Abstract
The effects and mechanisms of rare earth elements on plant growth have not been extensively characterized. In the current study,
Keywords
INTRODUCTION
Rare earth elements (REEs) include 15 lanthanide elements in group III A of the Periodic Table, exhibiting similar chemical and physical properties. In addition, yttrium and scandium have also been considered as REEs. Owing to their desirable properties and abundant sources in China, REE-based microfertilizers or additives have been utilized to enhance yield and quality of crops and vegetables (Hong et al. 2000; Wang et al. 2007).
Hormesis refers to concentration–response occurrence in various organisms, including numerous plant traits (Calabrese and Blain 2008). Hormesis is usually represented by two types of curves, including J-shaped curve showing a decrease below the control at low doses followed by an increase at higher doses, and inverted U-shaped curve showing an increase above the control at low doses followed by a decrease at higher doses (Calabrese and Baldwin 2003). The J-shaped curve is often followed immediately by an inverted U-shaped curve at a specific range of doses, together constituting so-called biphasic dose–response curves. Hormetic dose–response curves are widely used to determine the efficacy and hazard of pollutants (Qin et al. 2010). However, the underlying mechanism is not fully understood.
REEs are mainly taken up by roots and transported to other parts of plants, potentially contributing to hormetic effects in various biological parameters (Ouyang et al. 2003). REE-dependent stimulatory effects on cell proliferation and division as well as plant growth have been previously documented (Hagenbeek et al. 2000; Wu et al. 2001; Hong et al. 2003; Dai et al. 2008). However, REEs have also been shown to displace biometals in membrane metallobiomolecules, metallic proteins and enzymes, leading to mineral nutrient imbalance, membrane permeability, altered conformation in biomolecules, and ultimately functional disorders (Qiu et al. 2005; Zeng et al. 2006). For example, REEs appeared to regulate plant growth by affecting distribution and contents of mineral elements such as Ca, Fe, Cu, K, P, and Mg in some plants (Hu et al. 2004; Wang et al. 2008). In addition, cell cycle progression was further proved to be altered with lanthanum (La) treatment in root tips of
Generally, root lengthening correlates with apical meristem activity and cell extension. In this study, roots of
MATERIALS AND METHODS
Plant Materials and La3+ Treatment
Seeds of
Measurement of Shoot Heights and Root Lengths
Lengths between apical buds and basal stems were measured as heights of the seedlings, and lengths between basal stems and primary root tips as root lengths. Eight seedlings were measured in each container, and three containers were prepared in each treatment.
Contents of La and Other Metals
Fresh roots were consecutively rinsed with 1 M HCl and distilled water. Digestion of samples was performed according to previous protocol (Wang et al. 2010). La, calcium (Ca), Ferrum (Fe) or Potassium (K) content was detected by inductively coupled plasma optical emission spectrometer (ICP-OES) and expressed as μg g−1 dry weight (DW). Certified standard samples (GBW07429) and triplicates of all samples were used to ensure accuracy and precision. The detection limit of 0.02 μg L−1 for this method was applied for all results.
Cell Cycle Progression and Proliferation Index in Root Tips
Approximately 50 fresh root tips (5 mm long) were cut from each control and treatment, and immediately fixed in 4% (v/v) formaldehyde in Galbraith buffer (Galbraith et al. 1983), supplemented with 1% (m/v) polyvinylpyrrolidone and 10 mM sodium metabisulfite (pH 7.0) for 30 min at 4°C. Samples were then washed thoroughly in pre-chilled Galbraith buffer for 10 min. Nuclei of fixed root tips were isolated using 1mL of the buffer by crushing them with a glass rod in Petri dishes on ice. The dishes were kept tilted on ice to yield efficient collection of isolated nuclei in the buffer.
