Abstract
Two almost completely exclusive fields in radiobiology deal with splitting doses of radiation and comparing the effect to a similar total dose given in one exposure. In radiotherapy, dose “fractionation” is used to “spare” normal tissue and in the low dose field, the adaptive response is well documented as a phenomenon where a small “priming” dose administered before the larger “challenge” dose reduces the effect of the large dose. There have been very few studies where these fields overlap, thus it is not possible to ascertain whether common or distinct mechanisms underlie both phenomena but this is certainly an interesting question and relevant to our understanding of high and low dose radiobiology. This paper presents data for three human cell lines with varying p53 status and radiation responses, treated at a range of times between first and second dose and for 3 different first doses (0.1, 0.5 and 2Gy). The data show that time between doses is critical. Protective (adaptive) effects were seen in each cell line but most prominently in the malignant HT 29 cell line. Surprisingly none of the cell lines showed pronounced split dose recovery. This suggests different mechanisms may underlie the two phenomena.
INTRODUCTION
It has been well documented that cells respond to the harmful effect of exposure to ionizing radiation by inducing protective mechanisms (Azzam et al. 1994; Alsbeih et al. 1999; Adelstein 2003). The classic adaptive response and induced resistance to clinical-sized priming doses from split-dose recovery are forms of induced repair responses previously described (Alsbeih et al. 1999).
The adaptive response is a biological phenomenon in which resistance to a challenging dose of radiation is established by a very small preceding “priming” dose (Olivieri et al. 1984; Wolff 1998; Kadhim et al. 2004; Tapio and Jacob 2006). The adaptive response was first hypothesized after observations that prior exposure to low doses of radiation left cells more resistant to the effects of a higher subsequent dose (Olivieri et al. 1984; Wolff 1998). This priming dose has been shown to increase cloning efficiency as well as to reduce mutation frequency and micronucleus formation (Smith and Raaphorst 2003; Zhou et al. 2003).
Split dose recovery (also known as Elkind recovery) is defined as causing a decrease in radiation effect if a single dose is split into two fractions separated by time. Split-dose recovery has been described in several cell systems and is at least partly due to the repair of single and double stranded DNA breaks (Elkind and Sutton 1959; Elkind et al. 1967; Hall 2000; Utsumi and Elkine 2001). Other hypotheses such as repair pool replenishment have been suggested (Orr et al. 1966a; Orr et al. 1966b; Malone et al. 1971; Laurie et al. 1972; Malone et al. 1972; Orr 1984). Inducible defenses such as those seen in classical adaptive response experiments have been suggested to be involved in split dose recovery as well (Seymour and Mothersill 1989; Mothersill and Seymour 1993). The result of this effect is that cell populations have a higher resistance to a large radiation dose if it is split into fractions with an interval between exposures to allow for recovery and repair. This effect is important in clinical radiation therapy, and is the basis for the fractionated regimes used in the treatment of malignancy. The molecular mechanisms governing this response have yet to be completely elucidated.
In a previous study, our group investigated these cell lines for the presence of an adaptive response (Ryan et al. 2009). A correlation was found between the presence of an adaptive response and hyper-radiosensitivity/increased radioresistance (HRS/IRR). HRS/IRR is a phenomenon in which cells are exquisitely sensitive to acute doses of ionizing radiation typically below 0.2–0.3 Gy, but then show increased resistance above this dose range (Mothersill et al. 2002). That is cell lines which had the largest hyper-radiosensitivity/increased radioresistance had the greatest radioprotection induced by the 0.1 Gy priming dose to subsequent high doses. A protective classical adaptive response was not observed in the radiosensitive HPV-G and HaCAT cell lines which are non-transformed keratinocytes. These lines classically show a bystander effect (Lambin et al. 1993) and this phenomenon is known to be associated with cell lines which do not show HRS/IRR or adaptive response (Lambin et al. 1993; Ryan et al. 2009). Clearly there is an interesting mechanism which directs cells towards HRS/IRR/AR responses or towards bystander responses. Our previous work with bystander effects and SDR suggest that SDR may be associated with cell lines showing pronounced bystander effects (Mothersill et al. 2002; Ryan et al. 2009), which would mean AR and SDR should not be associated even though it would seem logical to expect them to be related mechanistically.
