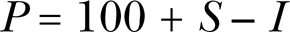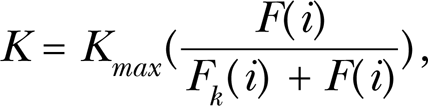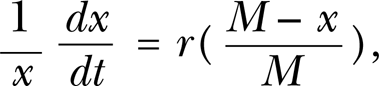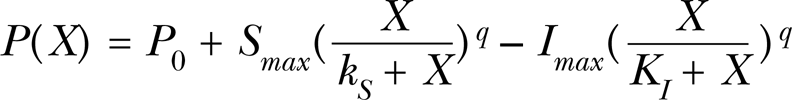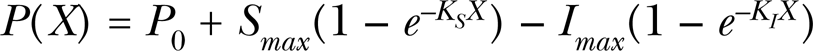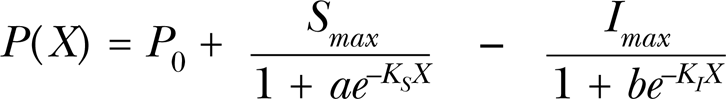Abstract
Allelopathy arises from the release of chemicals by one plant species that affect other species in its vicinity, usually to their detriment. Allelopathic effects have been demonstrated to be limiting factors for species distributions and ecological processes in some natural or agricultural communities. Based on the biphasic hormetic responses of plants to allelochemicals, ecological-limiting-factor models were introduced into the An-Johnson-Lovett hormesis model to improve modelling the phenomenon of allelopathic hormesis and to better reflect the nature of allelopathy as a limiting factor in ecological processes. Outcomes of the models have been compared for several sets of experimental data from the literature and good agreement between the models and data was observed, which indicates that the new models give some insight into the ecological mechanisms involved and may provide more options for modelling the allelopathic phenomenon as well as platforms for further research on plant allelopathic hormesis.
Keywords
INTRODUCTION
The statement of “the poison is in the dose” (Duke et al., 2006) is well known in the areas of environmental and medical toxicology and numerous examples illustrate that the dose-response relationship plays an important role for the environmental, medical and public-health issues. However, only in the past two decades, has the phenomenon of hormesis and, thus, stimulatory effect of a low dose of a toxicant, gained increased recognition (Calabrese and Mehendale 1996; Calabrese and Baldwin 2003; Duke et al. 2006). Hormesis has been found within all groups of organisms, from bacteria and fungi to higher plants and animals and the more efforts of hormesis with different toxicants and organisms will lead us to understand the implications of hormesis in all fields of science (Calabrese 2005).
Allelopathy refers to a phenomenon by which one plant directly or indirectly causes detrimental (or occasionally beneficial) effects on other plants through the release of allelochemicals into the environment (Rice 1984). Allelochemicals are well known to induce hormesis. Many studies have shown that allelochemicals can stimulate plant growth at low concentrations but inhibit plant growth at high concentrations (Rice 1984; Lovett et al. 1989). In fact, when the founder of allelopathy, Molisch (1937) coined the term allelopathy he well acknowledged the phenomenon of allelopathic hormesis by stating “… the often observed rule is confirmed that poisons and irritating compounds are harmful in higher concentrations, but stimulate in diluted form” (Molisch 1937, 2002; Chobot and Hadacek 2009). During the last two decades, the science of allelopathy has attracted a great number of scientists from diverse field worldwide, which is due to the prospects that allelopathy holds for meeting increased demands for sustainability in agriculture, for reducing environmental damage and health hazards from synthetic pesticides inputs, for reducing reliance on synthetic herbicides, and for finding alternatives to replace them.
