Abstract
The levonorgestrel intrauterine system (Mirena®) has been developed as a long-acting method of contraception. This medicated device, inserted into the uterus, delivers a small daily dose of a progestin, levonorgestrel, into the uterine cavity. It is one of the most effective methods of contraception and is approved in more than 100 countries for 5 years of use.
The levonorgestrel intrauterine system (LNG IUS) (Mirena®) has been developed as a long-acting method of contraception. This method, invented by T Luukkainen, was developed by the Population Council in the late 1980s and then with Schering Oy (formerly Leiras Oy) and Schering AG, with the objective of improving the contraceptive efficacy and decreasing the bleeding days of nonmedicated intrauterine devices (IUDs) [1]. This medicated device, inserted into the uterus, is one of the most effective methods of contraception and is approved in more than 100 countries for 5 years of use (Figure 1). In addition to its main indication for contraception, the advantages provided by the direct action of levonorgestrel on the endometrium have been studied in other medical conditions. By inactivating the endometrium, the LNG IUS is able to provide a range of other positive health benefits, such as treatment of menorrhagia and protection of the endometrium in women on estrogen therapy. These latter indications have been approved in 100 and 89 countries, respectively (Figure 1).

Approval of the LNG IUS for various indications in the world (as of February 2005).
Description of the system: what is the levonorgestrel intrauterine system?
The LNG IUS is a levonorgestrel (LNG)-releasing intrauterine delivery system (IUS) which, during a long period, releases small daily amounts of LNG into the uterine cavity (Figures 2 & 3). The system consists of a T-shaped plastic frame to the vertical arm of which a cylinder containing the active substance mixed with polydimethyl siloxane (PDMS) is attached (Figures 2 & 3). The total amount of LNG, a synthetic progestin, in the cylinder is 52 mg. The cylinder is covered by a rate-controlling PDMS membrane, which regulates the daily release rate of the hormone. The initial release rate is 20 μg/day, which declines to 11 μg/day at the end of 5 years [101]. The mean release rate is 14 μg/day over the 5-year wearing period. The distal end of the T-frame contains two removable threads, as well as barium sulfate, which makes it visible upon x-ray examination (Figure 4).

The levonorgestrel intrauterine system.
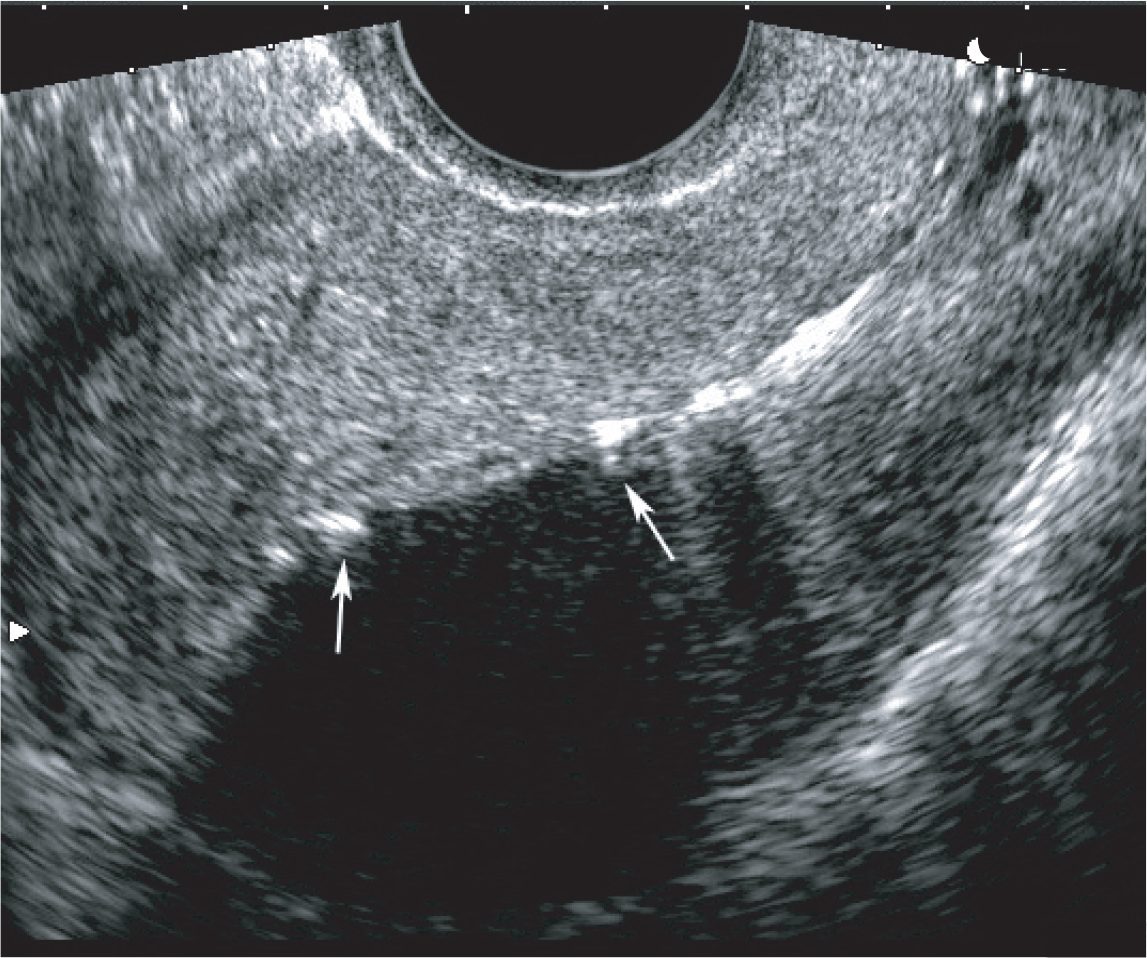
Levonorgestrel intrauterine system in the ultrasound examination.

Appearance of the levonorgestrel intrauterine system on x-ray examination.