These experiments were designed to examine how the adaptive response for three cell lines depended on the magnitude of the priming dose and the incubation period between priming and challenge doses. Cells were exposed to 0, 0.1, 0.5 or 2 Gy doses at various intervals prior to a 2 Gy challenge dose. The 0.1 and 0.5Gy doses would be regarded as “priming” doses while the 2×2Gy dose is a classic split dose used in radiotherapy.
MATERIALS AND METHODS
Cell Lines
HPV-G cells were originally obtained as a gift from Dr. J. Di Paolo, NIH, Bethesda, MD. These cells are non-transformed human keratinocytes immortalized by the HPV 16 virus (HPV-G), in these cells p53 has been suppressed by E6 protein but they express approximately 30% of the wild-type protein (Pirisi et al. 1992; Mothersill and Seymour 2001). HaCAT cells were originally obtained as a gift from Dr P. Boukamp, DKFZ, Germany. These cells are immortal but non-transformed human keratinocytes, with a point mutation in TP53 on one allele and a deletion of the gene in the other allele, therefore they half the gene dose of mutant p53 (Boukamp et al. 1990; Lehman et al. 1993; Datto et al. 1995). HT29 cells were obtained as a gift from Dr M. Joiner, Karmanos Cancer Institute, Wayne State University, Detroit, MI. HT29 cells are derived from a human colon adenocarcinoma, are radioresistant and have mutant p53 status (Popanda et al. 2000; Mothersill et al. 2004).
Cell Culture
All reagents for cell culture were obtained from Gibco (Grand Island, NY) unless otherwise stated. All cell culture was performed in a class two laminar flow cabinet. Cell stocks were maintained in 250 ml flasks in 30 ml of medium. The cell lines were all adapted over 6 months to grow in Dulbecco's MEM:F12 (1:1) containing 10% fetal calf serum, 10 U ml−1 penicillin and 10 μg ml−1 streptomycin sulphate, 25mM Hepes buffer and 1μg ml−1 Hydrocortisone (Sigma, St Louis, USA). Cells were incubated at 37°C with 5% CO2 in air and 95% relative humidity. Subculture was routinely performed when cells were 80–100% confluent using a 1:1 solution of 0.25% trypsin and 1 mM EDTA at 37°C.
Irradiation
Cells were irradiated in T-25 flasks six hours after plating using a cobalt-60 teletherapy source (Juravinski Cancer Centre, Hamilton, Ontario) at a flask to source distance of 80 cm and a field size of 35×35 cm. The dose rate during these experiments was approximately 1.7 Gy min−1. TLDs were used to confirm that the appropriate dose was delivered. Flasks were returned to the incubator immediately after irradiation.
Clonogenic assay technique
The sensitivity of HPV-G cells to direct irradiation or exposure to irradiated cell conditioned medium (ICCM) was determined by the clonogenic assay technique. A detailed description of these methods are given in Mothersill and Seymour (Mothersill and Seymour 1997). Briefly, sub-confluent flasks received a medium change the day prior to experimentation. Cells were removed from flask by incubation in a 1:1 solution of 0.25% trypsin and 1 mM EDTA. After detachment the cells were washed once, resuspended in fresh medium, and syringed gently to produce a single cell suspension. To determine the number of viable cells, an aliquot of the cell suspension was mixed with trypan blue exclusion dye and counted using a hemocytometer. Appropriate cell numbers were plated for survival analysis using the clonogenic assay technique of Puck and Marcus (Puck and Marcus 1956). The number of cells plated was selected to be within a range which ensured a linear relationship between the number of cells plated and the number of colonies returned. Cell cultures were incubated for 10 days at 37°C, 5% CO2 and 95% relative humidity. The cells were then stained with CarbolFuschin (Ricca Chemical Company, Arlington, TX) and colonies exceeding 50 cells were scored as representing surviving cells.