To demonstrate the existence of hormesis, dose-response curves that include several doses below the adverse effect concentrations must be generated (Duke et al. 2006). Of the disciplines involved in hormesis research, mathematical modeling is making increasingly significant contributions. More studies have been devoted to set up models to describe the relationship between the dose of toxicants and the response of organisms (Haanstra 1985; Streibig et al. 1993; Sinkkonen 2007). Mathematical models are generally classified into two types: empirical and mechanism-based models. The latter is often eligible to provide some insight into the underlying processes and has thus a more wide applicability. However, many dose-response models of hormesis are still at the stage of empirical modeling. One extensively used empirical model of plant responses to allelochemicals and other phytotoxins is the four-parameter log-logistic response function (Finney 1978; Streibig 1988). Mathematically it is a strictly monotone function, therefore, it cannot be used to describe low dose stimulatory effects. Brain and Cousens (1989) modified the four-parameter log-logistic function to exhibit the property of low-dose stimulation and high-dose inhibition. Cedergreen et al. (2005) developed a new, empirical four-parameter logistic model by further modifying Brain and Cousens’ work. Although, as a result of those endeavors, the improved models can be used to fit experimental data exhibiting hormesis and to estimate hormetic effects with lower concentrations, such empirical models are still limited in giving insight into the mechanistic processes involved.
An et al. (1993) developed a mechanism-based model to describe the responses of plants to allelochemicals. This model was named the An-Johnson-Lovett hormesis model by Liu et al. (2007). This model pioneered mechanistic modeling of allelopathic hormesis. In this model An et al. (1993) hypothesized that the characteristic response to allelochemicals is a result of the character of the allelochemicals themselves. An allelochemical is assumed to have two complementary attributes: stimulation and inhibition. As concentration changes, the relative dominance of stimulation and inhibition by the allelochemical changes, thereby determining the overall effect of the allelochemical. Accordingly, when the stimulatory attribute dominates, a biological response of stimulation appears, while a biological response of inhibition exists as the inhibition attribute is dominant. This can only be shown through the biological responses when an allelochemical acts on an organism. The authors defined such characteristics of an allelochemical as the biological property of an allelochemical, so as to distinguish it from physical and chemical properties (An et al. 1993). Recent experimental work with juglone (5-hydroxy-1,4-naphthoquinone) by Chobot and Hadacek (2009) supports this hypothesis. Juglone is a well known allelochemical with redox cycling properties (Rice 1984). Chobot and Hadacek (2009) suggested that the redox cycling properties of juglone may be contributed to its wide range of biological activities. Based on their experimental findings Chobot and Hadacek (2009) claimed that the hormetic effects of lower concentrations of juglone on germination of Sinapis alba may be caused by its antioxidant activities, while its pronounced prooxidative activities may contribute to the toxic effects of juglone at higher concentrations.
The mathematical expression of the An-Johnson-Lovett hormesis model is as follows:
where P represents the biological response to an allelochemical, S and I are biological responses to the stimulatory and inhibitory attributes of an allelochemical, respectively.
The critical part of the An-Johnson-Lovett hormesis model is to find a suitable function to describe the responses of stimulatory and inhibitory attributes. The modified Michaelis-Menten enzyme kinetics equation was employed in the An-Johnson-Lovett hormesis model to describe both stimulation and inhibition upon an assumption that both attributes have a sigmoid response to allelochemical concentration (An et al. 1993). However, biological and biochemical processes are complicated and plant species are subjected to various environmental conditions. Furthermore, allelopathy itself acts as a limiting factor in ecological processes (Muller 1969). Therefore, it seems unrealistic that one enzyme kinetics equation will fit all processes and situations. This has limited the applicability of the An-Johnson-Lovett hormesis model. The aim of the present study was to modify the An-Johnson-Lovett hormesis model by introducing ecological-limiting-factor and other sigmoid based equations in order to (1) expand its applicability, (2) better model the variety of allelopathic hormesis responses by different biological species, and (3) better reflect the nature of allelopathy as a limiting factor in ecological processes.
DESCRIPTION OF THE MODELS
Ecological-limiting-factor models
Organisms are affected by various environmental factors that directly or indirectly influence their growth, development, reproduction and distribution. Among these ecological factors are pesticides, temperature, moisture, and oxygen etc. In fact, organisms are simultaneously affected by many ecological factors during growth. Limiting factors are those factors that play a key role in affecting growth, survival, and reproduction of an organism. Any ecological factor has the potential to limit an organism, but one is usually the principle constraint at any given point in time and space, eg., water as a limiting factor for plant growth in arid regions and temperature as a limiting factor in cold areas (Kaiser et al. 1994; Cade et al. 1999). The Monod model and the Mitscherlich model are two commonly used ecological-limiting-factor models to describe the relationship between organisms and environmental factors.