The LNG IUS directly targets the endometrium by releasing LNG, a progestin derived from 19-nortestosterone with high progestational potency, that can be active at low daily doses, into the uterine cavity. The result is high local LNG concentrations that achieve uniform suppression of endometrial proliferation, inactive histology, thin epithelium and decidualization of the stroma. Such profound alteration of the endometrium creates an environment within the uterus that is unsuitable for sperm survival and fertilization and is thus a key mechanism of action of contraception [1,2]. In addition, the LNG IUS prevents fertilization via the local effects of the progestin on the cervix [1,2]. Several, but not all, studies showed that the cervical mucus is thickened, thus preventing sperm penetration and fertilization [3]. This mechanism of action is valued in countries where religious beliefs lead to a reluctance to use classic IUDs, thought to interfere with implantation.
Therefore, the effects of the LNG IUS are brought about by the intrauterine delivery of LNG, but detectable levels of LNG are also observed in the serum, although these are lower than with other combined or progestin-only contraceptives, and also lower than with Norplant®, a long-acting subdermal implant that delivers a low daily dose of the same progestin [3,4]. The systemic exposure is responsible for the hormonal adverse effects observed during the use of the LNG IUS, which, along with the release rate, are more pronounced during the initial months of use. Due to the mode of administration, the LNG IUS has some of the features of IUDs, such as a long wearing time, no need for daily attention and rapid reversibility, while some features are shared with other progestin-only contraceptives. Some characteristics are, however, unique to this type of delivery system.
By suppressing endometrial proliferation, the LNG IUS decreases menstrual blood loss and pain. In perimenopausal women, menorrhagia associated with uterine myomas and adenomyosis is frequent, and the system has also proven effective in the management of this condition. Indications for using the LNG IUS include contraception, treatment of idiopathic menorrhagia and endometrial protection during estrogen therapy, which have been approved in a large number of countries as described above (Figure 1). This article covers these indications, as well as providing an insight into the emerging uses of the system.
Mechanism of action for contraception
The contraceptive effects of the LNG IUS are brought about by multiple mechanisms acting together, and there is no single mechanism responsible.
Levonorgestrel is progestational, with an estimated potency ten times greater than that of progesterone. It also exhibits some androgenic properties [5]. Nilsson and colleagues studied the tissue concentration of LNG in women who volunteered to receive an IUS before a scheduled hysterectomy [6]. A total of nine women had an experimental model of the LNG IUS (with a release rate of 30 μg/day) inserted between 36 and 49 days prior to surgery. Direct intrauterine administration of LNG resulted in high endometrial concentrations, ranging from 470–1500 ng/g of tissue weight, while much lower concentrations were observed in adjacent myometrial and fallopian tube tissues (1.8–2.4 ng of LNG/g of tissue weight) [6].