Adaptive Response
For each dose point an appropriate number of cells was plated to ensure a linear relationship between the number of cells plated and the number of colonies returned. Cells were irradiated with the required priming dose (0.1 or 0.5 Gy) 6 hours after plating. At the same time controls were sham irradiated and all cultures were then incubated for various intervals (0, 1, 3, 7, 9, 24 or 33 hours) prior to receiving a 2 Gy challenge dose. Colonies were scored after cell cultures were incubated for 10 days at 37°C, 5% CO2 and 95% relative humidity. Colonies exceeding 50 cells were scored as representing surviving cells.
Split Dose Recovery
An appropriate number of cells were plated to ensure a linear relationship between the number of cells plated and the number of colonies returned. Cells were irradiated with two 2 Gy doses. The first was administered 6 hours after plating. Single dose irradiation controls were irradiated with 4 Gy administered 6 hours after plating. Absolute controls were sham irradiated. All cultures were then incubated for various intervals (1, 3, 7, 9, 24 or 33 hours) prior to receiving the second 2 Gy dose. Colonies were scored after cell cultures were incubated for 10 days at 37°C, 5% CO2 and 95% relative humidity.
Statistical analysis
Data are presented as means ± SEM for three independent experiments containing a minimum of three replicate flasks per experiment. Significance was determined using the unpaired
RESULTS
The response of HaCAT, HPV-G and HT29 cells to ionizing radiation is illustrated in Figure 1. The non-transformed cell lines (HaCAT and HPV-G) showed similar responses for all doses less than 2 Gy. HaCAT cells were more radiosensitive at doses greater than 2 Gy. Both of these cell lines had a linear quadratic dose response curve, indicating repair at low doses. The dose response curve for the malignant HT29 cell line showed a hyper-radiation sensitivity response at low doses (<0.5 Gy), but was more resistant to radiation at doses greater than 0.5 Gy.

Survival fraction for cell lines HaCAT (•), HPV-G (▪) and HT29 (△) after direct exposure to 60Co gamma radiation. Values are presented as the Mean ± SEM for n=9 from three experiment.