1 Monod Model
The Monod model is a biologically-based model commonly used for describing microbial growth and substrate degradations in applications such as batch and continuous fermentation, activated sludge waste water treatment, pharmacokinetics and plant physiology (Pirt 1975; Dette et al. 2003). The Monod model is normally expressed as:
where K refers to the biochemical response speed, i is the limiting factor, Kmax represents the maximum biochemical response speed, F(i) is the quantity of the limiting factor i, and Fk (i) is the quantity of the limiting factor when the maximum biochemical response speed is at the half of Kmax .
In an actual environment there may be a few limiting factors involved and their relative quantities could vary greatly. It may also be difficult to distinguish primary and secondary limiting factors. The Monod model is flexible in this regard and capable of dealing with a few limiting factors simultaneously, either with the same or different importance. For example, if two limiting factors are involved, i.e. i=2, then the model can be expressed as:
Such flexibility is a characteristic feature for the Monod model as an ecological-limiting factor model.
2 Mitscherlich Model
Mitscherlich's (1923) model proposes that, if no factor were limiting to growth, a certain maximum yield would be obtained, and that the biological response to addition of a limiting factor were proportional to the difference between current and maximum yield. The Mitscherlich model can be expressed as:
where K is the yield at a level of the i th limiting factor, F; Kmax is the maximum yield obtainable, and C is an experimental constant that describes the response of yield to increments of the limiting factor. The model was extensively used to describe the yield responses of crops to various nutrient deficiencies. The solution of the Mitscherlich model at the condition of K(0)=0 is
where i refers to the i th limiting factor. Other parameters are as above. The Mitscherlich model also is capable of dealing with a few limiting factors simultaneously just as the Monod model.
(3) Logistic Growth Model
Growth curves of animals or plants are usually sigmoidal, that is, their growth is initially slow, then accelerates, but slows down again in the end. For individual plants, tissues or organs, their growth speed will ultimately reach an upper limit owing to their physiological feedback in some certain growth stages. Although the growth and development of a plant can be classified into several levels such as cell, organ, individual and community it always exhibits a sigmoid curve on a certain level. The most pronounced S-shaped curve is a logistic growth curve, which was first proposed by P. F. Verhulst in 1838 to describe the succession of a community under resource-limited conditions. The Logistic growth model was employed here as an alternative to Michaelis-Menten enzyme kinetics equation based upon its feature of possessing a sigmoid response to a limiting factor.
Letting the upper limit be M and the inherent growth rate r, then the growth can be expressed by a logistic equation as:
where x refers to the value of a response parameter (e.g. height, weight or biomass) of an organism at time t. The relative growth rate is positively correlated with the remaining resources, but it is negatively correlated with environmental capacity. The result for satisfying the initial value is:
where a is the relative position of the curve to the origin, that is
x0 is the value of x at t=0.
New forms of the An-Johnson-Lovett hormesis model
The above three ecological-limiting-factor models were included in the An-Johnson-Lovett hormesis model, which yielded three corresponding new forms of the An-Johnson-Lovett model.
Model I: yielded from applying the Monod model, i.e.
Model II: yielded from applying the Mitscherlich model, i.e.
Model III: yielded from applying the Logistic model, i.e.
where the meaning of all parameters is the same as in An et al. (1993), i.e. P(X) is the biological response at concentration X of an allelochemical, P 0 is the control, Smax is the maximum stimulatory response, and KS is a constant that describes the response of stimulation to increments of the limiting factor. Imax and KI are the respective parameters of the inhibitory attribute. Parameters q, a, b are constants.
The general behaviors of the three new models are demonstrated through simulation by Matlab 7.x and fitted against the experimental data reported in An et al. (1993).