Intrauterine LNG is quickly resorbed by the capillary network of the basal endometrial mucosa and measurable levels of LNG have been detected in plasma 15 min after insertion [1]. Peak plasma LNG concentrations are detected within a few hours after insertion and thereafter they level off at 150–200 pg/ml (0.4–0.6 nmol/L), with some decline over 5 years of use [1,7,8]. With the subdermal implant, Norplant, plasma LNG levels are initially described at 400 pg/ml during the initial months and decline to 250–270 pg/ml at the end of the fifth year of use. With the LNG IUS, levels of LNG would correspond grossly to half those described with Norplant [4,7].
The high concentration of LNG in endometrial tissue induces dramatic changes in endometrial morphology, histology and biochemistry and these changes are independent of the menstrual cycle [9].
The endometrial changes associated with the use of the LNG IUS combine strong progestational effects, such as atrophy of the endometrial glands and decidualization of the stroma, with those of an intrauterine device, such as stromal inflammatory cell infiltrate and surface papillary formations [10]. The estrogen and progesterone receptors are downregulated, while the expression of some cytokines and growth factors show a variable response. The cyclical activity of the endometrium (eg., the proliferative and secretory phase) is rapidly lost after LNG IUS insertion and the endometrium becomes quiescent and nonresponsive to estrogen [9]. These profound changes in the endometrium are associated with frequently observed irregular bleeding or spotting during the initial months of using the LNG IUS. Numerous recent studies have tried to elucidate the mechanism of these bleeding disturbances, yet no single factor has been found to be responsible. It is likely that changes in growth factors, cytokines and enzymes involved in the modeling of the extracellular matrix, such as the matrix metalloproteinases (MMPs), as well as morphologic changes in the endometrial vasculature, together bring about these changes [9]. However, unlike the bleeding disturbances observed during the use of subdermal progestin implants, the irregular bleeding/spotting that occurs during the use of the LNG IUS is usually limited to the initial months. Once the endometrial effects are established, the bleeding pattern turns to gradual oligo- or amenorrhea. Using strict criteria for amenorrhea (90 consecutive days with no bleeding or spotting), approximately 20% of the LNG IUS users are defined to be in amenorrhea [11], and the mean number of 2 bleeding days per month at 1 year is significantly lower in LNG IUS users than in copper (Cu) IUD users [12]. However, the perceived amenorrhea rate is higher (~50%) [13,14], as light spotting is often unnoticed by women.
The LNG IUS also has a minor effect on ovarian function. After the first year, most (85%) cycles are ovulatory [1,2]. The initial months of use may be associated with more anovulatory cycles, yet the endogenous secretion of estradiol remains within the levels of a normal follicular phase [1,2]. The effects of the LNG IUS on ovarian function may be viewed as a dynamic process reflecting the systemic exposure to LNG. Therefore, as the serum LNG levels are higher during the initial months, after which time they gradually decline, the effect on ovarian function is also more pronounced at the start of therapy.
Contraceptive properties of the levonorgestrel intrauterine system
Cumulative experience from clinical trials and postmarketing studies with over 70,000 woman-years of observation has established the contraceptive efficacy of the LNG IUS, which is among the highest of reversible contraceptives. The gross cumulative pregnancy rate at 5 years is 0.5–1.1% [12,15,16]. Indeed, the contraceptive efficacy of the LNG IUS has been compared with female sterilization. The overall pregnancy rate is 0.16/100 woman-years; therefore, if 1000 women use the LNG IUS for a year, only two may become pregnant. The efficacy in contraception is similar throughout the 5-year lifespan of the system [16]. In Sivin and colleagues [15] and Andersson and colleagues [12], two large randomized, controlled trials compared the LNG IUS with Cu IUDs and found the pregnancy rate per 100 users was between 0.1–0.2 for the first year of use and 0.5–1.1 for 5 years of use.
The rate of ectopic pregnancy is also low, at approximately 0.6/1000 woman-years. An ectopic pregnancy rate of 2.6/1000 woman-years has been reported in women using no contraceptive method [17]. A recent study with Cu IUDs and inert (plastic) IUDs found an overall ectopic pregnancy rate of 0.8–1.0/1000 woman-years during 10 years of use [18]. Nevertheless, if pregnancy does occur during the use of the LNG IUS, the likelihood of the pregnancy being ectopic is high and therefore the location of the pregnancy should be established promptly [16]. In addition, counseling regarding the risk of pregnancy is important before insertion of an IUS.