Table 1 illustrates the relative change in the three cells lines pretreated with low-doses of radiation (0.1 or 0.5 Gy) prior to challenge dose compared to controls without pretreatment. Values greater than unity indicate greater cell survival for cells that received a small pretreating dose. For the non-transformed cell lines (HPV-G and HaCAT) pre-treatment with low-doses of radiation shortly before the challenge dose (≤ 3 hours) stimulated cell lines, leading to increased survival (0.1 Gy priming dose 3 hours prior to challenge,
Relative change in survival fraction for cell lines receiving priming dose (0.1 or 0.5 Gy) to unprimed controls. A significant change in survival after exposure to priming dose when compared to unprimed control is represented by ∗ (where

Summary Table for HPVG

Summary Table HaCAT

Summary Table for HT29
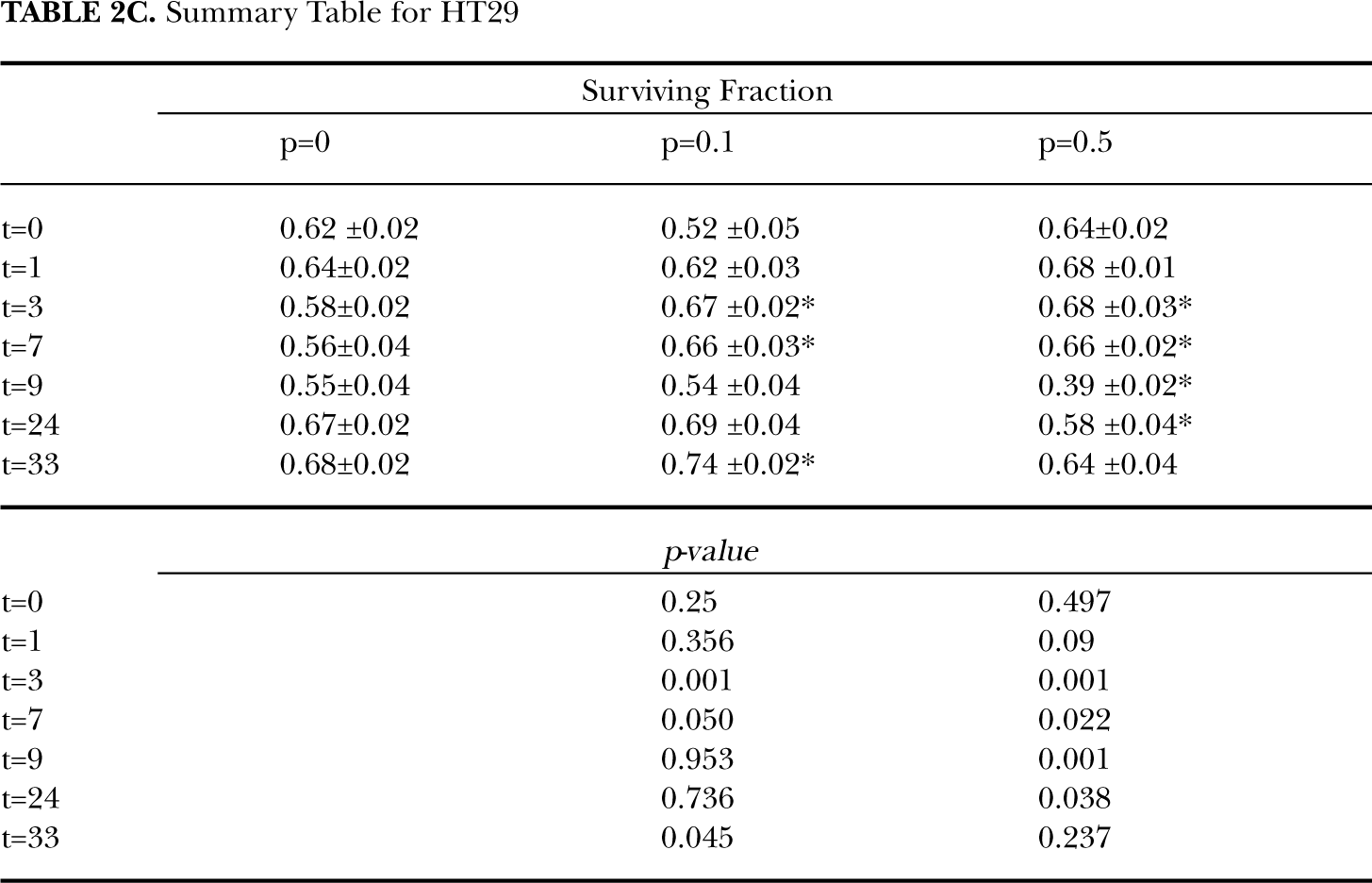
The relative change in survival for three cell lines exposed to three different priming doses (0.1, 0.5 and 2 Gy) at various incubation periods is illustrated in Figure 2. For HPV-G cells, small priming doses initially stimulated cell survival. Greater cell survival was observed for primed samples when compared to unprimed samples for incubations periods less than 3 hours for 0.1 and 0.5 Gy priming doses (at 1 hour SF0.1 + 2 = 1.19 ± 0.05,
Relative change in survival for cells with 0.1, 0.5 Gy or 2 Gy priming dose after exposure to 2 Gy challenge dose compared to cells which did not received sham prime irradiation. Three cell lines were tested, HPV-G (A), HaCAT (B) and HT29 (C). Values are presented as the Mean ± SEM for n=9 from three experiment.
The recovery response is a comparison of the survival at various incubation times to the response when treatment delivered as a single exposure. The response curve for cells exposed to different priming regimes (0.1 + 2 Gy or 0.5 + 2 Gy) follows a similar trend for all cell lines tested, that is a similar recovery response was observed for all cell lines when the total single radiation exposure was compared to treatments separated into two fractions irrespective of the priming dose.
