Given reported a 5% failure rate with an average follow-up of 7 years [45]. The success of this procedure is, in part, dependent on the integrity of the ligaments.
The high uterosacral ligament fixation has replaced this procedure in many US centers. This involves suspending the vaginal apex to the remnants of the uterosacral ligaments at the level of the ischial spines and incorporating the rectovaginal fascia and pubocervical fascia into the sutures. This maintains the vaginal axis in the midline and optimizes length; however, ureteric injury, as well as bowel dysfunction, is reported in 1–11% of patients [46].
Sacrocolpopexy
Abdominal procedures include either an open or laparoscopic sacrocolpopexy. This technique aims to attach the vaginal vault to the longitudinal ligament of the sacrum using a mesh. It has the advantage that it does not compromise coital function and may even elongate and restore function in those who have had compromised function from previous prolapse procedures. Several types of synthetic materials have been used including Mersilene©, Goretex©, Marlex and Prolene©, of which Prolene is most commonly used [47–50]. The open procedure has a high cure rate of over 90% [51,52]. Lefranc reported on a series of 85 cases and found that at median follow-up of 10.5 years, only 2.3% had recurrent vault prolapse [53]. However, the procedure is not without complications, which include major intra-operative bleeding and a 3.3% incidence of mesh erosion [54,55]. The other rare complication is osteomyelits of the sacrum, which may present as severe back pain.
In an attempt to minimize posterior-compartment weakness, it is often advocated that the mesh is placed as far back as the perineal body, although this can be technically difficult. In these cases, an alternative combined vaginal approach is proposed, allowing distal anchorage of the mesh. However, this has been associated with a high rate of sepsis and mesh erosion [56,57].
The laparoscopic route was first described by Nezhat and colleagues, and the results seem comparable to those observed with the abdominal approach [58]. There are no large, randomized trials with long-term follow-up to assess the objective and subjective cure rates of the laparoscopic route, although it has the obvious potential advantages of decreased hospital stay and recovery time, as well as an enhanced operative view and, thus, safer dissection. It is, of course, very dependent on the operator's skill.
Posterior compartment
Three different surgical approaches have been used for the repair of posterior compartment prolapse. Traditionally, gynecological surgeons have used a transvaginal repair involving levator plication, whereas colorectal surgeons use the transanal approach. In their retrospective study of 244 posterior colporraphies, Kahn and Stanton noted an increased postoperative incidence of constipation, incomplete bowel emptying, fecal incontinence and sexual dysfunction [59]. It is thought that levator plication is associated with an increase in dyspareunia, secondary to atrophy and scarring of muscle fibers. Despite this, 62% of patients reported an overall improvement and the vaginal defect was corrected in 76% of cases.
The transrectal approach involves an incision into the anterior rectal mucosa just above the dentate line. The submucosal plane is sharply dissected from the anal verge laterally and anteriorly. The dissected area is then plicated, involving the rectal muscle and rectovaginal septum. Sarles and colleagues reported on the outcome in 16 patients with difficulty in evacuation associated with rectocele [60]. A total of 15 patients reported an excellent outcome or significant improvement, and only one reported a poor functional result.
Two randomized trials have compared the vaginal and transanal approaches, and both have demonstrated better objective and symptomatic improvement in the vaginal than the transanal group [61,62]. In a study of 57 women with symptomatic rectoceles, Khan noted that 30% required further surgery in the transanal group versus 13% in the transvaginal group [61]. In a similar study, at 1-year follow-up, Nieminen and colleagues found that in the 30 women randomized to either group, 67% in the transanal arm and 7% in the transvaginal arm had persistent prolapse [62].
The principles of repair have changed over the last few decades and site-specific repairs have been advocated on the basis that rectoceles are due to fascial breaks. These breaks have been described as transverse or lateral separations of the rectovaginal fascia from the arcus tendineae, fascia pelvis, pelvic side wall and perineal body [63–65]. The most common break is the transverse separation of the fascia from the perineal body, resulting in a low rectocele. Repair of isolated defects in the fascia have been reported to cure rectoceles in 82% of patients, as well as resulting in an improvement in constipation, tenesmus and splinting of the vagina and perineum during defecation [59]. Abramov and colleagues retrospectively compared midline fascial plication with discrete site-specific repairs and noted a higher incidence of rectoceles following site-specific repairs (13 vs 32%) [66].
The use of mesh in the posterior compartment has been more controversial. The risks of mesh, particularly synthetic, include erosion, but it can also have detrimental effects on sexual function.
There are a few small studies reporting on the use of Marlex mesh for posterior-wall repair, which showed good anatomical results and improvement in defecatory function [67,68]. Kohli and Miklos reported site-specific repair of rectoceles augmented with dermal allograft in 43 women. A total of 33 women attended for follow-up, with a 93% anatomical cure rate as evaluated with the POP-Q [69].
In a woman who wishes to retain sexual function, the use of Prolene mesh in the posterior wall is inadvisable. Anatomical results are good but there is an unacceptable increase in the rate of dyspareunia, of up to 64% [70].
Conclusions
The procedure of choice for reconstructive surgery to the vagina should be tailored to the individual patient and should have a low risk of morbidity and mortality, while also having long-term durability. There is no single procedure that is able to satisfy all of these requirements, and more data are required on the evaluation of procedures in the form of randomized, well-designed trials with long-term follow-up using subjective and objective criteria.
Future perspective
In order for new therapeutic strategies to be developed, more research is required into the epidemiology, etiology and pathophysiology of POP, so that logical approaches may be developed to address structure and function.
Evaluation of surgical procedures for vaginal prolapse is hampered by the lack of large, randomized studies using both validated subjective and objective outcome measures. This is particularly relevant at the present time, when there are an increasing number of new techniques on the market, including graft materials, whose use is not supported by good-quality data. In order to carry out this future research, long-term studies should be promoted, using standardized definitions of cure and outcome assessment.
Executive summary
Assessment of prolapse should involve objective validated anatomical and symptom assessment, including quality-of-life issues.
The procedure should be tailored to the individual with attention to the anatomical and functional outcome, associated urinary, bowel and sexual dysfunction, and operative morbidity.
Sacrospinous fixation has a higher anterior-wall recurrence rate and sacrocolpopexy seems to result in a better long-term outcome.
High uterosacral ligment suspension is associated with increased rates of ureteric injury, and cystoscopy post procedure is advisable.
For posterior-wall prolapse, levator plication is associated with increased sexual and defectory dysfunction. The transanal approach may have a lower rate of sexual dysfunction than the transvaginal approach, but seems to result in a higher recurrence rate.
There is no strong evidence to support the routine use of biological or synthetic grafts for transvaginal prolapse repair and, if used, patients should be counseled regarding the risks of erosion, infection and dyspareunia.