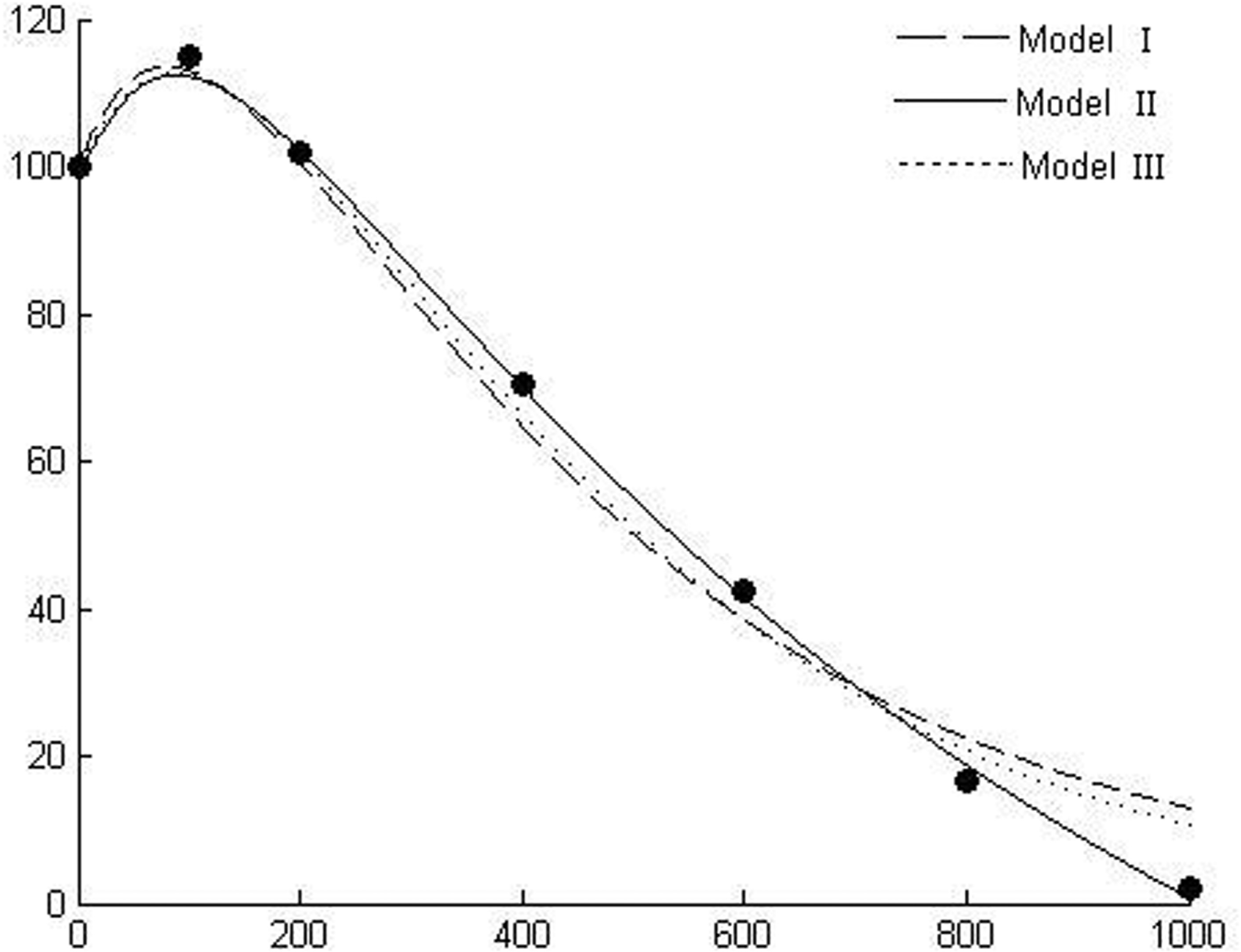
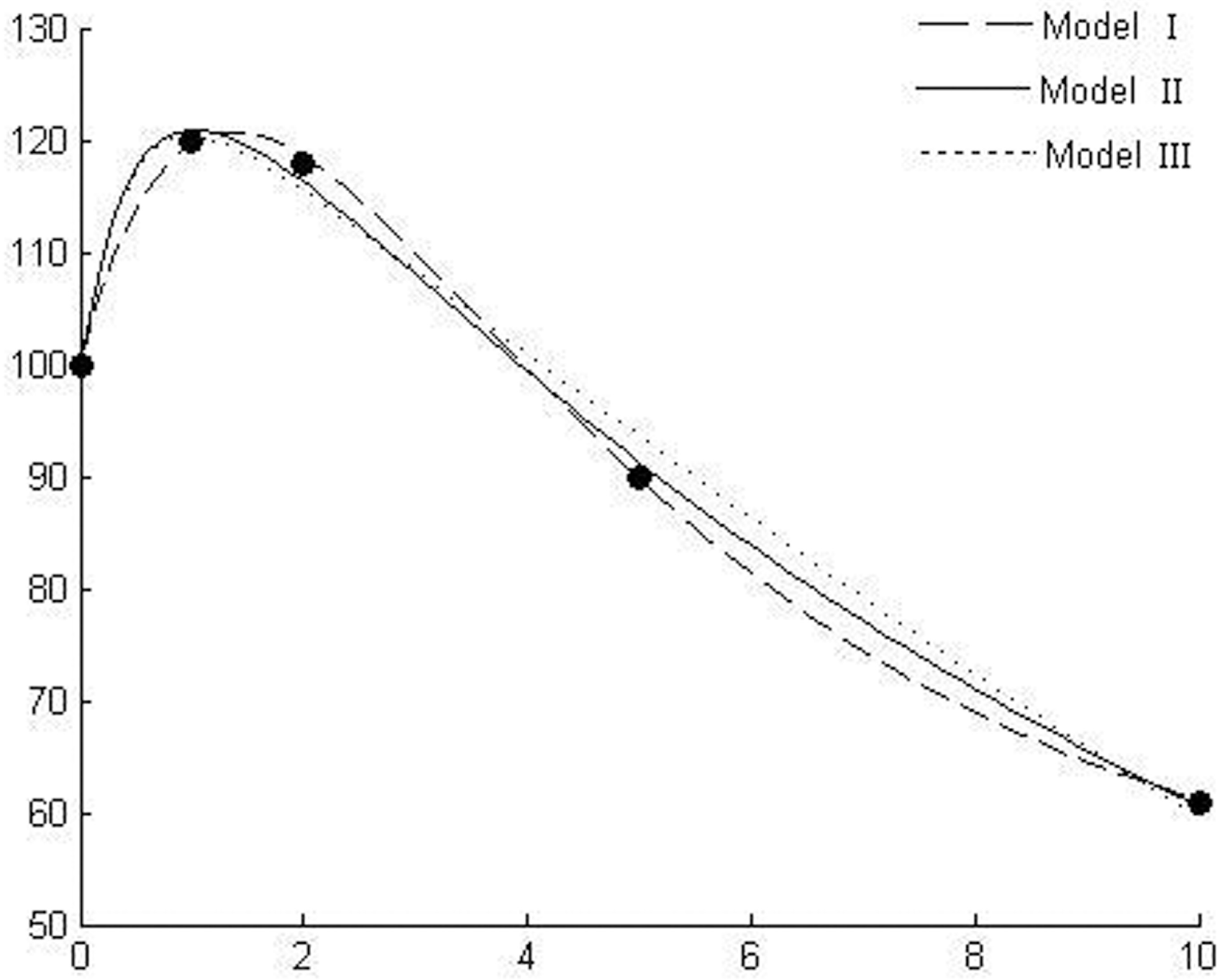
Response of radicle length of linseed (Linum usitatissimum L.) to benzylamine (data from Lovett et al. 1989).
RESULTS
All three models were able to describe the characteristic hormetic effect as the biological response to allelochemicals, i.e. stimulatory response at low doses and inhibitory response at high doses. The new models were also fitted against broad experimental data from Lovett el al. (1989), which were originally used by An et al. (1993) for the illustration of the An-Johnson-Lovett hormesis model. As in An et al. (1993), all direct measurements of biological responses have been converted to percent control values. All fitted curves were derived by minimizing the residual sum of squares for the difference between the actual data values and the values estimated by the three models. The parameters for models I-III determined using the Levenberg-Marquardt method for nonlinear least squares. The calculation processes were coded in Matlab 7.x.