Figure 3 shows the recovery factor for HaCAT, HPV-G and HT29 cultures which were irradiated with two 2 Gy fractions at various time intervals. The survival for the non transformed HaCAT and HPV-G cells initially decreased, however these cell lines demonstrated recovery when the doses were split by incubations greater than 9 hours. From Fig. 3 it can be seen that the HPV-G and HaCAT cell lines are initially more sensitive to the fractionated regime than the single dose exposure, however cell survival increases as the culture is allowed longer to recover between doses. A split dose recovery is observed for the HPV-G and HaCAT cultures with greater than 9 hours incubation between fractions.

The recovery factor for HaCAT (♦), HPV-G (▪) and HT29 (△) cell lines. The recovery factor is the ratio of split dose (2 × 2 Gy) to single fraction 4 Gy for the various timepoints. Values are presented as the Mean ± SEM for n=9 from three experiment.
Cell survival for the malignant HT29 cell line either exposed to a single or split 4 Gy dose did not differ significantly for incubations less than 7 hours between exposures. Increased cell-killing was observed in the HT29 cell line when the two fractions were separated by 9 hours (Recovery factor2+2 = 0.70 ± 0.03,
DISCUSSION
In these experiments two different dose-response curves were observed after cells were treated with direct 60Co radiation (Fig. 1). For doses lower than 0.5 Gy the non-transformed cell lines HaCAT and HPVG demonstrated a shoulder of sub-lethal repair for low doses, while the malignant HT29 cell line showed increased cell killing – a hyper-radiosensitivity response. This is in agreement with previous studies examining the low dose properties of these cell lines (Lambin et al. 1993; Mothersill et al. 2002). It is also worth noting that the malignant cell line is substantially more resistant to high radiation doses that the non-transformed “normal” cell lines.
Three cell lines of various radiosensitivity and p53 status were investigated in the current study. The response of these cell lines to the various treatment regimes was quite diverse, suggesting that the cell line used is a critical factor. The non-transformed, radiosensitive HPV-G and HaCAT cell lines did not demonstrate a classic adaptive response. These cell lines demonstrated a prompt transient stimulation of survival in these cells pre-treated with low-dose radiation prior to challenge dose. However, the kinetics are not those typically expected by adaptive response, which predicts increased cell survival in cells pre-treated with a small “priming” dose prior to a larger challenge dose. HPV-G and HaCAT cell lines did not exhibit classic split dose recovery for short incubations periods. Indeed, incubations less than 8 hours induced increased-sensitivity in these cell lines. On the other hand, the malignant, radioresistant HT29 cell line demonstrated a classic adaptive response/recovery. A similar recovery response was induced by 0.1 Gy and 0.5 Gy priming doses in all three cell lines; this would suggest that the magnitude of the priming dose is irrelevant for recovery in these cell lines (Fig. 2). This appears to be consistent with recent results published by Day et al. 2006, who observed that priming doses of 0.001 – 10 mGy followed caused a similar protective adaptive response.
HT29 was the only cell line of the three examined in this study to exhibit a classic adaptive response (Fig. 2C). This cell line demonstrated a radioprotective adaptive response 3 hours post-priming. This supports the theory that the adaptive response is probably dependent on de novo protein synthesis which results in upregulating DNA repair mechanisms (Joiner et al. 1996). Nonetheless, the adaptive response seems to diminish with increasing time interval between the priming dose and the challenge dose. This suggests that the protein synthesized subsequent to the priming dose is short lived and subject to quick degradation and therefore eliminates the effect shown by the priming dose.