Nuclear suspensions were filtered through nylon mesh of 25 μm pore size to remove gross particles and centrifuged at 700×
Determination of DNA-Protein Crosslink (DPC)
DPC formation was monitored based on fractionation of protein-bound and free DNA by potassium-SDS precipitation as described by Costa et al. (1996). Briefly, nuclei in root tips were isolated and purified as described by Gichner et al. (2004). The pelleted nuclei were lysed in 0.5% (m/v) SDS, and 0.5 ml of 100 mM KCl and 20 mM Tris (pH 7.5) were added to each tube. After 10 min incubation at 65°C, the tubes were placed on ice for 5 min, and then centrifuged at 6000×
HSP 70 Western Blotting Analysis
Crude extracts in root tips were prepared according to a method described by Romero-Puertas et al. (2004). Soluble protein content was determined by method of Bradford (1976) with BSA as standard. All operations were performed at 4°C. Sodium dodecylsulfonate polyacrylate gel electrophoresis (SDS-PAGE) and Western blotting of HSP 70 were conducted according to Wang et al. (2010) with minor modification. Total protein extract was mixed with lysis buffer (0.5 M Tris, pH 6.8, 20% (v/v) glycerol, 3% (m/v) SDS, 0.01% (m/v) bromophenol blue, 10% (v/v) β-mercaptoethanol), boiled for 4 min, and cooled on ice. Twenty six μg of total proteins per lane, together with PageRuler™ prestained protein ladder (Fermentas), were separated by 10% (m/v) SDS-PAGE. After electrophoresis, gels were either stained with Coomassie brilliant blue R-250 or transferred onto polyvinylidene fluoride membrane (Amersham). The membranes were blocked with 10% (m/v) non-fat milk/TBST buffer (50 mM Tris, 150 mM NaCl, pH 7.5, containing 0.05% (v/v) Tween-20) for 2 h. After washing, mouse anti-HSP 70/HSC 70 monoclonal antibody (SPA820, Stressgen Corp.) (diluted 1: 5000) was added and incubated overnight at 4°C. After washing, the membranes were incubated in goat anti-mouse IgG conjugated with horseradish peroxidase (Stressgen Corp.) (diluted 1: 25,000) at room temperature for 1.5 h. Bands were visualized by SuperSignal West Femto Maximum Sensitivity Substrate (Thermo Scientific) and exposed to X-ray film. Integrated densities in bands were quantificated by Image J software. The experiment was repeated 3 times.
Statistical Analysis
All statistical analyses were performed using SPSS 13.0 for Windows (SPSS, Chicago IL, USA). The data were presented as mean ± standard deviations of three replicates. Difference was considered as significant at
RESULTS
Contents of La, Ca, Fe and K Elements in Roots
ICP-OES was employed to monitor changes of La and mineral elements such as Ca, Fe or K in the roots. The results showed that contents of La in the roots increased with elevated extraneous La (Fig. 1A), which significantly enriched when the extraneous La was more than 1 mg L−1. Contents of Ca, Fe or K were initially reduced before rebounding with increased levels of La, displaying a characteristic U-shaped curve. Particularly, Ca or Fe contents were significantly enhanced at 16 mg L−1 of extraneous La. Generally, Low concentrations of La resulted in decreased contents of Ca, Fe or K, whereas higher concentrations led to opposite effects (Fig. 1, B–D).
Alteration in Cell Cycle Progression and Proliferation Index
Cell cycle phases and relative nuclear DNA contents were determined by flow cytometric analysis of nuclei released from the root tips (Fig. 2A). The ratios of G0/G1, S or G2/M phase displayed a biphasic curve in response to increasing levels of La. The G0/G1 phase ratios mirrored that of the G2/M phase, showing initial enhancement at 0–0.5 mg L−1, followed by a steady decline from 0.5 to 4 mg L−1 before the rebound. The S phase ratios, on the other hand, exhibited an essentially opposite pattern (Fig. 2B). Therefore, the cell cycles were likely arrested at G1/S and/or S/G2 interphases by La3+ in the root tip cells. Furthermore, the proliferation index displayed a pattern closely in parallel with the S-phase ratio, with initial reduction at 0–0.5 mg L−1, followed by enhancement from 0.5 to 4 mg L−1 and decline thereafter (Fig. 2 C). However, no significant elevation or reduction was observed in cell cycle phase ratios and proliferation index (Fig. 2 B, C).