Response of radicle length of white mustard (Sinapis alba L.) to gramine (data from Lovett et al. 1989).
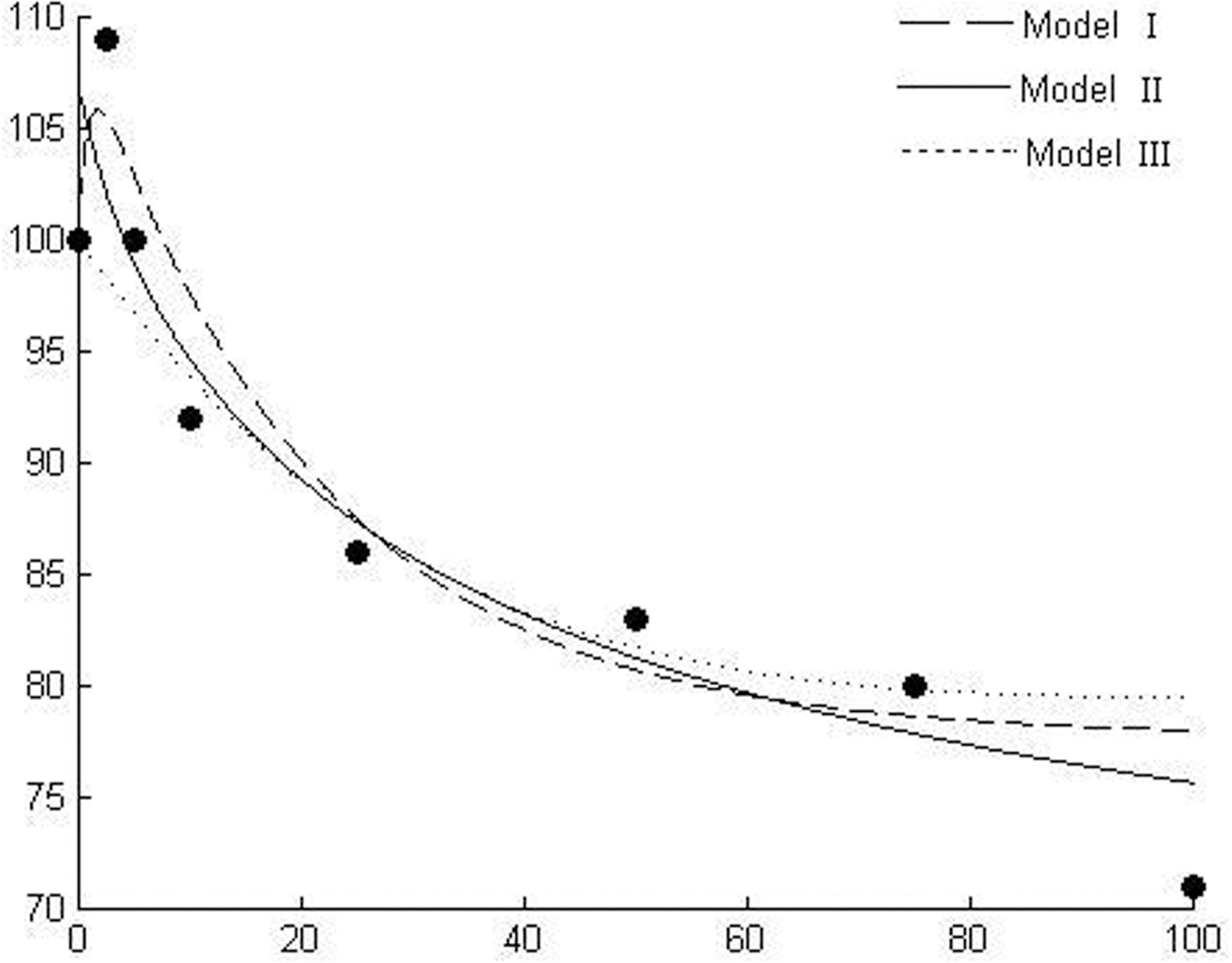
Response of radicle length of white mustard (Sinapis alba L.) to hordenine (data from Lovett et al. 1989).