In the current study, none of the cell lines showed split dose recovery. A recovery factor of 1.1 – 1.3 was observed for the non-transformed HaCAT and HPV-G cell lines when cultured for greater than 8 hours, however, this is likely a result of cell division. This is in agreement with earlier split dose recovery experiments with the HPV-G line (Mothersill and Seymour 2002). The formation of micro-colonies was observed in flasks plated with unirradiated cells over this time period (Fig. 4). For the 33 hour incubation time-point (38 hours post-plating) 33% of the HPV-G cells had doubled. Therefore part of the increased survival observed in the 33 hour split-dose recovery could be due to cell multiplicity. That is when cells were exposed to the second radiation dose, a third of the cells had divided therefore one would expect increased colony formation as two cells would need to be lethally damaged in the microcolony to result in no colony forming. Unexpectedly, no prompt repair of sub-lethal damage was observed in either of the sensitive cell lines (Fig. 3). Sub-lethal damage is used here as a classical radiobiological term (Elkind et al. 1967) used to explain why many mammalian cell lines show a “shouldered” dose response when the data are plotted in semi-log format. These cell lines showed an initial decrease in recovery factor indicating increased cell killing for doses with a short interval between fractions when compared to a single dose, a recovery factor of 0.9 and 0.8 were observed for a total 4 Gy dose over 3 hours for HaCAT and HPV-G respectively. It is possible that the initial 2 Gy dose had left the cells in a stressed state resulting in the generation of reactive oxygenation species which consequently resulted in increased sensitivity to subsequent doses. While this is perhaps speculative it is plausible and consistent with current paradigms which link low dose effects to generalized stress responses (Taghian et al. 1993). Cells typically cope with radiation-induced damage by activating repair mechanisms. This requires the synthesis of proteins which requires 2–3 hours. The observed increased cell killing for doses split by less than 3 hours may be a result of ineffective repair between doses compounded with the increased cellular stress. Alternatively, these cells may have been synchronized by the first 2 Gy treatment, and have entered into a sensitive phase of the cell cycle and therefore the second 2 Gy dose 3 hours later may induce-increased cell killing.
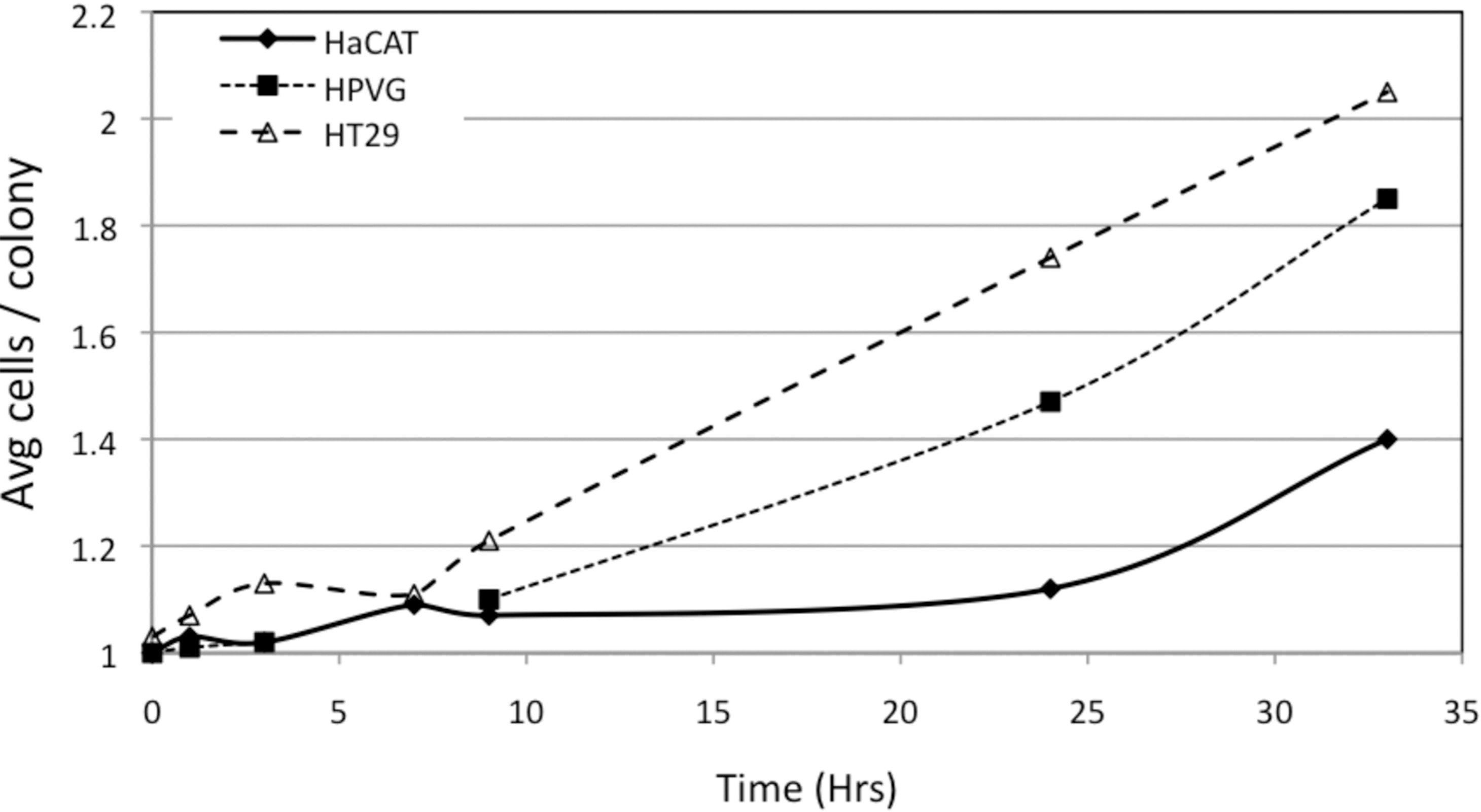
The average number of cells per microcolony plotted against time post plating for each cell line.