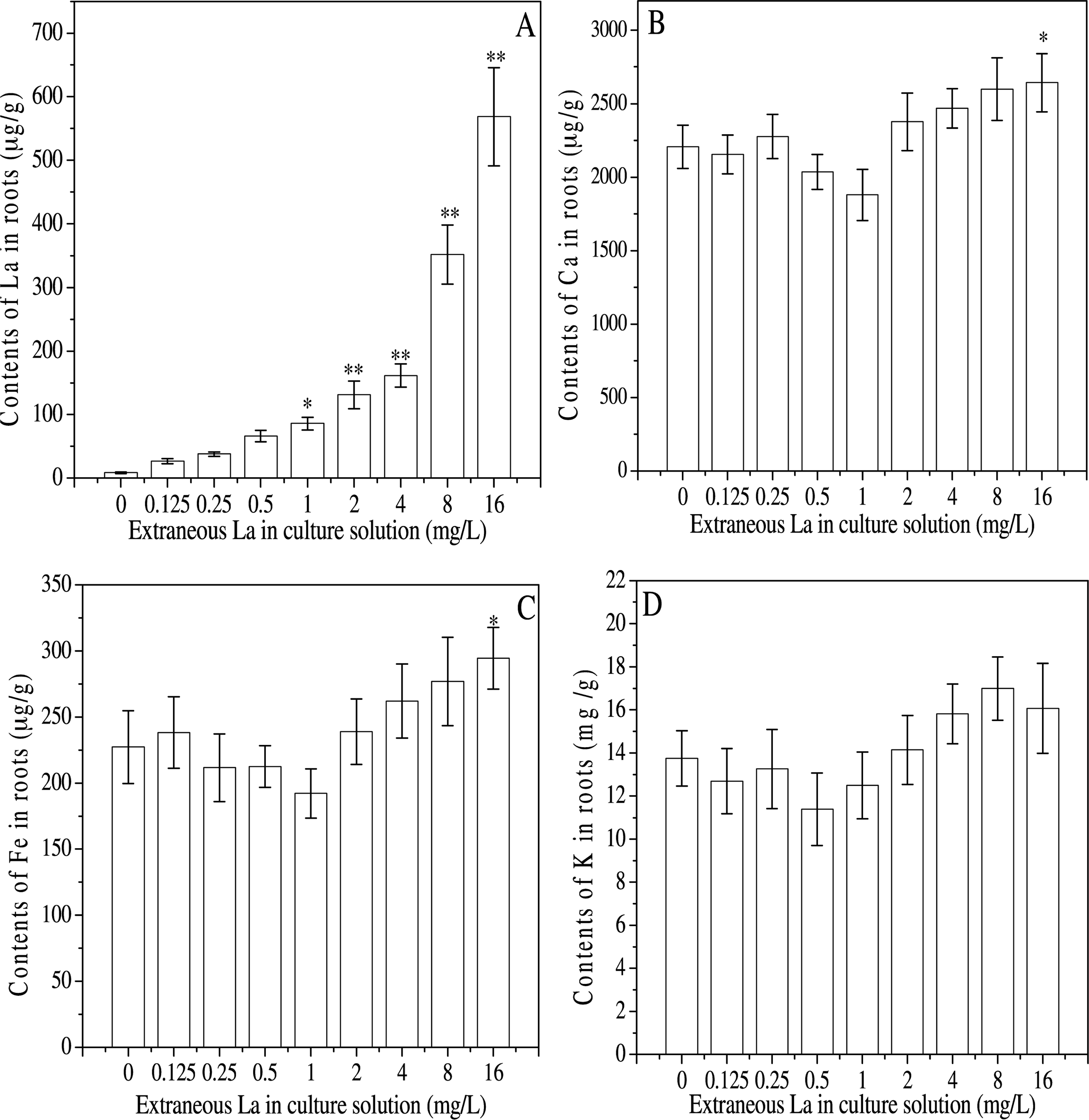
La (A), Ca (B), Fe (C), and K (D) contents in roots of

Histograms of relative nuclear DNA contents and cell cycles (A) obtained by flow cytometric analysis of nuclei released from root tips of
In addition, root lengths were found to be positively correlated with the S-phase ratios (
Changes of DPC and Seedling Growth
DPCs were involved in DNA replication and repair, which may interfere with cell proliferation. In the roots, contents of DPCs appeared to decrease at 0–0.5 mg L−1 of extraneous La, and then increased with the increasing La, which significantly elevated when the extraneous La was more than 4 mg L−1 (Fig. 3A). In addition, the contents of DPCs were well correlated with the S-phase ratios (
Moreover, root length fluctuated in parallel with the proliferation index, showing decreases at 0.125–0.5 mg L−1 followed by enhancement from 0.5 to 4 mg L−1 and subsequent decline (Fig. 3B).
Induction of HSP 70 Production
HSP 70 participates in many physiological activities, including cell proliferation. HSP 70 production in the roots was investigated by Western blotting method. The results showed that the relative HSP 70 levels decreased at 0–0.5 mg L−1 of extraneous La, and enhanced with the increasing La, showing a biphasic dose response. In addition, the HSP 70 levels were significantly elevated at 4–8 mg L−1 (Fig. 4).