Use of the levonorgestrel intrauterine system in nulliparous women
The use of the LNG IUS is generally not recommended as the first method of choice for young nulliparous women. With careful patient selection in women not at risk of sexually transmitted infections, the LNG IUS may be a good alternative. A recent randomized, comparative study examined the use of the LNG IUS in young (aged 18–25 years) nulliparous women, compared with the traditional choice of combined oral contraceptive pills [19]. Both methods were equally tolerated by the women, and there were no pregnancies in either group during a follow-up of 12 months. With the exception of the first 3 months, bleeding days were significantly fewer in the LNG IUS group. Dysmenorrhea was also alleviated to a greater degree in the LNG IUS group. A Dutch study involving mainly Cu IUD users (and a small cohort of LNG IUS users) found no differences between parous and nulliparous women with regard to IUD complications, such as pelvic inflammatory disease (PID) or expulsion [20]. Insertion of the LNG IUS appears to be feasible for nulliparous women, but this group report more pain during insertion [19]; therefore, special attention to proper pain relief should be paid.
Noncontraceptive benefits of the levonorgestrel intrauterine system: other therapeutic indications
Treatment of menorrhagia
Menorrhagia is a frequent complaint of women in their fertile years and in the perimenopause. It is defined as excessively heavy or prolonged uterine bleeding during menstruation, which may be caused by medical problems or hormone imbalance.
It can be secondary to uterine pathology (e.g., uterine fibroids, adenomyosis or endometrial pathology) or, rarely, to disorders of blood coagulation (e.g., von Willebrand disease), but frequently no underlying cause can be established. In such cases, the term idiopathic menorrhagia is used. Unlike dysfunctional uterine bleeding, menorrhagia is often associated with normal ovulatory function and a normal length of the menstrual cycle. The prevalence of menorrhagia is estimated to be between 5–15% of women in their fertile years and is highest in the late reproductive years. Annually, a high number of women undergo hysterectomy or other surgical procedures, such as endometrial ablation (EA) or resection (ER), for the treatment of this condition. The economic burden to society caused by menorrhagia is substantial, and it has been estimated that the health-related quality of life (HRQoL) of women with menorrhagia is reduced and comparable to that of chronically ill women [21].
Menorrhagia can be defined objectively as menstrual blood loss (MBL) exceeding 80 ml/cycle. The methods used for measuring the actual MBL are, however, cumbersome and not applicable for routine use. MBL can also be estimated using pictorial blood-loss assessment charts (PBAC) based on a bleeding diary. The correlation of PBAC with measured MBL is, however, less than perfect [22], and the woman's perception of the amount of MBL does not always correlate with measured levels [23]. In a clinical setting, the decisive role is usually the perception of the woman (whether she has excessive menstrual flow or not). Therefore, studies taking this approach yield the most clinically relevant results, although scientifically, studies employing more precise methods may be preferred.
The LNG IUS reduces MBL in both women with menorrhagia and those with normal MBL [1]. In women with menorrhagia (objectively measured median MBL of 176 ml/month), the measured MBL is reduced by 86 and 97% 3 and 12 months after the insertion of the LNG IUS, respectively [1,24]. The reduction achieved by the LNG IUS is superior to that observed with other commonly used medical therapies, such as tranexamic acid [25], nonsteroidal anti-inflammatory drugs [22] or oral progestins [26] and comparable with that observed after organ-preserving surgical techniques, such as ER [27] or EA [28].
Due to the effect of LNG IUS in decreasing excessive bleeding, this method may be of benefit in the prevention of anemia in some women [3,29]. This is particularly relevant in developing countries where there is a high prevalence of anemia [29]. Faundes and colleagues showed that LNG IUS users had significantly higher mean hematocrit and ferritin levels than IUD users or a control group of non-IUD users [29]. Andersson and colleagues found that LNG IUS users had an increase in hemoglobin of 1.6 g/l, while IUD users had a mean decrease of 2.6 g/l [12].
Importantly, the LNG IUS can also be used for patients who have menorrhagia due to inherited disorders of blood coagulation (such as von Willebrand disease) [30]. Concomitantly, with the reduction of MBL, dysmenorrhea and premenstrual syndrome (PMS), symptoms may also be reduced [7,12,31]. Sivin and Stern reported that IUS users were less likely to mention dysmenorrhea than Cu-T IUD users [31].
Barrington and Bowen-Simpkins studied a group of 50 women (mean age 40 years, age range 28–53 years) with a failed trial of medical therapy for menorrhagia, who were then treated with the LNG IUS, and showed an improvement or cure of PMS symptoms in 28 out of 50 women (56%) at 3 months postinsertion [32]. In addition, improvement of dysmenorrhea was noted in 80% of patients [32].