Effect of castanospermine on root growth of lettuce (Lactuca sativa L.) (data from Stevens and Molyneux 1988).

Emergence of wild oats (A. ludoviciana) in response to applied wheat straw leachate (data from Purvis and Jessop 1985).
Figures 1–6 show the behavior of the three models in comparison using six different experimental data sets and Table 1 gives the details of goodness of fit to experimental data. Figure 1 shows the dose responses of radicle length of linseed to benzylamine (Lovett et al. 1989). Figures 2 & 3 show the responses of radicle length of white mustard to gramine and hordenine, respectively (Lovett et al. 1989). Figure 4 shows the effect of castanospermine on root growth of lettuce (Stevens and Molyneux 1988). The experiment used concentrations of 0, 100, 200, 400, 800, and 1000ppb and did not show a stimulatory effect at low concentrations. An et al. (1993) claimed, based on their modeling outcomes, that it is possible that a stimulatory effect may occur at much lower allelochemical concentrations than those included in the experiment. Current simulating outcomes of the models also show stimulation at low concentrations. Figure 5 shows the emergence of wild oats (Avena ludoviciana L./Dur) in response to wheat straw leachate, which is known to contain allelochemicals (e.g., Lynch 1977). Figure 6 shows the response of a-amylase activity to scopolamine (Lovett et al. 1989).
Parameters of three new hormesis models and their goodness of fitting with experimental data from literature, i.e. Lovett et al. (1989), Stevens and Molyneux (1988), and Purvis and Jessop (1985)
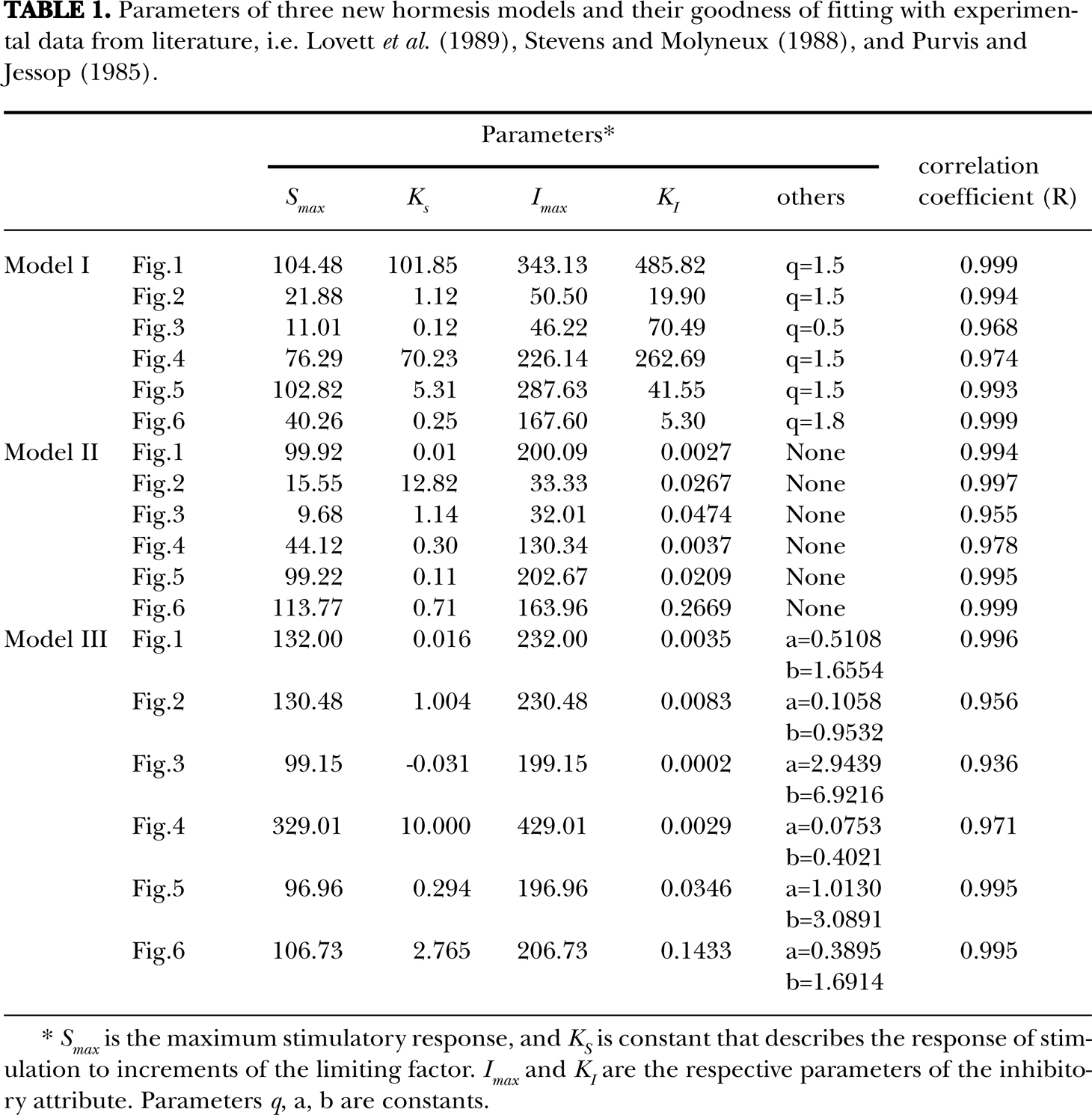
Smax is the maximum stimulatory response, and KS is constant that describes the response of stimulation to increments of the limiting factor. Imax and KI are the respective parameters of the inhibitory attribute. Parameters q, a, b are constants.
It is obvious that the behavior of the three new models is in a good agreement with a wide range of experimental data taken from the literature (Figures 1–6, Table 1). Three new forms of the An-Johnson-Lovett hormesis model accounted for more than 90% of variance in the observed dose responses, indicating a high degree of prediction and flexibility of the models (Table 1).
Response of a-amylase activity to scopolamine (data from Lovett et al. 1989).
DISCUSSION AND CONCLUSION