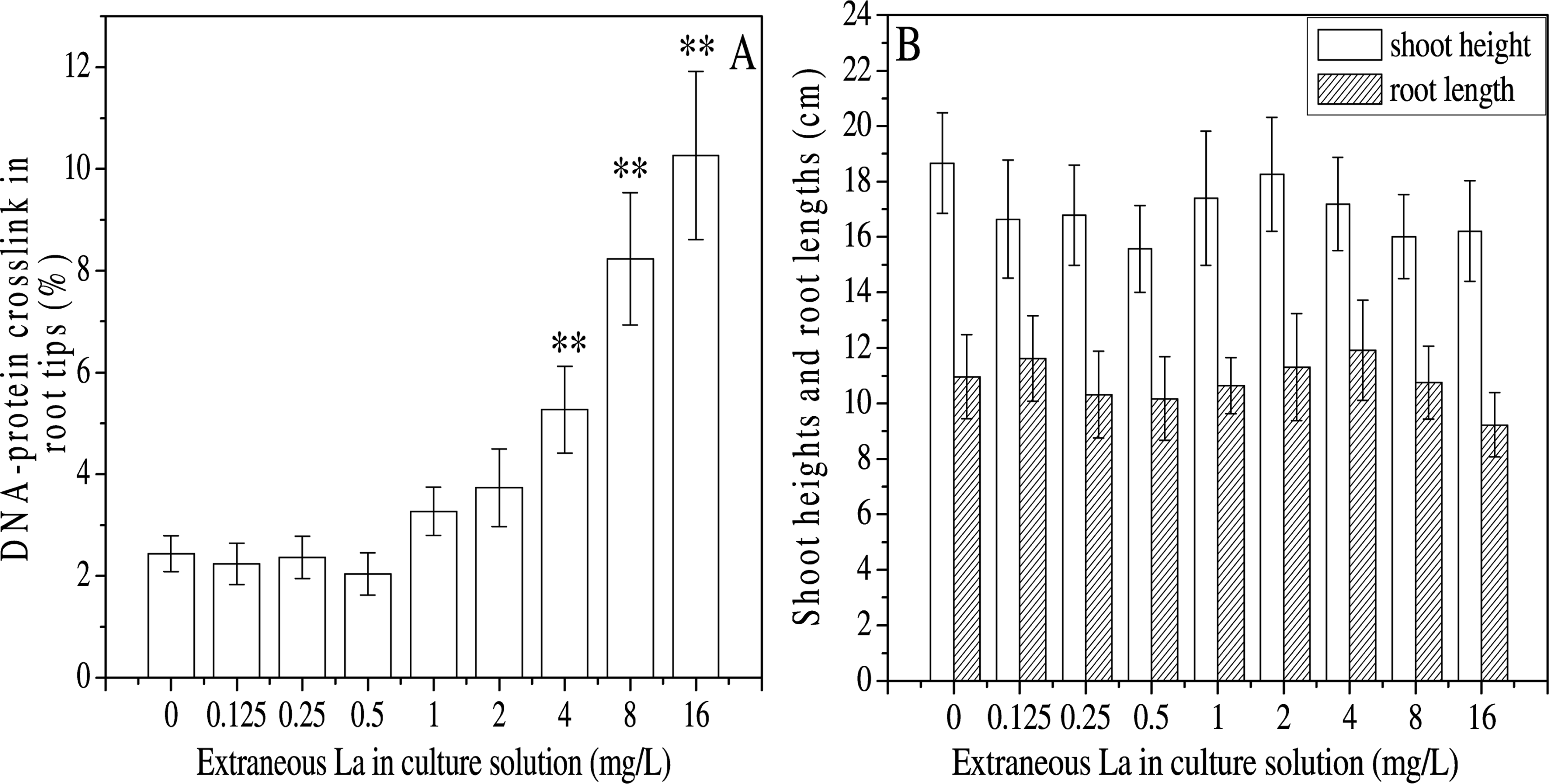
Contents of DNA-protein crosslinks in nuclei separated from the root tips (A), and shoot heights and root lengths (B) of
DISCUSSION
REEs mainly accumulate in plant roots as heavy metals. The content of La in the roots increased with elevated La concentrations, contributing to imbalance of Ca, Fe or K contents in the roots. The varying doses of extraneous La were responsible for the U-shaped dose response of mineral metal contents in the roots. Similar findings have also been reported for Cd on tomato plants. Low dose of Cd2+ caused a moderate Fe deficiency, whereas higher Cd2+ abolished any nutrient interaction due to excess Cd (López-Millán et al. 2009). Thus, REEs likely have similar properties in altering the balance of metal contents in plant tissues as heavy metals.