The malignant HT29 cell line did not exhibit split dose recovery (Fig. 3). No change in survival was observed when the two 2 Gy treatments were separated by less than 8 hours. The cell line was particularly sensitive when incubated for 9 hours between the two 2 Gy treatments. This is a rapidly dividing cell line, and the initial treatment may have synchronised the cells into a sensitive G2/M phase for the second dose.
Unlike the adaptive response, increased cell survival due to split dose recovery had a slower onset and was not observed for incubation times less than 8 hours. The effect was greatest in the more sensitive non-transformed cell lines. It has previously been reported that the split dose response was most effective 6 hours after irradiation (Taghian et al. 1993). It has been well documented that exposure to low doses of radiation triggers different responses in cells than those activated by high doses (Mothersill and Seymour 2006). The different kinetic responses observed for the two phenomena, the adaptive response and split dose recovery 1 could signify that these two phenomena are a result of different mechanisms activated within cells. For example the ATM cascade is a well documented response of cells to radiation induced DNA damage (Lobrich and Jeggo 2005; Pereg et al. 2006; Rashi-Elkeles et al. 2006). The activation of ATM signaling by ionizing radiation causes chk2 mediated cell arrest at G2/M, which has been shown to remain inactivated for doses below 0.4 Gy (Marples et al. 2003).
In summary these experiments showed no significant relationship between the adaptive response and split dose recovery at doses relevant in radiotherapy. Cells with the most pronounced adaptive response did not show split dose recovery at all, while the non-transformed cell lines showed no significant split dose recovery and a very transient adaptive response. It would appear that the time between pre-treatment and challenge doses is important. The kinetic response occurred in two phases, adaptive response/recovery was activated when the priming dose was administered shortly before the challenge dose (0–8 hours). When the two doses were separated by longer intervals, pre-treatment with low doses prior to challenge, caused increased cell killing – a negative recovery or sensitisation resulting from the fractionated dose.
The findings have implications for our understanding of mechanisms involved in recovery, repair and adaptive response.
Footnotes
ACKNOWLEDGEMENTS
This work was supported by NSERC Discovery and IRC grants and the Canada Chairs Programme, Bruce Power, CANDU Owners Group and OPG. The authors would like to thank Juravinski Cancer Centre Physics Department for assistance irradiating samples and dosimetry.