Allelopathic hormesis has gained increased recognition just in the past two decades, although Molisch coined the term allelopathy and acknowledged the existence of an allelopathic hormesis phenomenon more than 70 years ago. Various hypotheses on its physiological mechanisms are proposed, such as redox cycling properties of allelochemicals (Chobot and Hadacek 2009), “escape” or compensate for chemical stress, induced different defense systems, plant hormonal or non-hormonal mechanisms (Duke et al. 2006). Plants are regularly subjected to environmental conditions that are less than optimal and these collective stresses may interact in additive, synergistic, or antagonistic ways (Einhellig 1989). Muller (1969) stated that although allelopathy is not involving the depletion of a necessary factor it is depending upon the addition of a deleterious factor, which inevitably may interact synergistically with other stress factors (Sinkkonen 2006) and may become the limiting factors for species distributions and ecological processes in almost any natural or agricultural community (Muller 1969). The An-Johnson-Lovett hormesis model is a unique mechanistic model for describing allelopathic hormesis, which has become a platform for developing mathematical means to comprehensively analyze allelopathic hormesis data (An et al. 2005). This paper further expands the applicability of the An-Johnson-Lovett hormesis model by considering the nature of allelopathy as a limiting factor, an underlying ecological process for plant population and community, and successfully introducing ecological-limiting-factor models into it. The new models can be employed to simultaneously assess the synergistic effects of an allelochemical with other limiting factors such as temperature, moisture, and nutrients. This might better help increase our understanding of underlying mechanisms of allelopathic hormesis and better assess the potential benefits as well as risks of allelopathic hormesis.
Footnotes
ACKNOWLEDGMENTS
We thank the Foundation of Key Laboratory of Ecological Agriculture of Ministry of Agriculture (2009k01), the Natural Science Foundation for Doctoral Program of Guangdong Province (8451064201001009), President Fund of South China Agricultural University (2008K021), and National Natural Science Foundation of China (40876074), and the CSU Competitive Grant (2009) for the financial supports.