Western blotting and relative levels of HSP 70 in roots of
Ca uptake has been shown to be completely inhibited by a voltage-dependent Ca channel antagonist such as La in plants (Tammeba et al. 2004). In fact, La3+ can bind to surface-localized Ca2+ absorption sites with lower degrees of reversibility than Ca2+, thus blocking Ca2+ channels and disrupting the uptake of nutrient ions through Ca2+ channels (Hu et al. 2006). In the current study, increasing La induced a U-shaped dose response of Ca, Fe or K in the roots (Fig. 1), suggesting that low concentrations of La contributed to the reduction of these metals. Thus, lower concentrations of La appeared to function as Ca antagonist in the roots. Meanwhile, the U-shaped dose response curve of Ca content also indicated that higher La increased Ca contents in the roots. La at higher doses might inactivate the Ca channels in the membrane and promote the entry of metals such as Ca, Fe and K into the roots. However, La at lower concentrations also led to decreased contents of Fe and K. Whether La at low doses may act as an antagonist of Fe or K needs to be further investigated.
Mitotic activity, cell cycle progression and cell expansion are major factors involved in cell growth (Kononowig et al. 1992). Mineral elements such as Ca, Fe, K, Cu, Mg, etc are known to be essential nutrients for plant development. Absence or deficiency of these elements may have adverse effects on plant growth. In the present study, the concentrations of La resulted in the imbalance of mineral nutrients in the roots. Previous studies also showed that REE (s) regulated plant growth by affecting the distribution of mineral elements in crop seedlings (Wang et al. 2008, Wang et al. 2011). Likewise, the U-shaped fluctuation of mineral element contents is likely to affect cell proliferation and extension in the roots and shoots. Therefore, cell cycle progression and root lengthening may be altered due to nutrient imbalance in the roots subjected to La treatment.
The U-shaped dose response of HSP 70 parallels the patterns of mineral nutrients, S-phase ratios and proliferation index, but was opposite to those of the G0/G1-phase and G2/M-phase ratios in the root tips. Under normal conditions, Ca binds calmodulin and initiates a cascade of signal transduction events including HSP 70 induction. HSP 70 mRNA levels are rapidly elevated at early S phase, but subsequently decreased at late S and G2 phases (Milarski and Morimoto, 1986). In nuclei, the spatial and temporal association of HSP 70 and CaM has been shown to regulate cell cycle progression (Huang et al. 2009). HSP 70 proteins were found to be associated with the spindle, thus functioning in cell cycle progression (Vargas-Roig et al. 1997). Furthermore, HSP 70 appeared to positively correlate with cell cycle progression in human cells (Karlseder et al. 1996; Kim et al. 2006) and plant cells (Giménez-Abián et al. 2004). In addition, HSP 70 overexpression was particularly pronounced during S phase, leading to enlarged S-phase populations (Helmbrecht and Rensing, 1999). However, whether HSP 70 function in cell cycle progression of plants is not fully understood.
Our results suggest that the decreased HSP 70 levels may be involved in the retardation of root tip cell proliferation, since increased G0/G1- and G2/M-phase ratios and decreased S-phase ratios were observed at the 0–0.5 mg L−1 dose range. On the other hand, the enhanced HSP 70 at 1–4 mg L−1 may stimulate proliferation by decreasing G0/G1 and G2/M populations accompanied by increased S-phase ratios in the root tips. Therefore, the La-induced HSP 70 may play a role in the control of cell cycle progression in the root tips at 0–4 mg L−1 of extraneous La. Similar findings have also been reported in human lung cells (Bromberg et al. 2008)
DPCs are formed when proteins become covalently bound to DNA, and serve as bulky helix-distorting adducts to physically block DNA replication, repair and transcription (Barker et al. 2005). Thus, DPCs may interfere with cell proliferation and extension. In the present study, the DPC content was found to increase with the increase of extraneous La from 0.5 to 16 mg L−1. DPCs enrichment may be involved in the decline of the S-phase ratio at more than 4 mg L−1 La, leading to reduced proliferation index and root length.
Finally, our results suggest that the imbalance of mineral nutrients was involved in the induction of HSP 70, which in turn were associated with the cell cycle control. Together, the results in the current study provide useful insights into the functional mechanisms of hormetic effects of La3+ on plant cell proliferation and growth.
Footnotes
ACKNOWLEDGMENTS
We acknowledge supports from the National Natural Science Foundations of China (grant No. 20877032) and the Foundation of State Key Laboratory of Pollution Control and Resources Reuse of China (grant No. PCRRF08011). We also thank Ling Wang and Fan Hu in Center of Medical Analysis of Nanjing Medical University for cell cycle analysis in