Dysmenorrhea secondary to endometriosis has also been studied in small series [33,34]. Compared with expectant management after first-line operative laparoscopy treatment of endometriotic lesions, a significant reduction in dysmenorrhea (by visual analog scale) and the recurrence of dysmenorrhea was observed in patients who received the LNG IUS during the laparoscopy [34].
Obviously the improvement of PMS and dysmenorrhea is a positive side effect of the LNG IUS, but these symptoms are not an indication for an IUS in premenopausal women who do not need contraception or treatment for menorrhagia.
The LNG IUS has also been proposed in a small study of patients with small- or medium-sized uterine fibroids, excluding submucous fibroids and other fibroids distorting the uterine cavity [35]. A slight reduction in the size of fibroids during the use of the LNG IUS was reported by Grigorieva and colleagues [35], an effect presumed to be mediated by the induction of the insulin-like growth factor-binding protein-1. In another series of cases, Maia and colleagues showed that insertion of the LNG IUS after ER induced a high rate of amenorrhea in women with adenomyosis [36]. Indeed, after 3 months postablation of the endometrium, 38 out of 53 (72%) women who received the LNG IUS were amenorrheic, while only 8 out of 42 (19%) were amenorrheic in the control group who had only the ER (p < 0.001). At 1 year, 100% in the IUS group were still amenorrheic versus 9% in the control group.
More data are needed to confirm these findings in larger series. However, for patients with an enlarged uterine cavity (uterine sound length exceeding 10 cm) or for patients with fibroids distorting the uterine cavity, the LNG IUS appears to have limited use. In addition to reduced efficacy, there may also be an increased risk of spontaneous expulsion of the system.
Relevant guidance to aid in the decision-making process of selecting treatment for menorrhagia has been obtained from a recent large randomized study carried out in Finland, comparing the LNG IUS and hysterectomy. This study evaluated the HRQoL (using a validated Finnish version of the RAND 36-item short-form health survey) and cost-effectiveness of both methods over 5 years [21,37]. The study is of interest for its ability to compare two fundamentally different treatments: a conservative approach (the LNG IUS) and a radical approach (hysterectomy), for the same condition, and for the exceptionally low lost-to-follow-up rate of only 2% [37].
The change from baseline in various parameters evaluating the HRQoL of patients showed equal improvement in both groups, with the exception of physical functioning, which showed slight deterioration consistent with aging in both groups. Therefore, although complete amenorrhea was not achieved in every patient with the LNG IUS, the main outcome parameter of HRQoL was equal to that obtained with a radical approach, while avoiding major surgery in approximately 60% of patients [37]. The cost–utility analyses showed that the LNG IUS was about three times cheaper than surgery at 1 year of follow-up and still approximately 40% cheaper at 5 years of follow-up, taking into account both direct costs of healthcare as well as indirect costs such as productivity losses [37]. Although a considerable proportion of patients eventually underwent hysterectomy, the results should be viewed against the preconception that most of the patients who entered the study were referred to hysterectomy, and therefore might have been less likely to accept the possible adverse effects associated with the LNG IUS.
A review of trends in the number of hysterectomies performed in the UK for menorrhagia shows a decrease which coincides with the availability of the LNG IUS [38]; however, no decrease is seen in countries where ER and EA were used in the management of heavy menstrual bleeding [39].
Although this is only a hypothesis which requires to be confirmed, the LNG IUS, by its ability to treat menorrhagia, has the potential to reduce the number of hysterectomies carried out.
Despite the widespread traditional claim of no effect of hysterectomy on ovarian function, the LNG IUS versus hysterectomy study demonstrated a slight but significant increase in follicle stimulating hormone levels and in the occurrence of vasomotor symptoms in the group randomized to hysterectomy at 1 year after surgery [40].
Endometrial protection during postmenopausal estrogen therapy
Because of its strong antiproliferative action on the endometrium, the LNG IUS has been studied for the prevention of endometrial hyperplasia during estrogen treatment for climacteric symptoms. As progestin is recommended for this purpose in women with an intact uterus only, intrauterine progestin delivery should be a logical approach whereby the adverse effects caused by the systemic administration of progestins are minimized [41]. This approach also allows the free choice of estrogen with regard to type, dose and mode of administration (oral or transdermal). The downside of this regimen is the need for a small procedure to insert the LNG IUS. The insertion may be more difficult in women with an atrophic uterus secondary to hypoestrogenism and as such may not be acceptable for all women. On the other hand, this attitude may be changing due to the risks attributed to oral hormone therapy (HT) as reported in recently published large epidemiologic and randomized, controlled clinical studies [42,43]. The use of the LNG IUS is especially suitable for the perimenopausal phase (a phase beginning at the start of menstrual irregularity and ending one year after the final menstrual period), when contraception is still needed but the ovaries show variable activity with phases of both hypo- and hyperestrogenism [44].
The rationale for progestin use in combined HT of the menopause is to prevent estrogen-induced endometrial proliferation and cancer. Local release of a progestin in the uterine cavity may ensure the protection of the endometrium while avoiding the higher doses that are required with oral HT. The tissue concentrations of LNG in the uterus with local dosing far exceed the uterine concentrations when LNG is given systemically, which explains the marked endometrial suppression and amenorrhea. With LNG IUS use in women on estrogen therapy, the endometrium remains uniformly suppressed and nonproliferative even with high systemic levels of exogenous estrogen.
Several studies have shown that the use of LNG IUS in women receiving various types of estrogen therapy for menopausal symptoms is highly effective in preventing the occurrence of endometrial hyperplasia [3,45,46]. As with any continuous combined HT, the decreased incidence in endometrial hyperplasia as compared with estrogen given alone is also expected to decrease the risk of endometrial cancer [45]. The use of oral continuous combined HT regimens is limited to the postmenopausal phase only and, therefore, the IUS combined with oral or transdermal estrogen is one of the few methods available to avoid the withdrawal bleeding associated with sequential HT regimens. Furthermore, a non-bleeding pattern is established in a high proportion of women already at perimenopause [47]. However, spotting may occur as with other HT regimens, which should be mentioned during counseling. The LNG IUS is especially suitable for use together with transdermal estrogen delivery from patches or gels [46,48]. The transdermal approach for estrogen delivery may also be associated with a lower risk of thromboembolic complications such as deep venous thrombosis and pulmonary embolism [49]. However, transdermal estradiol combined with LNG IUS has not been studied in that respect.
Recent reviews and evidence from experimental studies have suggested that HT initiated at the time of menopause is associated with a lower risk of cardiovascular disease [50]. In contrast, if therapy is initiated over 10 years after the menopause, HT can be harmful, as the pre-existing plaques on the vessel walls may be more likely to rupture [50,51]. There is a growing body of evidence that each mode of delivery, type of estrogen and progestin, and timing of therapy have distinct beneficial and adverse effects, and the results pertaining to one regimen should not be extrapolated to other types of therapy. This likely applies to the unwanted effects of HT, particularly on breast cancer and cardiovascular events. The final effect of a HT regimen depends on the combined effects of each component of that particular regimen on breast tissue as well as on the cardiovascular system [50,52]. As far as breast tissue is concerned, the lowest impact can be achieved either with unopposed estrogen, with low doses of both estrogen and progestin and with progestins that induce programmed cell death (apoptosis) rather than proliferation in mammary epithelial cells [52,53].
As the effective lifespan of the LNG IUS is 5 years and the size of the uterus tends to decrease (involute) after the menopause, a system with smaller dimensions and with 3 years of usage time, together with a lower release rate of 10 μg/day (the menopausal levonorgestrel system [MLS]) has been studied [54,55]. With the MLS, the systemic LNG concentration is approximately 40% of that of the LNG IUS with 20 μg/day release rate [54]. The authors reported high acceptability and tolerability, although spotting was frequently observed during the first year of use [54,55]. The changes in the lipid profile during use of the MLS and estrogen therapy are very close to those achieved with estrogen-only therapy [54,55].
Other emerging therapeutic indications
The strong direct effects of LNG IUS on the endometrium, coupled with minimal systemic effects, have prompted many investigators to study the usability of the LNG IUS for indications other than those approved by the health authorities. These studies have involved the use of the LNG IUS for the prevention of endometrial hyperplasia during adjuvant anti-estrogen treatment with tamoxifen for breast cancer and for the treatment of various gynecologic conditions, such as endometriosis, endometrial hyperplasia and even frank endometrial carcinoma. While some of the above mentioned studies actually represent single case reports only, and are therefore of limited importance, preliminary results on the use of the LNG IUS in the treatment of endometriosis appear promising. A recent randomized, comparative study comparing the LNG IUS with gonadotropin-releasing hormone analogs (GnRH-a) showed equal improvement of endometriosis symptoms with both treatments [56], confirming the results of earlier observational studies [57,58].
Lockhat and colleagues, assessing the efficacy of the LNG IUS on symptoms of endometriosis, pain and menstrual loss, showed an improved score on the number of days with pain, significant from the sixth month of treatment (p < 0.05), and further improving at 12 months (p < 0.001) [59]. The symptoms continued to improve after the first 12–18 months in women who remained users of the IUS. The 11 women who discontinued the IUS over the first 12 months of use did so due to side effects related to progestin or irregular bleeding. The same authors also showed that the LNG IUS delivered LNG into the peritoneal fluid [60].
The effect of the LNG IUS in endometriosis is presumably due to the strong progestational effect on the endometrium, which also occurs in endometriotic implants. In line with this theory, a reduced progestational effect has been recently suggested to play a key role in the development of endometriosis [61].
These results support the need for further randomized, controlled trials to confirm the role of intrauterine delivery of LNG in women with endometriosis who do not wish to become pregnant.
Adverse effects of the system
Changes in bleeding patterns and enlarged ovarian follicles are the most frequent side effects of the method, occurring in over 10% of users.
Bleeding disturbances
The effects of Cu IUDs and the LNG IUS on the menstrual pattern differ. While the former is associated with an increased menstrual flow and increased dysmenorrhea in a substantial proportion of users, the latter is associated with reduced flow and less painful menstruation [1,12]. During the first 3 months after insertion, the number of bleeding/spotting days is higher in LNG IUS users compared with Cu IUD users, but thereafter the situation is reversed [12]. The median number of bleeding days per month at the end of the first year of use is only 0–2 in LNG IUS users [1,12], significantly less than the average of 5 days observed in Cu IUD users [1,12]. In randomized trials, the rate of discontinuation due to bleeding problems such as heavy or prolonged bleeding has been significantly higher in Cu IUD users compared with LNG IUS users (Table 1) [1,15]. Amenorrhea relatively frequently led to a discontinuation of LNG IUS use, especially in the early studies, but with better counseling this has even become a preferred state, as shown in more recent studies [14].
Cumulative gross termination rates (%) at 5 years for bleeding problems and amenorrhea in Cu IUD (Nova-T, n = 937) and LNG IUS (n = 1821) users.
Cu IUD: Copper intrauterine device; LNG IUS: levonorgestrel intrauterine system
Adapted from [12]: Contraception, 49(1), Anderson K, Odlind V, Rybo C: Levonorgestrel-releasing and copper releasing (Nova-T) IUDs during 5 years use: a randomized, comparative trial, 56–72 (1994) with permission from Elsevier
Enlarged ovarian follicles
Enlarged follicles may be accompanied with high estradiol values and are observed especially during the first year of use. This phenomenon, shared with other progestin-only contraceptive methods, is usually harmless, as the follicles are usually symptomless and resolve spontaneously within a few months in the vast majority of cases [62].
Device-related complications
The occurrence of device-related complications, such as expulsions and perforations, has been comparable between Cu IUDs and the LNG IUS. Pain at insertion was reported as moderate-to-severe in 18.7% of women who received the LNG IUS versus 10.9% in women receiving the Cu IUD for contraception [15]. Since then, a different prebent inserter with a rounded tip has been introduced to facilitate the insertion.
Pelvic inflammatory disease
A lower rate of pelvic inflammatory disease (PID) has been observed in LNG IUS users compared with Cu IUD users in some studies, but not in others [7,63]. Andersson and colleagues, in a randomized, comparative trial of 1821 women using the LNG IUS and 937 women receiving Nova-T®(a Cu/silver IUD), found that the incidence of PID was low with LNG IUS regardless of age, whereas in Nova-T users, the PID rate was increased in the younger age group of users, making the difference between the two devices significant (p < 0.01) [12]. The possible protective effect of the LNG IUS on PID may be due to the effects of LNG on the cervical mucus and endometrium. However, there is no definitive evidence of such protection and caution is warranted, especially in nulliparous women.
Hormonal effects
The hormonal adverse effects of the LNG IUS are, in general, mild in nature and more frequent at the start of therapy. The most frequent adverse effects observed in LNG IUS users are mood changes, acne, headache, breast tenderness, hirsutism and weight change. Most of these could be related to the androgenicity of LNG. Regarding weight change, it should be noted that no difference in the actual measured weight has been observed in randomized studies between Cu IUD users and LNG IUS users [64].
Long-term experience with the levonorgestrel intrauterine system
Since the initial launch of the LNG IUS in Finland in 1990, the use of the LNG IUS has expanded to other Nordic countries, other European countries (UK since 1995) and to the USA (since 2000). Currently, the LNG IUS is used in over 100 countries (Figure 1). The longest experience of LNG IUS use therefore derives from the Nordic countries, with some patients there using their third or fourth consecutive LNG IUS. The pattern of bleeding during the second 5-year segment is usually predominated by amenorrhea or oligomenorrhea. While, in the original studies, the majority of women using their first LNG IUS reported regular menstrual bleeding, the situation during the second LNG IUS is usually the reverse, with 60% reporting amenorrhea [64]. The increase in amenorrhea is not due to previous discontinuations for the same condition, as the discontinuation rate for this reason in the original study was only 6% during the first 5-year period (randomized, part comparative to Cu IUD) [12], and no discontinuations due to amenorrhea occurred in the second or third 5-year period [64]. Although the women who participated in the study were counseled regarding amenorrhea, this finding is in line with other surveys, which have observed that women are actually more ready to accept amenorrhea associated with contraception than healthcare providers [65].
The long-term use of the LNG IUS has revealed no major health problems [64]. The serum hemoglobin values continue to increase slightly throughout the use time [64]. Compared with Cu IUD users, body weight did not differ significantly in the LNG IUS users, as women in both groups gained approximately 0.5 kg in weight each year [12]. No significant effects on blood pressure [64] or bone mineral density [66] have been observed. Another study has found that the likelihood of uterine fibroids, dysmenorrhea and vaginitis during LNG IUS use was less than with Cu IUDs, whereas delayed follicular atresia, breast tenderness, headache and skin and hair conditions were more common among the LNG IUS users [31]. PID, other genital infections, urinary complaints, changes in papilloma smears and benign and malignant reproductive tract tumors were equally low in both groups [31].
For practical reasons, the removal of an old system and the insertion of a new one is usually carried out at the same visit. As the endometrium is already quiescent, the initial bleeding problems are then avoided [64].
Conclusion
The LNG IUS is one of the most effective methods of contraception available and is approved in many countries for 5 years of use. By inactivating the endometrium, the LNG IUS is able to provide a range of other health benefits, such as treatment of menorrhagia and associated anemia, a condition common in developing countries. This additional health benefit is perceived to be of high interest in these populations [29].
Clinical data gathered from large cohorts of users of the LNG IUS indicate a variety of health benefits of the medicated IUS in addition to its contraceptive efficacy. The safety of direct delivery of progestin into the endometrium has been well documented over many years of follow-up, and the health effects of the LNG IUS in estrogen replacement therapy support the rationale for its development for use in menopausal HT.
Future perspective
The high efficacy of the LNG IUS and its long duration of action present an additional contraceptive option for women. Compliance and effectiveness should be increased compared with daily or monthly contraceptives. This method should in the future replace tubal ligation in many women who have completed their family and require a long-acting method which is still reversible. Its direct action on the endometrium and the decrease in menstrual blood loss is an additional benefit in populations where women are anemic. It has been suggested that any new method of contraception should bring an additional health benefit to the users in order to increase compliance and improve health [67]. The LNG IUS showed such dual effects by reducing the incidence of anemia and menstrual blood loss while offering effective contraception.
In addition, given the recent debate raised by the Women's Health Initiative study results regarding the risk/benefit ratio of HT, and in particular the role of progestins, the potential advantages of delivering a low-dose of progestin directly into the uterus for protection of the endometrium are clear.
The LNG IUS has drawbacks, but some of the side effects are accepted by women when proper counseling is provided. In addition, technical difficulties in inserting the IUS in some women have been related to the size of the device. Improvement of the system and development of a smaller system more suitable to nulliparous or postmenopausal women is ongoing, and will expand its acceptability in these age groups.
Executive summary
The intrauterine system (IUS) delivering levonorgestrel (LNG) (Mirena®) has demonstrated its efficacy and safety in a large number of users all over the world.
The intrauterine delivery (IUD) of a small dose of a potent progestin maintains the endometrium in a nonproliferative state and reduces the number of bleeding days, thus preventing anemia.
The added medical benefits of this contraceptive method may increase compliance.
In addition, the IUD of a progestin allows the treatment of other medical conditions and prevention of endometrial hyperplasia in women taking estrogen therapy.
Although the circulating levels of LNG are very low, side effects related to the androgenic properties of LNG have been reported.
In addition, the insertion of the system needs a skilled health provider and may be difficult or painful when the uterus is small or atrophic. The development of smaller devices is warranted in these specific indications.
